Abstract
Objective
Methods
We collected clinical data on 227 patients with end-stage renal disease who were first diagnosed at our institution between February 1, 2023 and February 1, 2024, and were followed up regularly for 12 weeks post-AVF creation, with the diameter of the draining vein and the brachial artery volume flow (VF) recorded at 1 day, 2, 4, 6, 8, 10, and 12 weeks post-operation. We focused on the time of AVF maturation, and ultrasonographic diagnostic criteria 1 (the vein diameter was ≥4 mm, and the brachial artery VF was >500 mL/min, referred to as the “Rule of 4”) and ultrasonographic diagnostic criteria 2 (the vein diameter was ≥5 mm, and the brachial artery VF was> 500 mL/min, referred to as the “Rule of 5”) were used to evaluate the maturation of AVF. And compared with clinically maturation criteria to understand the correlation, consistency, and diagnostic efficacy.
Results
1. The venous diameter and brachial artery VF of AVF showed an upward trend, and increased significantly in 1 day to 6 weeks postoperatively (p < .05),especially between 1 day and 2 weeks, while no significant difference in the increases at 6-12 weeks; 2. Logistic regression analysis showed that venous diameter (OR = 19.589 , 95% CI 1.560-245.979, p = .021) and brachial artery VF (OR = 1.024 , 95% CI 1.005-1.042, p = .011) at 6 weeks were the independent correlates affecting AVF maturation; and the optimal thresholds were 4.08 mm (sensitivity 88.10%, specificity 70.00%) and 472.50 mL/min (sensitivity 93.20%, specificity 84.00%), respectively; 3. Compared with clinical maturation, the two ultrasonographic diagnostic criteria at 6 weeks postoperatively were correlated with clinical maturation, and the specificity of both was 100%, with “Rule of 4” having a sensitivity of 79.66%, an accuracy of 84.14%, and a concordance kappa = 0.633, while “Rule of 5″ had a sensitivity of 31.63%, an accuracy of 46.69%, and a concordance kappa = 0.169; and the area under the ROC curve (AUC) for “Rule of 4″ was higher than that for “Rule of 5” (0.898 vs. 0.658, p < .001).
Conclusion
Six weeks postoperatively may be the best timing for an ultrasound assessment of AVF maturation, and the “Rule of 4” ultrasound assessment criteria was more suitable for clinical judgment of AVF maturation.
Arteriovenous fistula (AVF) was the vascular access of choice for renal replacement therapy in patients with end-stage renal disease (ESRD). However, the rate of postoperative AVF immaturity was as high as 30%-60%.1–4 Color Doppler ultrasound was noninvasive and economical, and was the imaging method of choice for assessing and monitoring AVF function both pre- and post-surgery. The timing and criteria for ultrasound assessment and diagnosis of AVF maturation were still controversial, which may lead to over-intervention or untimely intervention in the AVF.5–7 In this study, we retrospectively analyzed the natural development of AVF in 227 ESRD patients post-surgery to find out the reasonable timing of assessing AVF maturation, and used two ultrasound diagnostic criteria to assess AVF maturation, and analyzed the correlation and consistency with clinical maturation.
Methods
This was a retrospective study of patients with ESRD who had their first AVF established at our Hospital from February 1, 2023, to February 1, 2024, and the study was approved by the hospital ethics committee (approval No. 2024ER15-1).
Inclusion criteria: (1) patients age >18 years; (2) preoperative ultrasound assessment of radial artery diameter >1.5 mm and diameter of cephalic vein behind the bundle arm >2.0 mm; (3) complete documentation of data during follow-up.
Exclusion criteria: (1) non-first-time establishment of AVF in the operated limb; (2) patients who were lost during follow-up.
Data collection
(1) General information: age, sex, body mass index (BMI), preoperative systolic and diastolic blood pressure, smoking history, primary disease, presence of comorbid diabetes mellitus, hypertension, and type of temporary vascular access. (2) Preoperative laboratory data: red blood cells (RBC), hemoglobin (Hb), platelets (plt), albumin (Alb), calcium (Ca), phosphorus (P), whole parathyroid hormone (iPTH), plasma plasminogen time (PT), activated partial thromboplastin time (APTT), triglycerides (TG), cholesterol.
AVF postoperative follow-up
Drainage vein diameter, brachial artery volume flow(VF) were recorded at 1 day, 2 weeks, 4 weeks, 6 weeks, 8 weeks, 10 weeks, and 12 weeks after completion of AVF, and time of first puncture of AVF and dialysis blood flow were collected.
Equipment and operation
All the included study subjects underwent the end-to-side anastomosis with cephalic vein-radial artery at the wrist by the same surgeon to obtain the radial-cephalic AVF, and the anesthesia was local anesthesia with lidocaine (1%).The vessels were evaluated and examined by the same sonographer using a Myeri DC-80 color Doppler ultrasound machine (line array probe at 14 Hz). artery diameter was measured 1 cm from the anastomosis, vein diameter was measured 5 cm from the anastomosis after 1 minute of arm-banding, and brachial artery VF was measured 3 cm above the elbow.
