Abstract

Introduction
The incidence and prevalence of diabetes are increasing in Canada. 1 Currently, one-third of the Canadian population is living with diabetes or prediabetes. Moreover, Canadian adults are now at 50% risk of developing diabetes during their lifetime. 1
Despite its serious complications and the advancement in diabetes care, optimal community-based care for patients with diabetes remains elusive. Indeed, it has been reported that only 13% of the Canadian community-dwelling patients met the triple target of glycemic control, lipids and blood pressure. 2 Furthermore, Al Hamarneh and colleagues 3 found that around half of community-dwelling patients with diabetes are not achieving their glycemic control target.
Such sobering statistics indicate the need for new and innovative ways to tackle one of Canada’s largest public health issues. 4
Pharmacists are highly accessible primary care professionals who see patients with diabetes frequently 5 and have strong interest in diabetes management. 6 Their interventions in patients with diabetes are well supported by high-level evidence in the literature.7-14 In fact, in their systematic review, Wubben and Vivian 7 reported great improvement when a pharmacist provides direct care to patients with diabetes. More recently, our group published the RxING and the RxEACH studies, where we reported large reduction in A1C (between 0.9% and 1.8%) in a short period of time (3-6 months) with independent pharmacist prescribing interventions.8,13,14
While there is strong evidence for the impact of pharmacist care in diabetes, implementation of this evidence is lacking. Perhaps practical implementation tools are needed. As such, we designed this registry to evaluate the impact of an implementation strategy (an online practice tool) for pharmacists (based upon the RxING study 8 ) on estimated cardiovascular (CV) risk in patients with diabetes.
Methods
The RxING Practice Tool was a prospective registry and practice implementation tool that was tested in community pharmacies and primary care networks (PCNs) across Alberta (for a list of the participating pharmacists, please see the acknowledgements section).
Patients were included if they had type 1 or type 2 diabetes and had at least 1 uncontrolled risk factor (A1C >7%, 15 blood pressure ≥130/80 mmHg, 16 low-density lipoprotein [LDL] cholesterol >2 mmol/L 17 or current tobacco use). We excluded patients if they were unwilling to participate/sign consent form, unwilling or unable to participate in regular follow-up visits or pregnant.
Recruitment
Pharmacists and pharmacy staff used proactive case finding to identify potential patients. Patients with physician-diagnosed diabetes were identified by reviewing prescriptions of antihyperglycemic agents such as metformin.
As part of routine care, pharmacists checked the most recent laboratory test results for the identified patients (through the provincial electronic health record) and measured their blood pressure. Then they checked whether patients met the inclusion criteria. Those who met the inclusion criteria were considered eligible and were invited to participate in the registry. Patients who agreed to participate were asked to sign a written informed consent form, and then they were enrolled in the registry.
The patient’s physician(s) received a letter from the pharmacist to inform them that the patient had agreed to enrol in this registry.
Intervention
The RxING Practice Tool is an online guideline-driven tool that helps pharmacists implement and document care of their patients with diabetes.
All enrolled patients received:
Patient assessment (blood pressure measurement according to Hypertension Canada guidelines, 16 waist circumference, weight and height measurements)
Laboratory assessment of A1C, nonfasting lipid panel (total cholesterol, LDL cholesterol and high-density lipoprotein [HDL] cholesterol) and kidney function and status (creatinine [and estimated glomerular filtration rate] and random urine albumin to creatinine ratio)
Individualized CV risk assessment and education regarding this risk using a validated interactive online tool 13 that explains the individual’s risk, the contribution of each risk factor to the overall risk and the impact of the intervention and controlling the risk factors on the overall CV risk (https://www.epicore.ualberta.ca/epirxisk/)
Treatment recommendations, prescription adaptation and prescribing where necessary to meet treatment targets. Pharmacists practised to their full scope (including prescribing medications and ordering and interpreting laboratory tests when needed).
