Abstract
Objective
To describe clinical outcomes associated with the use of the WRAPSODY Cell-Impermeable Endoprosthesis at a tertiary center in Western Australia.
Methods
Patients with recalcitrant occlusive disease in the venous outflow of their arteriovenous access circuits were treated with WRAPSODY. Patients were prospectively followed up to 12-month post-procedure. Study measures included 30-day adverse events, technical success, target lesion primary patency, access circuit primary patency, and assisted access circuit primary patency.
Results
Twenty-seven WRAPSODY devices were used to treat 15 consecutive patients. The technical success rate was 100%. No device-related adverse events were observed during the follow-up period. Two patients did not complete the full follow up. Patency rates at 3-, 6-, and 12 months for target lesion primary patency were 100% (15/15), 100% (15/15), and 100% (13/13), respectively. Rates for access circuit primary patency at 3-, 6-, and 12 months were 73.3% (11/15), 46.7 % (7/15), and 46.2% (6/13), respectively. Edge stenosis was observed in 33.3% (5/15) of cases and accounted for 5 of the 8 patients who experienced failed access circuit primary patency on angiogram. Primary assisted functional patency was 100% at 12 months.
Conclusion
WRAPSODY can be utilized safely and has durable patency in real-world patients with complex anatomical renal access stenotic lesions. The therapeutic benefits associated with the device may encourage broader use in clinical practice.
Keywords
Introduction
A permanent arteriovenous access circuit (autologous arteriovenous fistula [AVF] or a prosthetic arteriovenous graft [AVG]) is critical for the provision of long-term hemodialysis. However, once created the venous outflow section of the access circuit is vulnerable to progressive stenosis, which ultimately results in dysfunction or loss of the circuit.
AVFs and AVGs develop stenoses at key anatomical points (e.g., the cephalic arch in AVFs and the graft-venous anastomosis in AVGs) and central veins in both. 1 Standard of care involves percutaneous transluminal angioplasty (PTA) with or without bare metal stent placement. 2 However, both interventions are typically associated with low 12-month patency rates and high re-intervention rates.2–8 Although covered stents have shown superior patency rates compared to PTA,3–5 they are also associated with high re-intervention rates due to in-stent intimal hyperplasia, the inability to tolerate extreme anatomical demands of areas such as the cephalic arch, and edge stenosis9,10; furthermore, there are concerns related to their use, such as the risk of covering venous junctions, migration, difficulty re-canulating if they occlude, and, in particular, monetary costs.
Recently, a novel device, the WRAPSODY™ Cell-Impermeable Endoprosthesis, was designed to help overcome the limitations of interventions aimed at restoring patency of dialysis access circuits. 11 The inner spun polytetrafluoroethylene (PTFE) layer of WRAPSODY minimizes thrombus, the middle cell-impermeable layer prevents transmural cell migration, and its outer PTFE layer reduces inflammation. 11 Furthermore, the device has an optimized radial strength and compression resistance with flexibility to endure complex anatomy. The risk of edge stenosis has also been considered in its design through employment of softened, scalped end rows.
The clinical benefits associated with WRAPSODY’s design described in an animal model 11 have been confirmed in the first-in-human study. 12 However, a major criticism of the first-in-human study was that it was too controlled and patients were not reflective of a real-world population. As a result, additional evidence supporting the clinical utility of WRAPSODY is needed. To this end, we conducted a real-world, prospective study to describe the safety and effectiveness of WRAPSODY at 12-month post-deployment in Western Australia.
Materials and methods
Study design
Eligibility criteria.

AV, arteriovenous; AVF, arteriovenous fistula; AVG, arteriovenous graft; HD, hemodialysis; PTA, percutaneous transluminal angioplasty.