Endpoint definitions
(1) Clinical maturation of AVF: During 30 days, at least 6 successful punctures of the AVF for hemodialysis with a flow of >250 mL/min.8,10 This definition serves as a reference standard for comparison with ultrasound assessment of maturity. (2) Ultrasonographic diagnostic criteria 1: vein diameter ≥4 mm and brachial artery VF >500 mL/min, at 6 weeks postoperatively.
9
Ultrasonographic diagnostic criteria 2: vein diameter ≥5 mm and brachial artery VF >500 mL/min, at 6 weeks postoperatively. 10
Statistical analysis
Statistical analysis was performed using SPSS 25.0 software. Measurement information that conformed to normal distribution was expressed as mean ± standard deviation using the independent samples t-test, and those that did not conform to normal distribution were tested using the rank sum test (Mann-Whitney U test). Count data were expressed as frequency and % and were tested using chi-square. Logistic regression was used to analyze the relationship between each clinical data and AVF maturation; independence test and consistency test were used between ultrasound maturation criteria and clinical maturation definitions of AVF, and diagnostic tests were performed, and the difference between ultrasound criterion 1 and criterion 2 was further compared by using subjects’ work characteristic curves (ROC) to analyze the diagnostic efficacy of the diagnosis of AVF maturation. p < .05 was taken as a difference statistically significant.
Results
From February 1, 2023, to February 1, 2024, a total of 318 patients diagnosed with ESRD underwent their initial AVF creation. Out of these, 280 met the study’s inclusion criteria, , while 32 patients had incomplete postoperative follow-up data, and 21 patients were lost to follow-up. Consequently, 227 patients were finally included as study subjects. The screening process of the study population is shown in Figure 1. Flowchart for screening research subjects.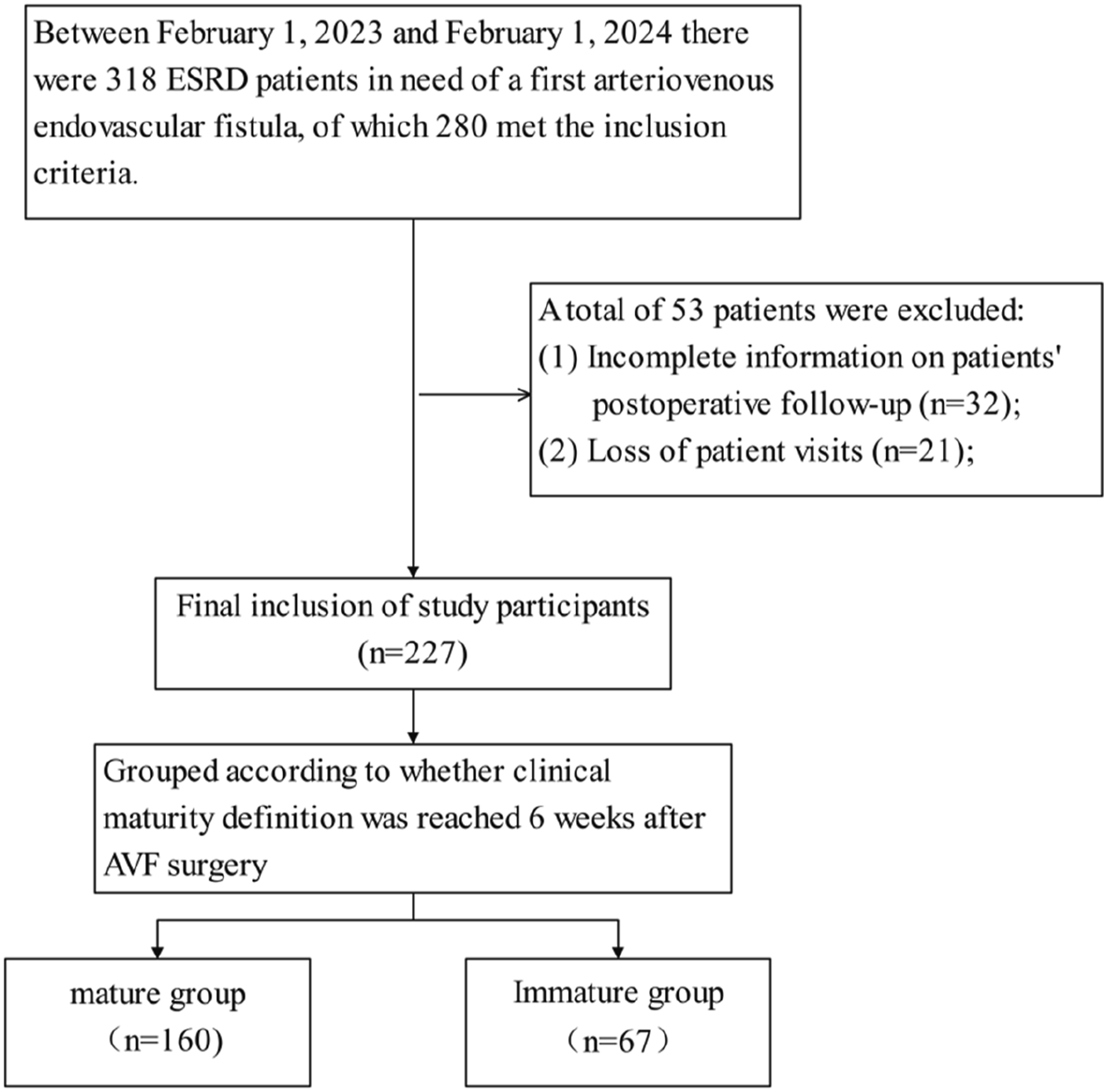
Baseline characteristics
The study population had an average age of 50.53 ± 13.86 years, with 89 (39.20%) males and 138 (60.79%) females. History of hypertension was 85 cases (37.44%), history of diabetes mellitus was 69 cases (30.39%), primary chronic glomerulonephritis was 104 cases (45.81%), and first hemodialysis access was central venous temporary access in 106 patients (46.69%), in which right femoral venous temporary access in 97 cases (42.73%), and right internal jugular venous temporary access in 9 cases (3.96%).