Regular follow-up visits at the pharmacist’s discretion to check on patients’ progress and provide ongoing care and motivation
Regular communication with the patient’s physician(s) after each contact with the patient as per usual pharmacist practice
Outcomes
The primary outcome was the change in estimated CV risk between baseline and the patient’s final visit. CV risk is defined as the risk for future CV events (e.g., coronary heart disease [CHD], stroke, peripheral arterial disease [PAD]) as calculated by validated risk assessment equations. CV risk was calculated using the EPI·RxISK™ Cardiovascular Risk Calculator (https://www.epicore.ualberta.ca/epirxisk/), which uses the United Kingdom Prospective Diabetes Study (UKPDS) risk assessment equation 18 for patients who have diabetes without other comorbidities. If the patient had other CV risk-modifying conditions (chronic inflammatory conditions, previous vascular disease or chronic kidney disease), risk was calculated using the UKPDS risk assessment equation 18 and the most appropriate risk assessment equation based on the patient’s medical history. The Modified Framingham risk assessment equation (Framingham risk score multiplied by 1.5) 19 was used for patients who have chronic inflammatory conditions, the SMART risk assessment equation 20 was used for those with previous vascular disease and the Framingham risk assessment equation 21 was used for those with chronic kidney disease. If the patient had diabetes and other CV risk-modifying conditions, the risk was calculated using all the respective risk assessment equations, and the risk assessment equation estimating the highest risk was used.
Secondary outcomes were the change in individual risk factors (A1C, systolic blood pressure, LDL cholesterol and tobacco use [self-reported abstinence]) between baseline and the patient’s final visit.
Analytical plan
Analysis was performed by using R 3.6.2 (The R Project for Statistical Computing, Vienna, Austria) and SAS 9.4 software (SAS Institute, Cary, NC).
Data were first screened to confirm that all the participants met the inclusion/exclusion criteria and provided informed consent. Once those conditions were confirmed, statistical analysis started.
Demographic information and clinical characteristics were analyzed using descriptive statistics. Mean (standard deviation) was used for continuous variables while frequency (percentage) was used for categorical variables. Statistical significance at the univariable level was assessed using t-test or Wilcoxon rank-sum test (when data were heavily skewed) for continuous variables and chi-square test or Fisher’s exact test (when small frequencies were present) for categorical variables (assumption of statistical tests was checked before performing them). The primary outcome was analyzed using a paired t-test. Multivariable linear mixed-effect model was used to adjust for centre effect and baseline characteristics. Secondary outcomes were analyzed using chi-square test and paired t-test as appropriate.
Trial and data management was performed by EPICORE Centre (https://www.epicore.ualberta.ca/).
RxING Practice Tool was approved by the Health Research Ethics Board of the University of Alberta (Pro00066764).
Results
We trained 82 pharmacists to use the tool; of those, 64 registered to use it. The practice tool was launched in May 2017, and the last patient was enrolled in November 2019. During that period, 36 pharmacists enrolled 157 patients (mean 4.4 patients/pharmacist). Patients were followed for a mean (SD) of 8 (5) months, the median number of visits per patient was 2 (interquartile range, 2-3) and the last follow-up visit was completed in February 2020. More than three-quarters (82%) of patients received at least 1 follow-up visit.
Patients’ demographic and clinical characteristics are presented in Table 1. They had diabetes for a mean (SD) of 9.6 (8.9) years, their mean (SD) age was 59.9 (14.3) years, 58% were male, 71% were Caucasian and 97.5% had type 2 diabetes (Table 1). The most common reported comorbidity was hypertension (73.2%), followed by dyslipidemia (66.9%), atherosclerotic vascular events (27.4%) and chronic kidney disease (CKD) (21.7%) (Table 1).
