Abstract
Purpose
To evaluate the method of thrombin injection under B-flow and ultrasound guidance (BUGTI) for the treatment of pseudoaneurysms.
Materials and methods
Twenty-one patients suffering from pseudoaneurysm (PSA) were retrospectively reviewed at the First Affiliated Hospital of Nanjing Medical University in Nanjing, China, from January 2018 to August 2019. The patients were treated using an ultrasound-guided injection of thrombin (500 IU/mL) combined with B-mode blood flow imaging (B-flow). The information on the PSA, including the size of the arterial rupture and sac, flow rate, thrombin dose, and treatment outcome, was recorded during the procedure. Follow-up evaluation was performed at 1, 3, and 6 months after the treatment. Pearson’s correlation analysis was performed among the characteristics of PSA and the dose of thrombin.
Result
The age of patients ranged from 34 to 80 years and averaged 62.8 years. The maximum cross-sectional area of PSA ranged from 208 to 1148 mm2. All patients were treated with thrombin injections. The dose of thrombin ranged from 300 to 1667 IU. No reperfusions were detected at follow-up 6 months, and the BUGTI treatment was successful in all 21 cases. Pearson’s correlation analysis demonstrated that the dose of thrombin was positively correlated with the width (r = 0.449, p < .05) and maximum cross-sectional area (r = 0.504, p < .05) of PSA.
Conclusion
Thrombin injection under B-flow and ultrasound guidance is a rapid and effective treatment for PSA. Additionally, the sac size could be used to estimate the dose of thrombin.
Introduction
Pseudoaneurysm (PSA) is a common complication of transarterial interventional procedures. 1 The puncture of the femoral artery is among the most frequent approaches. 2 Therefore, the incidence of femoral artery PSA becomes increasingly frequent with the widespread use of endovascular interventions. 3 Traditionally, PSA is treated by surgical therapy or local compression. 4 Although the surgery is effective, it has drawbacks of repeated trauma, pain, and other complications such as wound infection, nerve damage,5,6 and lymphatic leaks. 7 Besides, the treatment via local compression is often long, and its effect seems limited. 8 In some cases, the PSA sac becomes enlarged, 3 increasing the difficulty of operation during a subsequent treatment. Ultrasound-guided thrombin injection (UGTI) has become the most effective treatment for PSA. UGTI could reduce the patient’s trauma.6,8-10 However, in some cases, thromboembolic complications or recurrence of PSA after UGTI have been reported.11-13 Therefore, the objective of the present study was to determine the clinical value of UGTI combined with B-mode blood flow imaging (B-flow) in the treatment of PSA.
Methods
This study was approved by the institutional review board of the First Affiliated Hospital of Nanjing Medical University in Nanjing, China. Medical records of 21 patients admitted to the hospital from January 2018 to August 2019 were retrospectively reviewed. These patients were hospitalized due to local pain or palpable pulsatile mass in the inguinal region after an interventional puncture of the femoral artery. All subjects were diagnosed with PSA by clinical examination and ultrasound imaging. Patients with arteriovenous fistula and other vascular lesions such as multiple puncture injuries were excluded before the UGTI treatment, and digital subtraction angiography (DSA) was performed if the vascular lesion could not be confirmed. If the treatment with UGTI was feasible, appropriate preoperative assessments such as coagulation function and the distal blood supply of the affected limb were checked. For patients without a hemorrhagic tendency, the pts antiplatelets/anticoagulants were used as usual. Patients were instructed about the nature and risks of the PSA treatment and signed the informed consent.
Medication
A single-dose, 2000 IU, of lyophilized bovine thrombin (Changchun Leiyunshang Pharmaceutical Co., Ltd., Changchun, Jilin province, China) was dissolved in 4 mL of physiological saline to form a 500 IU/ml thrombin solution. The preparation was drawn into a 5 mL syringe.
Instrument and equipment
The patients were examined using a color ultrasound diagnostic instrument (GE LOGIQ E9, Chicago, IL, United States) equipped with an ML6-15 probe (6–15 MHz), 9L probe (5.0–9.0 MHz), and C1-6VN probe (3.0–5.0 MHz).
