Abstract
Objective
Subclavian artery aneurysms (SAA) are rare and usually involve the proximal segment of the artery. In this setting traditional surgical treatment or hybrid procedures are associated with significative morbidity rate so that new endovascular solutions are preferred.
Methods
Authors report a case of a patient with an intrathoracic left SAA involving the aortic arch’s outer curve. Patient’s comorbidities contraindicated open surgical treatment while anatomical issues were not favourable to hybrid or standard endovascular solutions. Thus a single-branched custom-made thoracic stent-graft (Castor, MicroPort Medical, Shanghai, China) was implanted.
Results
Technical success was achieved with no intra-operative complications and technical steps of the procedure are accurately described.
A pre-discharge CT scan showed the correct position of the endograft, complete aneurysm exclusion and branch patency.
Conclusion
Single-branched thoracic stentgrafts could be a good option in unfavourable anatomies with supra-aortic vessels disease involving the aortic arch, with good results in terms of morbidity and early outcomes.
Keywords
Introduction
Subclavian artery aneurysms (SAA) are rare but rupture is associated to high morbidity and mortality rates. 1 They are usually classified in intrathoracic and extrathoracic according to their position inside or outside the chest but the most frequent location is the proximal segment of the artery.
Open surgical repair and hybrid procedures are quite invasive so that surgical treatment is shifting towards total endovascular solutions.
Authors report a case of a patient treated with a total endovascular percutaneous approach using a single-branched custom-made thoracic stent-graft (Castor, MicroPort Medical, Shanghai, China) to exclude an intrathoracic SAA with unfavourable anatomy, involving both the aortic arch’s outer curve and the first segment of left subclavian artery (LSA). Technical steps of the procedure are described in detail. No ethical committee evaluation was needed for this case and the patient provided informed consent for the surgical treatment.
Case report