Baseline demographic and clinical characteristics
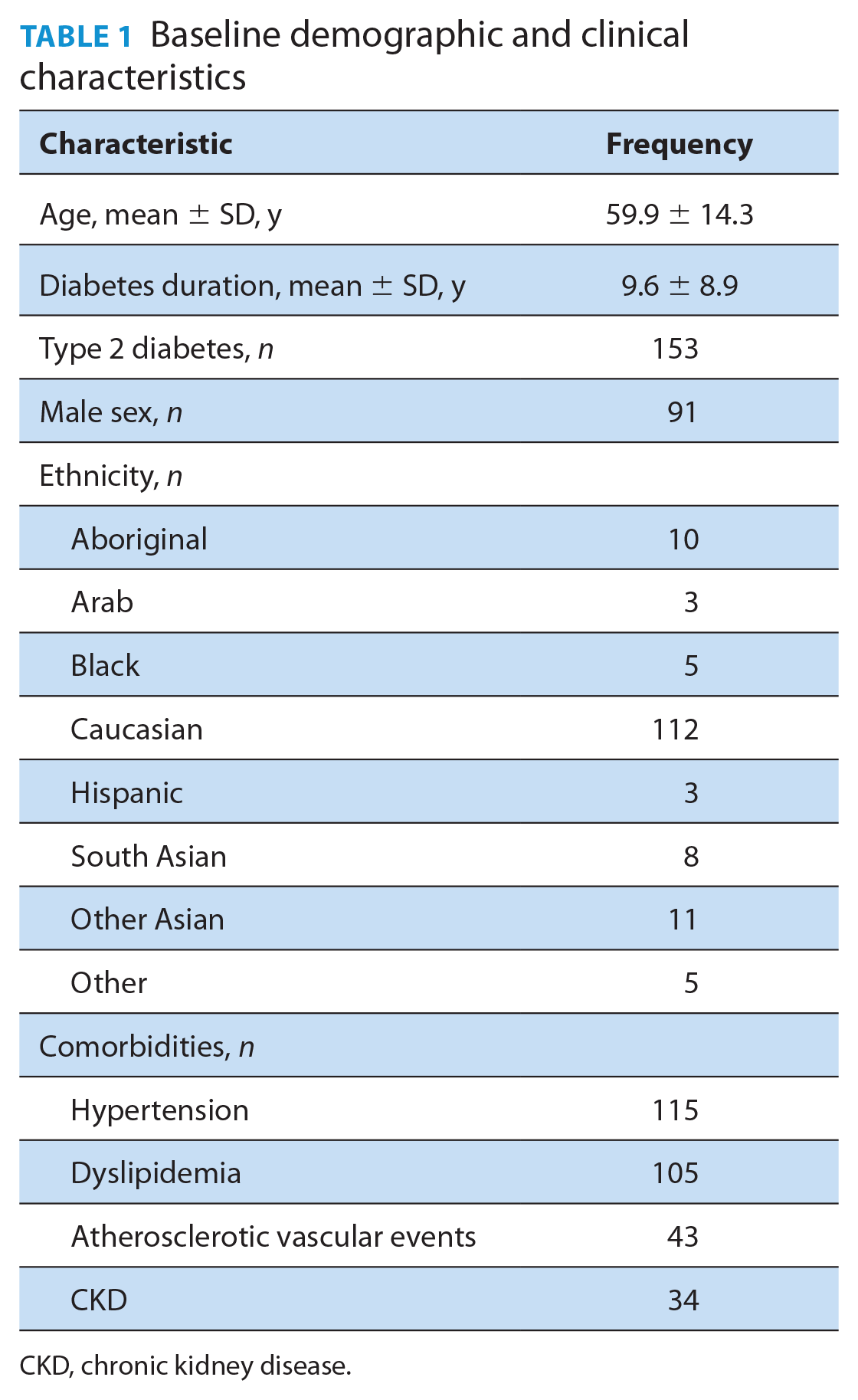
CKD, chronic kidney disease.