Surgical procedures
Patients were laid in the supine position with the inguinal area fully exposed and the affected limb straightened. Before the procedure, B-mode and color Doppler flow imaging (CDFI), pulsed-wave Doppler (PW), and B-flow ultrasound (US) were routinely performed to determine the location of PSA and the rupture. The information on PSA, including width and length of the arterial rupture and sac, the flow rate of the rupture, thrombin dose, treatment outcome, and the distance from the skin, was documented. The appropriate needle approach was selected and confirmed. After skin disinfection and draping, the sac was punctured with a 5 mL syringe under US guidance. When the needle tip reached the appropriate position within the sac, a probe was used to press the sac to decrease or stop blood flow in the PSA. The tip of the needle was close to the rupture. Thrombin solution was then pulsed injection into PSA until it was filled with thrombus. During the operation, thrombin was injected dynamically according to the real-time ultrasound imaging. The entire process was observed using B-flow. The pressurized probe was slowly released during the formation of the thrombus. The syringe was withdrawn when blood flow in the sac had stopped, as confirmed by CDFI and B-Flow. After the operation, B-flow and CDFI were again used to determine the absence of blood flow signal in PSA and the pulsation in the dorsal artery of the foot was present. The patients were required to stay in bed for 1–2 days. It should be mentioned that the inguinal area could not be compressed after the operation. Follow-up US was performed at 1, 3, and 6 months after the operation.
Statistical analysis
Pearson’s correlation analysis among thrombin dose, the size of the sac, arterial rupture width, sinus tract length, and maximum flow rate of the rupture was analyzed. All data were analyzed using the SPSS software.
Ethics approval
The study was approved by the Ethics Committee of The First Affiliated Hospital, Nanjing Medical University (Ethical approval number: 2020-SR-240).
Consent to participate
All patients signed the written informed consent.
Results
Patient characteristics.
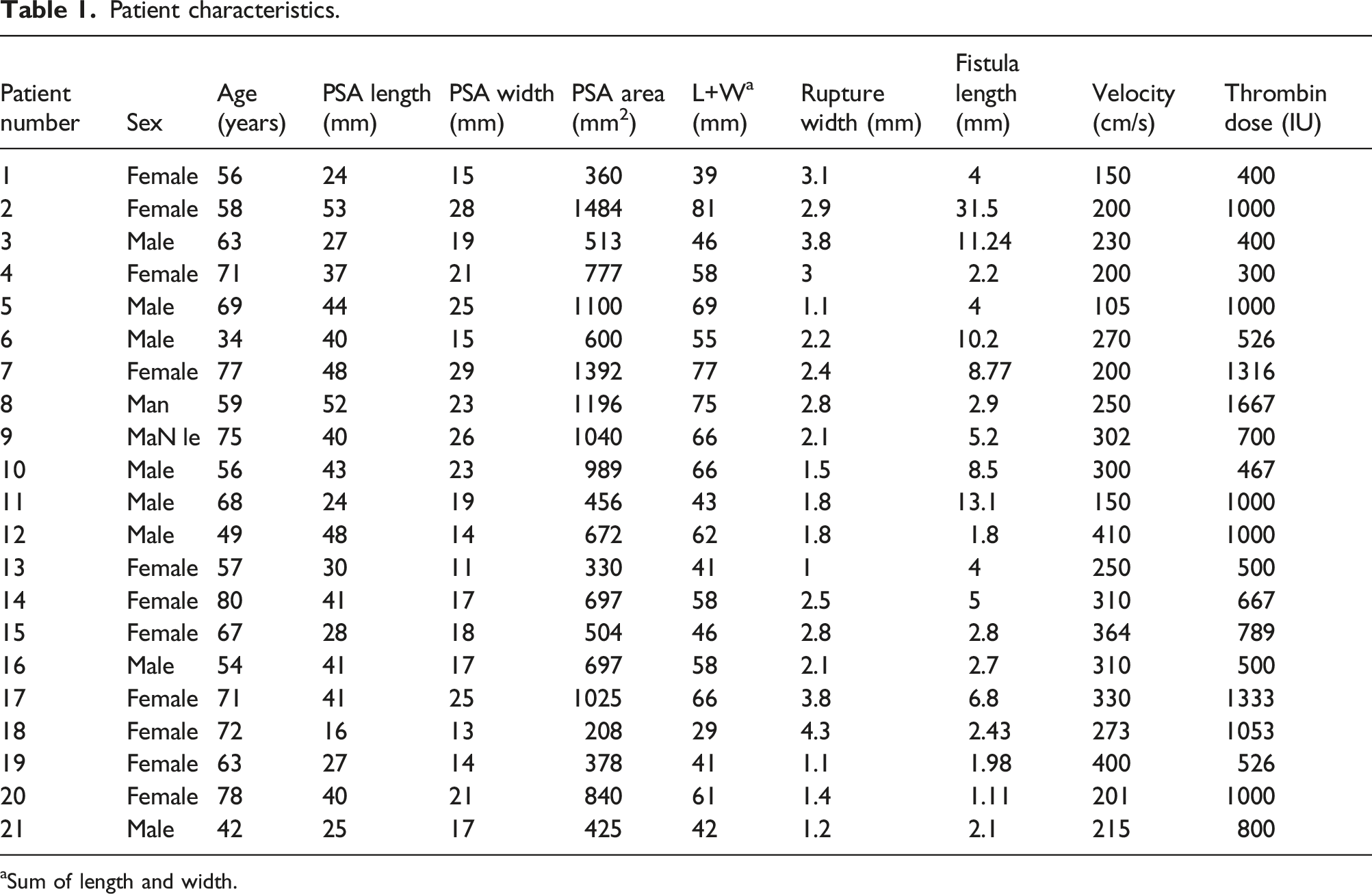
aSum of length and width.
