Abstract
Objectives:
The intestinal fatty acid binding protein (I-FABP) is a useful marker in the assessment of damages and inflammation to the small intestinal mucosa. We have investigated the influence of HIV infections and antiretroviral treatment on the small intestine mucosa aided by the evaluation of I-FABP concentration levels.
Methods:
We included in the study: 31 HIV-infected patients with antiretroviral therapy (ART) treatment (T), 32 HIV-infected patients not treated with ART (NT), and 18 healthy volunteers as a control group. The concentrations of I-FABP were measured in serum using a sandwich enzyme immunoassay kit: Human I-FABP.
Results:
The median level of I-FABP in the serum of HIV-infected patients was 2.10 ± 2.56 ng/mL and did not significantly differ between T and NT (2.32 ± 2.69 ng/mL vs. 1.89 ± 2.45 ng/mL). However, the I-FABP serum level was significantly higher in HIV-infected patients when compared to the group control (2.10 ± 2.56 ng/mL vs. 1.26 ± 1.18 ng/mL; P = 0.048). We recorded a correlation for treated HIV-infected patients between Viral load and I-FABP serum level (r = −0.66; P <0.05).
Conclusions:
Elevated serum I-FABP level in HIV-infected patients confirm that HIV causes damage to the intestinal mucosa. I-FAB is a useful marker in the assessment of intestinal damage in HIV-infected patients.
Keywords
Introduction
HIV infection leads to the damage of CD4+ T cells and numerous changes in different cells of the human body. Majority of the intestinal and peripheral CD4+ T cells are destroyed at all stages, in the acute and in the chronic phase of HIV disease.1,2 One of the main localizations for HIV activity is the immune system of the gastrointestinal tract (GI) and the majority of CD4+ T cells are located in the mucosa of the colon (gut-associated lymphoid tissue [GALT]), and there is a report which shows that lymph nodes contains more HIV-specific CD8+ cells than peripheral blood. 3 Furthermore, the gastrointestinal tract has the most substantial CD4+T cell reduction in the HIV-disease. The main mechanisms of the CD4+T cells depletion is apoptosis of uninfected bystander CD4+ T cells. 4
As mentioned above the HIV infects and destroys the immune system of the gastrointestinal tract; moreover, it is known that HIV causes structural abnormalities in the intestinal mucosa. 5 Thirty years ago, it was observed that HIV-infected patients had histological damages of the GI mucosa, malabsorption, and lymphocyte reduction. 6 There are many ways in which HIV causes structural abnormalities of the enterocytes. For example, the gp120 molecule of HIV can cause an increase of calcium concentration in enterocytes, which can lead to disturbances in the ionic balances in the enterocytes. 7
The HIV protein Tat causes inhibition of the uptake of glucose in the enterocytes. 8 Moreover, it has been suggested that HIV may lead to abnormal differentiation of enterocytes. 9 Furthermore, a few studies have shown that the viral replication correlates with the degree of inflammation of the GI.10,11
However, the apoptosis is the main mechanism causing mucosal damage during the acute stage of the infection and is induced by the HIV protein tat and perforin-expressing CD8+ T cells.12,13 During the chronic stage of the infection, there is an increase in the mucosal production of inflammatory cytokines such as o TNF- α, IL-2, IL-4, and IL-13 which in turn have an influence on epithelial apoptosis, tight junction composition, and epithelial permeability. 14
Damage of the mucosal barrier induces translocation of the bacteria and bacterial lipopolysaccharides (LPS). There are studies which show that microbial translocation is associated with chronic systemic immune activation.15,16 Moreover bacterial LPS stimulate the increase of cytokines, such as TNF, which promotes inflammation and HIV replication. 17
The process of microbial translocation influences the immune system activation in HIV-infected persons, resulting in an increase of T lymphocytes circulation, a high level of active T lymphocytes, polyclonal activation of B lymphocytes, and increased levels of proinflammatory cytokines.15,18
Effective antiretroviral therapy (ART) causes a reduction of microbial translocation and this is shown in a few studies with the aid of LPS and other bacterial markers. 19 As a result of the difficulty in obtaining clear images of damaged intestinal mucosa caused by HIV infection, researchers are looking for new markers to assess the damages of the small intestine.