Estimated CV risk was reduced from 22.1% (SD 18.5%) to 18% (SD 16.9%). After adjusting for baseline characteristics and centre effect, this corresponded to 19% relative risk reduction (p = 0.045) (Figure 1). There was a direct relationship between the reduction in estimated CV risk and the number of follow-up visits. Those who had the largest number of follow-up visits had the greatest reduction in CV risk as indicated by the regression coefficient (2 visits: –1.4; 3 visits: –2.5; 4 visits: –6; 5 visits: –6.7; 6 visits: –10).
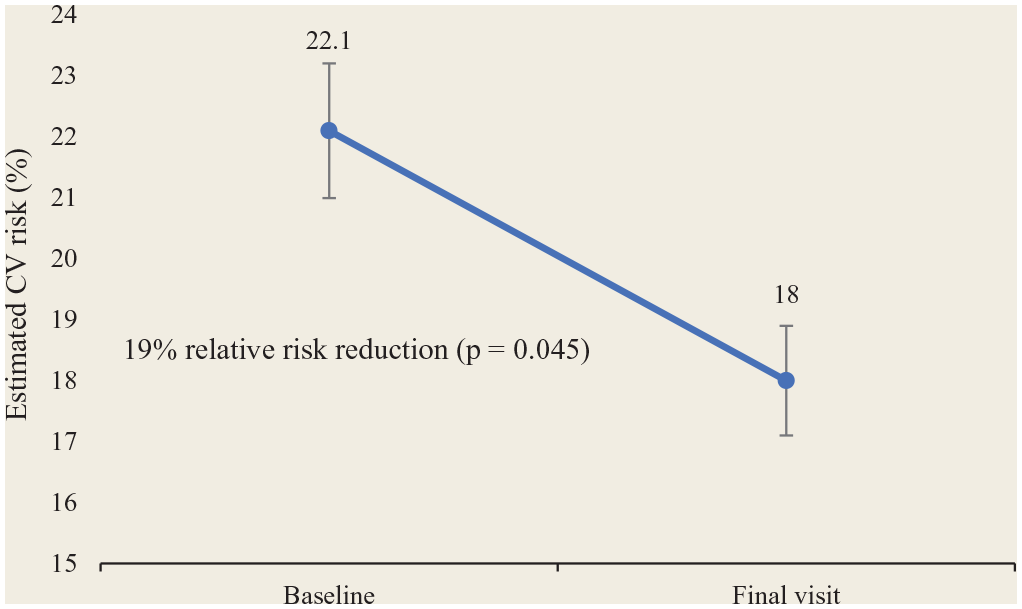
Change in estimated cardiovascular (CV) risk between baseline and the patient’s final visit
Significant reductions were observed in A1C and systolic blood pressure (Table 2). A1C was reduced from 8.6% (SD 2.1%) to 7.9% (SD 1.8%) (p = 0.006) and systolic blood pressure from 133.4 mmHg (SD 14.4) to 129.8 mmHg (SD 13.1) (p = 0.048). LDL cholesterol and tobacco use were not reduced significantly (Table 2).