All patients had a history of interventional surgery: 12 patients underwent radiofrequency ablation due to arrhythmia; one underwent coronary angiography; one underwent subclavian artery stent implantation; one underwent coronary angiography, coronary atherectomy and percutaneous coronary intervention; one underwent coronary angiography, percutaneous coronary intervention and intravascular ultrasound; one underwent coronary angiography and renal arteriography; one underwent electrophysiology examination and cardiac catheter radiofrequency ablation; one underwent renal artery sympathetic radiofrequency ablation; one underwent renal artery ablation; one underwent intracranial aneurysm embolization. After excluding the risk of bleeding, the patients were routinely treated with anticoagulant and antiplatelet drugs after the operation. Three patients had clinical symptoms at the puncture site 3 weeks after the operation, one showed symptoms after approximately 1 week, and 17 presented symptoms 1–2 days after the operation. After the diagnosis of PSA, patients were treated by thrombin injection under B-flow and ultrasound guidance (BUGTI). The PSA rupture in 16 cases was located in the right superficial femoral artery; 3 cases were in the right common femoral artery; and 2 cases were in the left superficial femoral artery.
One of the patients was an elderly male who underwent radiofrequency ablation due to atrial flutter and 2 days later experienced pain at the puncture site. Physical examination identified a pulsating mass in the left groin area and accompanying ecchymosis. The US detected a 40.2 × 19.5 mm anechoic mass in this region (Figure 1(a)). The mass was connected to the superficial femoral artery and had a rupture width of 2.1 mm. “Red-blue” blood flow signals in the mass were found by CDFI (Figure 1(b)). Additionally, PW identified the positive and negative bidirectional arterial spectrum at the rupture. The medical history, clinical manifestations, and ultrasound examination indicated a formation of the left superficial femoral artery PSA. B-mode ultrasonic image visualized an anechoic mass with mural thrombus in the inguinal region (Figure 1(a)), and CDFI showed the color blood flow signal at the rupture (Figure 1(b)). Due to the interference from a mural thrombus