Abstract
Objective
This study aims to evaluate the endoprosthesis complications in patients undergoing TEVAR for blunt traumatic thoracic aortic injury, through long-term clinical and diagnostic follow-up.
Methods
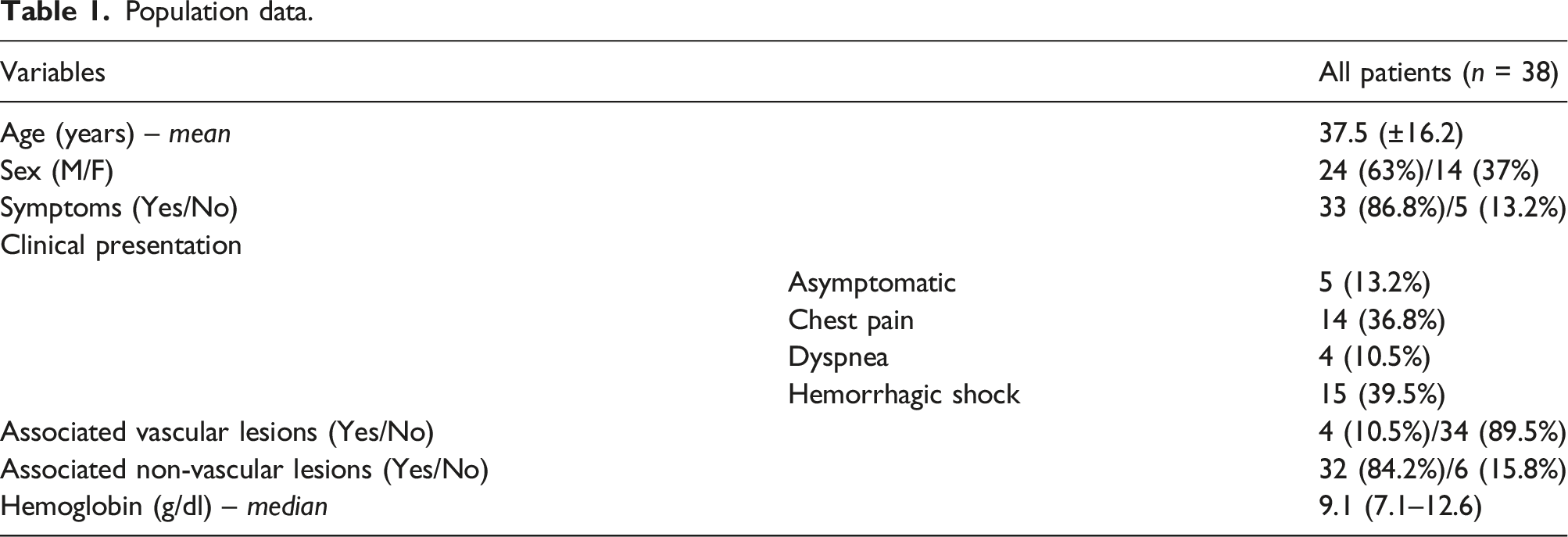
During the study interval (November 2000–October 2020), a total of 38 patients (63% male; average age 37.5 years) with thoracic aortic injury underwent thoracic endovascular aortic repair. Patients underwent routine follow-up with clinical examination and radiological evaluation (CT-angiography or MRI-angiography plus chest radiograph), scheduled at 1 month, at 6 months (only in the cases of thoracic aortic dissection), at 1 year after the procedure and every 1 year thereafter.
Results
Technical success was achieved in 38 procedures (100%). The TEVAR-related mortality rate was 0%. No immediate major complications related to the endovascular procedure were observed. The median duration of diagnostic follow-up was 80 months. A total of four procedure-related complications (10.5%) were identified at the follow-up. Three (7.9%) distal infoldings and collapses of the thoracic endoprosthesis and one (2.6%) type Ia endoleak were observed. No thrombosis of the prosthesis, nor signs of aortic pseudocoarctation were identified. No further complications related to endograft (endoleaks, infections, rupture, partial or complete thrombosis) occurred. No changes in the native aorta, stenosis, or increases in the endograft’s diameters were observed. A total of 20 patients (52.6%) underwent MRI-angiography examinations, while a total of 34 patients (89.5%) underwent chest radiographs at the follow-up. In all cases, CT-angiography examinations were performed at the follow-up.
Conclusions
Procedure-related complications were observed within one year of TEVAR, limiting concerns related to the durability of the prosthesis. No morphological changes in the aorta were observed despite long-term follow-up. The consequences of lifelong surveillance in terms of radiation exposure deserve special consideration, especially in younger patients treated for TAI.
Keywords
Background
Thoracic aortic injuries (TAIs) have been described mostly since 1900 as a consequence of road trauma. 1 Thoracic aortic injuries are defined as blunt when not caused by penetrative chest injury. To date, of all cases of blunt trauma resulting in substantial injury to the thorax, motor vehicle collisions account for a majority, followed by falls from height, pedestrian-automobile collisions, and crush injuries. Blunt TAIs occur in approximately 0.5%–2% of all nonlethal motor vehicle collisions and 10%–20% of all high-speed deceleration fatalities. About 80–90% of TAIs immediately result in the patient’s death from a grade 4 injury (rupture of aortic wall). In survivors, the lethality rate increases by 1–2% every hour afterwards and only 2% of the untreated patients survive beyond four months.1–3 Thoracic aortic injury is a “critical” injury with high mortality and an uncertain prognosis. The prognosis of the polytraumatized patients with TAI becomes worse if multiple injuries (abdominal, neurosurgical, bone, or thoracic) are associated. If possible, the patient should be referred to a trauma center. Patients need stabilization of vital functions and fast identification and treatment of the most serious and potentially fatal injuries. Computed tomography is the gold standard in the management of polytraumatized patients, if hemodynamically stable. Until a few years ago, only open surgery with extracorporeal circulation was used. This treatment is characterized by high morbidity, especially for subsequent anticoagulation therapies and coagulopathies in the polytraumatized hemorrhagic patients (pulmonary contusions, multiple fractures, limb injuries, cranial lesions, internal organs fractures such as liver, kidney, bladder etc.). In the last decade, open surgery has been replaced by endovascular treatment (TEVAR) with excellent immediate results (mortality reduction and few cases of major complications such as paraplegia, stroke or renal failure).4–6 This study aims to evaluate the endoprosthesis complications in patients undergoing TEVAR for blunt traumatic TAI, through long-term clinical and diagnostic follow-up. Computed tomography, magnetic resonance imaging, and chest radiographs have been used in the diagnostic follow-up.
