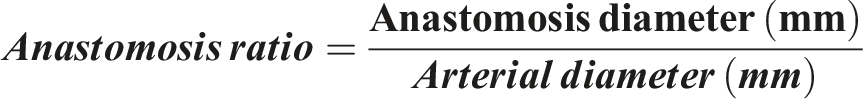Abstract
Objectives
The aim of this study is to assess the association between the anastomosis diameter enlargement and steal syndrome incidence in patients with upper limb arteriovenous fistula using ratios as reliable predictors.
Material and Methods
An analytical cross-sectional prospective study was conducted. A total of 49 patients with AVF hemodialysis access were recruited. Twenty-four participants with positive steal syndrome and 25 control were enrolled in the study. Anastomosis diameter, anastomosis diameter ratio, and volume flow ratio were measured ultrasonographically by two expert vascular sonographers. These clinical parameters were recorded and analyzed to assess the difference and association. Patient risk factors and steal syndrome association were emphasized.
Results
The study analysis indicates a strong association in the anastomosis diameter and anastomosis diameter ratio between steal and non-steal patients with a p-value ≤0.05. Additionally, there was a significant increase in the volume flow ratio in the patients with steal syndrome compared to the control group (p-value ≤0.05). There was a strong relationship between steal syndrome and the presence of peripheral arterial disease (73.9%, p = 0.001). The ICC index of absolute agreement between the two observers was ICC= 0.99 (95% CI 0.99–0.99, n = 10), indicating excellent agreement between observers.
Conclusion
Anastomosis diameter and volume flow ratio strongly suggest that steal syndrome is associated with the increased diameter of anastomosis. Patients with a≥1.05 anastomosis diameter ratio have a greater risk of developing steal syndrome than those with an anastomosis ratio of ≤0.8. In addition, patients with a volume flow ratio ≥0.98 have an increased risk of developing steal syndrome than those with a volume flow ratio of ≤0.75.
Introduction
Arteriovenous fistula (AVF) in the upper limb is considered the preferred hemodialysis access. AVF can be used for long-term access because it can provide better patency and fewer events of complications. However, fistula complications include aneurysms, infection, thrombosis, venous hypertension, and steal syndrome. Steal syndrome is a common problem in patients with arteriovenous access. Steal syndrome occurs when no vascular adaption after the dialysis access is constructed. It is defined as retrograde arterial flow distal to the anastomosis, which may lead to hand ischemia or “steal.” Steal syndrome can be divided into two categories, physiologic and distal ischemic steal syndrome (DISS). The physiological steal phenomenon may present in AVF cases as reversal flow in the distal outflow artery. DISS means that the patient presents with a symptomatic clinical steal which may develop into distal ischemia. The common symptoms presentation are nail changes, cold hand, muscle weakness, rest pain, fingertip ulceration, and tissue loss. The advanced stage of DISS could lead to severe morbidity and limb loss.1,2 It was stated that steal syndrome is less common when a radial artery is used (less than 1%) and in 5–10% of cases when the brachial artery is used. 1 Moreover, steal syndrome is more common in older women with diabetes and peripheral occlusive disease.1–3
The common causes of the steal syndrome phenomenon include the presence of arterial occlusive disease in the upper limb, which may limit the flow to the AVF access, increase the flow volume through the fistula, and absence of collaterals.1,3–7
Previous studies suggested a grading system for steal syndrome based on the symptoms. The patient normally presents with cool extremity with few symptoms and steal demonstrable by flow augmentation with flow reversal in the artery distal to the anastomosis; this is considered a mild steal (grade 1). When the patient develops intermittent ischemia only during dialysis/claudication can be graded as moderate (Grade 2). When the patient presents with ischemic changes and severe pain at rest or tissue loss, it can be graded a severe (Grade 3).3,4
Symptomatic steal syndrome can be diagnosed by an accurate history and physical examination. Additional imaging is required to confirm clinical steal symptoms, including an arteriogram and Duplex Ultrasound. Duplex Ultrasonography would provide anatomical and hemodynamical details of the clinical steal symptoms causes. The common finding is stenosis in the inflow artery, which will limit the flow distally. More specifically, spectral Doppler waveforms analysis and pressure assessment of the finger would provide more information regarding distal flow circulation.5–10 Finger pressure with and without compression of the access is helpful to make the diagnosis during the ultrasound scan. The change in finger pressure after AVF access compression is the key diagnosis of symptomatic steal syndrome. Retrograde flow or diminished or absent of radial and ulnar flow is another ultrasound finding. Angiogram is considered the gold standard for steal syndrome diagnosis and treatment. An angiogram would provide an excellent anatomical location of the length and severity of the stenotic lesion. Then, in the same setting, vascular stenotic lesions can be treated using balloon angioplasty.1–5
Some assumptions in the literature state that anastomosis diameter could be associated with stealing the arterial flow. No prospective studies have investigated this association in detail. Hence, it is assumed that enlarged anastomosis diameter is strongly associated with the occurrence of steal syndrome. The main aim of this study is to determine whether the increased diameter of the anastomosis is a cause and related to the incidence of distal steal syndrome.