The intestinal fatty acid binding protein (I-FABP) is a small intracellular protein with a mass of 15 kDa, which plays a role in the transport and metabolism of the long chain fatty acids. 20 I-FABP is present in large amounts in the cells of the mucosa of the small intestine. It is known that the level of intestinal-FABP in the circulation increases rapidly after damage to the small intestinal mucosa. 16 It is a marker which is useful in the assessment of damages and inflammation of the small intestine mucosa. There are many reports which state that I-FABP is a useful diagnostic marker for example in the diagnosis of: necrotizing enterocolitis, mesenteric infarction, or ulcerative colitis.21–23 However, the number of HIV-infection studies which use I-FABP as a marker of the intestinal damage in their research is limited, but it is increasing all the time.
It seems that the major arguments for the use of I-FABP in the study of HIV are: first, the marker appears in the blood, which facilitates measurement; and, second, the protein is located in the gastrointestinal tract, which is one of the principal places of HIV activity.
Aim of the study
To assess I-FABP levels in untreated and treated HIV disease.
Materials and methods
We carried out a case control study which consisted of 63 HIV-infected patients hospitalized at the Department of Hepatology and Acquired Immunodeficiencies at the Medical University of Warsaw (12 women, 51 men). We included in the study patients aged ⩾18 years with confirmed HIV infection. The exclusion criteria were as follows: diagnosis of intestinal disease or non-AIDS-related cancers, drug addiction, and pregnancy. We also included 18 healthy volunteers as a control group in the study (11 women, 7 men; mean age, 24.17 ± 0.7 years). Patients were divided in two groups: 32 HIV-infected patients not treated with antiretroviral therapy (NT) and 31 HIV-infected patients with a minimum of half a year ART treatment (T). The treatment consists of standard triple antiretroviral regimen. Each patient was examined and one blood sample from every patient was collected for testing. In the collected blood samples we marked: CRP concentration, the number of CD4+ T cells, CD8+ T cells and HIV viral load. Serum I-FABP concentrations were measured using a sandwich enzyme immunoassay kit: Human I-FABP was measured on a range of 0.156–10.0 ng/mL and sensitivity 0.063 ng/mL (Uscn Life Science, Inc.). Patient data were gathered from their medical history and from the hospital database. The results were statistically analyzed by Statistica 10.0 software (StatSoft). We used the Mann-Whitney test to find differences between groups of patients and Spearman rank correlation to find correlations between variables. P <0.05 was considered statistically significant.
Results
The mean age of the study group was 38.8 ± 10.0 years (age range, 23–70 years). Table 1 shows demographic data and laboratory tests in ART-treated and ART-non-treated HIV-infected patients. Treated patients were older and had higher levels of CD4+ T cell count than the non-treated patients (mean 42.61 ± 10.97 years vs. 35.03 ± 7.49 years and 446.81 ± 263.50 cells/µL vs. 237.76 ± 212.74 cells/µL). Furthermore, we observed that CRP levels and viral load were significantly lower in T group in comparison to the NT group (63.0 ± 11.80 mg/L vs. 29.59 ± 43.13mg/L and 10,795.79 ± 40,299.45 copies/mm3 vs. 252,455.50 ± 288,471.76 copies/mm3). The median level of I-FABP in the serum of HIV-infected patients was 2.10 ± 2.56 ng/mL and did not differ significantly between T and NT groups (2.32 ± 2.69 ng/mL vs. 1.89 ± 2.45 ng/mL). Figure 1 shows the comparisons of serum I-FABP levels between HIV-infected patients treated with ART, HIV-infected patients without ART, and the control group. The I-FABP serum levels were significantly higher in HIV-infected patients when compared to the control group (2.10 ± 2.56 ng/mL vs. 1.26 ± 1.18 ng/mL; P = 0.048). However, for the rest of the analyzed group we did not find any significant difference.
Demographic data and laboratory tests in HIV infected patients treated and non-treated ART.


(a) Concentration of I-FABP (ng/mL) in HIV-infected patients and controls. (b) Concentration number of I-FABP (ng/mL) in HIV-infected patients without ART and treated with ART. (c) Concentration umber of I-FABP (ng/mL) in group control and HIV-infected patients without ART. (d) Concentration number of I-FABP (ng/mL) in group control and HIV-infected patients treated with ART. P values were calculated using Mann-Whitney test. Each symbol represents an individual.
We recorded a weak negative correlation for treated HIV-infected patients between viral load and I-FABP serum level (r = −0.66; P <0.05) (Figure 2). Nevertheless, there were no significant correlations between I-FABP serum level and CD8 count in both groups of HIV infected patients (T and NT).