Methods
Study design
This study is a multi-center (Circolo Hospital and Macchi Foundation of Varese, Italy–Mater-Domini University Hospital of Catanzaro, Italy), retrospective analysis of prospectively collected data of consecutive patients with TAI, who had undergone, from November 2000 to October 2020, thoracic endovascular aortic repair. Inclusion criteria, met by all patients, are I) blunt traumatic thoracic aortic injury (TAI); II) treatment with thoracic endovascular aortic repair (TEVAR); III) evaluation by a multidisciplinary team of vascular surgeons, interventional radiologists, and anesthetists; IV) age ≥18 years. Exclusion criteria are (I) missed clinical and radiological follow-up at 6 months after the endovascular treatment; (II) death after TEVAR of associated traumatic injuries (intracerebral hemorrhage, abdominal hemorrhage, etc.). In all cases, a computed tomography (CT) angiography and a total body CT scan (multislice CT Lightspeed Plus, GE, Milwaukee, USA; Aquilion 64, Toshiba, Tokyo, Japan) were performed after trauma. Classifications by Ishimaru and Criado7,8 were used to describe aortic anatomy, defining the five possible landing zones of the thoracic aorta during the endovascular repair. The study was approved by the Institutional Review Board and an informed consent was obtained by each patient to allow a retrospective data analysis for research purposes.
Treatment
The endovascular procedure was performed in dedicated angiographic suites. A retrograde approach with surgical exposure of the common femoral artery has been preferred. In the cases of iliac arteries obstruction or small vessel diameter, a vascular access by common iliac artery or abdominal aorta was used. In all cases, general anesthesia was performed with cardio-respiratory monitoring and short-term antibiotic prophylaxis (cefazoline 2g or vancomycin 1g). Fluoroscopic monitoring in anterior left-angled projection was performed during the release of the prostheses. Various models of endografts were used (Medtronic Talent, Medtronic Valiant, Medtronic Valiant Captivia, Gore Tag, Gore Excluder, Gore C-Tag, and Cook Zenith TX2). We have overestimated the diameter of the prosthesis by 20% compared to CT-angiography measurements. A controlled hypotension (90 mm/hg) was maintained during procedures. After endoprosthesis’ positioning, we assessed the subclavian artery’s revascularization by Doppler-US examination to exclude any sign of ischemia. Post-procedural complications (incomplete endoprosthesis’ sealing or endoleaks) were immediately treated and, if necessary, left subclavian artery (LSA) embolization were performed. Associated vascular lesions, like traumatic splenic injury, were treated simultaneously to ensure the best treatment and prognosis. A dual anti-platelet therapy (in most cases, acetylsalicylic acid 100 mg/day plus clopidogrel 75 mg/day) has been administered at least for 1 year after endovascular treatment.
Follow-up
