Abstract
Objective:
Surface-modified titanium material was prepared to explore its corrosion resistance and microbial adhesion.
Methods:
Atmospheric pressure plasma was used to modify the surface of pure titanium. The surface morphology and adhesion morphology of P. gingivalis (Porphyromonas gingivalis) were observed through morphology scanning electron microscopy. Reactive oxygen species (ROS) formation was detected using a fluorescence spectrophotometer. Electrochemical corrosion properties of the material were detected using simulated fluids and SAGF artificial saliva as electrolyte. Bacteria suspension absorbance was tested using an enzyme standard.
Results:
After plasma modification, the material surface exhibited numerous ROS formations and improved wettability. Compared with the control group, corrosion potential was higher but corrosion current was lower in simulated body fluids and artificial saliva. Bacterial-suspension absorbance was significantly lower than that in the control group on days 3 and 5 (P <0.05).
Conclusion:
The electrochemical and microbial corrosion performance of titanium can be improved by atmospheric pressure plasma. Therefore, this method can thus be used for the surface modification of oral and bone implant materials.
Keywords
Introduction
Pure titanium and its alloys are widely used in orthopedic fixation and oral planting materials because of their good biocompatibility and corrosion resistance. Titanium usually forms a dense inert oxidation film in air, and the high electrochemical impedance of the film endows titanium and titanium alloys with good corrosion resistance. 1 However, infections on and around titanium implants remain a difficult problem. Implant-associated infection may lead to implant failure for the formation of a bacterial surface biofilm and compromised immunity at the implant/tissue interface. 2 The bacterial biofilm on the implant surface offers protection from host defense and antimicrobial treatment. 3 Therefore, the antibacterial surface modification of Ti implants is significant in preventing the initial bacterial adhesion and endowing titanium with antimicrobial properties. 4 However, bone integration performance is poor despite the good biocompatibility of titanium. Given that the surface oxidation film is an inert material, combination with bone tissue to form chemical bonds is difficult. 5 As such, researchers have modified the surface of titanium by adding biological ceramics and metal ions or hydroxyapatite to increase biological activity and performance in bone integration.6–8 However, body or oral implants are easily susceptible to bacterial contamination and microbial corrosion. In recent years, the application of tooth gum, toothpaste, and tooth-whitening agents with fluorine has gained popularity.9–11 Studies have shown that the corrosion resistance of titanium declines significantly within a pH range of 3.2–7.8 and in high fluorine concentration.12,13 The poor corrosion resistance of titanium causes a high incidence of inflammation. Inflammation is particularly rampant in dental implants, in which copious normal flora abounds, thereby leading to eventual failure in implant operations. Microbes exert no direct effect on the corrosion of metallic implants but are capable of altering the local environment of such implants and promoting electrochemical corrosion. A study has shown that microorganisms are associated with implant infections and that the main pathogenic bacteria are viscous actinomycetes, gums, porphyrin, and the bacterium Streptococcus mutans. 14 Hence, electrochemical corrosion resistance and antimicrobial properties are important in implant materials, and surface modification to improve the corrosion resistance of implants under physiological conditions is important for the clinical success rate of titanium implant materials. The present study explores the use of atmospheric pressure plasma jet in the surface modification of pure titanium. By observing the bacterial adhesion and changes in the corrosion resistance of titanium, this study provides a basis for the surface modification of titanium implant materials.
Methods and materials
Material preparation
Pure titanium was cut into wafers with a thickness of 2 mm and diameter of 1 cm and polished with 600-mesh sandpaper. Samples with a roughness of about 50 ± 0.02 m – n were obtained. Samples were washed ultrasonically with acetone, ethanol, and deionized water for 15 min to remove organic matter from the surface. We used Ar + O2 (2%) as gas source, a 500 V DC power supply with a 35 mA output, and a gas flow rate of 300 L/h. The end of the plasma jet temperature was 38°C, and the jet was operated for 5, 10, and 15 min.
Characterization of samples
Surface topography was observed through scanning electron microscopy (SEM). The contact angle of the sample surfaces were measured by using a contact angle measurement instrument. The fluorescence intensity of the PBS leaching solution was tested by using a fluorescence spectrophotometer at an excitation wavelength of 488 nm and emission wavelength of 525 nm. The control group Ti consisted of pure titanium, whereas the experimental groups consisted of P-Ti5, P-Ti10, and P-Ti15, which were treated by low-temperature atmospheric pressure plasma for 5, 10, and 15 min, respectively.