and angle dependence of the Doppler effect, the blood flow signal in the PSA could not be displayed entirely in the same section. However, it was a lack of angle dependence in B-flow. In B-flow images (Figure 1(c)), the blood flow signals of the rupture and PSA could be detected in the same section. In the meantime, B-Flow could not suffer from blooming or wall overwriting as CDFI. Thrombin was injected under the guidance of B-flow and US. The B-mode and B-flow images of PSA during this procedure are illustrated in Figure 1(d) and (e), respectively. After the operation, the B-mode ultrasound image showed that the mass was filled with hypoechoic thrombus (Figure 1(f)). Blood flow could not be detected in the mass by CDFI (Figure 1(g)) and B-flow (Figure 1(h)). Moreover, the pulsation of the distal artery was adequate. Two of the 21 patients were treated by medical compression because the attending clinician did not have experience in UGTI. One week after compression, the US examination detected a few thrombus in the PSA. In one of the two patients, the sac size of PSA decreased from 48 x 12 mm2 to 30 x 11 mm2. However, the symptoms were not alleviated. In the other patient, the sac size of the PSA enlarged from 18 x 12 mm2 to 27 x 14 mm2. After the BUGTI therapy, the blood flow in the PSA stopped, and the patient had no discomfort. Ultrasound images of PSA before, during, and after the operation. (a), (b), and (c): the B-mode, CDFI, and B-flow images before the procedure, respectively; (d) and (e): B-mode and B-flow image during the operation, respectively; (f), (g), and (h): the B-mode, CDFI, and B-flow images after the procedure.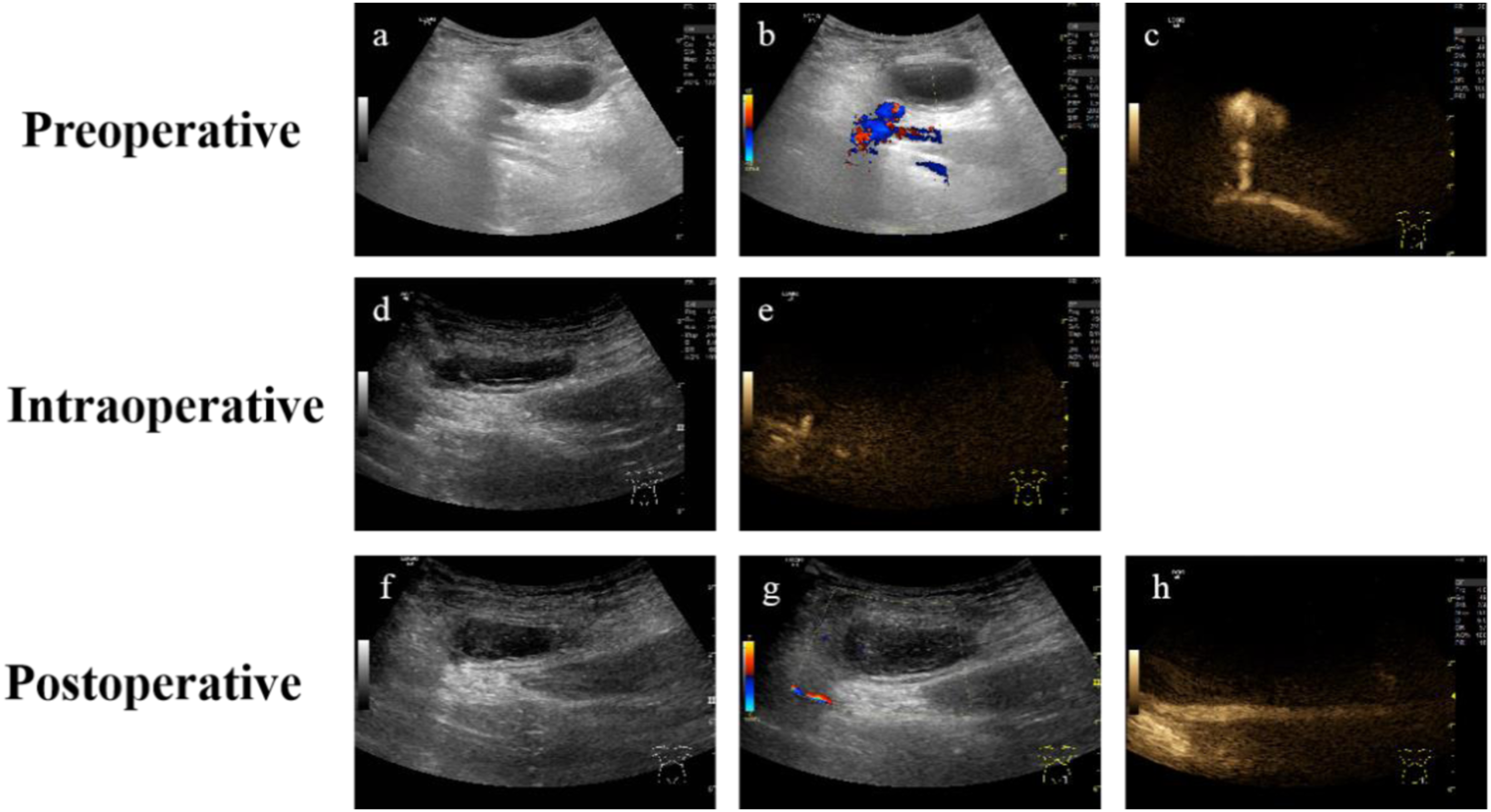
During the review, it was found that the pseudoaneurysm of all 21 cases after the operation presented a hypoechoic mass with no blood flow signal, and the bilateral dorsal pedis artery and symmetrical skin temperature of the lower extremities were normal. At the third-month follow-up, all these sacs are full of thromboses, and the size of the sac shrinks more than in the beginning. There was no incidence of recurrent PSA during the 6-months follow-up, and the thrombus mass was absorbed gradually to disappear. Thus, the treatment of PSA by BUGTI was successful in all 21 cases.