A 57-years-old male patient was incidentally diagnosed with a LSA aneurysm of the proximal segment (Figure 1). CT scan showed an intrathoracic LSA saccular aneurysm involving the outer curve of a type I aortic arch. Aortic proximal diameter was 28 mm in zone 2 (according to Ishimaru classification) with 14 mm distance between left carotid artery and LSA origin. Aortic diameter was 26 mm on descending aorta at the level of the intended distal landing zone. Subclavian artery diameter was 13 mm just caudally to the aneurysm and total subclavian length to vertebral ostium was 36 mm. A total endovascular solution with a custom-made Castor (MicroPort Medical, Shanghai, China) single –branched unibody thoracic stent-graft and distal side branch relining with a balloon expandable stent was planned. The procedure was performed by experienced vascular surgeons in a dedicated vascular operative room equipped with a mobile C-arm (Ziehm Imaging GmbH, Nuremberg, Germany), an echo duplex scan (Esaote AU 5, Genova, Italy). The intervention was completed under general anaesthesia, through echo-guided percutaneous puncture of both femoral arteries (Standard Proglide Preclose technique) and left proximal brachial artery (single Proglide). Pre-operative 3D CT reconstruction shows the intrathoracic left subclavian artery aneurysm involving the outer curve of the aortic arch.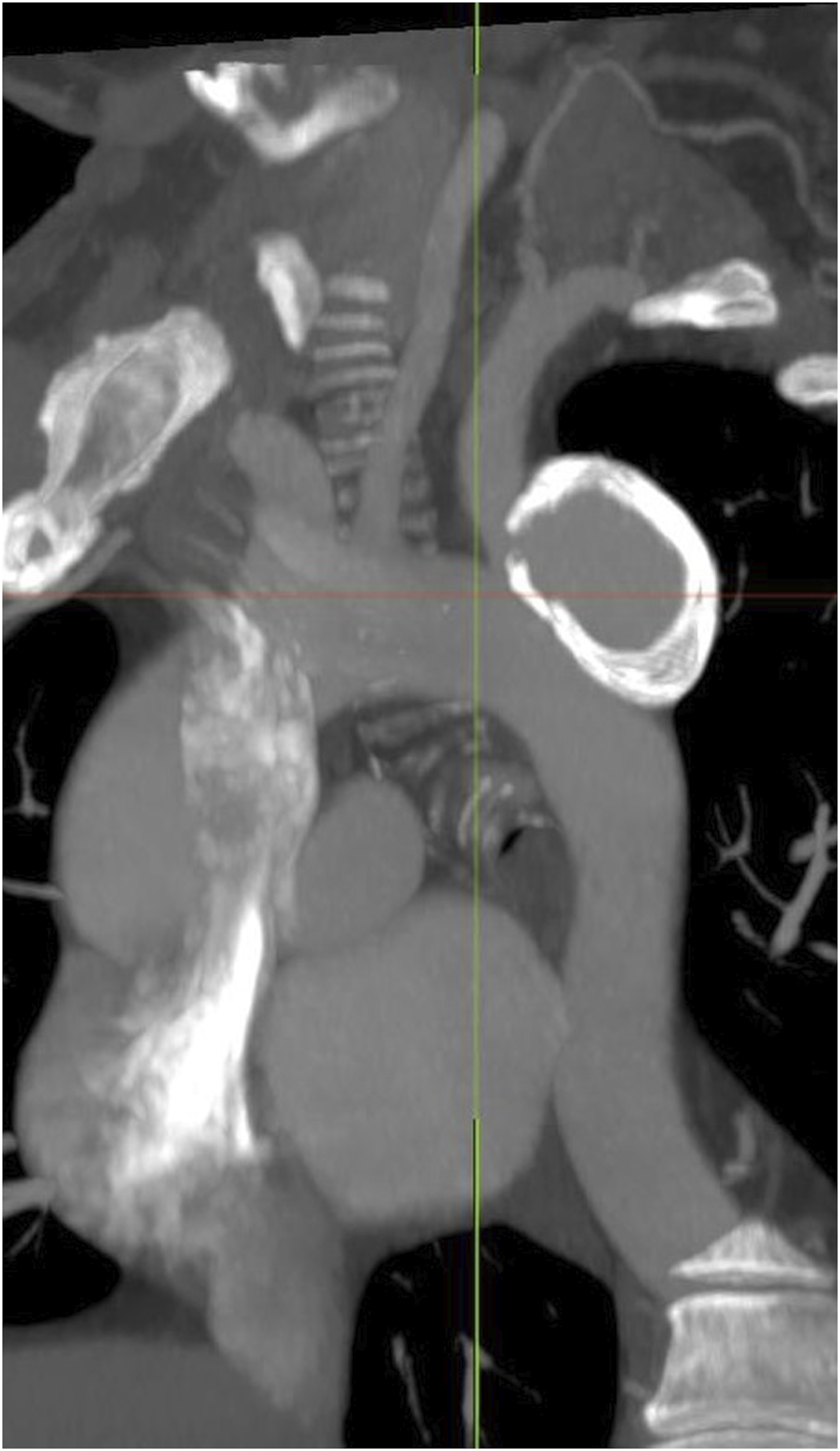
The Castor unibody single branch endograft (MicroPort Medical, Shanghai, China) customisation involved main-body proximal-end to side-branch distance, distal side-branch diameter and total main-body length, respectively 15 mm, 12 mm (the largest diameter available) and 120 mm (the shortest length available). A main body proximal diameter of 34 mm and distal of 28 mm was selected.
According to endograft IFU a 6 Fr - 155 cm selective catheter was advanced from brachial to right femoral access over an axillary-femoral through-and-through hydrophilic guidewire. The traction wire of the side branch was then retrieved through the catheter from the axillary access and the graft advanced in the ascending aorta over a coaxial super stiff guidewire (Lunderquist, Cook Medical Inc., Bloomington, IN) simultaneously retrieving the branch wire from above.
Following standard procedural steps, the device was positioned along the descending aorta and the outer sheath retracted.