Material and methods
This prospective cross-sectional study was completed during a 12-month interval (November 2020 to October 2021). Arteriovenous fistula patients referred to the vascular ultrasound department with clinical symptoms of steal syndrome or hand ischemia were enrolled in the study. The study was explained to all participants, and all participants were provided with written informed consent prior to the ultrasound scan. The inclusion criteria were referral with mixed grades of symptomatic steal syndrome, and patients could provide consent by themselves or an authorized guardian. The exclusion criteria were patients unable to sign the consent form, follow-up patients who had been previously examined in the vascular lab and diagnosed with steal syndrome, and patients with a low volume flow (<300 mL/min) in either an afferent artery or efferent vessel. The collected data included age, gender, risk factors, symptoms, and previous vascular surgeries. The Research Ethics Committee and the Institutional Review Board from our University were obtained.
Ultrasound systems and data collection
All participants were scanned using a high-end conventional ultrasound machine, including Supersonic Aixplorer (Supersonic Imagine, Aix-en-Provence, France) and iU22 Philips machines (Philips iU22 XM, Eindhoven, Netherlands) that use linear transducer (3.0–7.0 MHz). Two accredited vascular sonographers conducted ultrasound scanning with more than 10 years of experience in medical sonography. The vascular sonographer performed an interobserver reproducibility study on 10 patients to validate the measurements and assess the level of measurement agreement.
Based on the inclusion/exclusion criteria, 49 consecutive AVF patients with different anatomic configurations were enrolled in the study. Twenty-four patients with clinical steal symptoms and 25 AVF control group (negative steal). The control group was selected based on the following criteria: - Patient with Mature AVF fistula on regular HD. - No previous history of clinical steal symptoms. - Patient with normal flow volume >500 mL/min.
Several parameters were assessed, including anastomosis diameter, Peak systolic Velocity (PSV), flow volume (FV), and distal flow waveforms. In addition, the anastomosis ratio and flow volume ratio were calculated to compare with the control group.
Examination technique
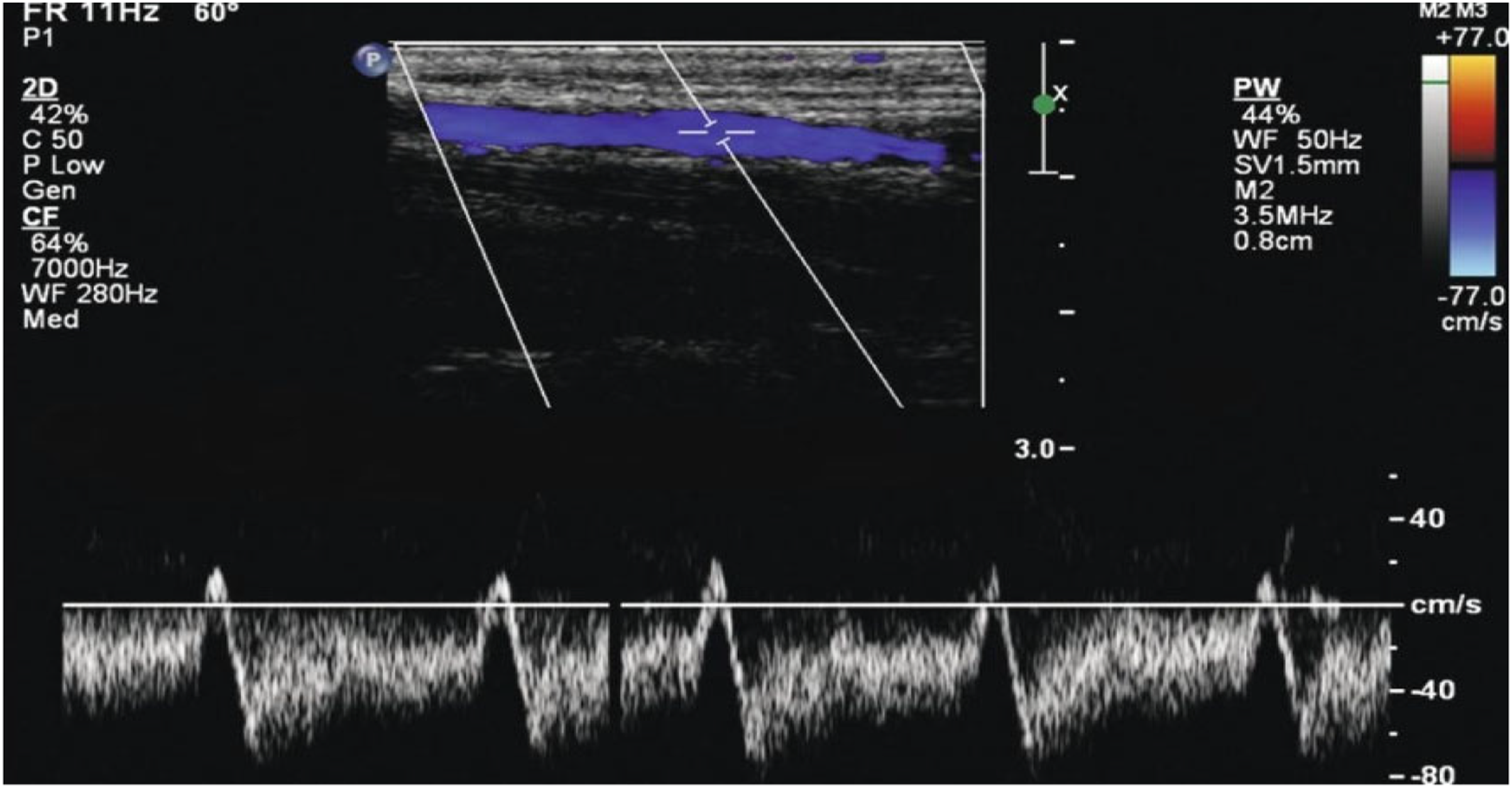