Patients underwent routine follow-up with clinical examination and radiological evaluation (CT-angiography or MRI-angiography plus chest radiograph), scheduled at 1 month, at 6 months (only in the cases of thoracic aortic dissection), at 1 year after the procedure and every 1 year thereafter, according to recommendations of Society for Vascular Surgery. 9 A different schedule was used according to previous recommendations by other international societies. 10 In all cases, a clinical and radiological follow-up was performed every year after endovascular treatment.
Statistical analysis
Data were maintained in an Excel spreadsheet (Microsoft Inc, Redmond, Wash) and the statistical analyses were performed on a per-protocol basis, using SPSS software (SPSS, version 22 for Windows; SPSS Inc, Chicago IL, USA). Kolmogorov–Smirnov test and Shapiro–Wilk test were used to verify the normality assumption of data. Categorical data are presented as frequency (percentage value). Continuous normally distributed data are presented as mean ± standard deviation. Continuous not normally distributed data are presented as median (interquartile range: 25th and 75th percentiles—IQR). A p-value of <0.05 was considered statistically significant for the aforementioned tests.
Results
During the study interval (November 2000–October 2020), a total of 38 patients (63% male; average age 37.5 years) with TAI underwent thoracic endovascular aortic repair. One patient was excluded from the study because he had a <6 months follow-up and two patients were excluded from the study because they died after treatment of associated traumatic injuries.
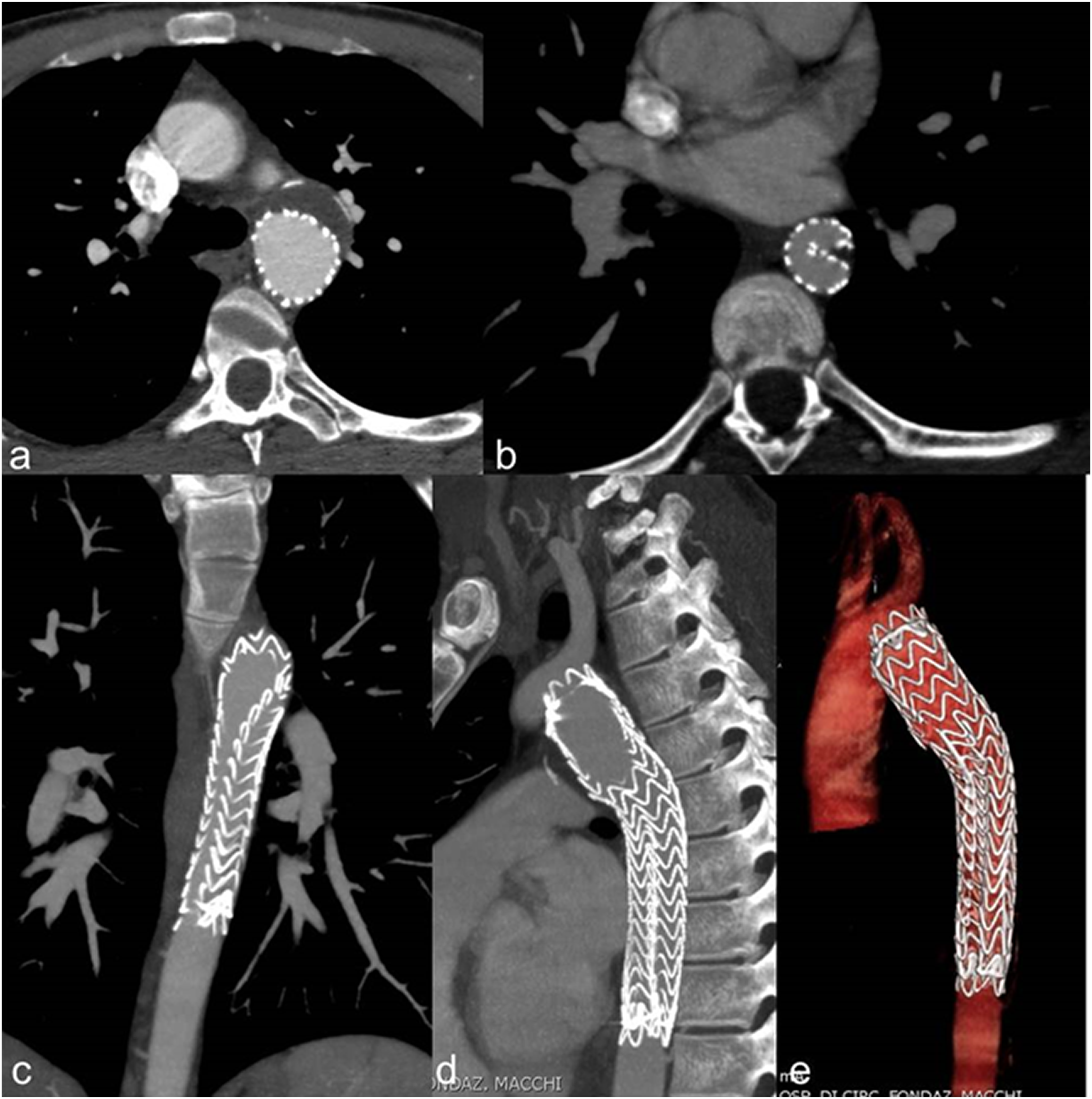
Thirty-two of the 38 patients underwent diagnostic CT immediately after the trauma, while 6 underwent CT during the weeks/months after. The other 6 patients had unrecognized post-traumatic aortic lesions: one of them underwent a CT scan for the onset of recent chest pain, while in the other five cases patients were asymptomatic (Figures 1 and 2). The clinical presentation was chest pain in 14 patients (36.8%), dyspnea in four patients (10.5%) and hemorrhagic shock in 15 patients (39.5%). 10.5% of the patients had associated vascular lesions, while 84.2% of the patients had associated non-vascular lesions. The median value of hemoglobin count at clinical onset was 9.1 g/dl. Population data are reported in Table 1. First case-polytrauma patient (2011) with an incidental finding in 2012 of a chronic PSA. CT angiography with axial images (a, b) showing voluminous chronic PSA at the isthmus level, in the presence of parietal lamellar calcifications. Sagittal MIP, (c) and 3D (d) reconstructions, plus angiography (e), confirm voluminous chronic isthmic PSA. Advancement of the endoprosthesis (f). Angiography demonstrates the exclusion of the PSA and the correct positioning with the regular expansion of the endoprosthesis in the descending aorta (g). First case-polytrauma patient. Follow-up with CT angiography at 6 months (asymptomatic patient). Axial images (a) and 3D (b), paracoronal MIP (c) and sagittal MIP (d) reconstructions highlight a partial collapse of the endograft in its most distal portion. Population data.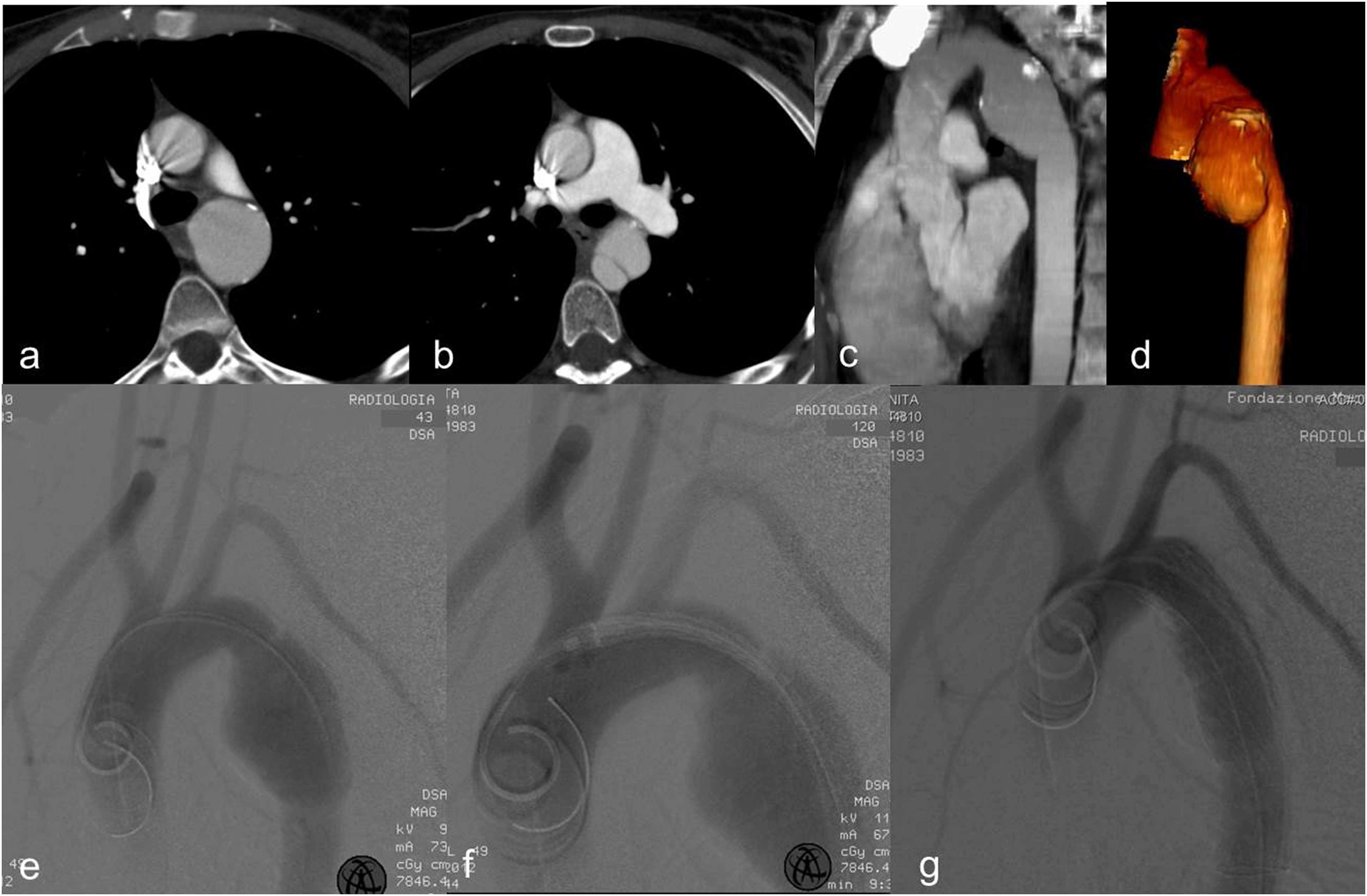
Lesion data.

Endovascular procedure data.