Then the traction wire was de-twisted from the main body rotating the main device in order to ensure right orientation of LSA side-branch along the aortic outer curve (Figure 2(A) to (B)). (A) Intra-operative procedural steps during stent-graft deployment: side-branch traction wire was twisted around the proximal segment of the stentgraft and required several rotation manoeuvres to free it from proximal main-body, (B) The stentgraft is advanced towards the aortic arch with proximal markers positioned along the outer curve. (C) After complete outer sheath retrieval the side-branch is positioned into LSA while the graft is advanced towards arch’s outer curve to gain as much proximal sealing as possible, (D) Main body deployment allows withdrawal of the traction wire and delivery of the side branch.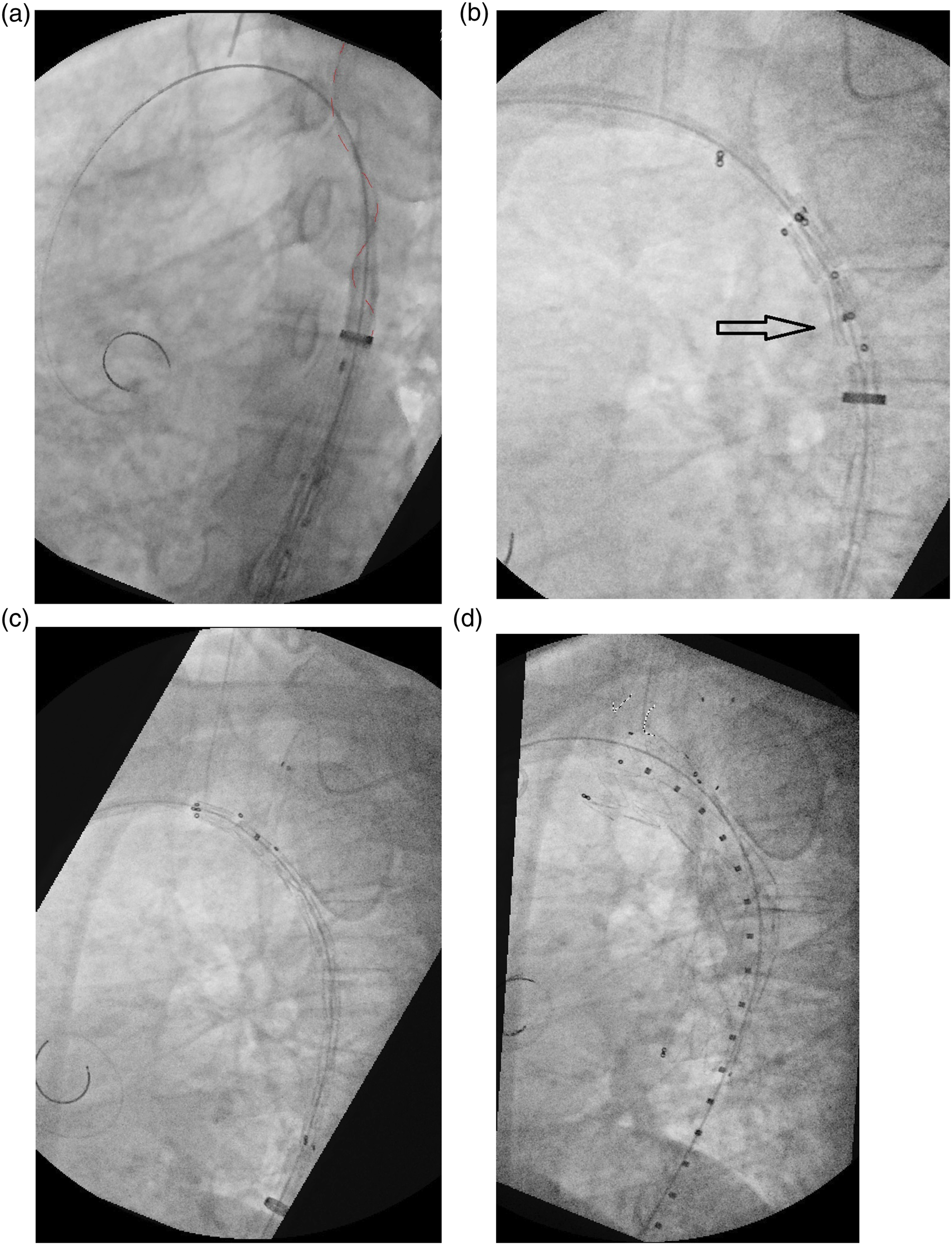
A diagnostic angiography was performed through a pigtail catheter inserted from left femoral access to confirm orientation and stentgraft position.