The patients were examined in a sitting position, where the limb abducted to around 90° with the arm elevated and resting on an armrest. The study protocol included storing images of the anastomosis in two views: longitudinal and transverse sections. All diameter measurements were taken using the transverse section. Also, the anastomosis diameter was documented in both longitudinal and transverse sections. Spectral Doppler was used during the protocol to determine the flow direction, particularly in an arterial segment distal to AVF anastomosis. FV and PSV were also measured using the spectral Doppler with the color Doppler technique. PSV was assessed at the anastomosis site, measuring any velocity >400 cm/s indicated stenosis. The Doppler sample was set at an angle of 60° or less to reduce the measurement error of velocities. Color Doppler was used during the protocol to assess the presence/absence of flow and aid the position of the spectral Doppler when quantifying stenoses in the arteries. To determine any proximal arterial inflow stenosis, all the proximal vessels were scanned, including subclavian, axillary, brachial, radial, and ulnar arteries. In addition, superficial veins, including Cephalic and Basilic, were scanned. The measurements were performed three times at the same site to reduce the chance of errors and avoid bias. Examples of measures of anastomosis diameter and reversal flow waveforms are shown in Figures 1 and 2. Transverse section of Anastomosis diameter measurements by using ultrasound B-mode imaging. Spectral pulse shows reversed flow in distal region of radial artery.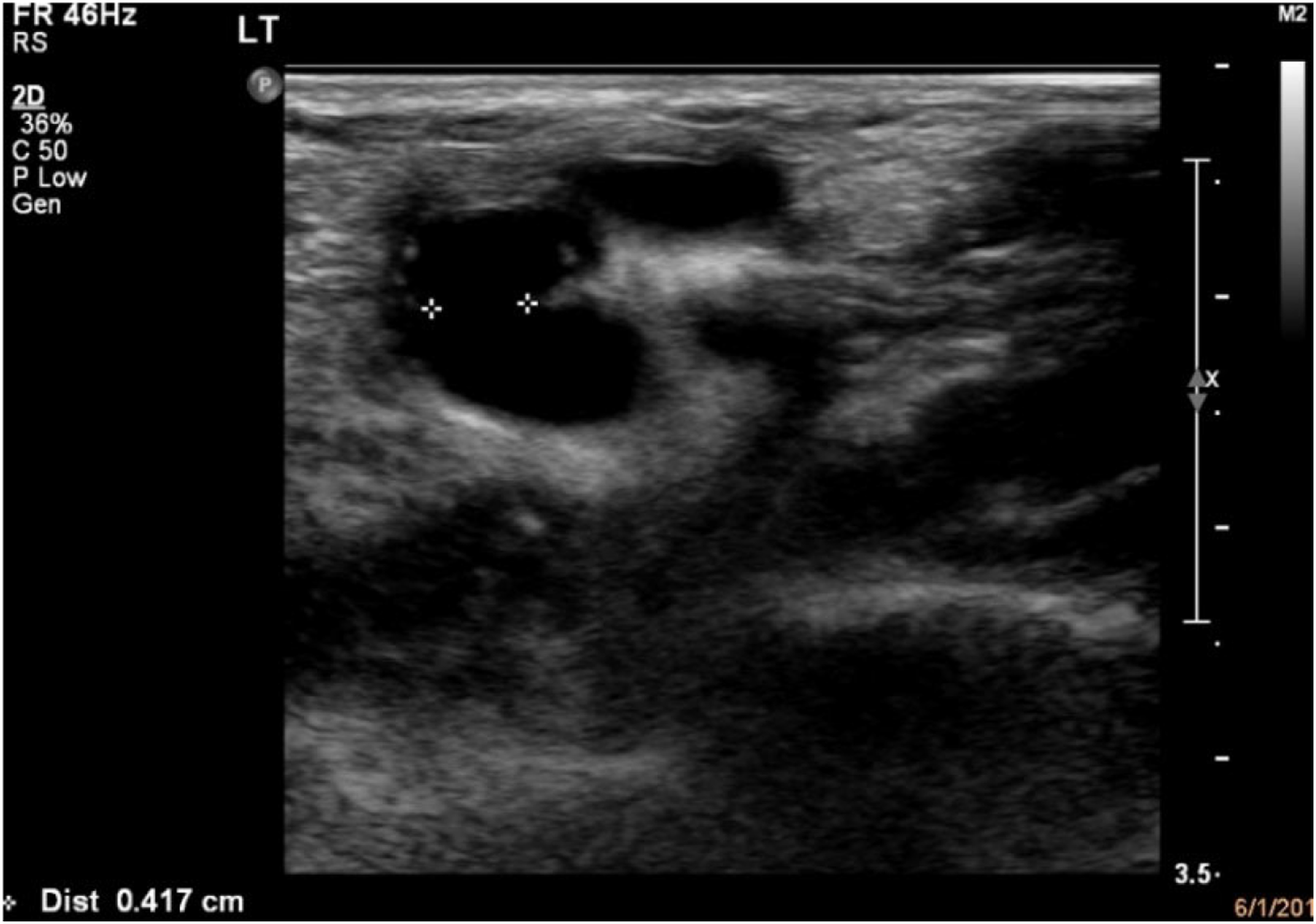
The diameter ratio was measured between the anastomosis and the arterial diameter to assess the difference and predict any signs of steal syndrome. Using the following formula, the flow volume ratio was calculated using the anastomosis diameter and the distal arterial diameter
The volume flow ratio (ml/min) was calculated using the following formula
Statistical analysis and data management
Statistical Packages for Social Science ((IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.)). The primary objective of this study was to determine the relationship between the anastomosis diameter and the steal phenomenon. Descriptive statistics for frequencies and percentages were used to present categorical data, whereas mean and standard deviation were used for continuous variables after testing the normality of data (Shapiro–Wilk test; p > .05). Student’s independent t-test was used to compare the difference between continuous variables, and any relationship between categorical variables was tested using Pearson’s chi-square (χ2) test. The independent samples t-test was performed to compare the anastomosis ratio in patients with positive steal syndrome to negative cases. Additionally, the flow volume ratio was used to compare the flow volume ratio in patients with positive steal syndrome to those with negative steal syndrome. A p-value ≤ 0.05 was considered statistically significant.
