Abstract
Objective
To report and compare neoaortoiliac system reconstruction and cryopreserved human allograft in treating aortic graft infections.
Methods
We retrospectively analysed the data of the patients treated for aorto graft infections between January 2015 and May 2021 in our hospital. The clinical data, diagnostic procedures, and surgical options were evaluated. The primary endpoint of this study was the 30-day and 1-year mortality; secondary endpoints were major postoperative complications.
Results
We retrospectively reviewed a series of 31 consecutive patients (28 males; median age 72 years, range, 50–87 years) with aortic graft infection treated with NAIS (n = 20, 65%) or cryopreserved allograft (n = 11, 36%). The clinical presentation included fever attacks in 18 (58%) patients, abdominal pain in 15 (48%) patients, haemodynamic instability in 6 (19%) patients, and haematemesis in 2 (7%) patients. The median operative time of the NAIS was longer than CHA without a statistically significant difference (458 min vs. 359 min, p = .505). The postoperative morbidity for all patients was 81%, with no significant difference between NAIS and CHA groups (85% vs. 73%, p = .638). There was no limb thrombosis of the new reconstructions. Limb loss occurred in 4 (13%) patients, including 2 (10%) NAIS patients and 2 (18%) CHA patients. One NAIS patient developed complications in the form of a distal (femoral) disruption of the vein 15 days after surgery. There were no significant differences between NAIS and CHA groups in ICU stay (12 vs 8 days, .984) but in hospitalization (22 vs 33, p = .033). The most common bacteria isolated were staphylococci strains in 15 (48%). In 13 (36%) patients, candida was positive. The in-hospital 30-day and 1-year mortality for all patients was 16% (5/31) and 29% (9/31), with no significant differences between NAIS and CHA at 30 days (25% vs. 0, p = .133) or 1 year (35% vs. 18%, .429). Five NAIS patients died during the hospital stay; three of them had end-of-life decisions. After a median follow-up of 16 months (1–66 months), 12 (39%) patients died, including 9 patients with NAIS and 3 with CHA reconstructions. The causes of death included overwhelming sepsis in 5 (42%) patients, graft disruption in one (8%) NAIS patient, non–small cell lung cancer in one (8%) patient, COVID-19 in one (8%) patient and unknown causes (8%) in one.
Conclusions
Non-staged neoaortoiliac system reconstruction and cryopreserved human allografts show comparable short- and midterm results for treating aortic graft infections. However, both procedures remain challenging with high morbidity and mortality rates.
Introduction
Aortic graft infections continue to have high mortality and morbidity rates despite the significant progress in surgical treatment, perioperative care, and antimicrobial therapy. During the last decades, many studies considered removing the infected graft material and in situ reconstruction with biological material as the first-line treatment for aortic graft infections.1,2 Although autogenous deep femoral veins and cryopreserved human allograft showed the lowest reinfection rates (<10%), the studies comparing these two methods are rare. 2
Despite their similarities in the surgical techniques, the need for a femoropopliteal vein in the neoaortoiliac system reconstruction expands the surgical field and may prolong the operating time and create potential difficulties in controlling body core temperature. However, the influence of these differences in the outcome of both surgical options has not been studied sufficiently.
This study aims to report our experience in treating aortic graft infections and compare the short- and midterm results of treating aortic graft infections with the use of neoaortoiliac system (NAIS) reconstruction and cryopreserved human allograft (CHA).
Methods
The institutional review board of the Charité – Berlin university of medicine approved the study. All consecutive patients with aortic graft infections after EVAR and open aortic repair treated with neoaortoiliac system (NAIS) reconstruction or cryopreserved human allograft (CHAs) from 2015 to 2021 in a single-centre institution were included in this study. The patients with aortic graft infections who underwent extra-anatomic reconstruction or in situ reconstruction with synthetic grafts were excluded. Data was prospectively collected in a database and retrospectively analysed.3,4
All patients underwent computed tomography (CT) scans at presentation. Esophagogastroduodenoscopy (EGD) was obtained in the cases of suspected aorto-duodenal fistula.
In addition, fluorine-18-fluorodeoxyglucose positron emission tomography (FDG-PET) was performed if CT failed to confirm the diagnosis of aortic infection.
Preoperative collected and analysed data included patient demographics, cardiovascular risk factors, clinical presentation, imaging, previous aortic pathology and previous procedures.
Moreover, we calculated the timeframe between the initial aortic intervention and the development of aortic graft infection. The physical status of all patients was assessed preoperatively with the American Society of Anaesthesiologists (ASA) score. Intraoperative details that were taken into consideration included: the surgical approach, duodenal repair, use of aortic cross-clamping, graft and stent-graft removal or not, estimated blood loss and operative time.