The treatment paradigm in the authors’ unit, prior to availability of WRAPSODY was to treat lesions on their first presentation with plain old balloon angioplasty (POBA) and proactively recall for an angiogram at 3 months to confirm that the lesion was angiographically and functionally treated. If lesions reoccurred within 3 months or more than once, a drug-coated balloon (DCB) was used for subsequent interventions. Uncovered stents were considered after multiple interventions had failed or inadequate response to dilation occurred (defined as residual stenosis of >50%) but they were avoided, if possible, due to the absence of evidence to support any long-term benefit. Covered stents were used rarely, due to concerns over cost, inability to manage complex anatomy (e.g., cephalic arch), lack of robust evidence supporting improved patency, and risk of shuttering junctions and collaterals within the central veins. Covered stents were primarily reserved for situations where vascular rupture or dissection had occurred. As a result, this paradigm meant that most patients had multiple interventions over extended time intervals, with one example patient in the study cohort having 9 interventions within a 2-year period.
The unique design features of WRAPSODY and encouraging first-in-man results 12 prompted consideration of its use for patients whose lesions were recalcitrant to the existing treatment regime with the hope that a longer patency rate would be beneficial in the longer term. For the purpose of this study, the treatment paradigm was modified as follows: patients were first managed via POBA before transitioning to a DCB as a second intervention; if restenosis reoccurred a third (or later) time, or if inadequate response to dilation occurred (defined as residual stenosis of >50%), or if the patient presented with acute thrombosis due to the target lesion, then WRAPSODY was prioritized over standard covered stents. Following any intervention for recalcitrant access circuit stenosis, the standard of care in the authors’ unit was to reschedule a planned re-look angiogram at 3 months irrespective of symptoms. Angiographic follow up in these patients was considered to be within normal care and allowed careful monitoring of outcomes.
This study was conducted in accordance with the Declaration of Helsinki and was approved by a local ethics committee (GEKO approval number: 48976). All patients provided written informed consent prior to enrollment.
Procedure
All procedures were performed in a hybrid endovascular suite under local anesthesia with or without sedation. Access was established percutaneously with ultrasound guidance followed by a diagnostic fistulogram to identify the target lesion(s). During each procedure, concomitant lesions at the swing segment, anastomosis, or the arterial limb were assessed. Target lesions were then crossed using a variety of wire/catheter combinations before pre-dilation with plain balloon venoplasty sized to the diameter of the surrounding vein.
The access venotomy was progressively upsized using sequential access sheaths before they were exchanged for WRAPSODY, which was inserted directly into the vein without a sheath in a bareback fashion. It was found that dilation to three French sizes smaller than the recommended sheath size for the selected device was required to allow bareback insertion, the same size sheath could then be replaced to maintain access and hemostasis after the WRAPSODY delivery system was removed.
Each device was oversized according to the manufacturer’s instructions for use and deployed to sufficiently extend beyond the length of the pre-treated lesion. Post-dilation was performed in accordance with the instructions for use at the same or lesser diameter to the WRAPSODY diameter. All patients were discharged within 24 h of the procedure.
Follow-up
Prospective follow-up visits for all patients occurred at 3-, 6-, and 12-month post-procedure with additional visits as needed for assessment and treatment of access circuit function. Follow-up visits consisted of a physical examination and assessment of dialysis parameters. Surveillance using catheter-based angiography was also performed at each follow-up visit to assess visual patency of the device and aligned with the pre-treatment intervention regimen for each patient.