Electrochemical corrosion experiment
With pure titanium samples as the control, the corrosion resistance of the samples was evaluated at 10 min after plasma processing. Simulated body fluids (SBF) and SAGF artificial saliva were used as electrolytes. The samples were immersed in a water bath with a temperature of 37°C under natural ventilation conditions.
Bacterial adhesion experiment
P-Ti10 was considered as the experimental group, and Ti is the control group. Gum porphyrins were selected as the experimental strains of the bacterium groups. A total of 20 µL of gum porphyrin liquid was added in each group. Three holes were repeatedly set in each group. A culture medium containing 1% sucrose and a BHI culture medium containing 1% glucose were also used. On days 1, 3, and 5, the floating bacteria were washed with PBS. The measured absorbance value was 420 nm. The morphology of bacteria adhesion was observed through SEM.
Statistical analysis
The absorbance difference among groups was analyzed at various points by single factor variance analysis by SPSS 17.0 statistical software.
Results
Figure 1 shows the SEM results and contact angles of the Ti and P-Ti specimens. The Ti surface is flat, whereas those of P-Ti5, P-Ti10, and P-Ti15 appear rough. The deionized water contact angles of the specimens are as follows: Ti θ = 76.9 ± 2.30; P-Ti5 θ = 69.9 ± 2.21; P-Ti10 θ = 60.2 ± 2.21; and P-Ti15 θ = 61.3 ± 1.78. The fluorescence intensity of the leaching solution increased after subjecting the solution to low-temperature atmospheric pressure plasma for 10 and 15 min. Afterwards, fluorescence intensity reached the same level as that in Figure 2.

SEM images of the material and the contact Angle. A : Ti. B : P-Ti10.
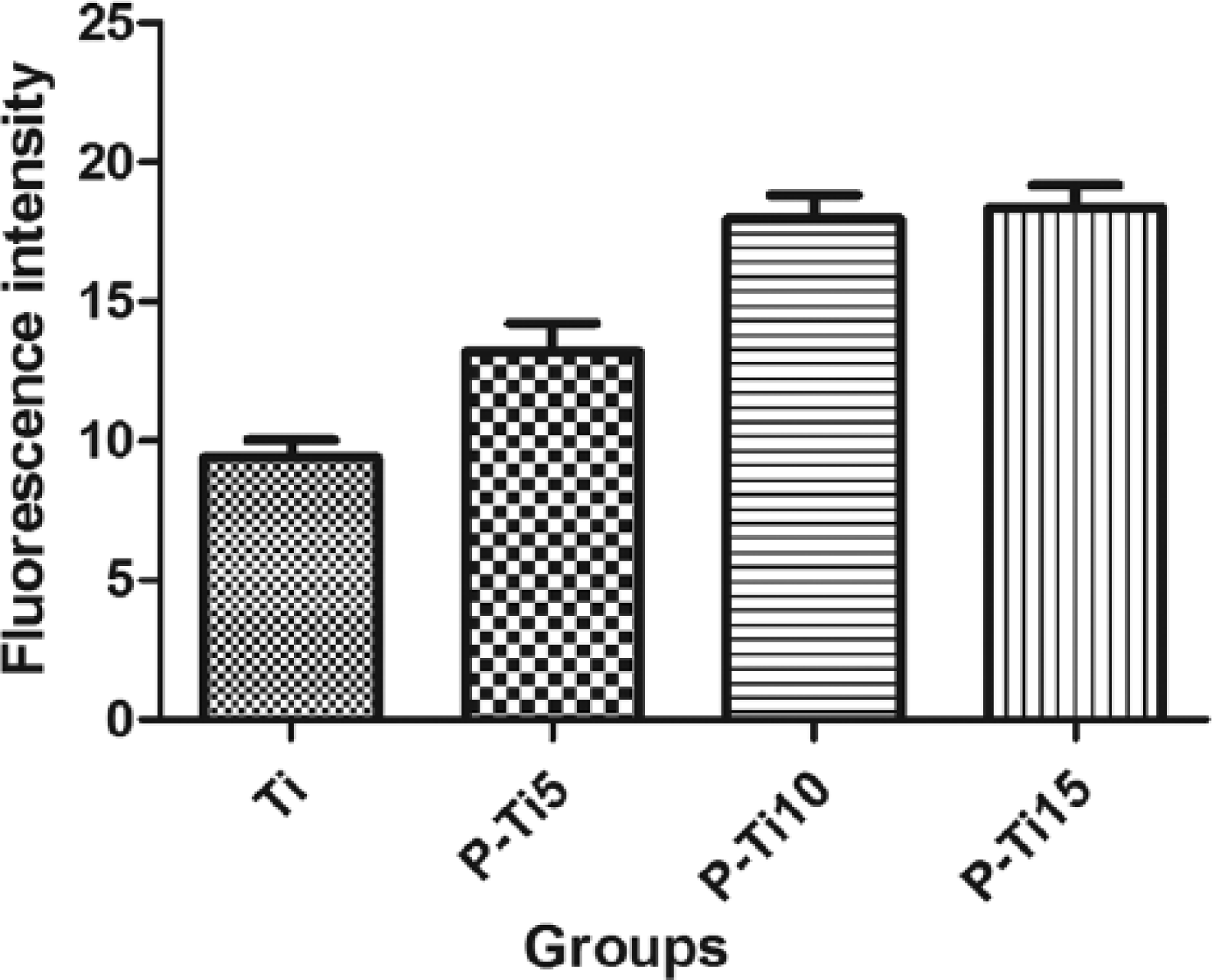
Fluorescence intensity between groups.
The corrosion potential (Ecorr) and corrosion current (Icorr) are shown in Table 1. The corrosion potential of P-Ti10 is higher than that of pure titanium, and the corrosion current of P-Ti10 is lower than that of pure titanium. This finding indicates that P-Ti10 presented better corrosion resistance in SBF and SAGF artificial saliva than pure titanium.
Ecorr and icorr values of group P-Ti and group Ti in SBF and artificial saliva.

The adhesion morphology of each group on days 1, 3, and 5 is shown in Figure 3. Bacterial clumps began to form on the surface of pure titanium on day 3, and P-Ti10 exhibited relatively low bacterial adhesion. On day 5, the formation of the bacterial clumps also began on P-Ti10, but the quantity of the clumps was obviously lower than that in the pure titanium group. The absorbance difference among the groups was analyzed at various points by single factor analysis using the SPSS 17.0 statistical software. The results are shown in Table 2. On day 1, no significant differences were found among groups. On days 3 and 5, the bacteria adhesion of group P-Ti10 was obviously lower than that of group Ti.