Result
Baseline characteristics of the study groups.
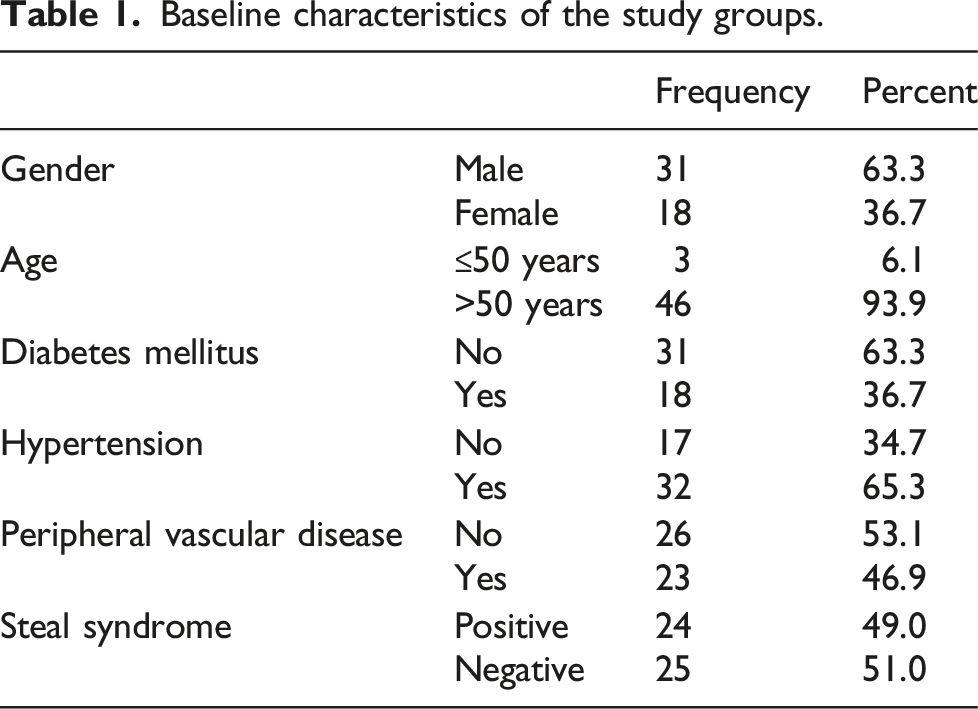
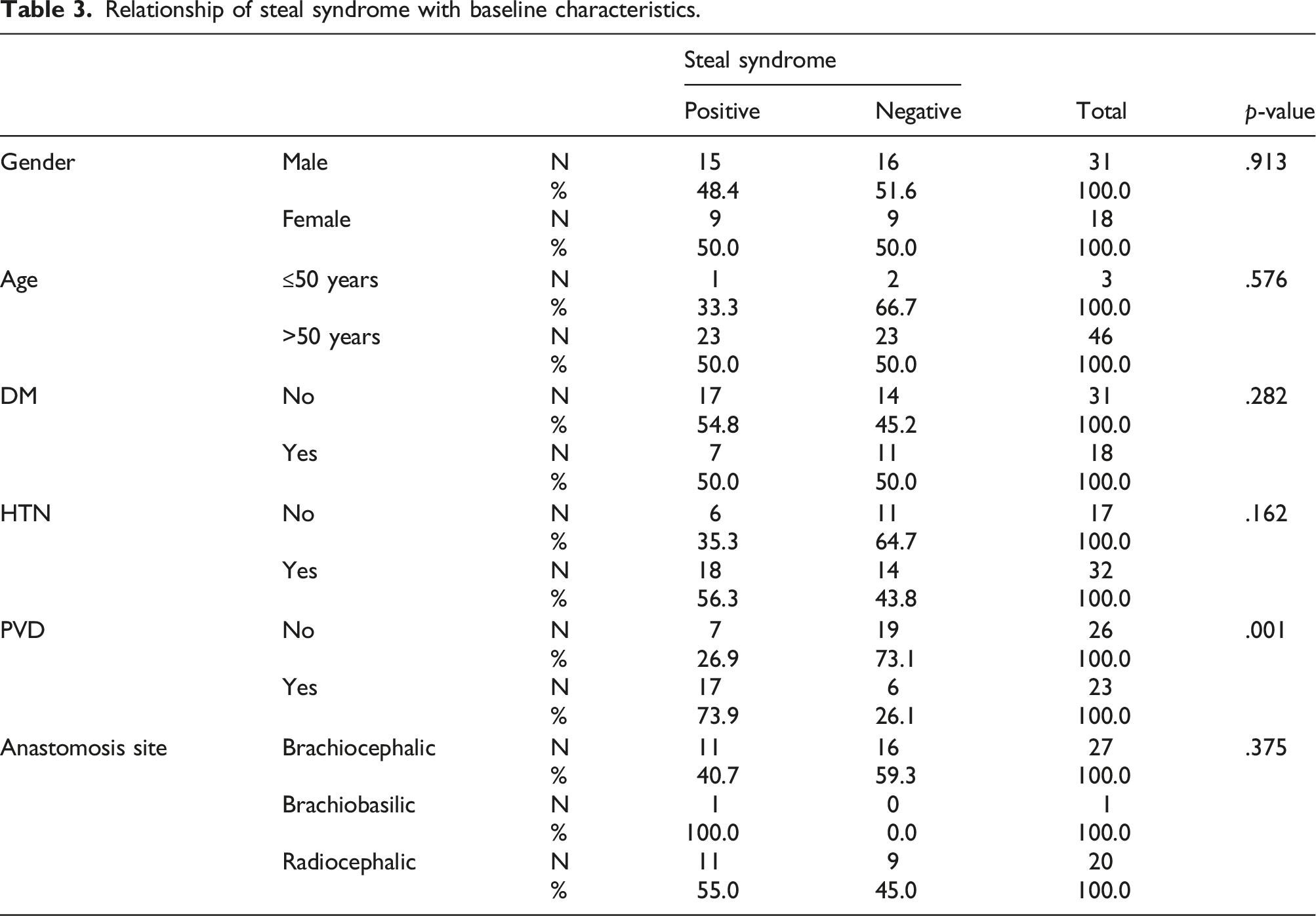
Regarding fistula anatomic configuration and symptomatic steal syndrome, our analysis revealed that 11 (55%) were radiocephalic and 11 (40.7%) were