Abstract
Objectives
Intravascular ultrasound (IVUS) is a uniquely objective but underutilized imaging modality in the interventional treatment of peripheral arterial disease treatment. IVUS has been shown to improve device size selection and diagnose occult pathology difficult to see with routine angiography. We hypothesize that the use of IVUS in lower extremity endovascular intervention improves accuracy in stent and balloon size selection and minimizes contrast use.
Methods
This is a retrospective case series performed at a single-institution outpatient center by two vascular surgeons from July 2016 through July 2017. We identified 94 total IVUS-assisted procedures. We collected data regarding demographics, balloon and stent size, IVUS-determined vessel diameter, pre-procedure and post-procedure ABI values, and contrast used during the procedure. An independent core laboratory was further utilized to characterize all IVUS and angiographic size vessel measurements.
Results
For the 94 lower extremity IVUS-assisted interventions, the average patient age was 70±9 years old, with an average contrast use of 37.1 mL of Omnipaque 300. There was substantial improvement in pre- and post-procedure ABI (0.7±0.23 to 0.94±0.21). On analysis of average intraluminal IVUS vessel diameter to balloon angioplasty used, we had a correlation in size of 0.96±0.16 in our series. Additionally, in eight instances IVUS allowed for identification of a flow-limiting dissection with subsequent stenting.
Conclusions
Routine use of IVUS offers a powerful diagnostic paradigm that has the potential to significantly decrease contrast use and to improve objectivity in choosing appropriate vascular tools to maximize intra-procedural efficacy. The discovery of adverse findings not initially noticed on angiography further reinforces the value of IVUS utilization.
Introduction
There is a plethora of published literature pertaining to the use of intravascular ultrasound (IVUS) in assisting with placement of coronary artery stents. However, there is significantly less published literature on the utilization of IVUS for aiding office outpatient lower extremity endovascular revascularization for peripheral vascular disease. IVUS has increasing possibilities for accurate delineation of infrainguinal anatomy and is apt for complementary use in angiography. High-quality imaging and minimization of contrast are paramount goals in improving endovascular techniques, improving long term outcomes, and is ripe for the outpatient setting as peripheral arterial interventions continue to migrate from a hospital to outpatient/office-based environment. IVUS can facilitate more accurate stent size selection and balloon angioplasty size selection in endovascular procedures. This can be most pronounced for tibial level interventions, where sizing by traditional angiography likely underestimates the vessel size.
Several studies have shown increased accuracy and dependability of IVUS in comparison to various modes of angiography. IVUS was shown to produce significantly different data pertaining to the measurement of the aortic diameter compared to the standard computed tomography angiography (CTA) sizing measurements. 1 It has also been shown to be more sensitive compared to multiplanar venography in identifying iliofemoral vein stenosis amenable to treatment and that the use of IVUS in these cases often leads to a change in the treatment plans. 2 IVUS has been predictive in lower extremity endovascular interventions to reduce complications and amputation rates. 3 One study has shown higher primary patency demonstrated in patents with TASC A-C lesions of the femoral and popliteal artery measured with IVUS and treated with stents. 4 Another study published by Kumakura et al. 5 showed that utilization of IVUS for stenting of the iliac arterial anatomic region had favorable 15-year patency in all TASC categories. Growing literature suggests that the use of IVUS-assisted interventions is associated with very good results and has the potential to improve outcomes. In this study, we describe our institution’s experience with IVUS for endovascular lower extremity revascularization in the office outpatient setting to further investigate its potential to improve patient outcomes.
Our overall hypothesis is that IVUS improves accuracy in pre-intervention stent size and balloon angioplasty size, and significantly decreases contrast use as a complementary tool with angiography. All procedures were performed in an ambulatory, office-based setting where we believe this modality is prime for use in terms of minimizing morbidity, optimizing safe outcomes, and increasing efficiency of readily available endovascular treatment tools.
Methods
This study was approved by the Institutional Review Board of St Joseph Mercy Health Center, Ann Arbor, Michigan. This study is a single institution case series performed through retrospective chart review by assessing CPT codes to identify all lower extremity endovascular arterial interventions performed from July 1, 2016 through July 31, 2017 with intravascular ultrasound assessment through the IHA Vascular and Endovascular Surgeons’ outpatient angiography suite by two high volume, board-certified vascular surgeons.
