Abstract
Objectives
Non-pneumatic compression devices (NPCDs) rely on shape-memory alloy technology that allows patients to ambulate and remain active during lymphedema treatment. This study examines the effect of the NPCD on foot and ankle swelling.
Methods
This was a prospective, non-randomized study of patients with phlebolymphedema (venous insufficiency-related lymphedema) treated with a novel NPCD for 4 weeks. Ankle and foot circumference (cm), and volume measurements (ml water displacement using immersion) of the foot and ankle were obtained at enrollment, 2-weeks, and 4-weeks, and ambulation was tracked with a wearable exercise monitoring device.
Results
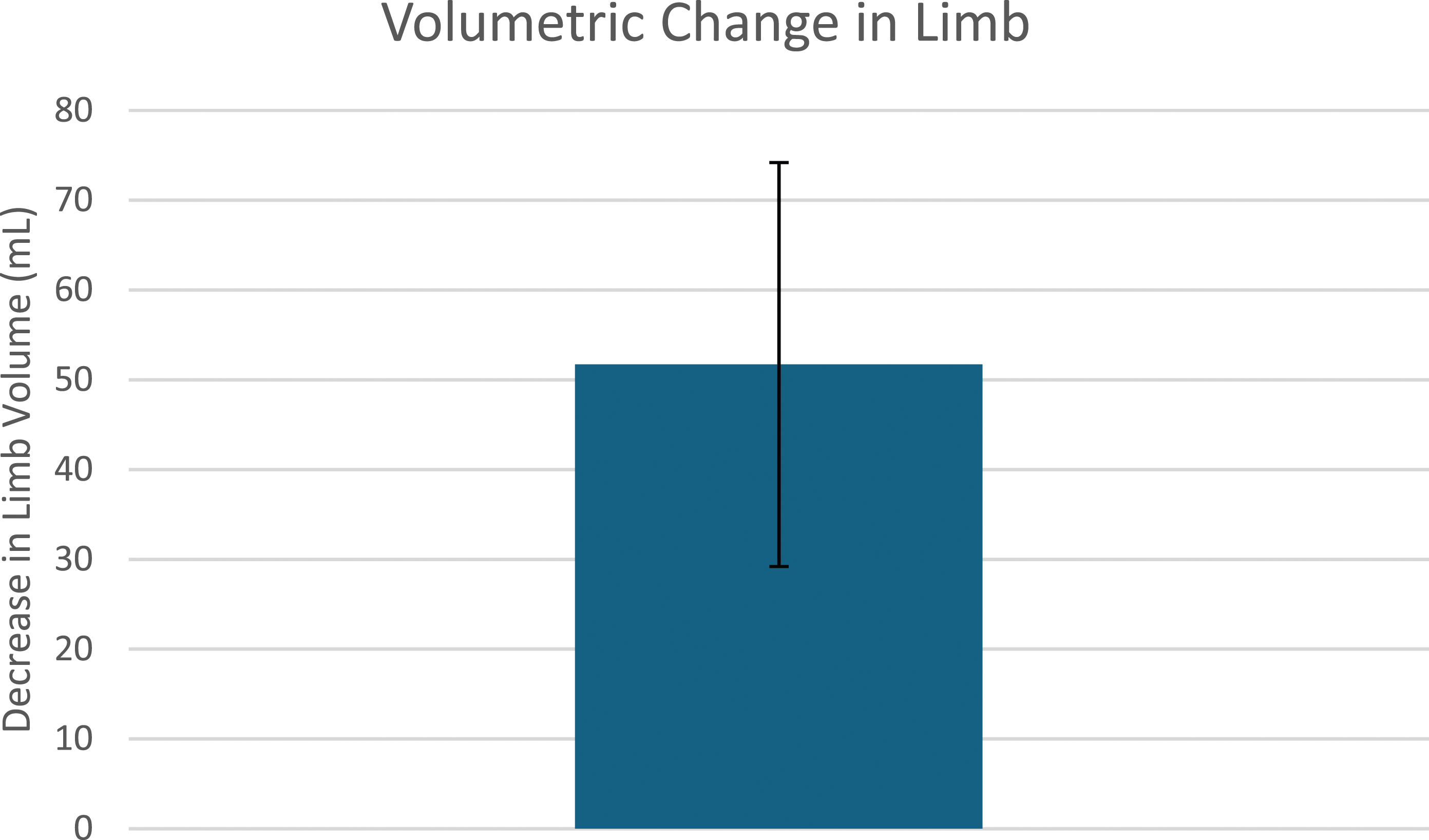
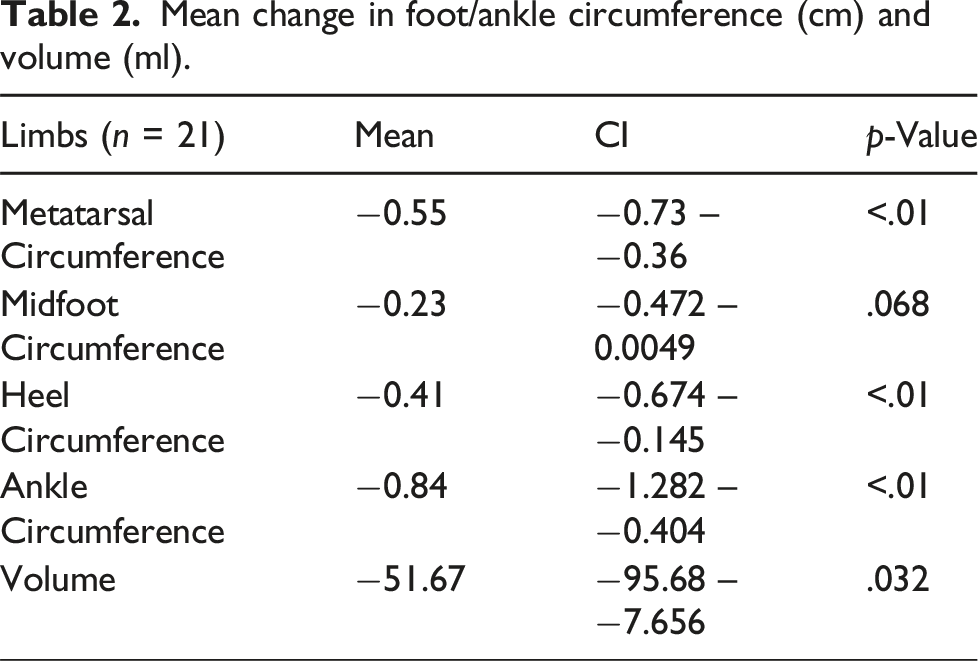
This study included 13 patients (21 limbs). A significant reduction in circumference (ankle (−0.84 ± 0.22 cm, p < .01) and metatarsal bed (−0.55 ± 0.95, p < .01)) and volume (foot/ankle (−52 ± 22 mL, p = .032)) was observed. Patients reported a mean of 8100 steps per day (±6100).
Conclusions
NPCD results in significant reduction in edema in the foot and ankle despite garments lacking a foot piece. Patients remained mobile while adhering to therapy.
Keywords
Introduction
Lifestyle-limiting edema due to chronic venous insufficiency and lymphedema has a prevalence of 28% of the US patient population. 1 Chronic venous disease associated lymphedema, or phlebolymphedema, is associated with a decrease in quality of life, impaired mobility, and a higher incidence of lower-extremity wounds and associated morbidity.2,3 Standard therapy for the treatment of edema includes both compression stockings and basic pneumatic compression therapy.4,5 Advanced Pneumatic Compression Devices (APCDs) deliver compression via air pumped into separate compression chambers which inflate sequentially peripherally to centrally, facilitating movement of fluid centrally. APCDs have been shown to be effective in treatment of lower-extremity edema, but their use is limited by the need for external power and consequent restriction of patient mobility. Consequently, this limits the effect of the calf-muscle pump function in clearing fluid from the interstitium as it remains inactive during APCD therapy in a mandatory recumbent position. 6 Additionally, APCDs do not have a static compression component and rely on sequential compression alone to achieve results.