Analysis of the rise in venous diameter and brachial artery VFat various postoperative periods
The increase in range was statistically significant during the periods of 1 day to 2 weeks, 2 to 4 weeks, and 4 to 6 weeks postoperatively(p < .05), especially between 1 day and 2 weeks. while there was no significant difference in the range of increase at the stages of 6 to 8 weeks, 8 to 10 weeks, and 10 to 12 weeks( p > .05) (Figure 2(a), (b)). Since there was no significant increase in AVF draining vein diameter and brachial artery VF after 6 weeks, it appears that 6 weeks postoperatively may be the optimal time to assess maturation. Trends in the diameter of the draining vein and the rise in brachial artery blood flow at different time intervals postoperatively. (a) Changes in the diameter of the draining vein: V1-V7 indicate the increase in the diameter of the vein from the diameter of the posterior cephalic vein of the fasciculus arm before surgery to 1 day after surgery, from 1 day to 2 weeks after surgery, from 2 weeks to 4 weeks, from 4 weeks to 6 weeks, from 6 weeks to 8 weeks, from 8 weeks to 10 weeks, and from 10 weeks to 12 weeks, respectively; and (b) Changes in the increase in the brachial artery blood flow: Q1-Q6 indicate the increase in the blood flow of the brachial artery on the 1st day after surgery, from 2 weeks to 4 weeks, from 4 weeks to 6 weeks, from 6 weeks to 8 weeks, from 8 weeks to 10 weeks, and from 10 weeks to 12 weeks, respectively. weeks to 4 weeks, 4 weeks to 6 weeks, 6 weeks to 8 weeks, 8 weeks to 10 weeks, and 10 weeks to 12 weeks, respectively. *:p < .05. **:p < .01. ***: p < .001. ****: p< .0001. ns: p > .05.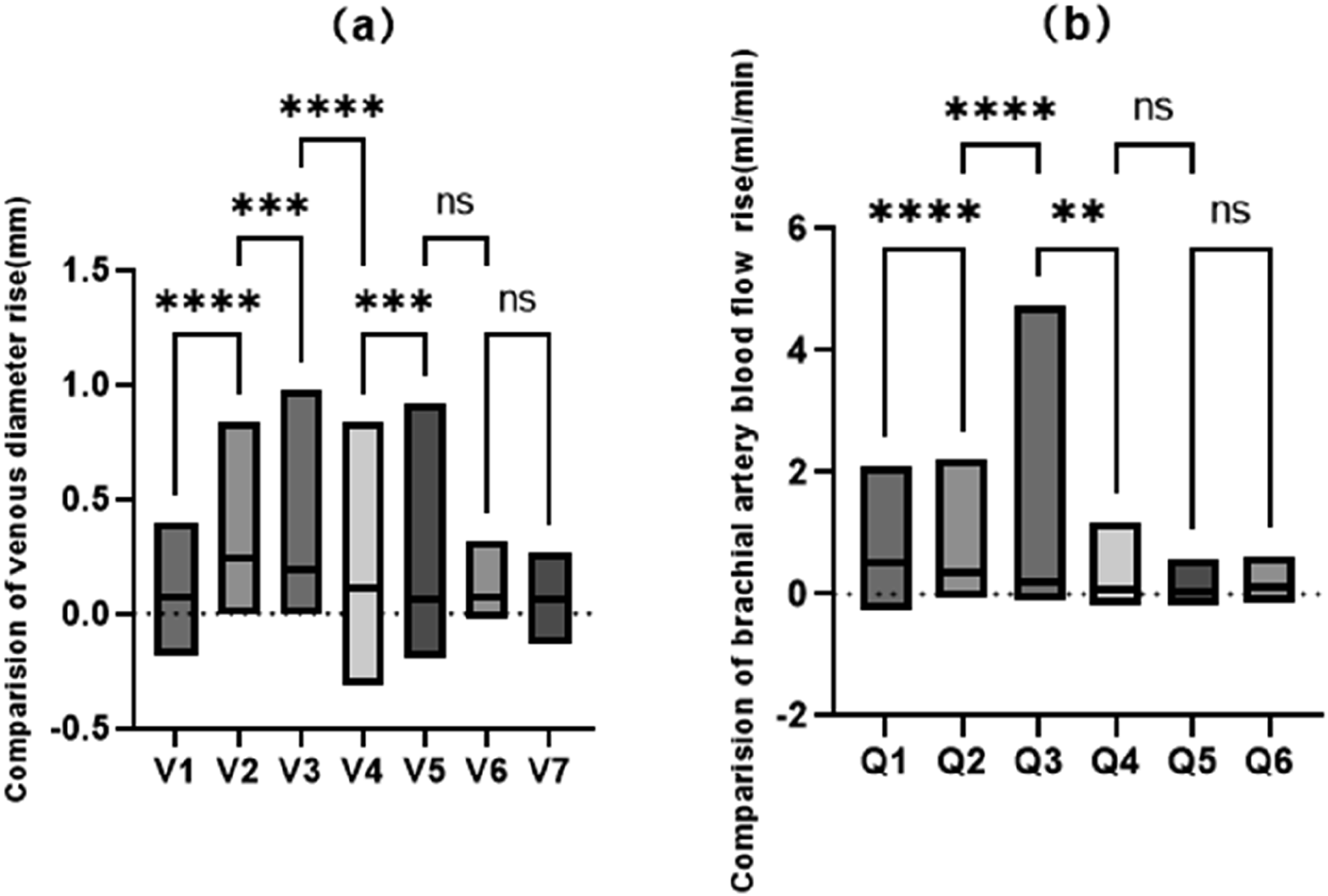
Analysis of factors influencing the clinical maturation of AVF
Comparison of parameters between patients in the clinically mature and non-mature groups.
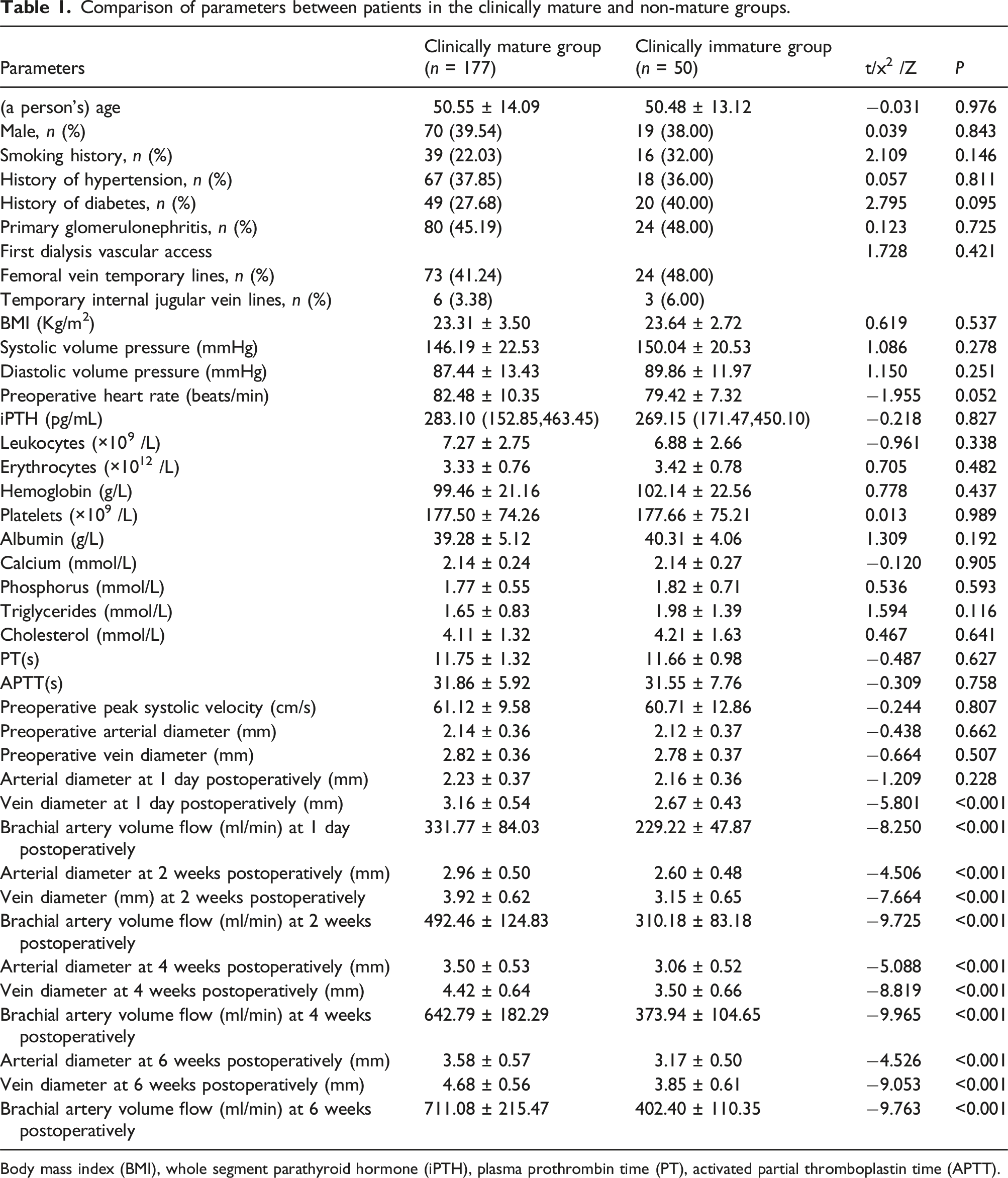
Body mass index (BMI), whole segment parathyroid hormone (iPTH), plasma prothrombin time (PT), activated partial thromboplastin time (APTT).