Changes in individual risk factors
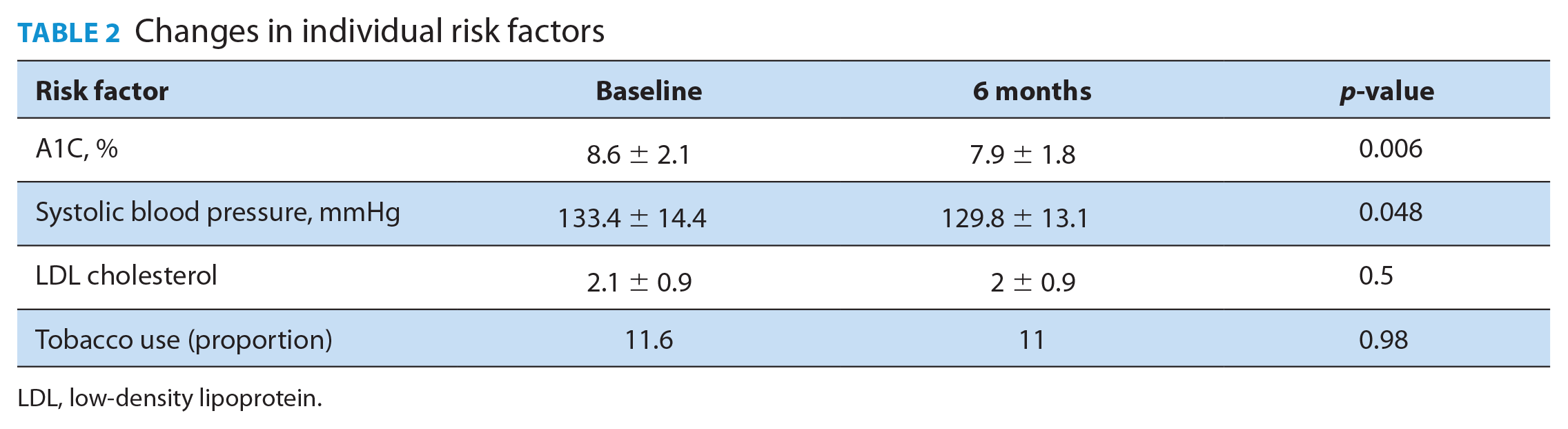
LDL, low-density lipoprotein.
The most implemented pharmacist intervention was lifestyle education and advice (44%), followed by medication/dose change (18%), lab assessment (16%), adherence assessment and improvement (13%) and referral to other health care providers (9%) (Table 3). Very minimal self-reported adverse events were noted during the study. Indeed, the number of patients who reported having hypoglycemic events at baseline was reduced significantly after receiving the pharmacist intervention.
Pharmacist interventions
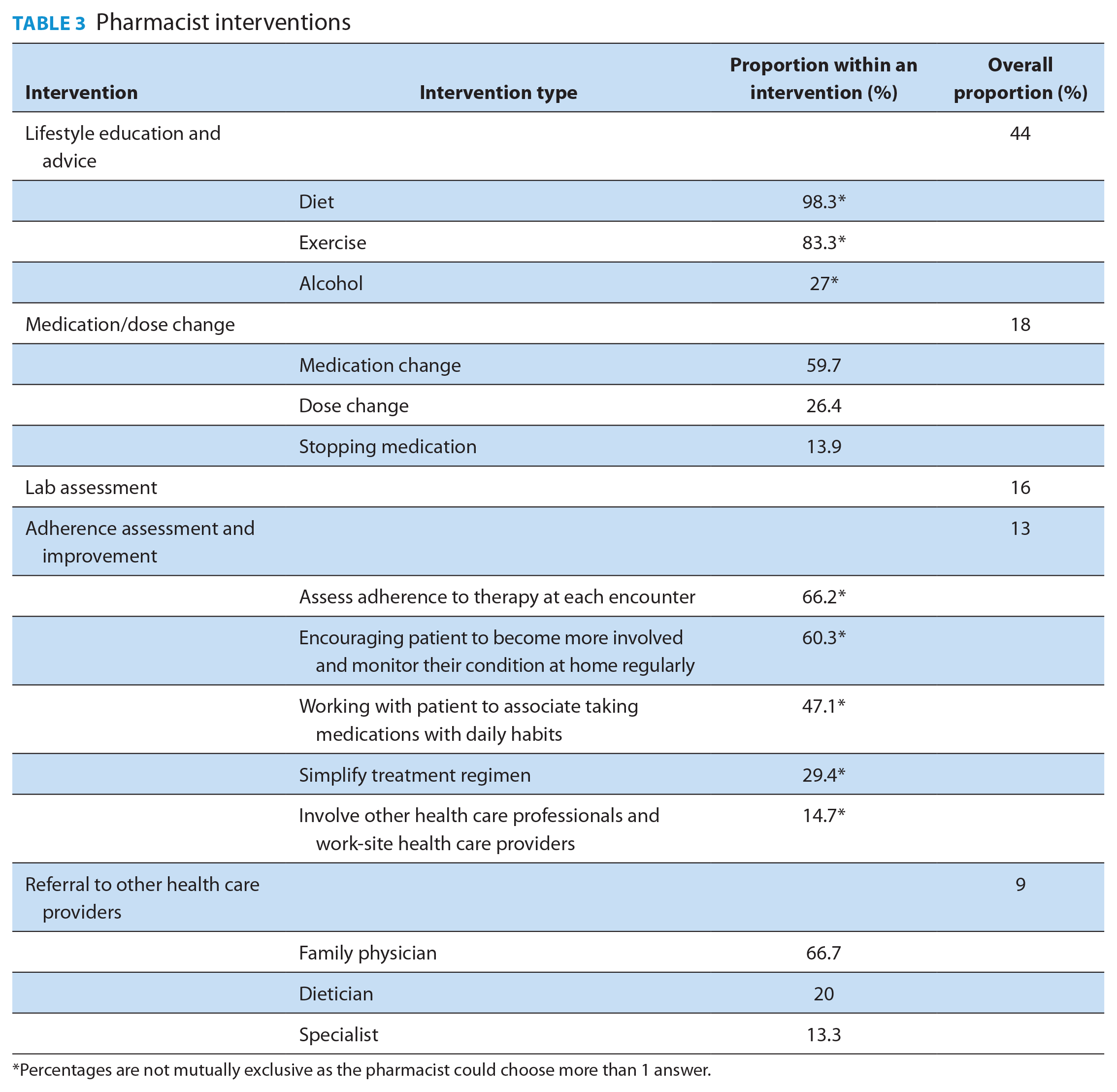
Percentages are not mutually exclusive as the pharmacist could choose more than 1 answer.
Discussion
Patients cared for by pharmacists who used the RxING Practice Tool showed a significant reduction in their estimated CV risk (19% relative risk reduction; p = 0.045). Significant reductions were also observed in A1C and systolic blood pressure. However, the uptake of our implementation strategy was poor.
Our findings are consistent with the findings of Tsuyuki and colleagues, 13 who evaluated the impact of pharmacist intervention (assessment, prescribing and follow-up) on CV risk in patients at high risk for CVD (including those with diabetes). They reported that such intervention was associated with significant reduction in estimated CV risk and significant improvements in all individual risk factors.
Medication/dose change was the second most implemented intervention, which underlines the value of pharmacist prescribing. These interventions would have not been possible if the pharmacists were not able to prescribe. This is consistent with what has been reported in the literature, where better outcomes were observed when pharmacists had prescribing authority.7,8,13,14
Notably, patients had better outcomes when they had more follow-up visits. Such observation highlights the importance of regular follow-up visits in patients with diabetes. This is consistent with what has been reported in the literature underlining the vital role of regular follow-up visits in achieving the treatment targets and maintaining the improvements in patient outcomes.22-24
It is noteworthy that patients’ outcomes improved when the pharmacists used the RxING Practice Tool. However, only 44% of trained pharmacists actually used the tool and enrolled patients. This, combined with the need for multiple extra prompts from the research team to conduct any follow-up visits, despite the system-generated reminders suggests that our implementation strategy was lacking in certain aspects. Further investigation to characterize those aspects is currently underway.
This implementation study is not without limitations. The fact that fewer than half of the trained pharmacists used the tool could affect the generalizability of the results. However, despite that, the findings were consistent with what has been reported in the literature. The study team monitored the study sites against source documents to prevent any bias that could have been introduced from the fact that the pharmacists who delivered the interventions were the same ones who conducted the assessment and entered the data into the study system.