Study measures and analysis
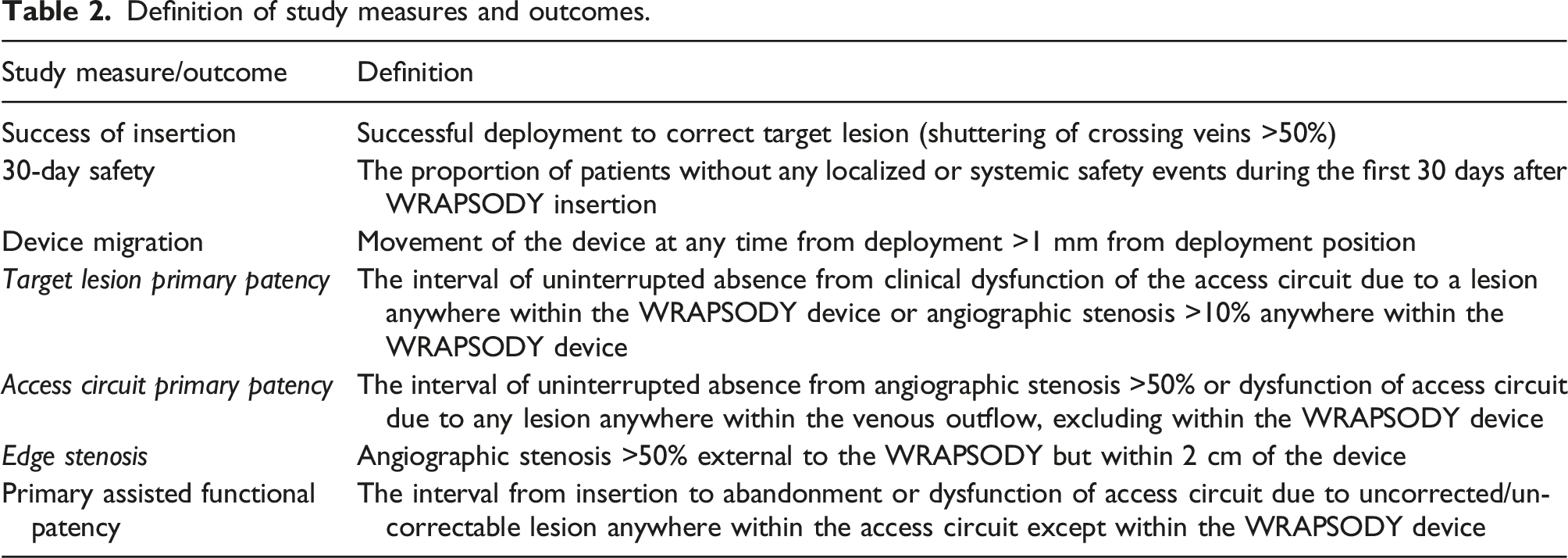
Definition of study measures and outcomes.
Descriptive statistics were used to summarize results; continuous measures were described using mean and standard deviation; categorical variables were described using counts and proportions. As this study was purely observational, no hypothesis testing was performed.
Results
Clinical outcomes
Patient demographics and lesion characteristics.

CFV, common femoral vein; EIV, external iliac vein; IV, innominate vein; IVC, inferior vena cava; LIV, left innominate vein; LSCV, left subclavian vein; RIV, right innominate vein; RSCV, right subclavian vein; RIV, right innominate vein; SCV, subclavian vein; SFA-GSV, superficial femoral artery-great saphenous vein.
aOther reasons for renal failure included Alports syndrome (n = 1), focal and segmental proliferative glomerulonephritis (n = 1), immunoglobulin A nephropathy (n = 1), microscopic polyarteritis nodosa (n = 1), and polycystic kidney disease (n = 1).
Three patients presented with acute occlusion of their fistulae due to a lesion that had undergone multiple interventions per the pre-study protocol of management. These patients underwent open thrombectomy prior to treatment of the distal target lesion.
Twenty-seven devices were used to treat all 15 patients. On average, 1.8 devices were used per patient (range: 1–3). Anatomical, clinical, and procedural success were achieved in all cases. One patient had protrusion of a cephalic arch endoprosthesis into the subclavian vein (>50%), resulting in shuttering of the drainage of the axillary vein at the confluence. No further intervention was required in this patient, and no adverse clinical or radiological sequelae arose during the follow-up period. This protrusion event occurred in the first patient enrolled in the series and did not occur in subsequent cases due to subsequent modification of the delivery technique.
No patient experienced an adverse event related to the procedure during the 30 days following the intervention and no stent migration was observed during the follow-up period. One patient was lost to follow-up after 6-months due to a personal decision to stop receiving dialysis and one patient died during the follow-up period (at 7 months) from factors unrelated to renal access. Both patients had patent access circuits at time of their loss from follow-up.
Target lesion primary patency and access circuit primary patency over the follow-up period.