Logistic regression analysis of clinically mature and non-mature groups.
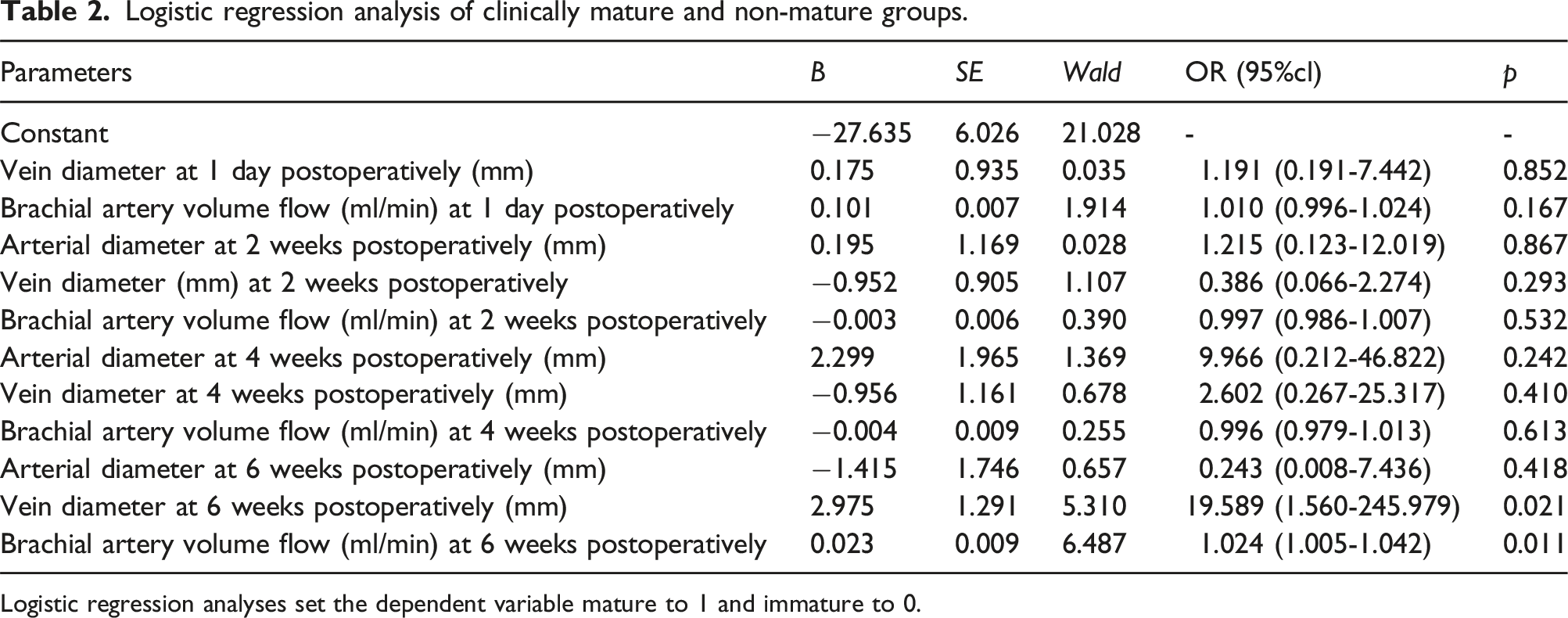
Logistic regression analyses set the dependent variable mature to 1 and immature to 0.