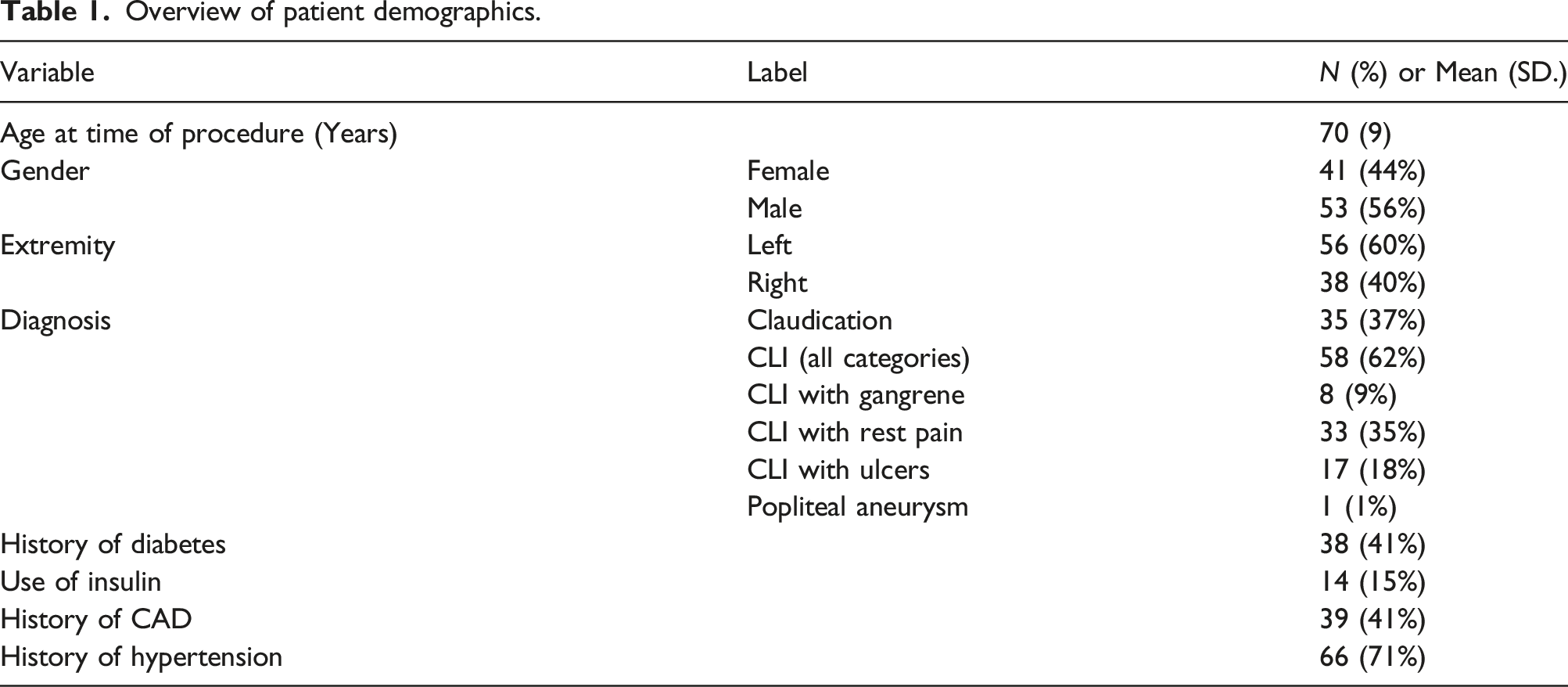
There were a total of 94 separate procedures on 79 total patients. The subject population included all patients undergoing a lower extremity endovascular office outpatient procedure involving at least one of the following anatomic levels—iliac, femoral, popliteal and/or tibial—with the procedure also including assessment by IVUS at one of these anatomic levels. For this study, the possible procedures performed were among the following: diagnostic angiography, balloon-angioplasty intervention, atherectomy with balloon-angioplasty, and/or stenting. Our exclusion criteria were patients who are less than 18 years of age. The patients were selected for IVUS based upon concern about the patient’s GFR, delineation of anatomy as experience grew with outpatient IVUS, and for close evaluation of the target vessels if angiography was unclear. Interventions with IVUS assessment for venous anatomic segments were not included in this study. Patients treated while inpatient were also not included in this study. All angiograms were performed with a Siemens Cios Alpha mobile C-arm.