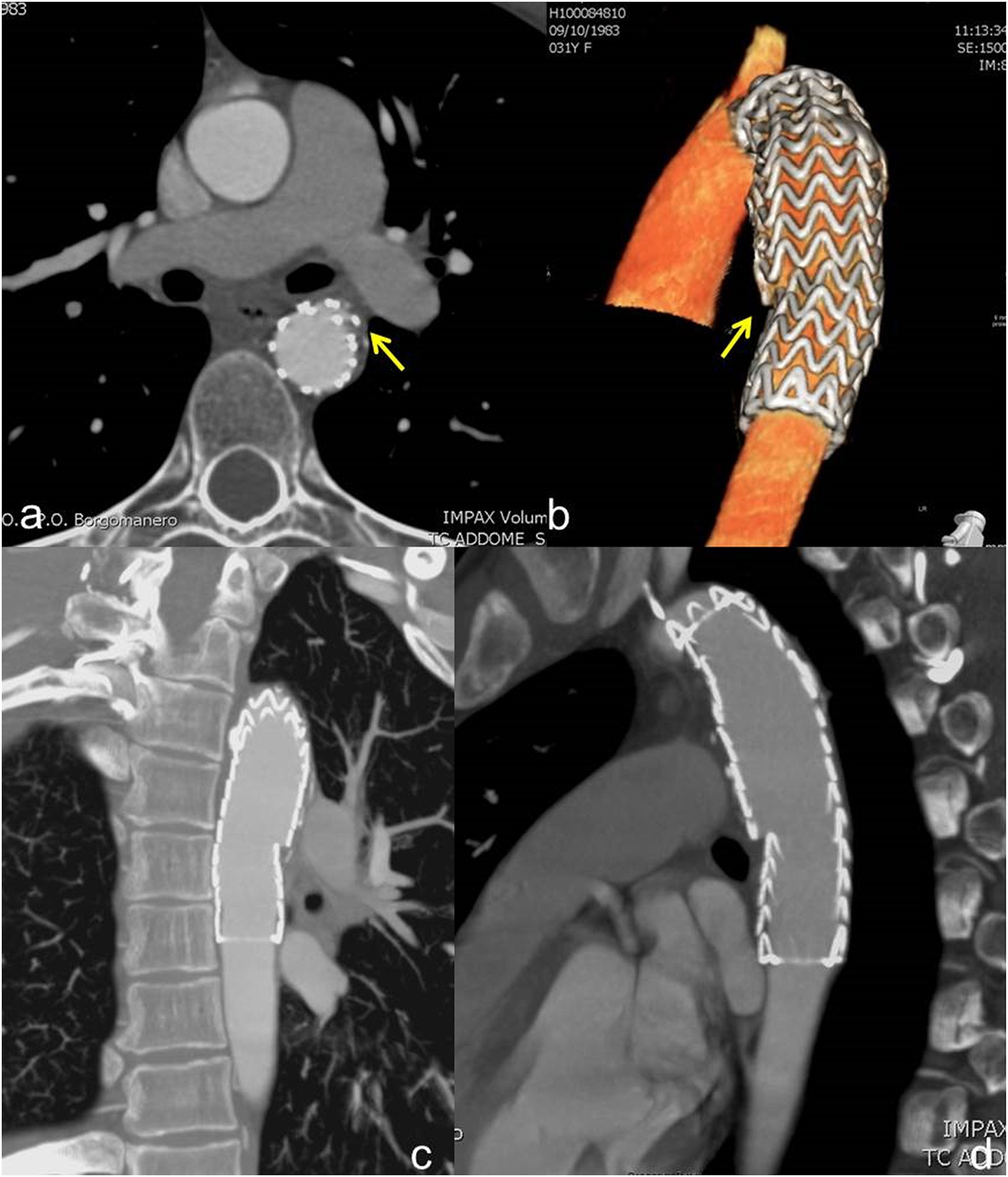
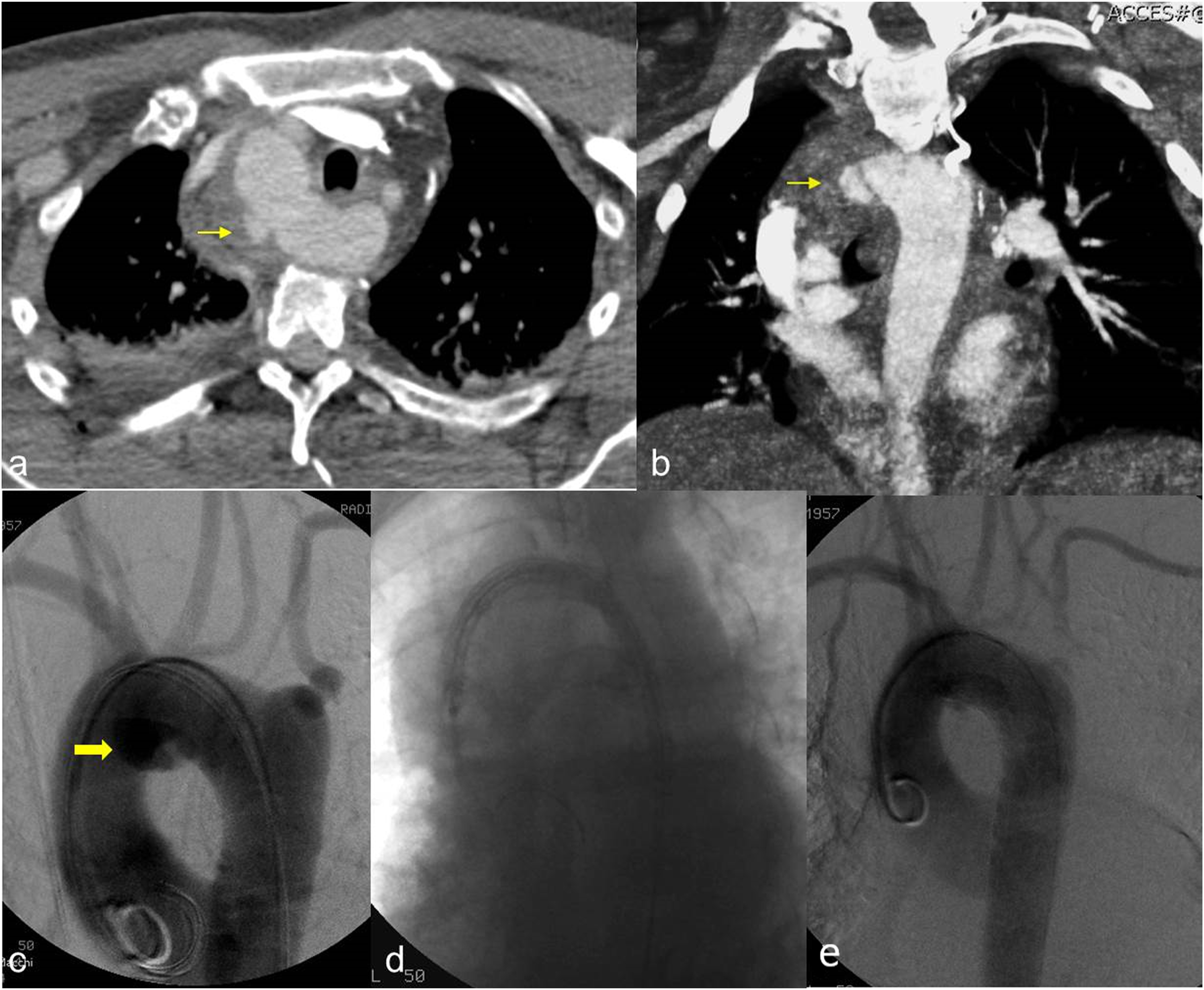
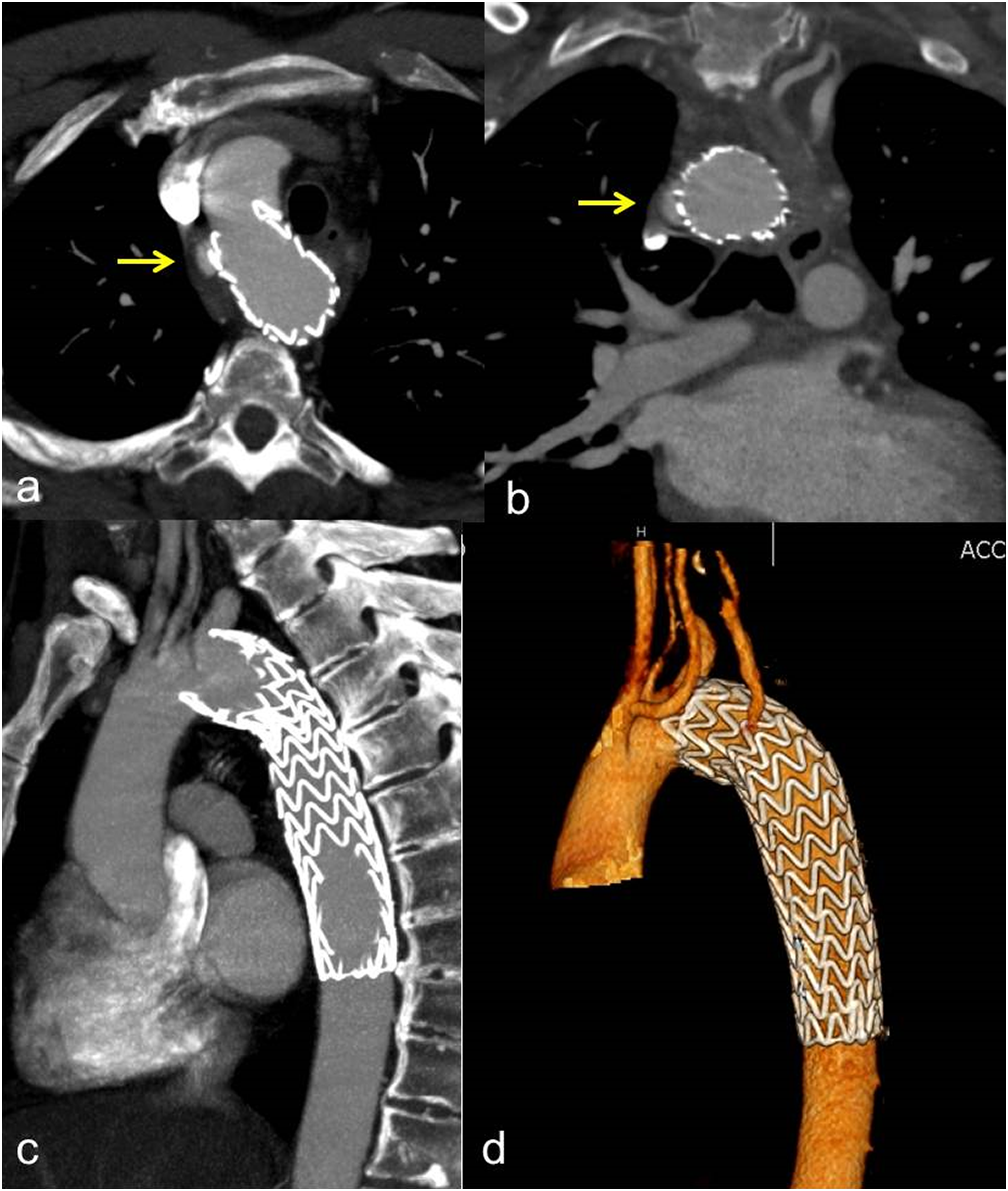
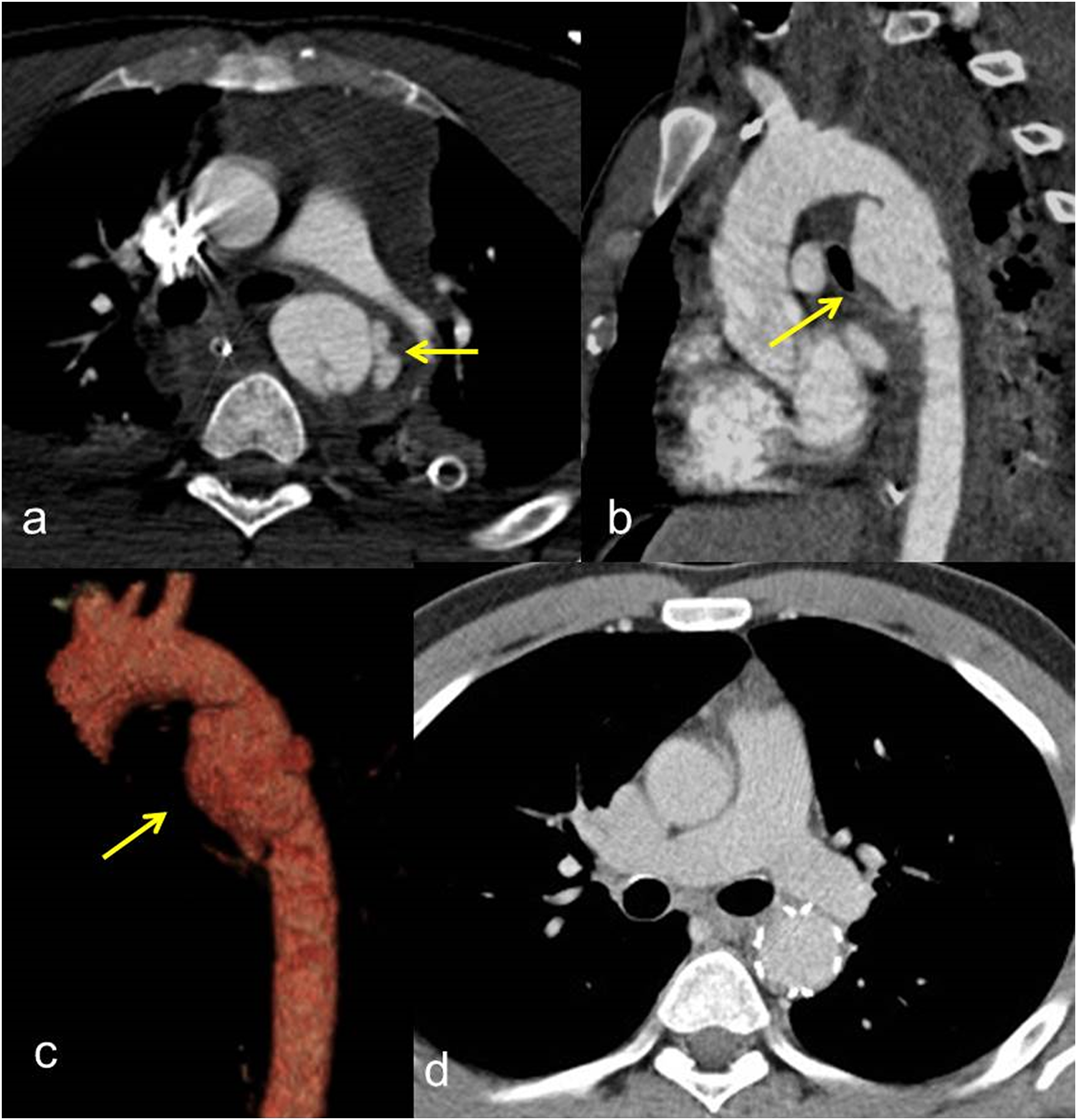
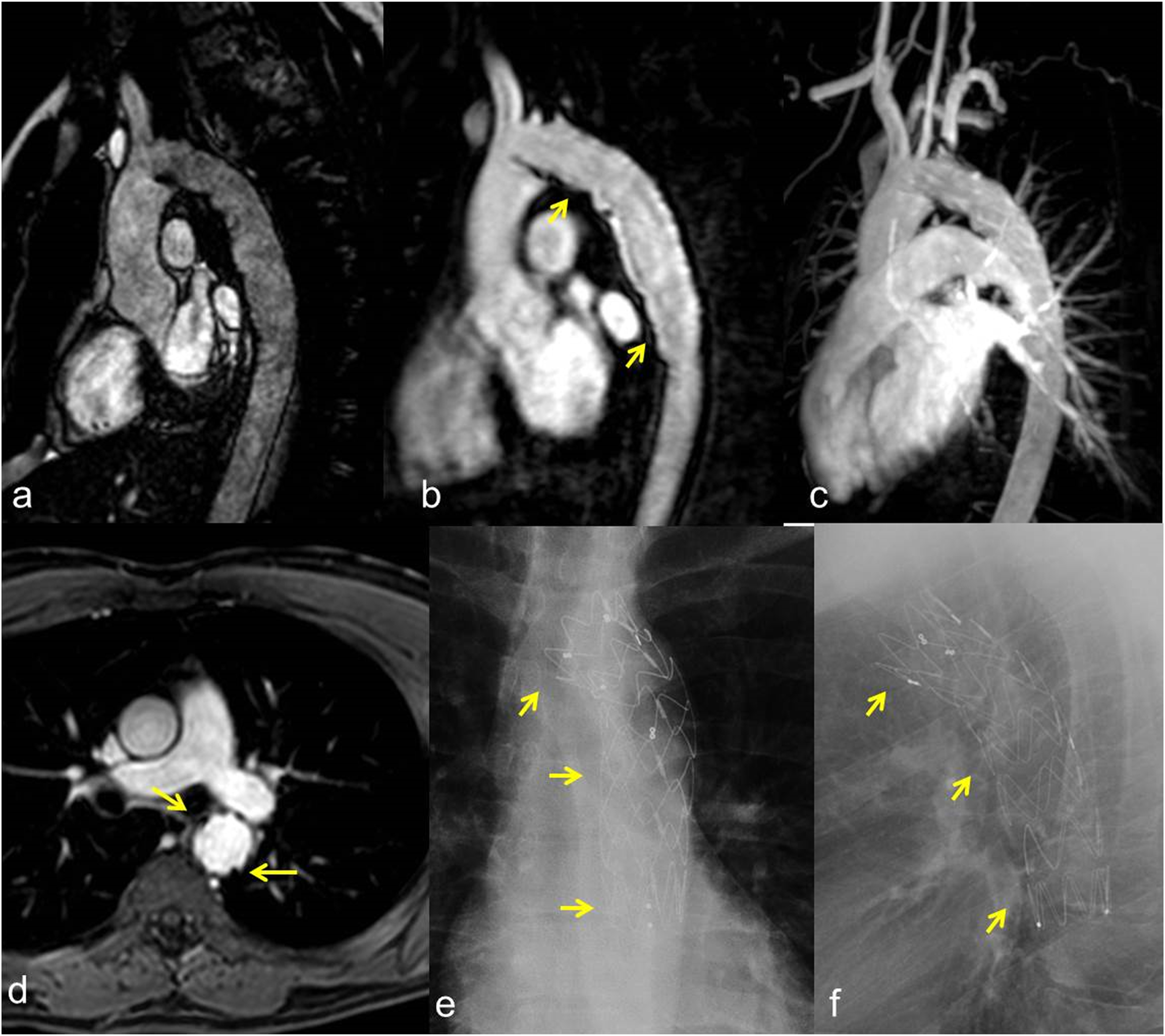
Technical success was achieved in 38 procedures (100%). The TEVAR-related mortality rate was 0%. No immediate major complications related to the endovascular procedure (such as malposition, dislocation, aortic, or iliac–femoral axis laceration) were observed. The median duration of diagnostic follow-up was 80 months (IQR, 68.2 months). In all cases, the aortic hematoma and hemothorax were completely solved. A total of four procedure-related complications (10.5%) were identified at the follow-up. Three (7.9%) distal infoldings and collapses (Figures 3 and 4) of the thoracic endoprosthesis and one (2.6%) type Ia endoleak were observed. No thrombosis of the prosthesis, nor signs of aortic pseudocoarctation were identified. One of the collapses occurred 1 month after the procedure, while the second and the third were identified at the 6-months and at the 1-year follow-up. Type Ia endoleak was identified at the 6-months follow-up (Figures 5 and 6). No further complications related to endograft (endoleaks, infections, rupture, partial, or complete thrombosis) occurred. No changes in the native aorta, stenosis, or increases in the endograft’s diameters were observed. A total of 20 patients (52.6%) underwent MRI-angiography examinations, while a total of 34 patients (89.5%) underwent chest radiographs at the follow-up (Figures 7 and 8). In all cases, CT-angiography examinations were performed at the follow-up. All patients discharged from the hospital were still alive at the time of their last follow-up. Outcomes and follow-up data are detailed in Table 4. Second case-polytrauma