The area under the Receiver Operating Characteristic(ROC) curve (AUC) of the vein diameter and brachial artery VF at 6 weeks postoperatively were 0.853 and 0.932 (95% confidence intervals of 0.795∼0.911 and 0.891∼0.972, respectively; p < .001), and the optimal thresholds for the two were 4.08 mm (sensitivity 88.10%, specificity 70.00%) and 472.50 mL/min (sensitivity 93.20%, specificity 84.00%) (Figure 3). ROC curves of draining vein diameter and brachial artery blood flow at 6 weeks postoperatively.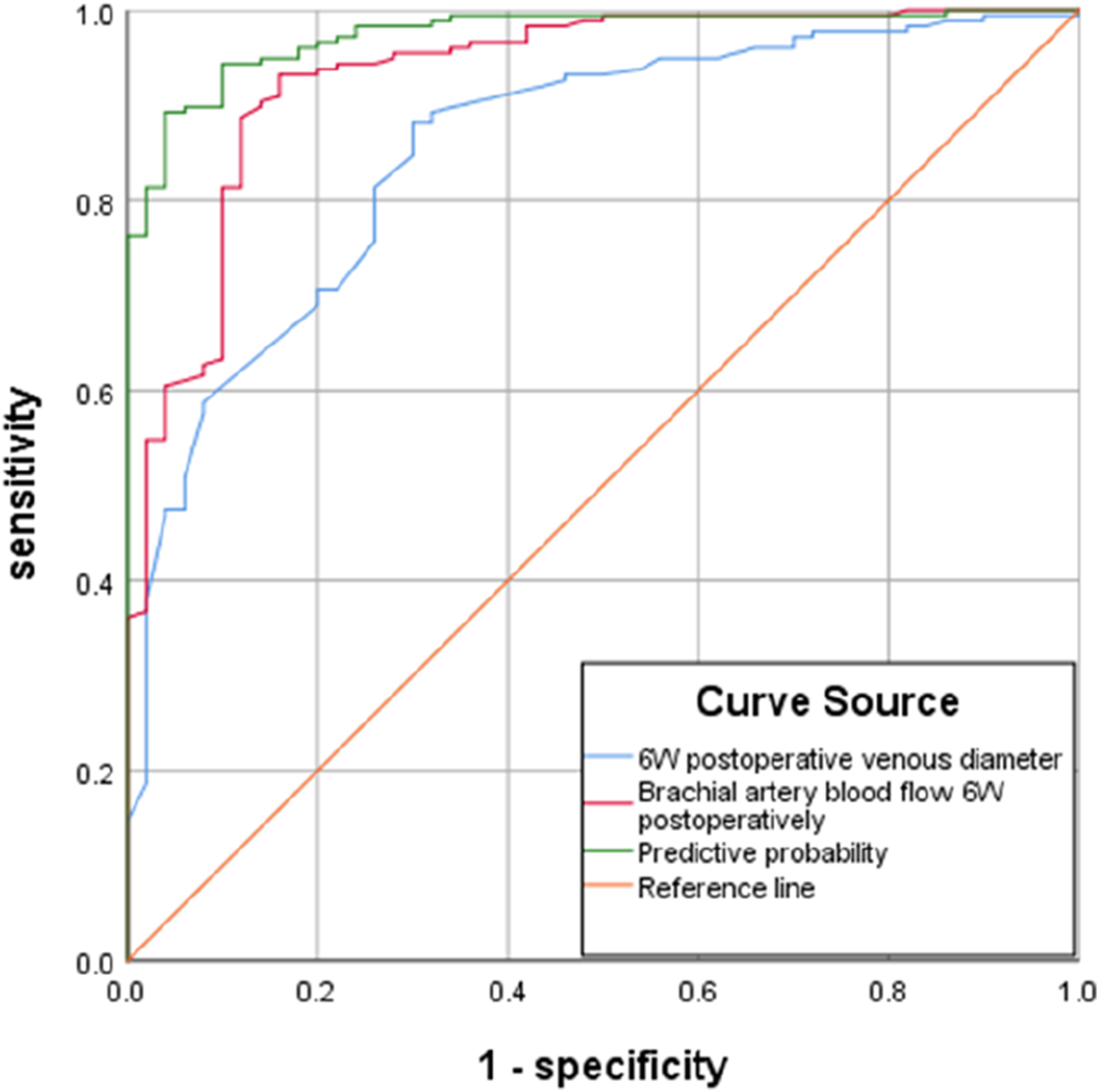
Diagnostic tests for two ultrasound criteria
At 6 weeks postoperatively, There were 141 cases that met the criteria for determining maturity by ultrasonography 1 (62.11%, 141/227).Ultrasound criterion 1 diagnosed AVF maturation with a sensitivity of 79.66%, specificity of 100%, accuracy of 84.14%, Yoden’s index of 79.66%, positive predictive value of 100%, and negative predictive value of 58.13%, and in comparison with clinical maturation, the test of independence, χ2 = 105.130, p < .001, and rp = .707, showed that there was a correlation between ultrasound criterion and clinical maturation, and it was a moderate correlation. The consistency test kappa = 0.633 (p < .001), indicating good agreement between ultrasound criteria 1 and clinical maturity.
At six weeks, 56 cases met ultrasonographic criterion 2 (24.67%, 56/227) for determining maturity. Ultrasound criterion 2 diagnosed AVF maturation with a sensitivity of 31.63%, a specificity of 100%, an accuracy of 46.69%, a Yoden index of 31.63%, a positive predictive value of 100%, and a negative predictive value of 29.23%. In comparison with clinical maturity, the test of independence χ2 = 21.000, p < .001, rp = .408, the ultrasound criteria were correlated with clinical maturity, but in a low degree. The test of consistency kappa = 0.169 (p < .001) indicated that there was a slight consistency between ultrasound criteria 2 and clinical maturation.