Dayspring® (Koya Medical) is an FDA-cleared prescription-only, non-pneumatic compression device (NPCD) (Figure 1). It covers the full leg from ankle to hip crease and is also available in a half-leg from ankle to tibial tuberosity. The Dayspring garment does not rely on a pump for air insufflation of sequential chambers as seen in the APCD; rather it contains shape memory alloy-based actuators made with Nickel/Titanium (Nitinol) programmed by a portable and rechargeable controller to apply rhythmic, sequential gradient compression from the distal to the proximal end of the limb. The NPCD itself is powered by a phone-sized battery which can be carried by a strap worn around the patient’s shoulder or neck. Hence, patients using the NCPD Dayspring device are untethered to an external power source and are able to remain active and mobile during compression treatment, most importantly being able to walk while receiving compression therapy. Koya Dayspring Wearable NPCD device. Sourced from Rockson, S.G., Karaca-Mandic, P., Nguyen, M. et al. A non-randomized, open-label study of the safety and effectiveness of a novel non-pneumatic compression device (NPCD) for lower-limb lymphedema. Sci Rep 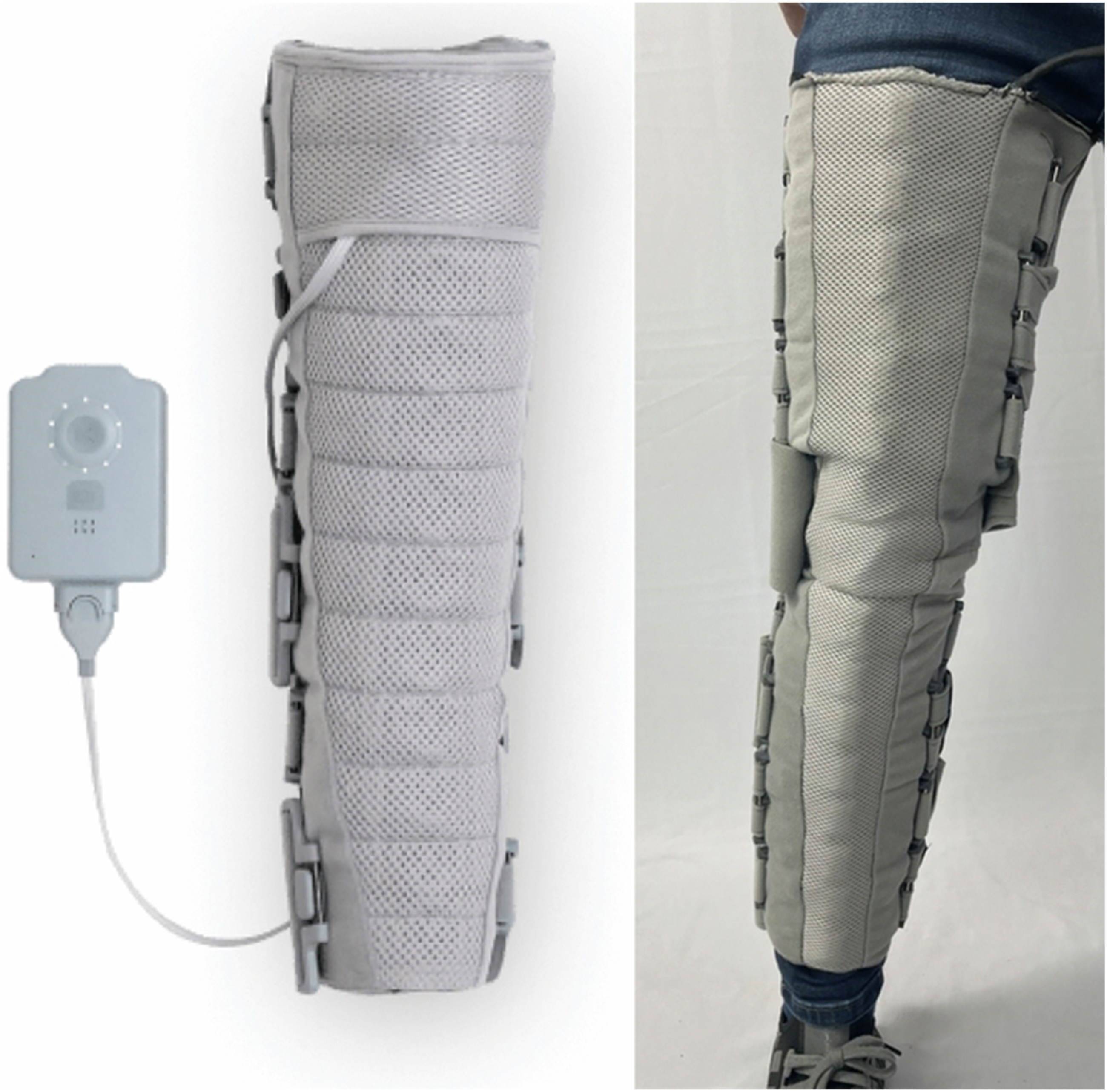
Compared to pneumatic compression or APCD, Dayspring has a unique multi-modal approach to compression; its mobile-powered garment provides static as well as active sequential compression, while allowing for the activation of the calf pump through exercise, and increased range of motion and activity. This results in enhanced lymph flow from the superficial lymphatics to the deeper, central lymphatics. Dayspring has been associated with improvements in quality of life and reduction of edema in patients with both upper- and lower-extremity lymphedema, but its effect on edema extending into the foot has not been well described.