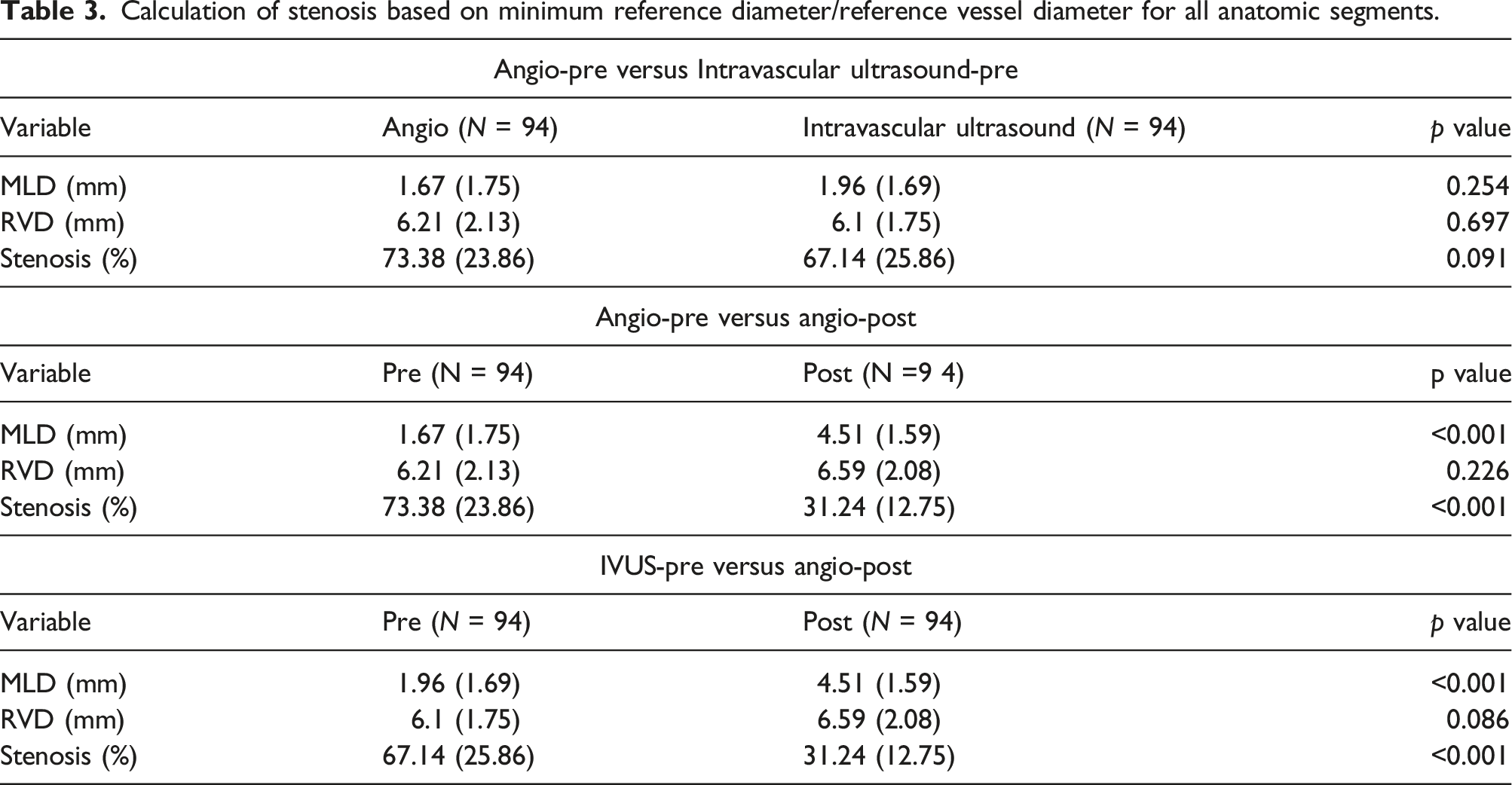
Notably, all angiographic films and IVUS studies were separately queried through a core imaging laboratory (SYNTACTX) to individually calculate all angiographic and IVUS parameters which included the following findings: Lesion location (external iliac, common femoral, superficial femoral, popliteal, tibeo-peroneal trunk, anterior tibial, and posterior tibial). Angiography and IVUS measurements are explained in the included Appendix and listed: Minimum lumen diameter (MLD), reference vessel diameter (RVD), (%) diameter stenosis, and lesion length. For post-treatment angiography: MLD, RVD, and (%) diameter stenosis. For IVUS-pretreatment: MLD, ML area, RVD, RV area, (%) diameter stenosis, and (%) area stenosis.