The main body was then pushed forward while pulling the traction wire to drive the side-branch into LSA origin (Figure 2(C)). Stentgraft’s side-branch pre-release prevention design avoids accidental side branch opening before main body delivery is completed.
The graft was then rapidly deployed “gently” pushing the graft towards the outer curve and finally, the side branch opened pulling out the traction wire. (Figure 2(D))
Angiographic control showed a side branch type IB endoleak. As planned, a distal relining was performed with a balloon expandable covered stent VBX 11 L X 29 mm (W. L. Gore & Associates, Flagstaff, AZ), deployed antegrade from left femoral access. To avoid distal migration after primary delivery of the undersized stent an angioplasty balloon catheter (Mustang, Boston Scientific, Marlborough, MA) was inserted retrograde through brachial access and inflated just distal to the vertebral artery before stent release (Figure 3). The VBX stent was then flared with a 14 mm non-compliant balloon (Atlas Gold, BARD Peripheral Vascular Inc., Tempe, AZ) to achieve complete distal sealing. Final angiography confirmed aneurysm complete exclusion with no endoleak. Distal branch relining with a 11 mm VBX L balloon expandable covered stent was planned to correct type IB endoleak on LSA. A semi-compliant balloon was inserted via retrograde from humeral access to avoid stent migration.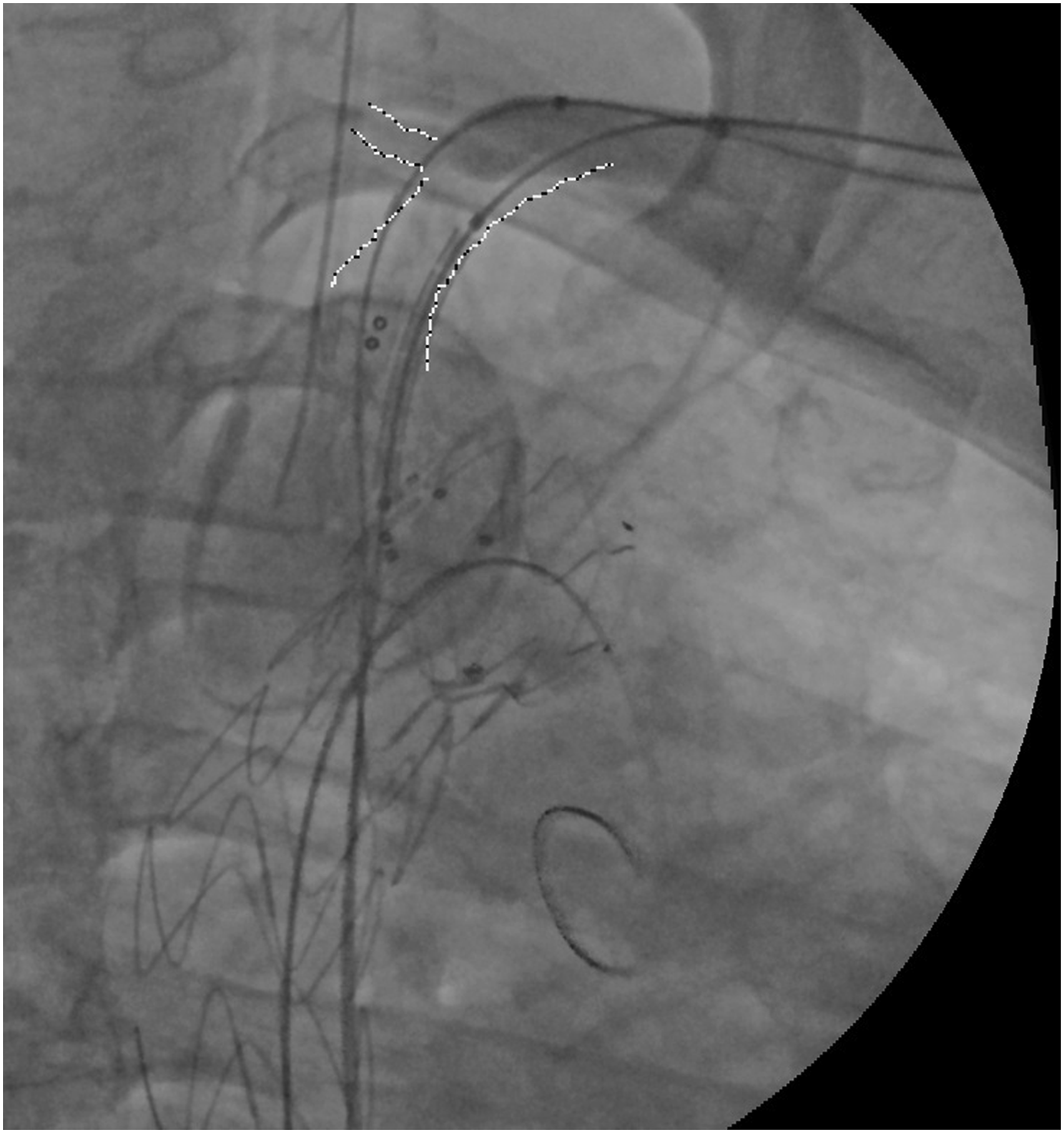