patient. CT angiography (a, b) demonstrates Psa at the isthmic level, with associated hemomediastinum. Angiography (c) demonstrates the correct positioning of the endoprosthesis which appears regularly expanded. CT angiography at 1 month after TEVAR (D, E) shows correct expansion and patency of the prosthesis, which in the most cranial images presents a partial collapse despite being open. Second case-polytrauma patient. Follow-up with CT angiography at 12 months (asymptomatic patient). Axial images (a, b) confirm the correct expansion and patency of the prosthesis except for a partial collapse of the prosthesis in its most caudal portion. Paracoronal (c) and sagittal (d) MIP reconstruction, plus 3D reconstruction (E), demonstrate the collapse of the prosthesis in the descending aorta. Third case-polytrauma patient. CT angiography with axial images (a) and paracoronal MIP reconstruction (b), plus angiography (c), showing an acute PSA (arrow) at the concavity of a right-sided aortic arch. Advancement of the endoprosthesis (d). Angiography demonstrates the exclusion of the PSA and the correct positioning with the regular expansion of the endoprosthesis in the descending aorta; the left subclavian artery was intentionally covered (e). Third case-polytrauma patient. Follow-up with CT angiography at 6 months (asymptomatic patient). Axial images (a) and MIP paracoronal reconstruction (b) showing a millimeter type IA Endoleak (arrow). Parasagittal MIP (c) and 3D (d) reconstructions confirm the correct positioning with the regular expansion of the endoprosthesis. Fourth case-polytrauma patient. CT angiography with axial images (a) and parasagittal MIP (b) and 3D (c) reconstructions highlighting an acute PSA (arrow) at the aortic isthmus. Follow-up with CT angiography at 60 months