OD values on different time points (x ± s, a = 0.05).

P <0.05 vs. group Ti.

Bacterial adhesion SEM images.
Discussions
Ti surface treatment technologies are employed to improve its osteointegration property for biomedical application. Classic Ti modifications usually encompass the following two main aspects, that is, changing the surface morphology structure and then optimizing the surface chemical composition and mechanical properties. Sandblasting, 15 acid etching,16,17 anodization, 18 and plasma spraying are common methods for Ti surface modification. 19 The methods are capable of cleaning surface contamination and altering chemical composition and surface topography. These modification methods can induce a certain roughness of hierarchical porous morphology characteristics and the anatase crystalline phase on the titanium surface. Numerous works have focused on the biological properties in adhesion and osteointegration regardless of the inflammatory response around the implant. The absence of infection is one of the necessary factors to obtain the best osteointegration.
In the present work, we prepared a new modified titanium material with anti-inflammatory properties. Low-temperature plasma jet technology was employed in the modification of pure titanium. Changes in the performance of the titanium surface were evaluated by using Ar + O2 (2%) as gas source, a 500 V DC power supply with a 35 mA output, and 300 L/h gas flow rate. The surface roughness of titanium increased, and the active oxygen concentration reached its highest when titanium was treated with plasma for 10 min. The electrochemical corrosion experiment confirmed the increase in the corrosion resistance of the experimental and control groups in artificial saliva and simulated body fluids. The bacterial adhesion experiment confirmed that the sample inhibited bacterial adhesion after plasma treatment. In turn, this finding confirmed that low-temperature atmospheric pressure plasma can be used as a method for the modification of bone materials and oral implant materials.
Footnotes
Acknowledgements
The authors are grateful to the Project of Shandong Province Higher Educational Science and Technology Program (no. J14LC56).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