Technical success was achieved with no intra-operative complications. Total operative time was 110 min, total fluoroscopy time 30 min and total contrast medium 110 mL.
A CT scan performed on the second post-operative day confirmed complete aneurysm exclusion and branch patency (Figure 4). Post-operative stay was uneventful and the patient was discharged on third post-operative day with single antiplatelet therapy for life as standard treatment. A 6 months and 1 year CT angiogram follow-up was planned. Post-operative CT 3D reconstruction showing the correct position of the stentgraft, patency of all the supra-aortic trunks and complete aneurysm exclusion.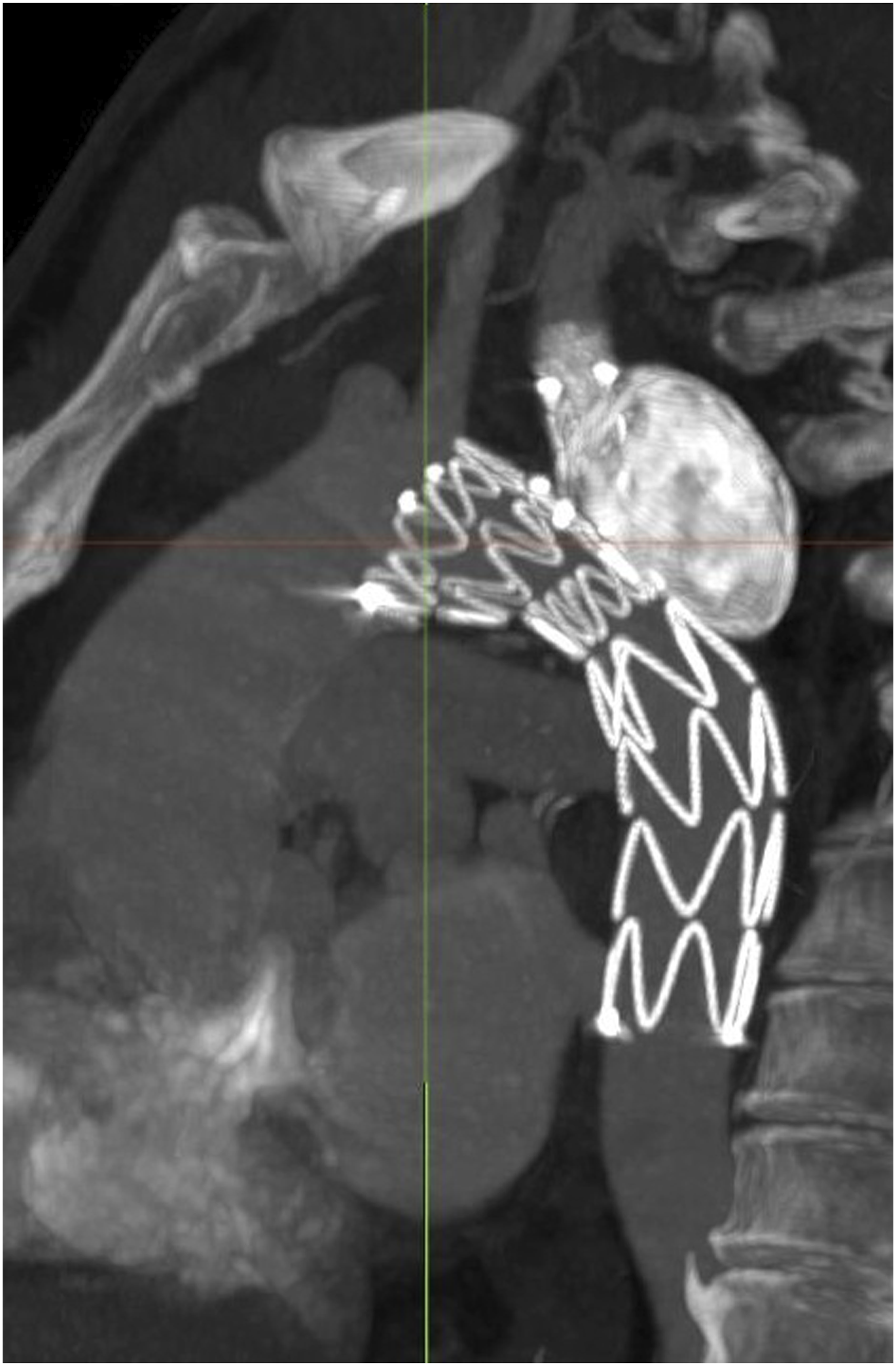
Discussion
Nowadays total endovascular solutions are often the first line strategy for LSA aneurysms and advanced research is done in the field of branched stent-graft specifically designed for the aortic arch.2,3
Preservation of LSA during TEVAR has generated debate, especially in terms of prevention of stroke and spinal cord ischemia, 4 but there are no recommendations on the most effective technique to achieve this goal.
Hybrid treatment (TEVAR + LSA surgical revascularisation) is probably the widely used technique. It consists of zone 2 TEVAR with previous LSA surgical bypass, usually between left common carotid and LSA distally to vertebral origin. Management of LSA presents some drawbacks regarding the surgical procedure itself (stroke, bleeding, nerve injuries) and is debated in its indications.5,6
A recent systematic review and meta-analysis stated that LSA revascularisation during TEVAR was associated with a lower rate of perioperative stroke (5.4%) compared with LSA coverage (7.8%) and Authors recommend LSA revascularisation whenever possible. Although relevant (16 cohort studies with 2591 patients included) no RCTs on the subject are reported. 7
Other authors suggest revascularisation in selective subgroups, while ESVS and SVS guidelines recommend LSA revascularisation in all patients unless in emergent situations.8,9
Parallel grafting, with the use of a stent alongside the main graft to maintain branch vessel patency could be another option and is usually performed in urgent settings. This technique can be adopted only in specific anatomies and is usually associated with higher incidence of gutter-related endoleak, along with problems of parallel grafting itself (friction and stent fracture, occlusion). 10
Recent papers have highlighted the non-inferiority of chimney endografting compared with carotid-subclavian bypass but this technique is still considered off-label for all thoracic stents on the market. 3
Finally, branched thoracic stentgrafts could be an option, but to our knowledge the majority of them are currently under evaluation or used in highly specialised centres in investigational settings. 11
Left subclavian artery intentional occlusion was excluded in our patient for the young age and active working life and is usually not taken into consideration at our division for elective surgery.