No patients developed fistula dysfunction (i.e., any one of inclusion criteria 2–5 as outlined in Table 1). However, 8 patients underwent proactive intervention based upon angiographic findings of stenosis in the access circuit. In 3 cases the stenotic lesions involved either the juxta anastomotic region or areas disparate from the WRAPSODY device, the remaining 5 cases were classified as edge stenosis. Inflow edge stenosis occurred in 4 cephalic or basilic vein lesions and at the outflow edge of 1 centrally deployed device. In all cases, the edge stenosis appeared within the first 3 months post-deployment. One case of the 4 inflow stenoses and the one case of outflow edge stenosis were felt to be residual venous disease as the vein was extensively narrowed prior to the treatment. The 3 remaining cases of inflow stenosis were pro-actively treated with another WRAPSODY device or DCB at 3 months and did not return during what remained of the follow-up period (Figure 1). Treatment of a patient with a left brachiocephalic arteriovenous fistula with a pre-existing subclavian vein stent who presented with new stenosis at the cephalic arch. (A) Immediately post treatment with a single WRAPSODY device, deployed with some backward traction. Inflow (leading) edge of the WRAPSODY device is marked with an arrow. (B) Edge stenosis at the inflow edge seen at 3 months post insertion of single cephalic arch stent. (C) No reoccurrence seen 12-month post-deployment.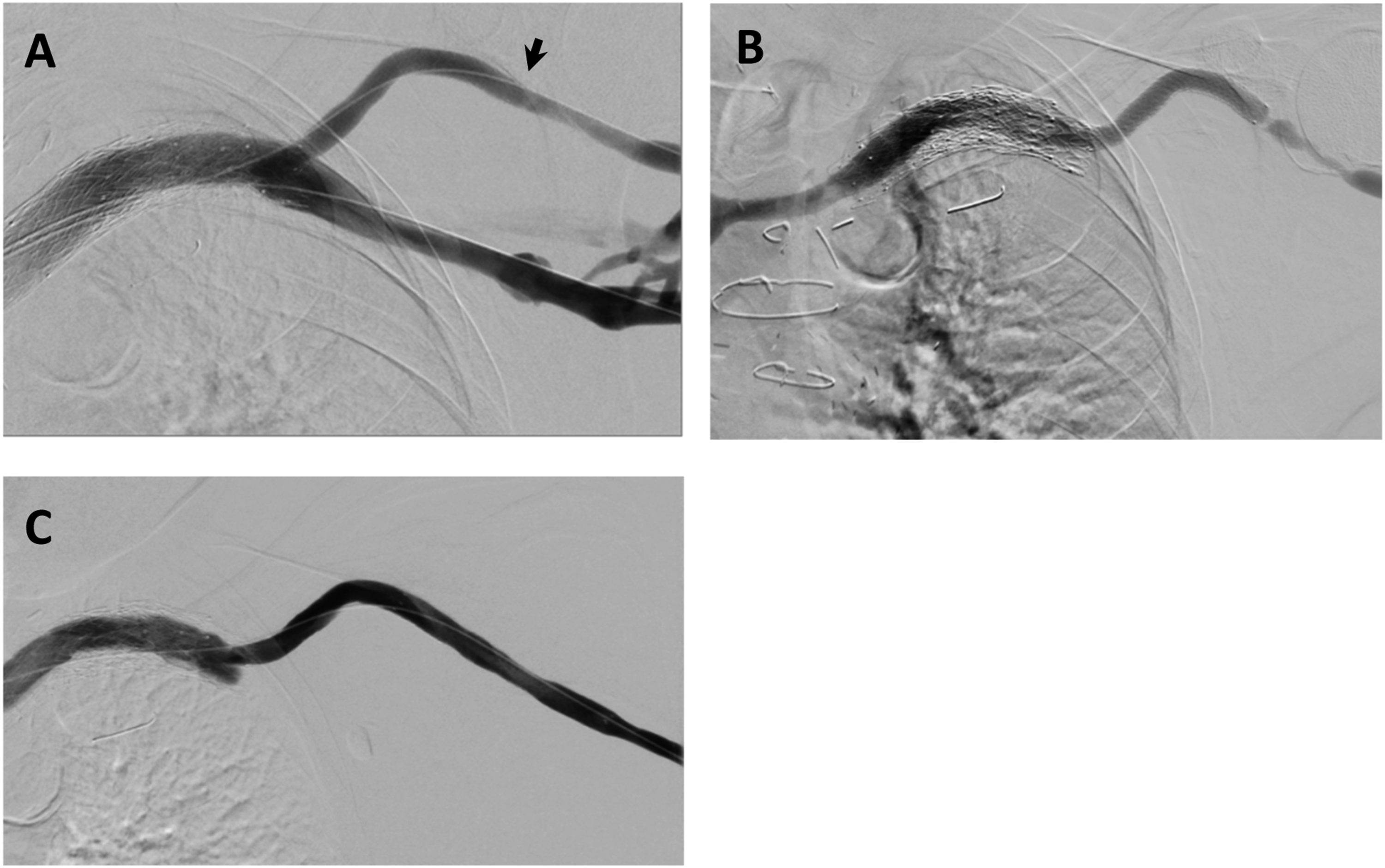
Description of practical experience