Correlation coefficients (r) among the length, width, and area of the PSA, diameter of rupture site, fistula length, flow rate, and thrombin dose.
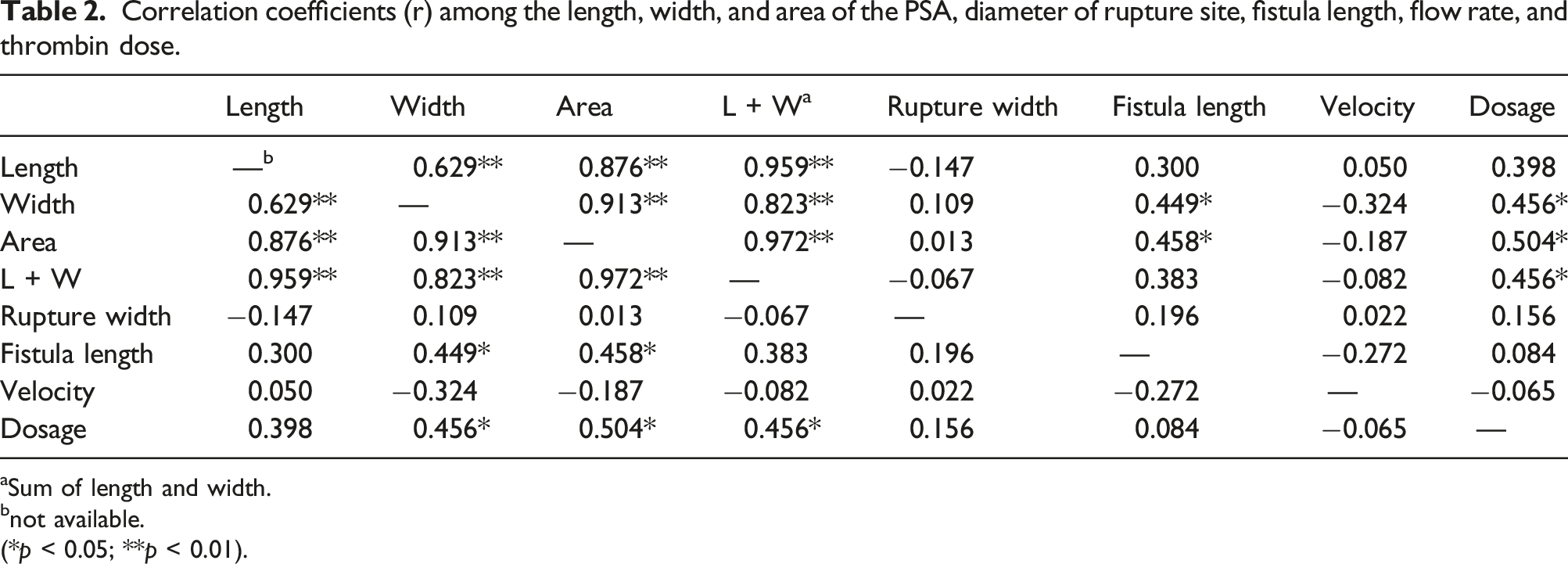
aSum of length and width.
bnot available.
(*p < 0.05; **p < 0.01).