Correlations: (a) CD4 and I-FABP levels at the HIV-infected patients, (b) viral load and I-FABP level at the HIV-infected patients, (c) CD4 and I-FABP levels at the HIV-infected patients treated ART, (d) viral load and I-FABP level at the HIV-infected patients treated ART. r values were calculated using Spearman’s correlation test.
We also examined the influence of two protease inhibitors Lopinavir and Ritonavir on the I-FABP serum level. We compared the control group with patients treated with Lopinavir/r and Ritonavir, and also compared patients treated with other drugs to those treated with Lopinavir/r and Ritonavir. But both comparisons were not statistically significant (Figure 3).
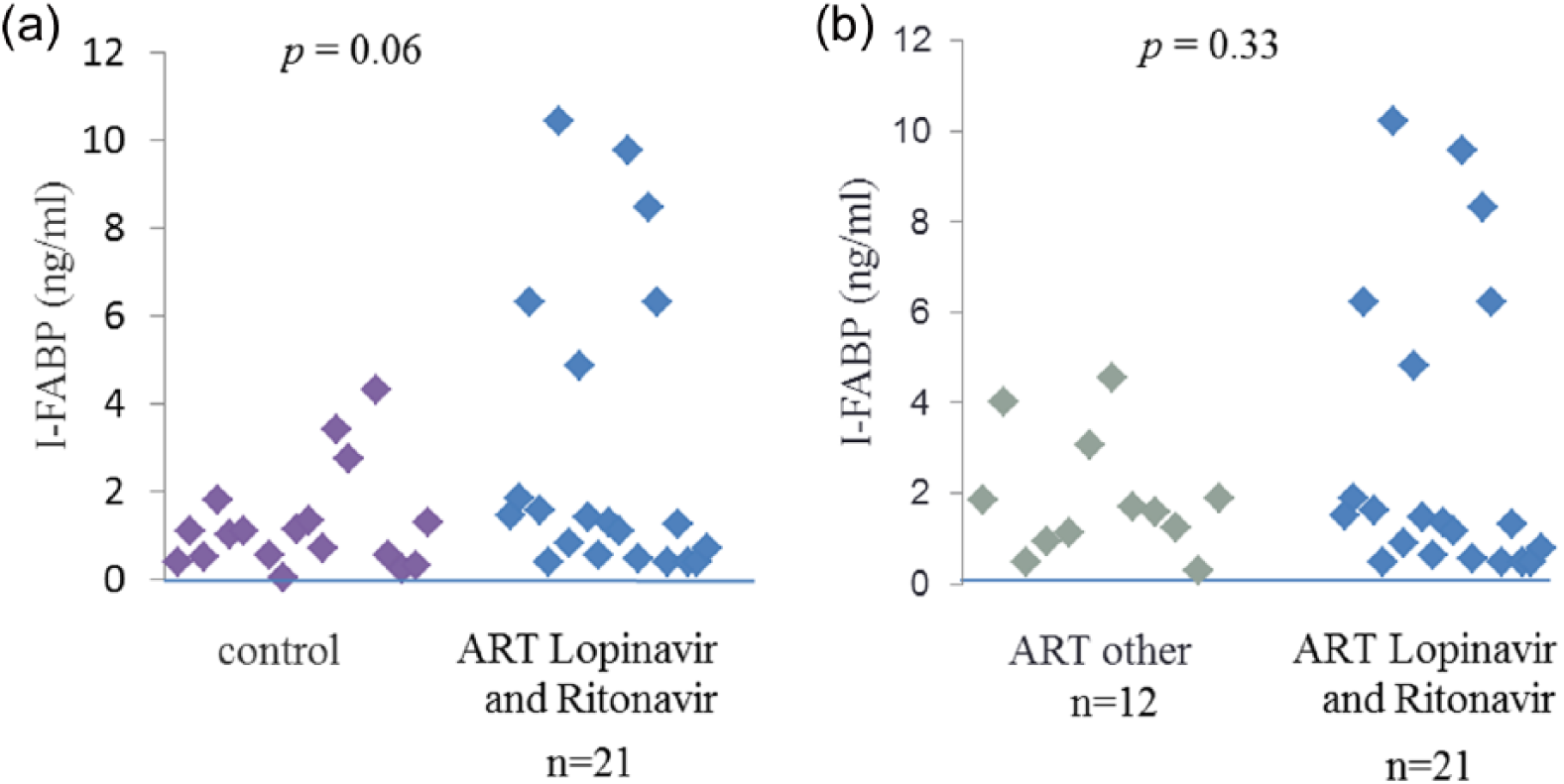
(a) Concentration of I-FABP (ng/mL) in HIV-infected patients treated with Lopinavir and Ritonavir and controls. (b) Concentration of I-FABP (ng/mL) in HIV-infected patients treated with Lopinavir and Ritonavir and treated with other drugs. P values were calculated using Mann-Whitney test. Each symbol represents an individuals.