An example of a complex case is shown in Figure 2. In this example, three WRAPSODY devices were used to treat a tightly stenosed lesion in the cephalic arch extending into an aneurysmal section of the vein. In this case, the last (inflow end) device was deployed such that it did not have full vessel wall apposition. No further intervention was required at any time during the remainder of the follow-up period for this patient. Treatment of a patient with a left brachiocephalic arteriovenous fistula that presented with a stenosis at the cephalic arch with aneurysmal segment. (A) Fistulogram taken prior to the intervention, (B) after deployment of WRAPSODY to treat the lesion, (C) fluoroscopy of treated segment at 6 months, and (D) 12-month post-deployment.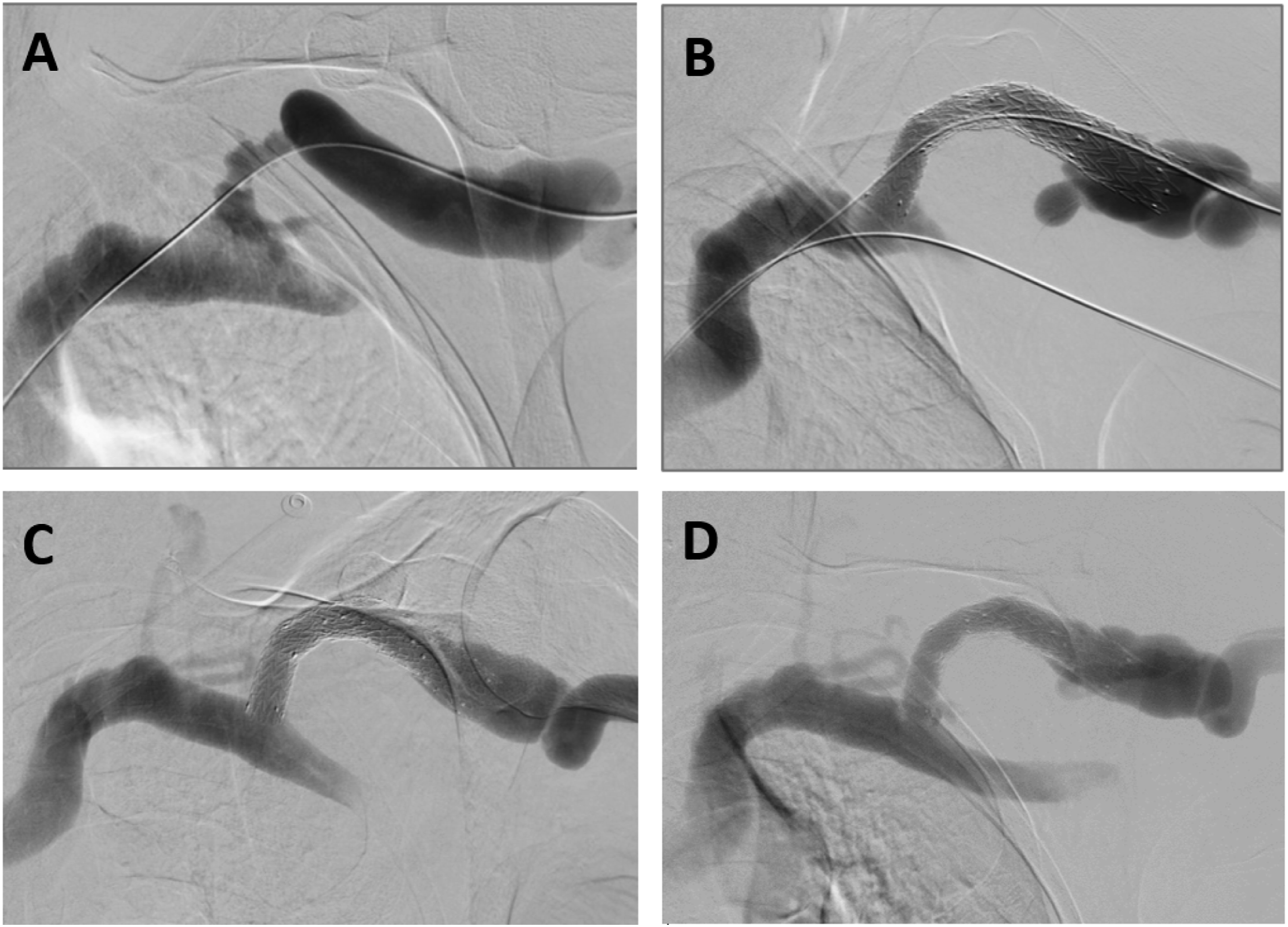
The WRAPSODY delivery system has a one-handed ratchet style bi-axial delivery. The French size of the device may be a concern to some operators. However, the unique hydrophilic delivery system with its smooth taper makes it ideally suited for bareback delivery and there were no issues or complications with this approach. Progressive upsizing of an access sheath to three French sizes smaller than the recommended delivery sheath to dilate the puncture site before introducing WRAPSODY was suitable. After deployment and removal, the original access sheath was replaced again with good hemostasis in all cases.
The bareback approach used meant that the procedure rarely required an access puncture size larger than 8-French, even when large caliber devices (up to 16 mm) were used. Purse string sutures (3/0 nylon) were inserted into the skin around the puncture and removed during the following dialysis session. As this technique was found to be sufficient early in the study, it became the standard method used. Cutdowns were only used on the first two patients and on patients where thrombectomies were conjointly required.
The technique of barebacking the device meant that it was not possible to undertake angiography around the device using the same access site. Consequently, it was routine to co-puncture the basilic vein when precise deployment was essential, such as placement in the cephalic arch, or contralateral subclavian vein in the case of central stenosis. This basilic puncture allowed a wire to be placed into the at-risk crossing vein to mark the location of the ostium and allowed precise deployment.
WRAPSODY is designed to allow firm backwards traction as it is partially deployed without the device falling off or unsheathing from the delivery system. This is advantageous as it allows the device to be partially deployed forward to the desired landing zone and then “snugged” back into the desired position. However, excessive back tension should be applied with caution as the authors (RGB and RA) believe this may be a contributing factor to the edge stenosis observed in some of the treated lesions where the back tension stretches the vessel causing some distortion that recoils when the device is fully deployed.
When a stent was placed within the cephalic arch and innominate vein/superior vena cava, every attempt was made not to encroach more than 25% into the traversing vein. However, in the single case when this occurred early in the study, and placement was observed on angiography to significantly shutter the traversing vein, no significant adverse effects were evident to the flow in that vessel and there was no need for further intervention. This may have been due to the scalloped crenulations at each edge of each device acting to ensure some flow was always maintained around the device.