demonstrates the exclusion of the PSA and the correct positioning with the regular expansion of the endoprosthesis in the descending aorta (d). Fourth case-polytrauma patient. Follow-up with MR angiography plus chest radiograph at 72 months. TRUFI sagittal localization sequence (a). 3D Flash MR angiography sequence (b) and MR angiography reconstruction (c) showing the correct positioning and the regular patency of the endoprosthesis (arrows) in the descending aorta. T1 axial post-contrast sequence (d) demonstrates the regular expansion of the endograft in the absence of endoleaks (arrows). Chest radiograph with PA (e) and lateral (f) projections highlight the complete expansion of the endoprosthesis and the regular morphology of the endoprosthetic meshes (arrows). Outcomes and follow-up data.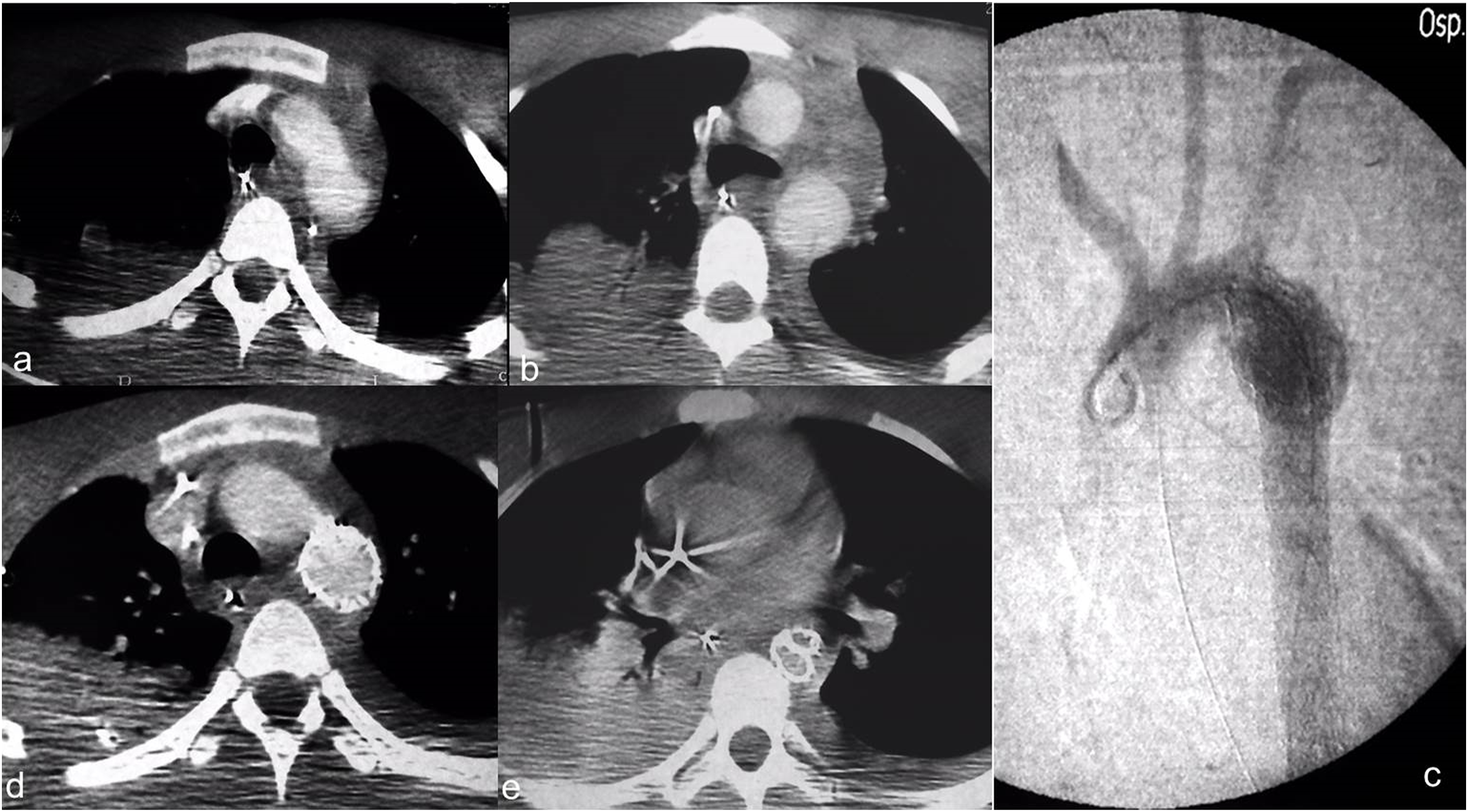

Discussion
In the last twenty years, the endovascular treatment of TAI became the treatment of choice compared to “open” surgery, as it reduces mortality and morbidity.10-13 Currently, there are studies with a small number of cases because the incidence of TAI is low, except for a few multicenter studies or work performed by trauma centers, which describe acute complications with medium-term follow-up after TEVAR. TEVAR for TAI treatment is feasible and safe also in patients with associated lesions and in young patients. Long-term results are scarce in the literature, but it is equally true that the late complications’ rate is relatively low.12,14–18 Therefore, the most important findings of our study are the length of long-term follow-up and the absence of significant morphological changes of the native aorta determined by mechanical forces exerted by the endograft. The most recent guidelines5,9,10 recognize that TEVAR for TAI is associated with