Brachiocephalic (Table 1). This study indicates that patients with a Radiocephalic fistula had commonly developed a steal syndrome.
Comparison of clinical parameters with steal syndrome.
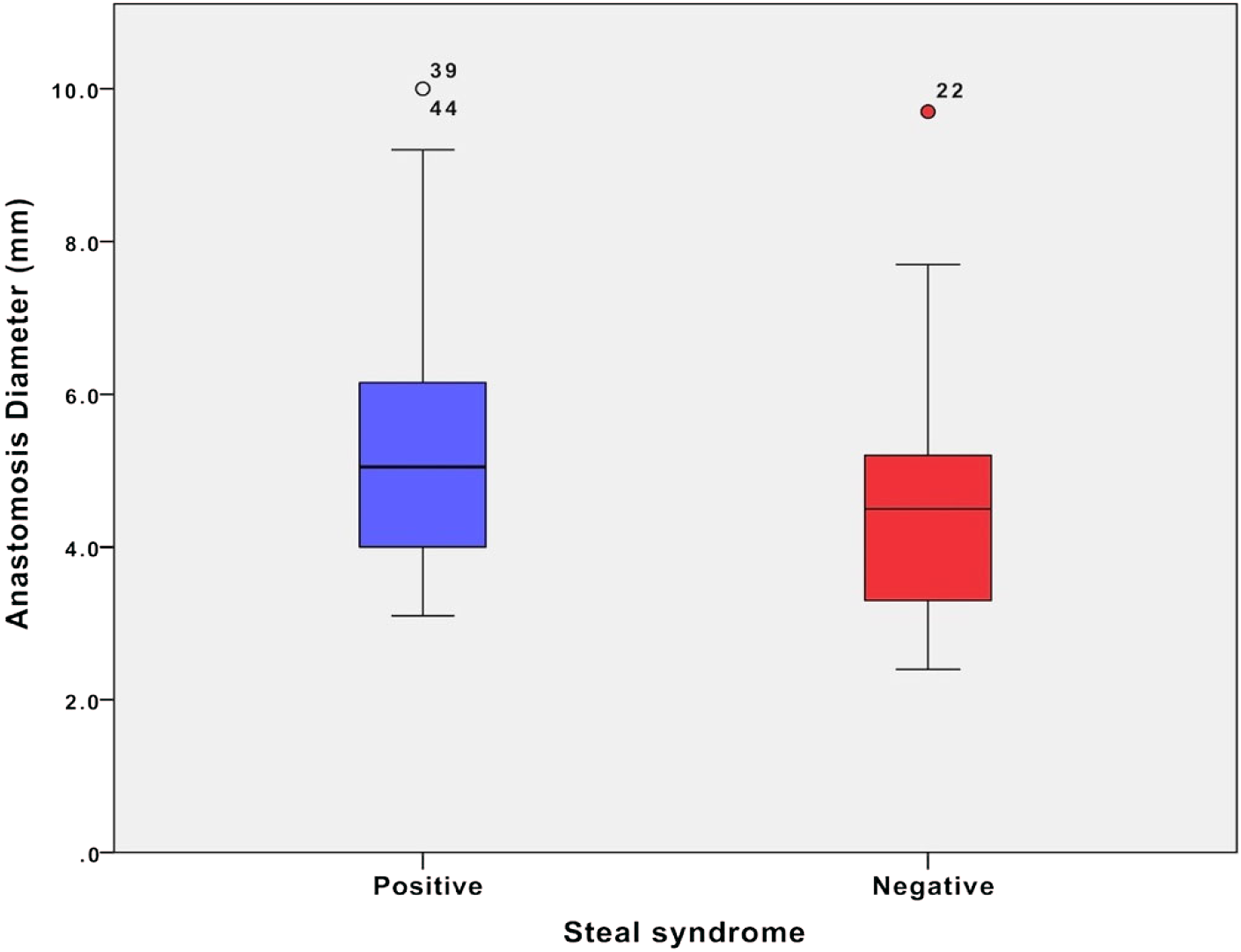
Anastomosis diameter relationship between positive and negative steal.