Demographic variables assessed included the following: age, gender, and claudication. CLI (differentiated by rest pain, gangrene or ulcers), diabetes, history of insulin dependence, hypertension, coronary artery disease (CAD), current smoking status, extremity laterality, use of balloon angioplasty, balloon size, pre-procedure ankle brachial index (ABI) recording with toe pressures within 60 days of the procedure, post-procedure ABI recording with toe pressures within 60 days of the procedure, pre-procedure and post-procedure creatinine, the contrast in milliliters (mL) utilized, total fluoroscopy time, total radiation in mGy, and the Dose area product in Gycm. 2
Angiography was analyzed using Medis QAngio and IVUS imaging was analyzed using Medis QIvus. Percent diameter stenosis was calculated as the ratio of the minimum luminal diameter (smallest luminal diameter along the treated lesion) to the reference vessel diameter (normal vessel diameter at the location of the minimum luminal diameter) [separately completed through SYNTACTYX core laboratory]. Prior to the analysis, all variables were inspected for outliers and missing values. There were no major outliers within any of the study variables. This resulted in a total sample size of 94 observations for this analysis. Descriptive statistics were provided for all study variables. Presented are means and standard deviations for continuous variables, and frequencies and proportions for categorical variables. Next, MLD, RVD, and stenosis measurements are compared between pre-angiogram, pre-IVUS and post-angiogram tests. Additionally, comparisons of stenosis values using diameter and surface area are presented. For these analyses, means and standard deviations are presented for each group, and the p-values come from t-tests. Lastly, pre-IVUS diameter and surface area measurements are compared by looking at their correlation with the post-angiogram diameter measurements. For these correlation companions, the p-values come from t-tests on the Fisher z-transformation of the correlation coefficient. Statistical software used was R version 4.0.3.
Results
Patient Demographics
Overview of patient demographics.
Pre-procedure and Post-procedure ABI data
Of the 94 procedures analyzed, 52 (55%) had both a pre-procedure and a post-procedure ABI (within 60 days of the procedure) recording on the intervention side and the average increase in the ABI value was 0.24 on the intervention side for these “complete” ABI data sets (Figure 1). For 14 of the 94 procedures, post-procedure IVUS was performed and was used to assess for a flow limiting dissection. In 6 of these instances no flow limiting dissection was identified. In 8 of these instances (9% of all procedures) a flow limiting dissection was identified utilizing IVUS after inadequate visualization on angiography and subsequent intervention was performed with stenting. Comparison of Pre-procedure to Post-procedure ABI Measurements. Error bars indicate 1 standard deviation from the mean.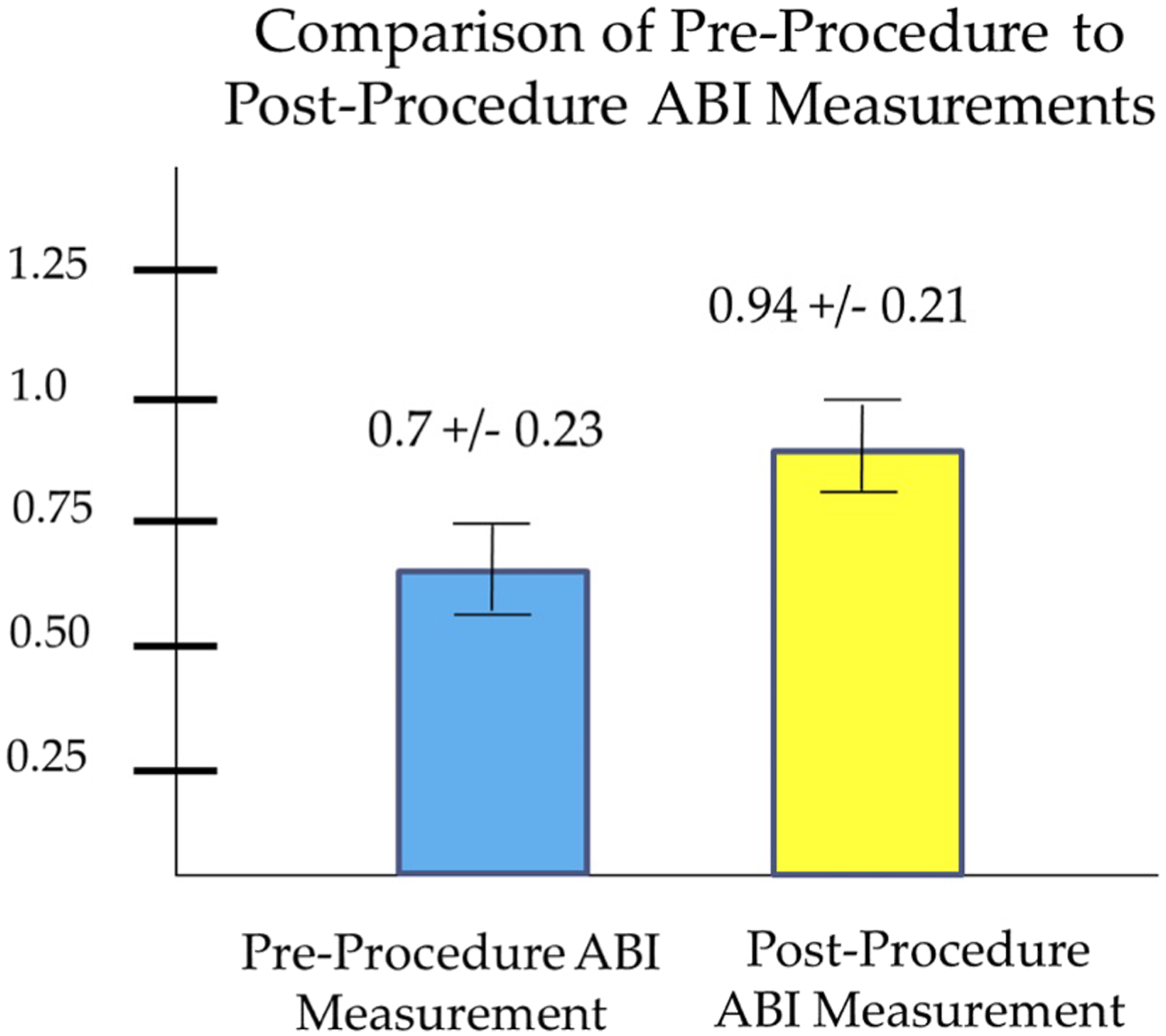