better immediate technical results than traditional surgery, especially immediately after the procedure, where survival is the main purpose of the intervention. However, the long-term durability and efficiency of this less invasive treatment over open surgical repair remain uncertain. Besides, a recent systematic review found no RCTs conducted to determine whether the use of TEVAR for the treatment of blunt TAI is associated with reduced mortality and morbidity when compared to conventional open repair. 19 Another comprehensive evidence is from a systemic review by Murad et al., 20 which was based on 27 comparative observational nonrandomized studies with very low-quality evidence suggesting that, compared with the open repair or nonoperative management, endovascular repair of thoracic aortic transection is associated with better survival. There are two negative aspects related to the long-term follow-up of TEVAR for TAI. The first concern is the limited number of reported cases with long-term follow-up and the second is the low participation of patients in the follow-up.11,15,21–28 Furthermore, only a few of these studies specify the participation of patients in follow-ups with clinical evaluation and CT angiography. Instead, our study presents a collection of consecutive cases with long-term follow-up and with a low rate of withdrawal thanks to the strong adherence to scheduled checkups. These are two key points because literature data enlightens that it’s difficult for authors to continue obtaining data from patients (only 30–65% of patients continue follow-up).25, 26 In our study, none of the patients came from a catchment area far from our centers. This logistical aspect and the patient recruitment capacity may have been the two decisive factors in achieving this result.