Discussion and conclusions
PSA, known as a false aneurysm, develops from a defect in the wall of the artery, which causes constant communication between the artery and the adjacent cavity.1,14 PSA may be pulsatile like a true aneurysm, but it does not have a three-layer structure consisting of the adventitia, middle-layer elastic fibers, and intima in contrast to a true aneurysm. 15 PSA is a frequent complication of radiologic and cardiologic catheterizations. Its high incidence will likely continue to increase due to several factors, such as the increased number of interventional surgery procedures, longer operations, and larger sheath sizes (7F or higher) used.2,16
Most of the patients in the current study showed ecchymosis at the punctured site. Pulsatile masses could be palpated, and the systolic murmur was present. A hypoechoic sac in the vicinity of the impaired artery was identified by B-mode US imaging, and red-blue turbulent blood flow signals could be seen by CDFI. Moreover, swirling high-resistance flow and “to and fro”-type wave could be detected by PW in the neck or close to the neck.
3
Besides, the bloodstream ejected into the mass from the artery was visible by B-flow (Figure 2). Compressing the proximal artery could reduce the size and tension of the mass. Moreover, the murmur of blood vessels was relieved. The features of PSA detected by CDFI (a), PW (b), and B-flow (c).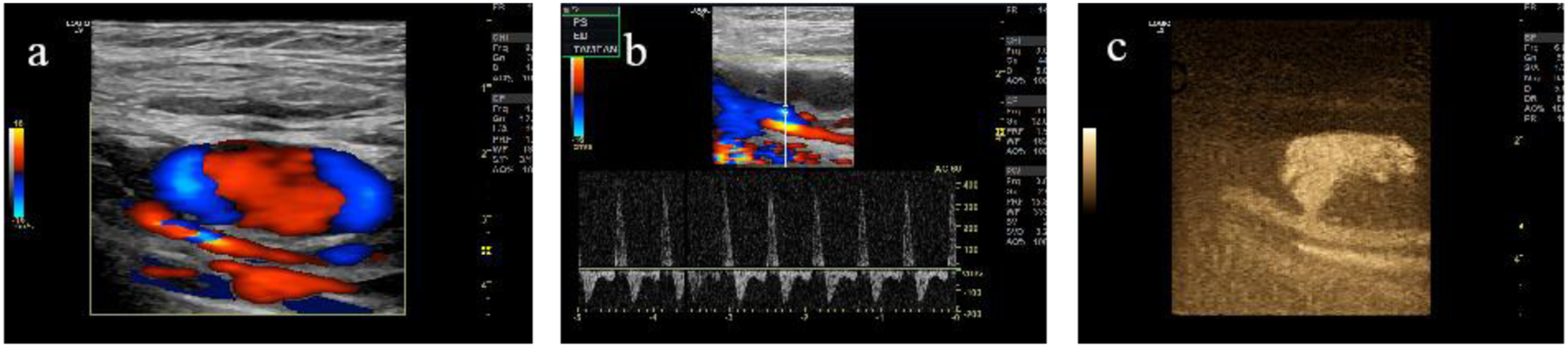
A giant aneurysm may cause compression syndrome in adjacent nerves and ischemia of remote tissues.17,18 Due to low-velocity blood flow, mural thrombosis often develops. Mural thrombus migration may lead to the embolism of distal arteries and induce corresponding symptoms. PSA may also rupture and bleeding due to trauma or increased internal pressure. 19 Therefore, once PSA appears, it should be treated promptly to prevent the severe disease.
Currently, three treatment options for iatrogenic femoral artery PSA are available, inlcuding local compression, surgical intervention and UGTI. Local compression is considered safe and effective, but prolonged local compression always leads to pain and discomfort. Therefore, local compression often fails when patients could not tolerate the pain. Besides, lengthy compression may result in the necrosis of local soft tissue and venous thrombosis. The therapeutic outcome of local compression for PSA located near the external iliac artery or for a large or complex PSA is not ideal. Surgical treatments of femoral arterial PSA, such as stent-graft repair, coil embolization and resection, could be employed.8,20 However, surgical treatments are expensive and traumatic. UGTI is among the most effective methods for the treatment of PSA and has the advantage of generating less trauma and producing the desired curative effects at a low cost. The success rate of UGTI in the treatment of PSA is as high as 95–100%, 21 which is significantly higher than that of compression alone (74–93%). 22 However, in some cases, thromboembolic complications or secondary recurrence may develop after the UGTI treatment.