Open surgical approach was ruled out because of high morbidity related to the recent PTCA procedure performed for symptomatic ischemic cardiopathy.
Considering standard hybrid options and chimney endografting, authors were concerned about the short proximal landing zone. Hybrid treatment (TEVAR + carotid-subclavian bypass) was ruled out because of the short proximal sealing in zone 2 related to involvement of both the arch and proximal LSA. Using parallel grafting technique, the short overlap between the aortic and the chimney graft was considered prone to proximal endoleak development. In addition, the acute angle between LSA proximal segment and aortic arch was deemed at risk of kinking and occlusion of the chimney stentgraft. Parallel grafting with retrograde perfusion of LSA (periscope technique) was excluded and is performed only in urgent cases or as a bail-out manoeuvre at our centre.
Fenestrated devices specifically designed for the aortic arch (semi-custom) and custom made thoracic endografts were also excluded. Authors were concerned about adequate sealing of large fenestrations for the extensive SAA involvement of the external curve of the aortic arch. 12
Endograft in situ laser fenestration and surgeon modified solutions could be another possible choice for this specific case but the elective setting, concerns about long terms results and low expertise with this technique led authors to rule out this solution.13,14
The Castor custom-made thoracic endograft with mono-branch is recently available on the European market and patient’s anatomy was considered favourable for its use. Castor stentgraft allows customisation in a wide range of combinations concerning mainbody proximal diameter (26–44 mm), distal diameter (20–44 mm), branch distal diameter (6–14 mm), mainbody length (60–210 mm), branch length (25–45 mm), mainbody proximal end-to-branch distance (5–30 mm). Although, not all these combinations are possible and not all were available in Europe when the treatment was performed.
Literature reports good early outcomes with this endograft for treatment of aortic type B dissection or intramural haematoma. These experiences are mainly from Asia, where LSA is revascularised in highly selected cases during TEVAR procedures.15–17 To our knowledge, there are still no published experiences reporting treatment of aortic arch or proximal LSA aneurysms with this device.
Authors believed that stentgraft’s specific customisation could allow a short thoracic aorta coverage along with LSA and vertebral artery sparing, ensuring a stable implant and a durable sealing minimising the risk of paraplegia and neurological complications.
Technical success was achieved without intra-operative complications and the procedure was completed percutaneously, even if accurate procedural steps were necessary to avoid complications in such a hostile anatomy. Several rotation manoeuvres were needed to de-twist the traction wire from the proximal segment of the stentgraft. Care was taken to “sit” the branch towards the proximal side of LSA ostium to achieve the correct position of the graft and gain as much proximal sealing as possible. Lastly, a planned relining of the side branch was performed with a 11 mm VBX L balloon-expandable stent allowing good flexibility and large distal flaring for adequate sealing on LSA.
Conclusions
Nowadays endovascular procedures offer a wide range of possible solutions to treat aortic arch and LSA diseases and are generally associated with low morbidity and mortality rates. Hybrid solutions are still a valuable option but technological advances are now offering new tools to treat these pathologies with total endovascular solutions. Single-branched thoracic stentgrafts could be an option in unfavourable anatomies, as supra-aortic vessels disease involving the aortic arch, with good early outcomes. Further investigation is needed to confirm early promising results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