ROC curves of the diagnostic efficacy of two ultrasound criteria
The AUC of ultrasound criterion 1 was 0.898 (95% confidence interval 0.859∼0.937, p < .001); the AUC of ultrasound criterion 2 was 0.658 (95% 95% CI 0.584∼0.732, p = .001), so the diagnostic efficacy of ultrasound criterion 1 was superior to that of ultrasound criterion 2 (Z = 12.752, p < .001) (Figure 4). ROC curves of the diagnostic efficacy of the two ultrasound criteria.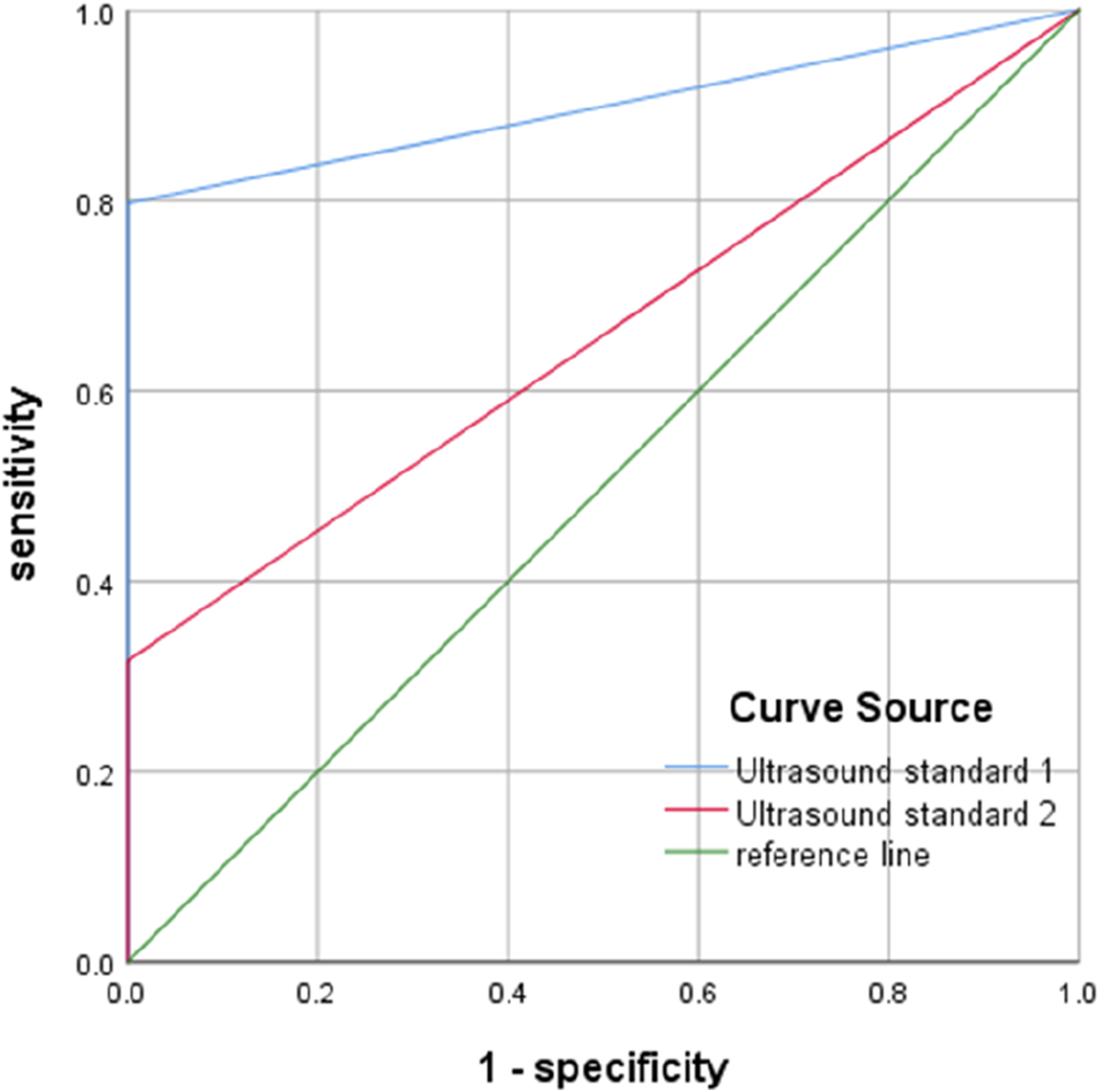
Discussion
According to worldwide hemodialysis vascular access guidelines or expert consensus, ultrasound measurements of vein diameter and brachial artery VF were both key indicators for assessing AVF maturation, however, the timing and criteria to diagnose AVF maturation were not consistent.10–14 In Japan, the timing of AVF puncture was earlier, and its guidelines recommend that AVF be evaluated at 4 weeks postoperatively. The KDOQI guidelines for vascular access recommend 4-6 weeks postoperatively. 11 and the Chinese expert consensus recommends 6 weeks postoperatively. 10 Zhao et al 15 retrospectively analyzed 568 patients for AVF maturation at 6 months postoperatively and found that vein ≥3.9 mm and brachial artery VF ≥446.9 mL/min at 4 weeks postoperatively predicted natural maturation of the AVF, and therefore suggested that 4 weeks postoperatively may be the best time to assess AVF maturation as well as the time to assist in the treatment of AVF maturation. The study was only followed up once a month although it was until 6 months postoperatively, so it was not clear about the changes in AVF during shorter intervals after AVF surgery. In the present study, 227 patients were followed up once every 2 weeks and up to 12 weeks after surgery. The results showed that both the of vein diameter and the brachial artery VF showed an upward trend after AVF surgery, The range of increased were statistically significant in 1 day-2 weeks, 2-4 weeks, and 4-6 weeks, 6-8 weeks postoperatively (P < .05), indicating that AVF development was still evident at 4-6 weeks, and therefore premature or excessive intervention may occur if 4 weeks postoperatively was used as the time to assess AVF maturation. Meanwhile, the results of the present study showed that there was also no significant difference in the rate of increase in the three stages of 6-8 weeks, 8-10 weeks, and 10-12 weeks postoperatively, indicating that the AVF development was significantly slow after 6 weeks and that delaying the timing of assessment could not lead to a meaningful developmental space but instead might result in the failure to assist the AVF maturation in time to delay the patient’s hemodialysis treatment. Based on the above analysis, this study suggests that 6 weeks postoperatively can be the best time to assess AVF maturation, which is consistent with the assessment time mentioned in our access consensus.