Our findings add to the body of evidence that supports pharmacist interventions in patients with diabetes.7-14 The interventions, when implemented, were not only effective but also safe, as the number of patients who reported hypoglycemic events was reduced with the pharmacist interventions, and very minimal self-reported adverse events were observed during the implementation period.
Conclusion
To our knowledge, this is the first study to evaluate the impact of an implementation strategy for pharmacists on estimated CV risk in patients with diabetes. The patients’ access to care and outcomes improved when pharmacists used the RxING Practice Tool; however, less than half of the trained pharmacists used the tool in their practice, suggesting that our implementation strategy was lacking in certain aspects. Further investigation to characterize those aspects is under way.
Footnotes
Acknowledgements
We would like to acknowledge the support of the Consultation and Research Services Platform at The Alberta SPOR SUPPORT Unit in Data management and statistical services. Also, none of this could have taken place without the dedication and care of the RxING Practice Tool investigators, listed in descending order of recruitment: Derek Durocher (Shoppers Drug Mart #313, Edmonton); Rick Siemens (London Drugs #38, Lethbridge); Maricar Verdida (London Drugs, Airdrie); Adrian Azim (Shoppers Drug Mart #355, Calgary); Carlene Oleksyn and Jelena Okuka (Meridian Pharmacy, Stony Plain); Esmond Wong (Calgary Foot Hills Primary Care Network, Calgary); Jim Kitagawa (Pharmasave #345, Brooks); Taria Gouw (Apple Drugs, Bow Island); Robert Clermont (Skelton’s Pharmacy, Lethbridge); Gehan Rizkalla (Loblaws Pharmacy #4950, Leduc); Roberta Taylor (Roots & Berries Pharmacy, Maskwacis); Rob Hozack (Redcliff Pharmasave, Medicine Hat); Nadine Winsor (Safeway Pharmacy, Calgary); Nader Hammoud (Shoppers Drug Mart #2326, Calgary); Erin Foster (HealthCheck Pharmacy, Leduc); Melissa Mauriello (Mint Health + Drugs, Canmore); Anita Dobson (Medicine Shoppe #212, Calgary); Nermen Kassam (Pharmacy Plus Ltd, Calgary); Lonni Johnson (Rita’s Apothecary, Barrhead); Leslie Leontowich (Melrose Drugs, Aidrie); Nick Leong (Shoppers Drug Mart #328, Edmonton); George Leung (Costco Pharmacy 1069, Okotoks); Jodi Mills (Shoppers Drug Mart #2440, Edmonton); Brad Couldwell and Sam Turner (Shoppers Drug Mart #321, Calgary); Mike, Pat and Vanda Kinshella (Value Drug Mart, Peace River); Ignatius Anane-Ansah (Wood Buffalo Primary Care Network, Fort McMurray); Todd Pranchau (Shoppers Drug Mart #2450, Sylvan Lake); Randy Howden (Medicine Shoppe, Calgary); Janice Chua (Landmark Pharmacy, Calgary); Ankita Karla (Pharmasave #393, Calgary); Trish Molberg (Sandstone IDA Pharmacy, Calgary); Gordon Morck (Strathmore Value Drug Mart, Strathmore); Rita Lyster (Pidsadowski’s IDA Pharmacy, Westlock); Anita and Reid McDonald (Sunset Ridge Pharmacy, Cochrane); Leanna St. Onge, Otti Gohrbandt and Chelsey Collinge (Co-op Pharmacy, Rocky Mountain House); Darsey Milford (Turtle Mountain Pharmacy, Bellvue).
Funding:
We would like to acknowledge the funders of the RxING Practice Tool: Merck Canada. Our funders did not have any role in the study design, collection, analysis, interpretation of the data, writing the report and the decision to submit for publication.
Financial Disclosure:
Drs. Al Hamarneh, Tsuyuki and Okada declare they have no relevant financial interests.