The mean difference in the anastomosis ratio in positive steal was 1.05 (SD ± 0.26), and negative steal was 0.8 mm (SD ± 0.24). Statistically, there was a significant difference in the anastomosis diameter ratio between the two groups, p = .001. Box plot shows a large overlapping difference between the two groups, Figure 4. Anastomosis diameter ratio relationship between positive and negative steal.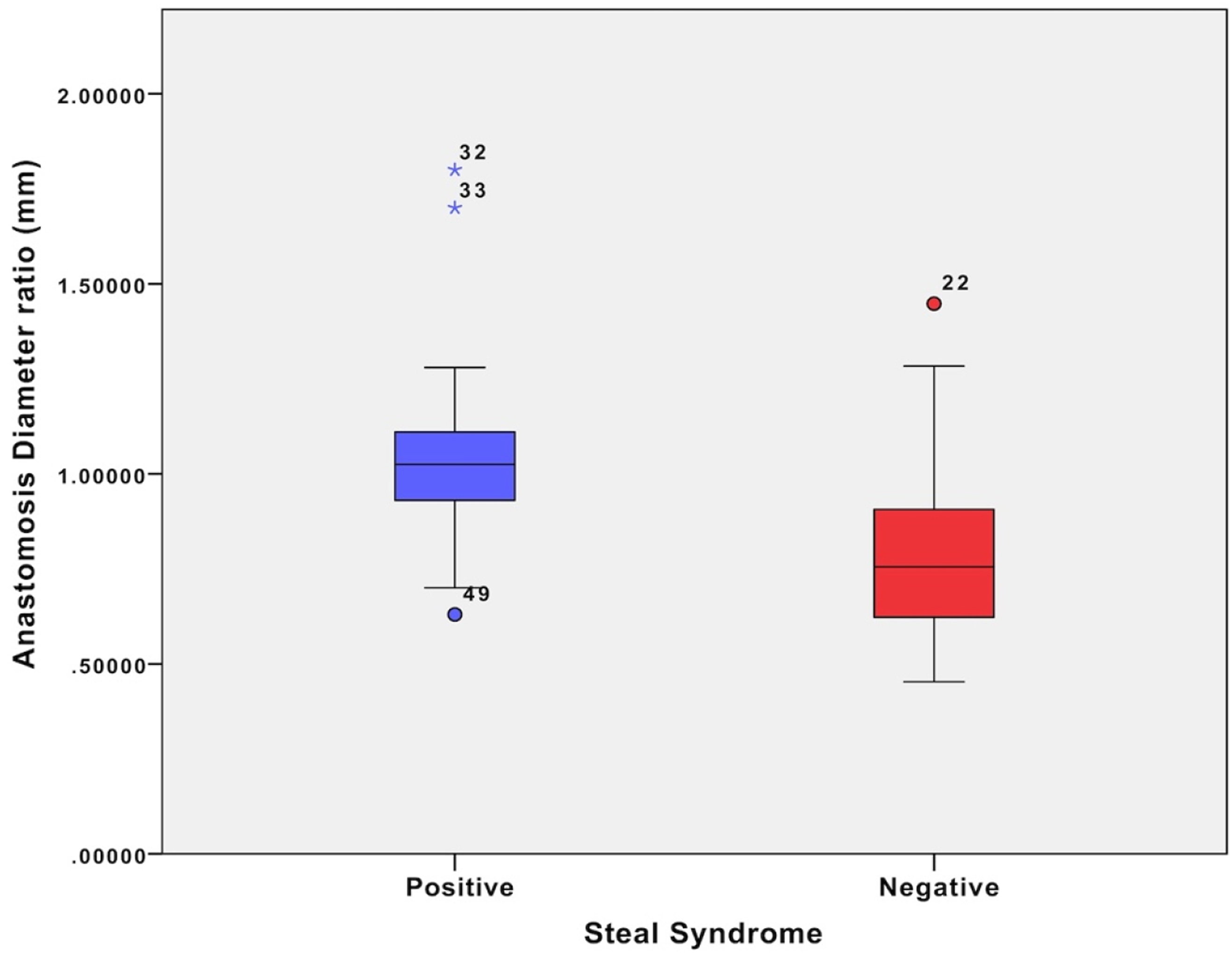
Furthermore, the flow volume ratio difference between positive and negative steal is assessed in Table 2. The mean flow volume ratio difference in positive steal was 1.29 (SD ± 1.46) and 0.753 (SD ± 0.137), p-value = .001.