Comparison Between the Vessel Diameter Measured by IVUS and the Balloon Angioplasty Size Selected
Average Intravascular ultrasound vessel measurements compared to balloon angioplasty size utilized across all anatomic segments.

Radiation Exposure and Contrast Use
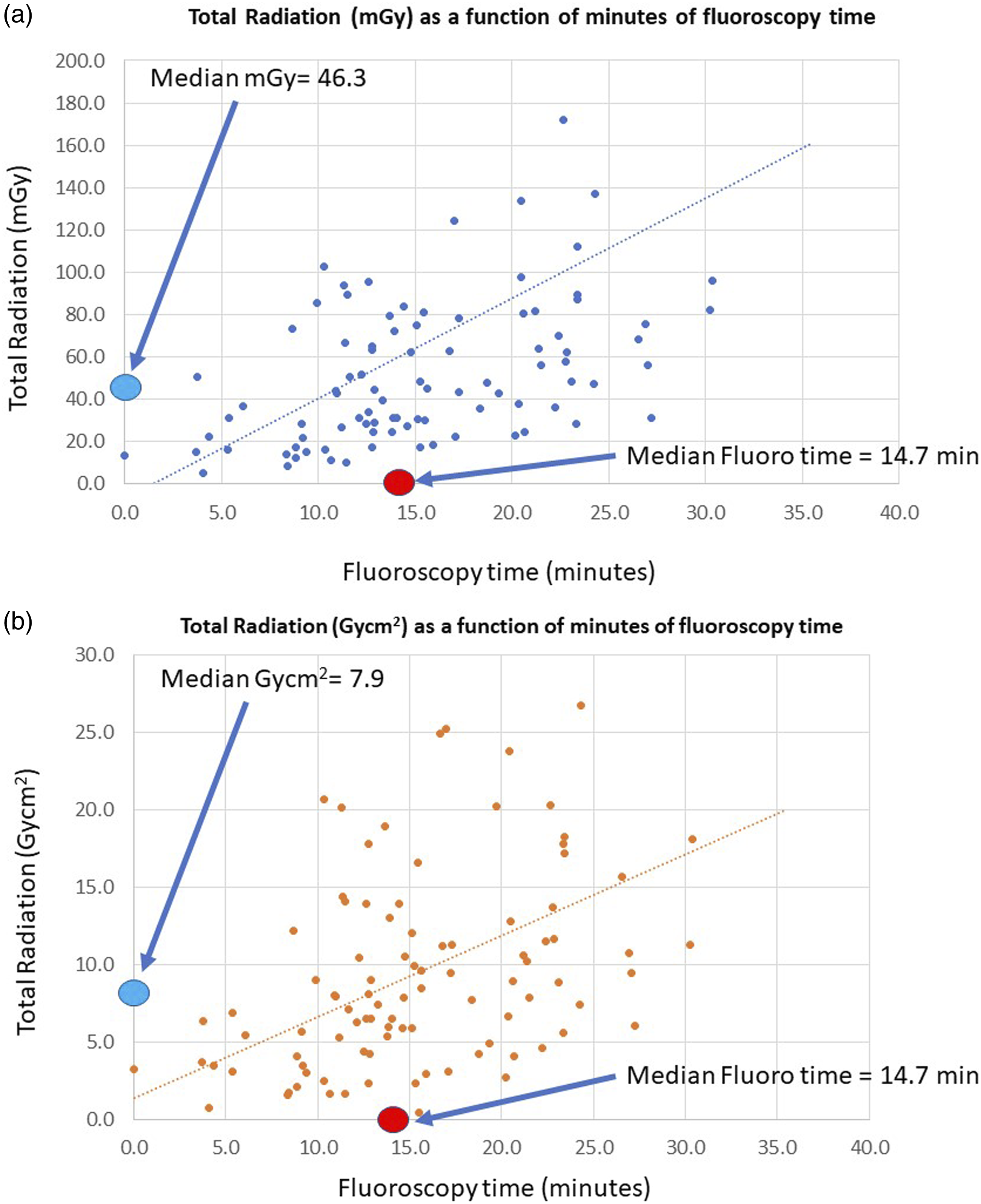
The average contrast used for these 94 procedures was 37.1 mL. The median fluoroscopy time for these 94 procedures was 14 min, 7 s. The median radiation exposure was 46.3 mGy and the DAP (dose area product) was 7.9 Gycm.
2
Scatter plot imaging of fluoroscopic values and time are shown as scatter plots as a function of time in Figures 2(a) and (b). Total radiation for infrainguinal arterial intervention as a function of time by 2a) mGy, 2b) DAP (Dose Area Product, Gycm
2
).
Comparative analysis of pre-angiogram and pre-IVUS vessel measurements to post-angiographic results.
Calculation of stenosis based on minimum reference diameter/reference vessel diameter for all anatomic segments.
Evaluation of IVUS diameter v. surface area measurements as a means of stenosis calculation with post-intervention completion angiography.
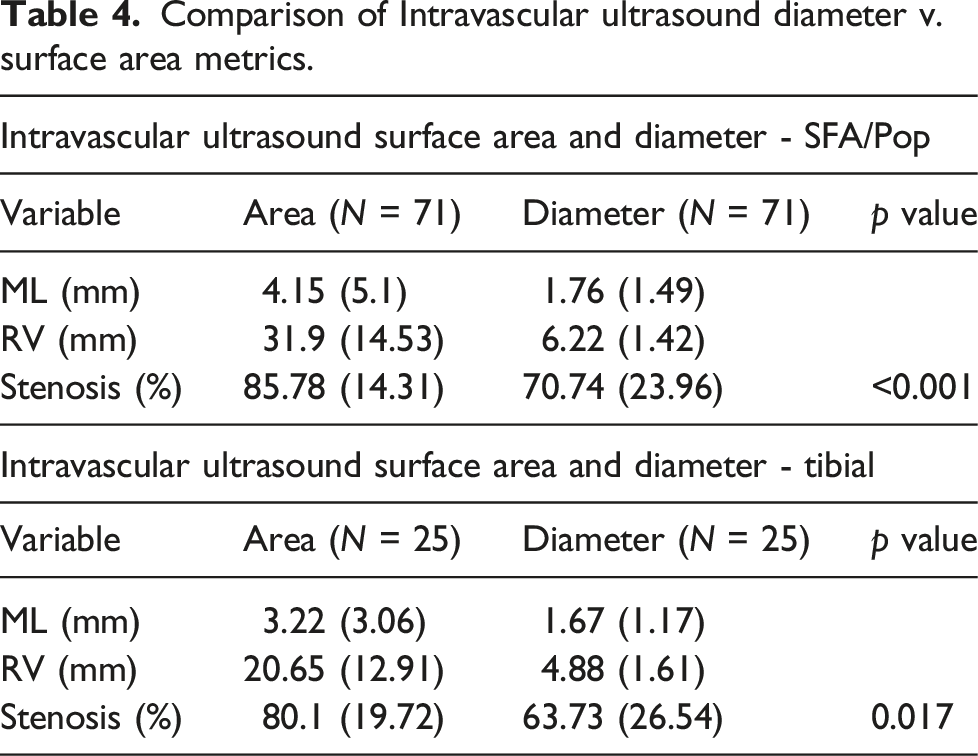
Comparison of Intravascular ultrasound diameter v. surface area metrics.