B-flow represents a novel technology for diagnosing vascular diseases using digitally encoded ultrasound, which is independent of angle and is less affected by the presence of gas. 23 Given the high resolution and high frame frequency used in B-flow, the echo signal from the tissue and blood flow could be used to generate an image. The information on blood flow, mural thrombus, and the relationship between the vascular wall and surrounding tissues could be displayed. Therefore, B-flow has significant advantages in diagnosing vascular lesions.24,25 In the area of PSA with a high flow velocity (such as artery rupture), the color bleed artifact is often present in the CDFI imaging. 26 In the low-velocity areas of PSA, the blood flow could not be fully displayed by CDFI due to the angle limitation. These problems could affect the accurate estimation of the position and width of rupture, and the location of the thrombus in the PSA. Compared with CDFI, B-flow could better display the blood flow in PSA with mural thrombus. 27 The location and amount of thrombosis in the PSA were visualized by BUGTI, which could help the surgeon to adjust the injection angle and thrombin dose promptly. This technique could prevent the possibility of the embolization of the peripheral artery due to an excessive dose of thrombin or the recurrence of PSA due to an insufficient amount of thrombin.
The 21 patients included in this study ranged from 34 to 80 years, with an average of 62.8 years, and 12 patients were older than 60. This may indicate that elderly people are more susceptible to the development of PSA. This relationship between age and PSA may reflect the higher incidence of cardio- and cerebrovascular diseases in the elderly, requiring more interventional therapies. Additionally, atherosclerosis could increase vascular fragility, and the decreased elasticity of arteries in the elderly population may contribute to their susceptibility to PSA. 28
The effect of dual anticoagulation or antiplatelet drugs on PSA is still controversial. It has been reported that anticoagulation and antiplatelet therapy may induce PSA formation. 29 However, several studies have demonstrated that the incidence of PSA complications in the patients receiving dual anticoagulation and the antiplatelet therapy is not significantly different with that in patients not receiving this type of treatment. 30
Although it has been reported that a low dose of thrombin could reduce the risk of peripheral arterial embolism, it may increase the risk of PSA recurrence.31,32 In clinical practice, it is necessary not only to estimate thrombin dose based on the size of PSA, but also to take into account the structure of the PSA visualized under B-flow. After pressing the rupture, B-flow could confirm that the blood flow signal in the PSA disappears or is significantly reduced. In the present investigation, the appropriate needle angle was selected according to the actual situation, and thrombin solution was injected pulse-wise into PSA. We found that the dose of thrombin was positively correlated with the width, the sum of length and width, and maximum cross-sectional area of PSA. Therefore, the thrombin dose could be estimated according to the sac size of PSA. Although there was no correlation between fistula length and thrombin dose in this study, it has been reported that the length of the fistula correlated negatively with the complications of peripheral arterial embolization. 11 Therefore, the shorter the fistula, the more care should be exercised. A suitable injection dose could be applied by observing blood flow and thrombosis in the PSA by B-flow in real-time. This approach could avoid complications due to a too low or too high dose of thrombin.
Two of the 21 patients received compression therapy 1 week after the occurrence of PSA. The two patients were followed up for 5–7 days by US, which showed that the PSA volume was slightly decreased in one patient and was increased in the second patient. Both patients were experiencing pain. After the BUGTI treatment, the blood flow signal in the PSA disappeared. Importantly, no recurrence of PSA was found in the follow-up examinations. The remaining 19 patients were treated by BUGTI at the time when PSA was diagnosed, and none of them developed complications. These results indicate that BUGTI is a rapid and efficient method for treating PSA, which could effectively reduce the pain and shorten the duration of hospitalization.
The limitation of this investigation is its design as a retrospective single-center study with a small sample size. Moreover, the effect of the needle tip’s position inside the PSA during thrombin delivery on the treatment of PSA was not investigated. Further research is required to identify the key factors affecting the outcome of PSA treatment in order to provide precise individualized therapy and prevent complications. Besides, if the size of the PSA is remarkably large with complications, including surrounding tissue vascular dysfunction and skin necrosis, we would recommend surgical repair because evaluating revascularisation or amputation is essential.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the Natural Science Foundation of Jiangsu Province, China (Grant No. BK20160576)