Relationship of steal syndrome with baseline characteristics.
Reproducibility (interobserver) study
The level of agreement between observers using ICC. (a) The degree of absolute agreement among measurements. (b) Estimates the reliability of single ratings. (c) Estimates the reliability of averages of k ratings.

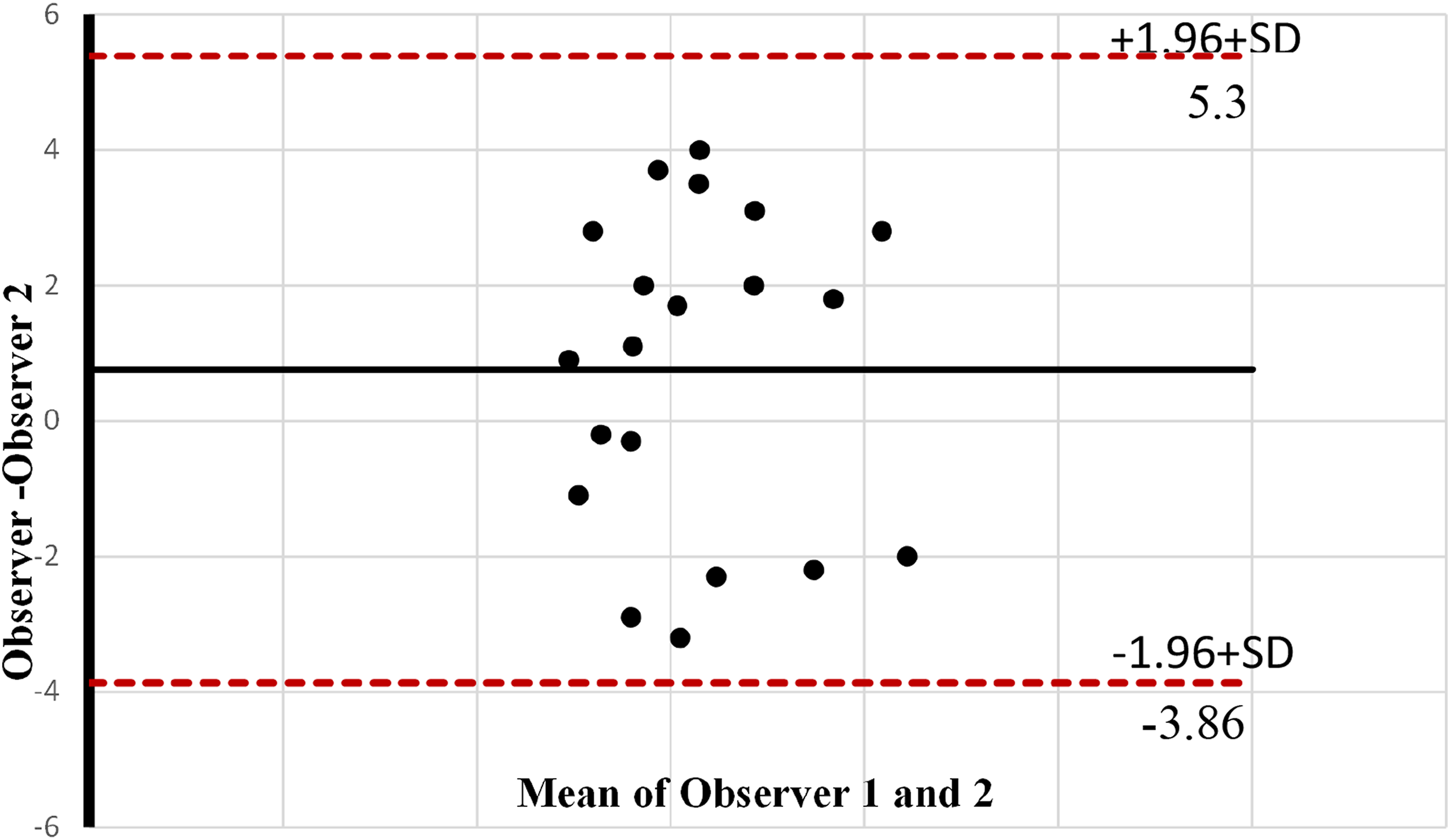
Bland–Altman graph displays the agreement level between two vascular sonographers in measuring the anastomosis diameter. Horizontal lines represent the mean difference (red dashed), and at the limits of the agreement (black Line).
Discussion
There are no previous studies investigating the association between increased anastomosis diameter and incidence of steal syndrome. The present study found a significant association between anastomosis diameter enlargement and the steal syndrome manifestation. A large anastomosis diameter would increase the risk of blood stealing through the fistula, and flow diminished distally due to reversal of flow, as illustrated on the Doppler spectral sonogram (Figure 2). Furthermore, anastomosis diameter ratio with the inflow arteries and flow volume ratio have demonstrated a considerable difference between positive and negative steals. These additional ultrasound parameters support the hypothesis of this study that increased anastomosis diameter is related to the increased incidence of distal steal syndromes.
Some previous studies suggested that a combination of or any arterial stenosis, arterial calcification, and retrograde flow would induce distal ischemia. It was also stated that a large anastomosis diameter could be one of the causes of steal syndrome.5–6,8–10 The reason is that high blood flow volume is augmented via AV anastomosis of a large vessel, especially in the elbow, where a brachiocephalic fistula or graft is used.