Discussion
Results from this non-randomized, real-world, prospective study demonstrated that WRAPSODY can be used to treat complex vascular access lesions safely and effectively, as evidenced by the absence of adverse events, and high technical success rate (100%) and primary patency rates (100% 12-month TLPP). Importantly, findings from this study align with clinical outcomes reported in the first-in-man study, 12 which suggest that results may generalize to real-world patients with lesions located in complex anatomical sites (e.g., cephalic arch and central veins) where standard interventions typically have poor outcomes. Six patients in this study had central venous lesions. In our experience, standard interventions (angioplasty or DCB) for central lesions are generally associated with poor patency rates. Furthermore, to our knowledge, there is limited evidence to support stent placement compared to angioplasty alone. 13
To avoid loss or abandonment of the renal access circuit, recalcitrant access circuit stenosis often necessitates multiple returns to the interventional theater for treatments over prolonged periods of time. Among the 15 patients enrolled in this study, the average number of interventions required per patient was 3.3 in the preceding 12-month period. This is considerable as these patients are already a vulnerable population and many live in remote areas. Consequently, the need for repeat interventions exacerbates the already substantial negative impact of the disease on their quality of life and is a significant economic burden to the healthcare system. Interventions that can help reduce the frequency of care and increase patency rates are essential for this patient population.
Renal access circuit restenosis or failure occurs despite stent deployment for several reasons such as inaccurate device placement, migration, and or compression/fracture of device due to anatomical forces. Intimal hyperplasia within the device and edge stenosis externally are also common causes of failure. In-device restenosis is an important cause of failure of covered stents. Many postulate that it results from migration of intimal hyperplasia from the edges of the stent inward, toward the middle of the stent. However, prior evidence suggests that it is probably a result of direct ingrowth of cells through the permeable PTFE lining of most covered stents. 11 No cases of in-device stenosis of any degree were observed in this study, which would support the capability of the WRAPSODY impermeable layer in preventing this occurrence.
Edge stenosis, defined as a stenotic lesion fully external to the device but located at least partially within 2 cm of either the inflow or outflow edge, is another common occurrence in covered and uncovered stents. It is associated with reduced patency and increased failure of access but is poorly understood. Mechanical forces at the transition point between the rigid device and soft vein, particularly at sites of high mobility (e.g., cephalic arch), may also contribute to edge stenosis. Furthermore, intimal hyperplasia may occur either as a primary or secondary event and oversizing the stent may also encourage stenotic development.
WRAPSODY has been designed to mitigate the risk of edge stenosis through its softened end rows and scalloped edge. However, in this study edge stenosis was still observed in five (33%) cases. We believe this study to be unique in prospectively evaluating the outcomes of covered endograft insertion for AV access circuits with angiography at 3, 6, and 12 months where there was no observed dialysis circuit dysfunction. All other published studies have relied upon ultrasound or clinical findings only when the dialysis circuit failed or was dysfunctional. Two of the five cases in this series would not have been detected on ultrasound due to their central anatomical location and none of the patients presented with fistula dysfunction in our series. Where there was primary intervention of observed stenosis it is uncertain what the trajectory of these lesions would have been if left untreated. However, in one case the observed edge stenosis at 3 months was left untreated and no further progression of the edge stenosis appeared, the circuit was still functional at 12 months with no further intervention required.
Although previous studies have not evaluated prospective angiographic edge stenosis rates for covered endoprosthesis in AV circuits, some studies have reported “treatment area primary patency” based upon lesions causing fistula dysfunction. 14 The average 12-month treatment-area primary patency rates are notably poor in previous studies of other devices and ranged from 15.7 to 26.9% of which the majority were due to edge stenosis as well as in-device stenosis.14,15 While it seems unlikely, if our series is analyzed as worst-case scenario for treatment area primary stenosis where each prospectively observed edge stenosis treated were considered a dysfunctional circuit, and the two patients lost to 12-month follow-up are presumed to have resulted in edge stenosis causing clinical dysfunction at 12 months, the patency rates would have been 53%. Even at this patency rate, these results presented herein demonstrate a far greater patency rate than observed in other studies using other devices.
Given the comparatively low rate of edge stenosis compared to historical studies the design of WRAPSODY appears to have ability to mitigate the condition. It is possible that the edge stenosis observed may have been but related to the nuances of deployment. To successfully and accurately deploy WRAPSODY at a junction (e.g., where the cephalic vein joins the axillary/subclavian vein), the device is deployed forward of its intended sight and then “snugged” back into position and held with moderate backward tension whilst the rest of the device is deployed. The design allows over half of the device to be deployed whilst traction is applied and facilitates a precise positioning. It is possible that this backward tension may encourage a localized “rucking up” or concertinaing of the device into the non-stented vein, forming a mechanical intussusception. Supporting this belief is that all three cases of inflow edge stenosis occurred when only a single device was deployed, whereas, in all cases where a second WRAPSODY was placed at the leading edge of the first device without any back tension, no edge stenosis was observed. Furthermore, all cases detected in this study occurred rapidly and were present at first angiography at 3 months, suggesting a mechanical issue. If an additional WRAPSODY was placed at 3 months, no further edge stenosis was observed over the next 9 months.