7
Unlike APCD garments, the Dayspring garment does not encompass the foot and ankle. This study aims to specifically examine the effect of NPCD on foot and ankle swelling (Figures 2 and 3). Decrease in limb and foot circumference following utilization of the NPCD device. Decrease in limb volume following utilization of the NPCD device.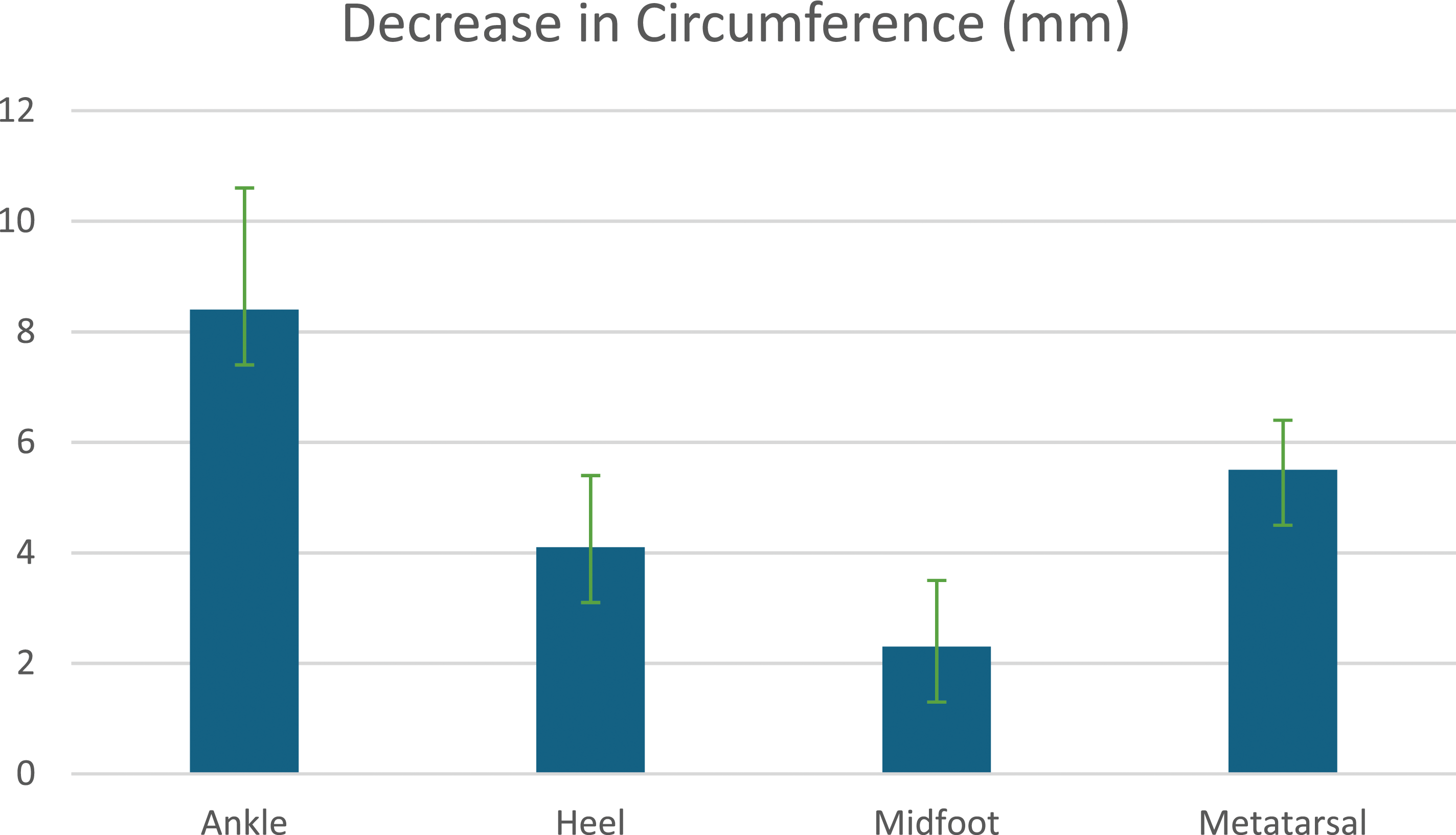
Methods
Patient cohort
This study included the population undergoing treatment with NPCD therapy (Dayspring®, Koya Medical) from August to December 2023. Subjects 18 years or older with venous insufficiency-related lower-extremity lymphedema, or phlebolymphedema, were included. This study excluded those patients with any contraindication for compression therapy/increased venous or lymphatic return, conditions that would prevent safe and effective use of a compression device (i.e., cellulitis, and open or healing wounds), acute thrombophlebitis, advanced peripheral arterial disease, recent pulmonary embolism/DVT, pulmonary edema, uncompensated congestive heart failure, or uncontrolled asthma.