A long-term comparison, in terms of clinical outcome, between TEVAR and surgery is still absent. Patel et al. 25 reported that the risk of treatment failure was higher in the TEVAR group, but this had been determined only by re-treatment that occurred in the first year; after that, no long-term complications were observed. A retreatment rate of 5.8% was estimated in patients with a follow-up lasting longer than 3 years after treatment with TEVAR. This rate is higher than that of surgical treatments but is lower than that of TEVARs for non-traumatic diseases.11,15,16,25–28 Endovascular treatment in these patients had excellent results and low mortality. 25 In our series, we have recorded a low rate of late complications. Because we had a wide range of endografts, we were able to treat virtually all diameters of the native aorta. Furthermore, hypovolemic shock affects the diameter of the descending aorta in trauma victims; therefore, the choice of the endograft was made taking this aspect into account. Prosthetic collapses were not treated because patients were clinically asymptomatic; at CT examination the prostheses presented a slight collapse in the most distal part, while they were open and well expanded in the proximal tract. The aortic tracts distal to prostheses were regularly opened without signs of ischemia of the splanchnic circle and lower limbs. The type I-A endoleak was not treated for the small size and the unfavorable anatomy of the aortic arch; monitoring over time with close follow-up was performed without evidence of progression.
Given that TEVAR is no longer restricted to a cohort of older patients, the consequences of lifelong surveillance deserve special consideration, especially in younger patients treated for TAI.9,29,30 As stated in the recommendations of the Society for Vascular Surgery, 9 it will be necessary to show that the benefits of surveillance after TEVAR are justified by the risks and costs over time. Complications prevail in the short term and include unsuccessful exclusion and endograft infolding. However, long-term device behavior is still uncertain so that surveillance regimens in those patients may be relaxed yet not completely stopped. 31 According to recommendations of the Society for Vascular Surgery 9 and previous recommendations by other international societies, 10 radiological follow-up was performed with yearly lifelong CT-angiography. In young patients, follow-up made by CT is worrying about the annual cumulative exposure of ionizing radiation. A single CT scan of the chest exposes the patient to approximately 7 mSv of ionizing radiation. 32 A single exposure to 10 mSv of ionizing radiation causes cancer in one patient every thousand. In a 40-year-old patient (life expectancy of at least 30 years) who underwent annual chest CT follow-up, the cumulative dose will be approximately 210 mSv; this would significantly increase the risk of developing malignant conditions such as cancer and leukaemia. 33 Hence, MRI has been proposed as a valid alternative in patients with endografts compatible with the magnetic field, associated with a chest X-ray. Rasche et al. 34 evaluated 20 patients who underwent an endovascular procedure for TAI with CT and MRI. In this study, MRI performed for the evaluation of endoprostheses (MR-compatible) showed the presence of signal artefacts, up to complete signal voids, as the main limitation. Another limitation is the lack of long-term follow-up and complications. The authors concluded that MRI can give a global assessment of the complications of the thoracic endoprosthesis and proposed MRI as an “imaging” examination to identify complications related to TEVAR. However, given the limitations of a poor long-term follow-up period in the study, MRI was not proposed as a standard, but as a valid alternative. MRI plus chest radiograph can highlight endoleaks, alterations in the structure of the endoprosthesis and its dislocation over time.5,10,11
Knowledge of the long-term behavior of devices is the main cause for concern, preventing the interruption of surveillance regimes. Morphological changes of the aorta due to age can still occur over the years, resulting in endograft-related complications.35,36 In our experience, we have not observed aortic morphological changes with a median follow-up duration of 80 months. In light of these late results, TEVAR may be considered effective and long-lasting.
Among TEVAR complications, the main ones described are the collapse and the poor positioning of the endograft resulting in endoleak. Our experience is in agreement with literature data, as we have found three endograft collapses and one endoleak during follow-up, resulting in 10.5% of procedure-related complications. The two main factors associated with an increased risk of long-term procedure-related complications are the mismatch between the endograft and aortic diameters and the bird-beak configuration, defined as a wedge-shaped gap between the undersurface of a thoracic endograft and the lesser curvature of the arch.35,37 Furthermore, in our case series patients with the aforementioned procedure-related complications were clinically asymptomatic, like that of Jonker’s series. 36
A classification scheme for grading the severity of the aortic injury has been proposed by Azizzadeh. 38 Clinical practice guidelines of the Society for Vascular Surgery for the endovascular repair of traumatic thoracic aortic injury 5 suggest expectant management with serial imaging for type I injuries. Riesenman et al. 26 opted for medical non-operative management in three cases with first degree lesions, but a pseudoaneurysm developed six months after the follow-up (one-third of cases). Marone et al. 16 delayed four stable injury treatments, but then all TAIs were treated when patients showed signs of instability. More recent data show that TEVAR remains equivalent in outcomes to medical management for minimal injuries. 39 The optimal management for minimal blunt traumatic TAI remains controversial. In our case history, we have immediately treated four cases of type I TAI in patients with severe chest pain. This approach can be criticized because it exposes the patient to the risks related to the endograft; however, complications related to the endograft are low and the purpose of treatment is survival, despite recent literature data would have modified our strategy. Further evidence is needed to establish the best treatment for type I TAI.
Limitations of the study are the lack of a control group to compare long-term outcomes of the open surgical repair, the retrospectivity of the analysis and the scarcity of data in the literature, necessary to evaluate the congruence and the consistency of the data presented.
Conclusions
Thoracic endovascular repair (TEVAR) is a safe and effective treatment for blunt traumatic thoracic aortic injury (TAI). Procedure-related complications were observed within one year of TEVAR, limiting concerns related to the durability of the prosthesis. No morphological changes in the aorta were observed despite long-term follow-up. The consequences of lifelong surveillance in terms of radiation exposure deserve special consideration, especially in younger patients treated for TAI, making MR-angiography plus chest radiograph a possible alternative to CT-angiography that needs to be tested in further comparative studies.
Footnotes
Author contributions
RM and DL conceived the study concepts and design. RM, DL, RS, MUB, MP, and GC performed literature research and assisted in data collection. RM, DL, and AMI were in charge of manuscript editing. MP performed the statistical analysis using the provided database. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent to participate
Written informed consent for patient information and images to be published was provided by the patients or a legally authorized representative.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