It is possible that placing a second WRAPSODY device might have increased the likelihood of extending it more distally down the arm to vessels where less of a torsion effect would be observed, thereby reducing the risk of edge stenosis. However, additional assessments in a larger cohort of patients are needed to confirm whether routinely deploying a second WRAPSODY, not under tension and/or if a more distal extension into the upper arm, prevents or reduces the occurrence of edge stenosis. Instructions for use recommend a 2 mm oversizing of the device to vein diameter. An important reason for such oversizing is to prevent migration in high pressure systems; however, it is possible that a lesser oversize can be used safely in stenotic lesions and may also reduce the risk of edge stenosis. Further investigation regarding the optimal sizing of WRAPSODY in renal access settings is warranted.
This study was subject to certain limitations. While all treated lesions were representative of challenging real-world lesions with limited therapeutic options, the number of patients assessed was small. Furthermore, the proactive approach to follow up with angiography and treatment of lesions based upon angiographic appearance alone has the potential to bias the results. Patency outcomes following treatment have historically been reported according to clinical function of the dialysis circuit, ultrasound imaging, or flow rates (in accordance with prior guidelines from the Kidney Disease Outcomes Quality Initiative 16 ). Consequently, reporting angiographic abnormalities might have implied a worse outcome if there was more conventional follow up. There was no fistula dysfunction in any patient evaluated. The decision to treat was based solely on the physician’s clinical assessment. It is recognized that this may have resulted in some lesions being treated that may not have developed into dialysis circuit dysfunction but reflects the desire to ensure best possible outcomes for a vulnerable patient group. Undoubtedly, patients in this study received close attention during the follow-up assessment, which may have positively influenced their outcomes.
Another limitation was the low number of AVG cases despite the small sample size overall. This was partly due to the local practice of striving for native vein options as a primary treatment approach rather than synthetic grafts. Despite having a practice of ∼200 primary formation cases per year, the percentage of AVGs formed is <1%, which limits inferences that can be made.
Overall, findings from this real-world study suggest that WRAPSODY is a safe and effective treatment for AV access circuit stenoses. The high TLPP and absence of device-related adverse events observed following procedures in anatomically complex veins among patients that underwent multiple interventions in the preceding year demonstrate the clinical utility of WRAPSODY in real-world practice. These results may help adopt broader use of WRAPSODY thereby optimizing the clinical management of patients with stenosis in their AV access circuit.
Supplemental Material
Supplemental Material - Patency rates following treatment with the WRAPSODY™ cell-impermeable endoprosthesis for recalcitrant renal access circuit dysfunction: Results from a tertiary Australian centre
Supplemental Material for Patency rates following treatment with the WRAPSODY™ cell-impermeable endoprosthesis for recalcitrant renal access circuit dysfunction: Results from a tertiary Australian centre by Richard G Bond, Rohan Arasu, Troy M Jantzen and Richard P Alley in Vascular
Footnotes
Acknowledgments
Merit Medical was involved in certain aspects of data collection. Editorial and administrative assistance in the preparation of this article was provided by Gloria DeWalt, PhD, an employee of Merit Medical.
Author contributions
This study was designed by RGB. Data were gathered by RGB and analyzed by all authors (RGB, RA, TMJ, and RPA). RGB and RA prepared the initial draft of the manuscript. TMJ and RPA assisted with writing and editing the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RGB: Paid consultant for Merit Medical Systems, Inc.; RA: Conflicts of interest: none; TMJ: Employee and stockholder of Merit Medical Systems, Inc.; RPA: Employee and stockholder of Merit Medical Systems, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