Each subject was enrolled during an initial screening visit and trained in the application of the NPCD. Those with bilateral leg swelling were also included and prescribed bilateral devices as needed. At the initial visit, limb girth and volume measurements were taken, and quality of life was measured using a standardized LYMQOL survey. Patients were instructed to wear the non-pneumatic compression device (NPCD), for 60 minutes each day. Girth and volume measurements were taken at the foot and ankle with circumference measurements and an immersion device (CanDo Baseline Volume Measuring, Foot Set, 5-inch x 13-inch x 9 inch cavity), respectively, at enrollment, 2-weeks, and 4-weeks after use of the NPCD. A wearable exercise monitoring device to monitor daily ambulation levels was given to each subject during the study duration. Quality of life data was collected using the LYMQOL validated scale at enrollment and again at 4-weeks. Adherence was defined as using device as instructed 1 hour per day for duration of study.
Data collection
Demographic variables included age, gender, race, ethnicity, medical comorbidities, prior imaging studies, previous surgical history, and smoking status. Average number of steps walked per day during the study was quantified by exercise monitoring devices, with patients divided into high activity (>6000 steps) and low-activity groups (<6000 steps). Primary outcomes included QOL and foot/ankle measurements. Data on reintervention, death, MI, DVT/PE, and readmission for infection or ulceration was also collected. The study was approved by the New York University Institutional Review Board, and informed consent was obtained from all patients.
Statistical analysis
The software packages used for data analysis for this retrospective study were R and STATA. Categorical variables were presented as proportions, normally distributed continuous variables presented as mean + standard error, and skewed continuous variables presented as median (interquartile range). Assumptions were checked; non-parametric alternatives (such as Mann–Whitney U test) and mathematical transformations (such as Box–Cox) considered as needed for skewed distributions. Univariate and multivariable analyses were performed with candidate variables and outcome measures. Statistical significance was tested using a two-sided alpha level of 0.05 and with appropriate multiple testing correction (Bonferroni or Benjamini–Hochberg) approach when needed, with each limb considered a unique observation.
Results
Demographics.

Mean change in foot/ankle circumference (cm) and volume (ml).
A mean of 8100 steps per day were taken (±6100 SD). Seven subjects took over 6000 steps per day (the high-activity group) (mean = 11,000; range: 6500–25,000), with six patients taking fewer than 6000 steps per day (the low activity group) (mean = 4200; range: 2400–5500). There was no statistically significant difference between the high-activity and low-activity groups in volumetric change (−60 mL vs −78 mL, p = .75) or limb circumference (ankle: −0.59 vs −1.29 cm, p = .28).
Discussion
The most common cause of lymphedema in the developed world is phlebolymphedema. 8 Chronic venous insufficiency has been estimated to affect as much as 50% of the adult population 9 and accounts for 2% of Western health care budgets. 10 Venous hypertension in patients with chronic venous insufficiency leads to increased hydrostatic pressure in capillaries, which in turn leads to increased capillary permeability, and extravasation of proteins into intracellular spaces. 11 The accumulation of proteins and macromolecules in interstitial spaces occurs more rapidly than lymphatic vessels can drain them, thus leading to the clinical development of lymphedema. 12 As such, treatment of lymphedema and chronic venous disease are synergistic. There exists a gap in adequate clinical treatments for both chronic venous insufficiency and lymphedema. This study demonstrated an overall improvement in edema at the level of the foot and ankle using a novel portable non-pneumatic compression device that applies static and dynamic sequential compression to the extremity while allowing patients to remain active, leveraging the benefits of the foot and calf muscle pump.