Discussion
There are many trials which demonstrate the damaging effect of HIV to the intestinal mucosa, the mucosal barrier and the microbial translocation due to damaged intestinal mucosa.1,2,24
Results of the study of Pelsers et al. show that the I-FABP serum level is significantly higher in HIV-infected patients when compared to the healthy control group. 25 If we get the same results in our analysis, then we can conclude that I-FABP is a useful marker in distinguishing between HIV-infected patients and healthy individuals. But is it enough to evaluate a small intestinal injury in HIV-infected patients? Two recent studies have shown a relationship between CD4+ cell counts and I-FABP levels in HIV infected patients treated with ART: the decrease of CD4+ cell count is correlated with an increase of I-FABP level.26,27 Our study did not show this correlation, but it can be the result of the differences in the study groups or non-effective treatment. Both the first study as well as our study included patients at the chronic stage of infection; the number of patients were 20, but they had been treated with ART for a median of 66 months. In our study the period of ART had a median of 6 months. The second study included 254 HIV-infected patients treated with ART. Differences such as a shorter period of ART therapy or the number of patients included in the studies, can have a big impact on the differences in our results.
Assuming the development of HIV infection causes damage to the intestinal mucosa, we checked if there was a relationship between I-FABP serum level and viral load, and we recorded a negative correlation, which did not confirm our conjecture. The decrease of the viral load, which is the result of effective ART treatment, correlates with increased levels of I-FABP in the patients’ serum. We can hypothesize that ART can cause an increase in the level of I-FABP as a side effect, or that the period of treatment is too short for a reconstruction of the small intestine mucosal barrier.
On one hand, the effective treatment should result in lower levels of I-FABP and on the other hand, without treatment I-FABP levels should be higher. And if not, why not? Successful treatment with ART and an increase of CD4+ cell count should protect intestinal mucosa. But recent studies have shown that the effective treatment with ART leads to a near complete recovery of CD4+ T cell count population in the blood. However, significantly lower number of CD4+ cells are located in the GI mucosa, despite long-term suppressive therapy.2,26
Other studies shown that HIV-infected patients with effective and long-term ART exhibited a decrease of I-FABP levels. Furthermore, the therapy caused a decrease of microbial translocation and immune activation.27,28 In the present study we compared the levels of I-FABP between the HIV-infected patients treated and not treated, but it did not differ significantly. The different findings in our study may be due to the fact that we are comparing two different groups of patients: treated with ART and not treated with ART. In previous studies, the same group of patients receiving ART was observed over a period of time. In addition, the time of the therapy was longer and study groups had greater numbers of patients.
It is known that therapy with protease inhibitors (PIs) has a negative impact on the digestive system and causes side effects, such as abdominal pain, diarrhea, and abnormal stools. The study by Wu X et al. showed that therapy with PIs especially with: Ritonavir and Lopinavir, significantly interrupted intestinal epithelial barrier integrity and induced apoptosis. 29 We compared HIV-infected patients on therapy with Lopinavir and Ritonavir to patients on therapy with other antiretroviral drugs, but the comparison was not statistically significant. It would probably be more valuable to compare the same group of patients treated with Lopinavir and Ritonavir over a period of time.
The title of our study is “The role of serum I-FABP concentration in assessment of small intestine mucosa among HIV-infected patients” and we particularly confirm this role.
On one hand, it is known that HIV causes structural damage to the intestinal mucosa and our study shows an increase of the I-FABP in HIV-infected patients. On the other hand, many trials show that treatment with increased lymphocytes CD4+T leads to lower level of the i-FABP. Our study did not show this correlation. The differences in our results to other studies may be the result of differing study groups or non-effective treatment. Following the review of current publications, alongside our own results, we can confirm that I-FABP is a useful marker to assess intestinal damage in HIV-infected patients.
However, our study has not brought anything new to existing knowledge on this topic. The issue of the use of i-FABP in HIV infections is very interesting and requires further research.
Footnotes
Acknowledgements
We are indebted to the patients and healthy volunteers who took part in this study. We would like to thank Alicja Fic from of Institute of Immunopathology at Medical University of Warsaw for her assistance with the I-FABP assays.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by a scientific grant for Young Scientists number 1M22/NM1/11 from the Medical University of Warsaw.