Steal syndrome is just one of the potential AV access complications; nevertheless, steal syndrome is relatively uncommon and occurs in 1%–20% of all fistula patients undergoing HD as a treatment for kidney failure. Although steal syndrome is rare, it’s still challenging to manage. It is essential to understand the causes of this phenomenon, the following of which have been recognized and analyzed in the literature review: lack of collaterals, proximal stenotic lesion, reduced arterial pressure, and distal arteriopathy.11–16
There was a noticeable increase in blood flow volume in the patient with positive steal syndrome. The mean flow volume in the fistula in patients with steal syndrome was 1016 mL/min, whereas the mean flow volume was 759 mL/min in patients without steal syndrome. Previous studies have noted the same findings, which showed that the mean fistula blood flow volumes in patients with steal syndrome were higher.15–21
The efferent volume flow rate is normally reported by the vascular laboratories when assessing the functionality of an AVF for hemodialysis. High volume flow rate is associated with steal syndrome, as shown in this study; however, having additional parameters such as volume ratio and anastomosis diameter ratio would provide more accurate normalized parameters by reducing the errors involved in the measurements. These parameters would be similar to other well-established ratios used in blood flow assessment studies, such as peak systolic indices and ankle brachial pressure index, known as ABI, which is a normalized ratio of ankle and brachial artery blood pressures. If validated in larger studies, these AVF ratios could become a standardized method of diagnosing steal syndrome.
Previous studies indicate that symptomatic steal syndrome is more common at the elbow when using the brachial artery and less common at the wrist.1–10 However, this study revealed that 55% of cases with the positive steal located at the wrist, and the other 40.7% were brachiocephalic.
The present study suggests that age may impact steal syndrome patients. There was a significant association between steal syndrome in elderly patients >50 years old. This was also noted in previous studies where patients above 60 years developed steal syndrome due to arterial disease, DM, and calcifications.18,19,22,23
Clinical implication
The result of this study has significant implications for predicting and preventing steal syndrome. In this study, we have used volume flow and diameter ratios to investigate the association between these parameters and the steal syndrome. The normalized ratio is in keeping with other blood flow measurement criteria, such as the peak systolic velocity ratio used for grading carotid stenosis and lower/upper limb arterial stenosis. To our knowledge, this first-time velocity ratio has been used to investigate the causes of steal syndrome in patients with an AVF.
The potential use of diameter ratio criteria would be to provide a predicted value of AVF anastomosis size prior to the fistula creation. Ultrasound can easily obtain this information during the pre-AVF arterial/venous mapping. This study shows that the probability of steal syndrome occurrence is less when the anastomosis diameter ratio is <0.8. However, this study suggests that when the anastomosis diameter ratio is >1.05, the risk of developing steal syndrome is more significant, especially in female patients who are >50 years of age and possess other risk factors, including diabetes mellitus/or PAD.
This research implies that the possibility of patients developing asymptomatic steal syndrome, mainly grades 1 and 2, may be predicted by calculating the blood flow volume ratio; when the blood flow volume ratio exceeds 0.99, the chance of steal syndrome occurring is more likely. When the ratio measures roughly around 0.75, the chance of stealing syndrome is lower. Steal syndrome following AVF creation has a great impact on patient morbidity and often requires revision of the anastomosis with a potential risk of fistula failure. Although this study has some limitations, the data shows that early prediction of diameter size for AVF creation could prevent or at least reduce the risk of developing steal syndrome.
Study limitation
The limitation of this study includes a small sample size. Another limitation includes the operator dependency; errors may arise while measuring blood volume flow, such as increasing 1 mm in diameter measurements being multiplied by a power of four. Accurate measurement of the anastomosis diameter is quite complex in patients with diffuse arterial calcification and proximal vein aneurysms. Future studies should have a larger number of participants to confirm the results of our small study. Additionally, the anastomosis angle should be investigated to see if there is any association with steal syndrome.
Conclusion
Steal syndrome is a potential complication of AVF. Early detection of steal syndrome will allow the corrective measures to be taken to prevent distal ischemia and loss of vascular access. The incidence of steal syndrome is expected to increase due to the recent increase in the population of HD patients. The study demonstrated that increasing the diameter of anastomosis leads to a rise in the risk of developing steal syndrome. For accurate assessment of the fistula, it is essential to use the anastomosis ratio and the volume flow ratio, which can be calculated to predict and prevent steal syndrome, specifically when it is asymptomatic. During the pre-assessment AVF mapping, the vascular and nephrology teams can obtain the optimum anastomosis diameter size by calculating the anastomosis ratio. The risk of developing steal syndrome is likely low when the anastomosis diameter ratio is ≤0.8. Furthermore, patients with ≥1.05 anastomosis diameter ratio have a greater chance of developing steal syndrome. Patients with a flow volume ratio ≥0.98 have a higher risk of developing steal syndrome than patients with a volume flow ratio of ≤0.75. Further studies are recommended to support this ultrasound evidence with clinical correlation.
Highlights
• Ischemic steal syndrome in patients with upper limb arteriovenous fistula access is a major complication. • Increased anastomosis diameter has an association with the incidence of steal syndrome. • Patients with DM and peripheral arterial disease are likely to develop Ischemic steal syndrome. • Anastomosis diameter and Volume flow ratios are reliable predictors for steal syndrome in AVF patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.