Compression and elevation are mainstays of therapy for both chronic venous disease and lymphedema. Compressive garments do not extensively compress the deep venous system and lymphatic systems. Instead, the primary mechanism of compression garments is to empty the superficial venous system into the deep venous system, increasing the venous ejection fraction of the calf muscle pump. They also have the secondary effect of stimulating microvascular and lymphatic circulation, additionally aiding in diminishing subdermal edema and improving microvascular circulation. 13 Exercise therapy also works according to the same principle, activating the calf-muscle pump system with corresponding decreases in edema and limb volume. 14 Within this study, patients were able to maintain significant amounts of mobility and exercise, with patients taking an average of 8600 steps per day. Patients were successfully able to remain mobile and participate in activities of daily living while adhering to therapy. These results highlight the importance of exercise and mobility, which activate the foot and calf-muscle pumps, and their synergistic effect on edema reduction.
Previous studies comparing NPCD to APCD have demonstrated increased effectiveness of NPCDs in the upper extremity in breast-cancer related lymphedema 15 and also shown significant reduction in edema in the lower extremity. 16 Advanced pneumatic compression devices provide sequential compression of the extremity but rely on an air pump powered through an electrical outlet to inflate individual air chambers, hence limiting patient mobility. Unlike APCDs, the NPCD is a portable device that provides both sequential compression and static compression. In the lower extremities, the NPCD enables exercise and mobility that stimulates lymphatic and venous return through recruitment of the calf-muscle pump system. Indeed, in our study all patients remained active as measured by an electronic physical fitness monitor; even the lowest activity group was ambulating > 2000 steps per day, and the highest group recorded up to 25,000 steps per day. The calf muscle pump is responsible for the majority of venous return to the central venous system, with both a high capacitance and an ejection fraction of 65%. 17 Exercise increases the ejection fraction and thus overall venous return throughout both the leg and foot in patients with venous insufficiency.18,19 This beneficial effect of exercise on the calf-muscle pump system has been demonstrated in venous stasis as well as lymphedema, likely due to the interrelated nature of the venous and lymphatic systems and the effect of tissue edema and distension on both skin fibrosis and pain. 20
Unlike the APCD, the NPCD garment does not encompass or provide direct compression to the foot and ankle, as this would limit adorning footwear and mobility. One might speculate that the absence of a dedicated foot garment may lead to distal swelling. On the contrary, our study shows that the NPCD device showed reduction in foot and ankle edema as measured by metatarsal circumference distal to the device itself (Figure 1), in an area with no active pressure applied. The mechanism by which the NPCD achieves this decrease in foot and ankle edema may be attributed to its effect on permitting the calf muscle pump to function and increase overall venous ejection fraction and return. In turn, this results in decreased foot and ankle venous pressures.7,21 The NPCD aids in the return of lymph and venous flow back to the deeper venous and lymphatic system, enabling increased calf muscle pump return that is synergistic with the additional opportunity for exercise afforded by the mobile nature of the device. However, this mechanism does not fully explain the effect of the NPCD given there was no difference in swelling reduction between the high activity and low activity groups, suggesting that more activation of the calf muscle pump at a certain point did not yield more of a benefit. Further study into this effect is necessary to elucidate this. Finally, the fact that patients had improved outcomes while maintaining activity and mobility while using the NPCD may explain the improvement in quality of life.
Limitations of this study include its small sample size, and a larger cohort would be necessary to further evaluate this effect, especially to distinguish differences in high-mobility vs low-mobility cohorts. An additional limitation includes the non-blinded nature of this study. Furthermore, this study was a single-arm study with a lack of a comparison arm to evaluate the effect of non-pneumatic compression in conjunction with standard of care as opposed to standard of care alone. Nevertheless, the use of an immersion device to accurately measure foot and ankle volume (in addition to girth measurements at the ankle, forefoot, and midfoot level) is an important objective method of assessing the impact of the intervention on foot/ankle edema. This may represent an avenue for future research and investigative studies. Additionally, although patient adherence was self-reported, the use of mobility monitoring devices allowed for precise recording and capture of patient activity.
Conclusion
This study shows that NPCD results in significant reduction in edema in the foot and ankle, by volume and girth, as well as improvement in QOL. Specifically, despite the garment lacking a dedicated foot piece, foot and ankle swelling is significantly reduced with NPCD use. Importantly, patients were successfully able to remain mobile and participate in activities of daily living while adhering to therapy. These results highlight the importance of movement and exercise, an active calf-muscle pump, and its synergistic effect on edema reduction.
Footnotes
Author contributions
All authors contributed to the study design and conception. Data collection was conducted by AM and SB. Data analysis and the initial draft of the paper were performed by AR and TM, with revision and final approval of the paper by all authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AR, AM, TB, and SB have no conflicts of interest. TM serves as chief medical officer for Koya Medical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