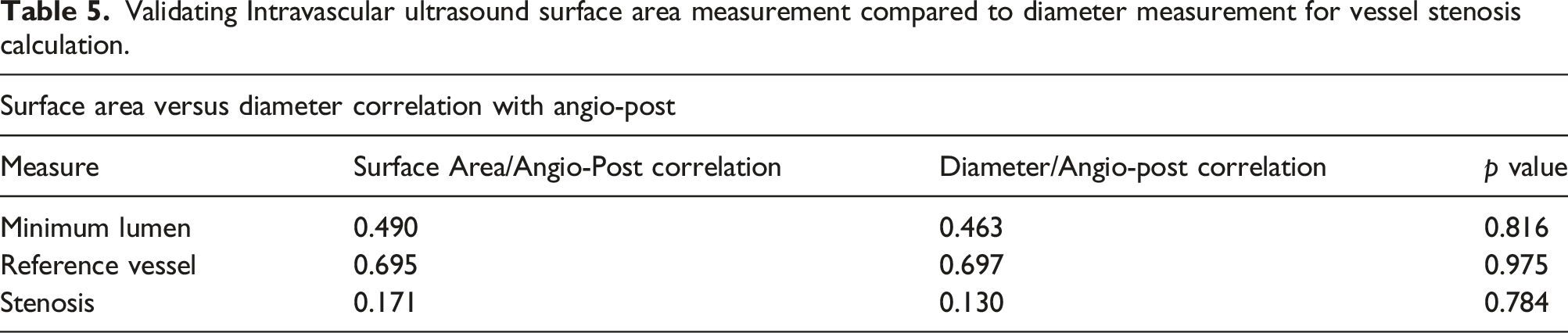
Validating IVUS surface area compared to diameter as for vessel stenosis.
Validating Intravascular ultrasound surface area measurement compared to diameter measurement for vessel stenosis calculation.
Discussion
Our single center case series, performed through retrospective chart review, identified 94 procedures for office outpatient lower extremity endovascular intervention that were assisted by IVUS. Short-term outcomes were good based upon ABI data, as expected for these percutaneous peripheral interventions. The majority of our cohort was notably physiologically and anatomically complex, with 62% presenting with critical limb ischemia (27% with tissue loss). With these complex patients undergoing interventions in an office-based setting, minimization of morbidity and maintaining a high level of patient safety and comfort is ever more paramount as nationwide trends show a shift toward outpatient vascular care. In concert with our clinical findings on a case-by case basis, IVUS will typically show a greater percent stenosis than typical 2-dimensional angiography when using surface area measurements, a testament to the robust imaging data delineating accurate plaque stenosis, morphology, and ability to image in 360°. This tool is particularly well-suited in our modern shift to the outpatient realm, as its non-invasive, uncomplicated implementation has augmented the accuracy and efficacy of traditional treatment tools—particularly the sizing of appropriate balloons and stents to avoid “under treatment” secondary to fear of dissection or vessel perforation.