In 2006, the KDOQI recommended the use of the criteria of vein diameter ≥6 mm and brachial artery VF >600 mL/min (“Rule of 6”) to diagnose AVF maturation, and this criterion had been widely adopted in clinical practice previously but has been questioned by many scholars in recent years.16–19 Retrospectively analyzed the AVF data of 202 patients and found that the mean values of venous diameter and brachial artery flow in only 14% of clinically mature AVFs met the “Rule of 6” criteria, so they suggested that this criterion was too strict for diagnosing AVF maturation. 20 The Chinese vascular access expert consensus proposed that the diameter of the vein diameter ≥5 mm and the brachial artery VF >500 mL/min (“Rule of 5”) was the criteria for diagnosing AVF maturation. 10 One study analyzed and compared the diagnostic criteria of “Rule of 6” and “Rule of 5,” and found that both of them can better predict the first successful clinical puncture, with diagnostic efficacy of 0.75 and 0.74, respectively. 21 In the present study, we found that the vein diameter and the brachial artery VF at six weeks after surgery were independent correlates of AVF maturation, and the optimal thresholds for both were 4.08 mm and 472.50 mL/min, which were comparable to the results of the study conducted by zhao et al, 15 but the mean value did not reach the “Rule of 5,” and 68.37% of patients with clinically mature AVF did not meet this criterion either, which showed that the “rule of 5” has also been challenged in clinical application.
Ferring et al 22 proposed the diagnostic criteria of vein diameter ≥4 mm and brachial artery VF >500 mL/min (“Rule of 4”), which was reported to have higher sensitivity (89% vs. 68%) but lower specificity (42% vs. 70%) for diagnosis of AVF maturation compared to the former “Rule of 6.” The study did not evaluate the diagnostic efficacy of the two diagnostic criteria, while the authors still suggested that the “Rule of 4” could minimize the need for unnecessary interventions in the early stages of AVF. Of course, it might also delay the necessary interventions. The Chinese scholar zhu et al 23 further compared the two diagnostic criteria of “Rule of 4” and “Rule of 5,” and found that both of them showed a low correlation with clinical maturation, but the sensitivity (67.34% vs. 42.86%) and diagnostic efficacy (AUC 0.837 vs. 0.668) of the former were better than those of the latter. Therefore, Zhu et al 23 suggested that the “Rule of 4” diagnostic criteria might be more suitable for clinical judgment of AVF maturation in Chinese patients, but none of the reports that analyzed the performance of the diagnostic criteria above adequately elucidated the timing of diagnosis or evaluation. In our study, based on the trend of AVF growth postoperatively, 6 weeks should be the best timing of assessment AVF maturation, and we found that the specificity of “Rule of 4” and “Rule of 5” ultrasound diagnostic criteria both was 100%, while the “rule of 4” had a higher correlation with clinical maturation, and the sensitivity (79.66% vs. 31.63%) and accuracy (84.14% vs. 46.69%) were higher, and the diagnostic efficacy was higher, as well as superior diagnostic efficacy (AUC 0.898 vs 0.658).
Conclusions
The six-week postoperatively was the dominant stage of natural development of AVF, Consequently, six weeks postoperatively may be the best timing for an ultrasound assessment of AVF maturation. And the “Rule of 4” ultrasound assessment criteria was more suitable for clinical judgment of AVF maturation. In addition, the study also presented several limitations. Firstly, being retrospective and single-center in nature, it may introduce selection or information bias and limit the generalizability of the findings. Secondly, while a 12-week follow-up duration is adequate for assessing AVF maturation, it may not fully capture the long-term efficacy of the diagnostic criteria. Therefore, in the future, we aim to conduct multi-center prospective studies with extended follow-up periods and larger sample sizes to improve validity and reliability.
Footnotes
Acknowledgments
We gratefully acknowledge all the participants and investigators for their contribution to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