To the point of specifically evaluating vessel diameter, intraluminal diameter, and percentage stenosis, 0.96 was the average ratio of balloon diameter selected to maximum IVUS vessel measurement recorded in this series. Current literature does not suggest what the appropriate or “best” ratio should be. However, our data do demonstrate that our surgeons can reproduce close correlation between the maximum IVUS vessel measurement and the balloon angioplasty size selected in a safe manner with no episodes of perforation and often times surprising the surgeon (particularly in the tibial arteries) with larger diameter balloons appropriately based on IVUS. We feel this not only contributed to more rapid diagnosis compared to conventional angiography, but many times obviated contrast use (particularly germane in patients with suppressed GFR endemic in this CLI population) and likely prevented undertreatment with smaller balloon diameters. This enabled robust procedures down to the submillimeter level, where balloon inflation with appropriate atmospheric pressure obtained precise diameter measurements using surface area measurements. Investigating the optimum ratio that maximizes patency while avoiding an increase in dissection may be a useful goal of future research regarding IVUS, as would be validation comparing pre- and post-IVUS results as part of a multi-surgeon or multi-institutional prospective study to decrease user bias. For eight patients (9% of the total procedures performed), a flow limiting dissection was identified by IVUS after inadequate visualization on angiography and subsequent intervention was performed with stenting. Thus, the use of IVUS changed our management in a minority of cases while maximizing treatment efficacy.
Additionally, our data demonstrates that IVUS lowers contrast use and can potentially prevent the unwanted renal side effects of contrast exposure. We are able to compare our contrast usage per procedure to average amounts documented by Napoli et al. Their study specifically looked at contrast use in CT angiography compared to digital subtraction angiography (DSA) with revascularization. In that study, 135 mL was the average contrast volume for diagnostic DSA alone and 186 mL was the average contrast volume for DSA including some type of intervention. 6 The average contrast used in our study was 37.1 mL. As values from the literature are significantly higher than this, it suggests that IVUS not only augments visual information and detail from angiography, but can also potentially decrease amount of contrast used. As this is a retrospective study, we do not know how much contrast would have been needed without the assistance of IVUS, and it is difficult to find an appropriate control group with a similar level of complexity to our study cohort. We typically found our contrast-to-GFR ratio to be less than 1:1. Our standard procedure is to prepare a 100 mL solution consisting of 50 mL Omnipaque 300 and an equal portion of heparinized saline, a portion of which is used for the endovascular balloon insufflator. We did not find the need to use CO2 angiography for any of these patients, nor did our patients experience any documented episodes of contrast-induced nephropathy. We found that IVUS allowed our complex interventions to be done without the clinical stress of inducing contrast-induced nephropathy.
This imaging supplement was very well suited to treating the difficult lesions typically found in our CLI-heavy population. Lastly, in comparison to other investigators, we found the combination of IVUS in this study coupled with a Siemens mobile C-arm afforded excellent overall visualization with the lowest total mGy and DAP values in comparison to published values for similar infrainguinal procedures.7,8 In an outpatient setting, we were able to consistently achieve outstanding, remarkably low radiation values. We believe this paradigm of IVUS with low-dose radiation represents a huge advancement in mitigating the undeniable occupational hazard of scattered radiation for our peripheral arterial interventionalists (and patients) while still attaining excellent technical results.
This is a retrospective manuscript with clear limitations, the most obvious is lack of a consistent post-intervention IVUS cohort in all cases, precluding further validation of post-IVUS interventions with conventional angiography as a control in this setting. We compare our contrast use to values found in the literature but do not have an adequate control group from our own study. However, we show the very low use of contrast in this study does not undermine its clinical utility, particularly in the obvious circumstance of a patient with decreased GFR requiring outpatient, complicated percutaneous revascularization. The significance of close correlation of balloon size to vessel diameter seems intuitive but is not clearly proven or defined, and a larger case series best collected prospectively between more institutions would reduce provider bias and shed more light on the efficacy of IVUS head-to-head with angiography in terms of accuracy with diagnosis and treatment.
Conclusion
Our data show that IVUS-assisted lower extremity endovascular interventions can produce successful initial results with very close correlation of balloon size matched to vessel diameter measured by IVUS. This case series was obtained through retrospective analysis so causation cannot be inferred. However, this study suggests that IVUS is a very safe, likely underutilized tool for lower extremity revascularization in the office outpatient setting where contrast use, procedure length and efficacy, patient safety, and minimization of radiation to the provider are at a premium. We found surface area measurements to be statistically more profound than diameter measurements, with implications for “borderline” cases in the outpatient setting particularly questioning intervention necessity. These advantages translated into greater confidence in management of commonly used endovascular treatment tools.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