Every patient underwent a multi-disciplinary discussion and a decision-making process with vascular surgeons, radiologists, and anaesthetists in order to enable a choice between the available surgical options. However, emergency patients needed urgent decision-making by the vascular surgeon and anaesthetist and depended on the surgeon’s preference in most cases. The factors that impact the choice of surgical approach included the urgency of the operation, the availability of cryopreserved human allografts or deep femoral vein, and the patient’s comorbidities. In addition, because CHAs need more time to be obtained, NAIS was preferred in emergency cases. On the other hand, cryopreserved human allografts were the first options whenever they were available and feasible. We received CHAs from the Tissue Bank of the Charité – Universitätsmedizin Berlin within 12–24 h of our request. ABO matching between donors and recipients was obtained in all CHA cases.
Non-staged NAIS approaches were obtained in all cases. Accordingly, two separate teams were involved and worked concurrently. One team dissects femoropopliteal veins, which are left in situ until needed, and the other team performs the abdominal part of the operation.
Additionally, all obtained CHAs and autologous vascular segments were prepared and tailored on another table while operating surgeons were operating on the patient on the main table. Collateral branches of the CHA and NAIS were ligated using through-and-through polypropylene sutures.
The abdominal steps of the operation were similar in both CHA and NAIS patients. A median laparotomy was routinely performed; infrarenal aortic clamping was performed whenever possible. However, supra celiac clamping was obtained in the cases of EVAR infections to remove the stent-graft. In all cases, prosthetic graft material was removed completely, and infected native vessels and periaortic tissues were debrided to obtain as normal a macroscopical tissue as possible. All the specimens were cultured to test for aerobic and anaerobic bacteria and fungi. All CHA and NAIS reconstructions were implanted in situ. The length of the vascular segments and anastomosis was optimized appropriately without tension. Drains were routinely positioned. The infected field was covered with pedicled omentum in the abdomen and sartorius muscle flaps in the groin.
Postoperatively, all patients were admitted to the intensive care unit (ICU), received low-molecular-weight heparin twice per day during the hospital stay. In addition, the NAIS patients were administered the anticoagulant Phenprocoumon upon discharge for 3 months to lower the risk of additional deep venous thrombosis. CHA patients and NAIS patients were treated with aspirin as part of a general medical treatment for cardiovascular disease. No immunosuppressive agents for CHA patients were used.
Empiric intravenous antibiotic therapy was administrated until results of cultures were available. All patients received postoperative intravenous antibiotics and antifungal treatment for 4–6 weeks, followed by oral antibiotics for 3 months
Follow-up examinations included clinical visits, echo-colour-Doppler ultrasound, or computed tomography angiography within 30 days after the treatment, and from then on twice per year.
Primary outcomes were overall survival and in-hospital and 1-year mortality. Secondary outcomes included major perioperative and late complications and reinterventions.
Statistical methods
We descriptively analysed the data and reported the results on an individual level. We presented categorical data as frequencies (percentages) and continuous data as median (range). Endpoints analyses between subgroups were compared using the Chi-square test or Fisher exact test, as deemed appropriate. Survival curves were computed using Kaplan–Meier. All statistical analyses were performed using IBM SPSS Statistics 25 software.
Results
Between February 2015 and May 2021, a total of 713 abdominal aortic repairs, comprised of 393 open procedures and 320 EVARs, were performed in our institution. A total of 56 patients were operated on for aortic graft infection, accounting for about 7.7% of all abdominal aortic repairs in our hospital. Of those, 31 patients (28 males; median age 72 years, range, 50–87 years) underwent in situ aortic repairs with NAIS (n = 20, 65%) or cryopreserved allograft (n = 11, 36%). The aetiology of the initial procedures included AAA in 21 (68%) patients and Leriche syndrome in 10 (32%) patients. Previous procedures included primary open aortic repair in 23 (74%) patients, EVAR in 4 (13%) patients, and open conversion after EVAR in 3 (10%) patients. One (3%) patient with a mycotic aneurysm had had no previous operations. Two (7%) patients had had early (<4 months) infections and 28 (90%) late (≥4 months) infections.
On average, the interval between initial aortic procedure and AGI was 49 months (range, 1–262 months) with no statistical difference between EVAR (median 35 months, range 5–71 months) and open aortic repair (median 49 months, range 1–262 months) and p = .402.
Eight (26%) patients had aorto-duodenal fistulas at presentation, including 3 (10%) patients with true fistula and five (16%) patients with paraprosthetic–enteric fistula.
Descriptive characteristics and clinical presentation.
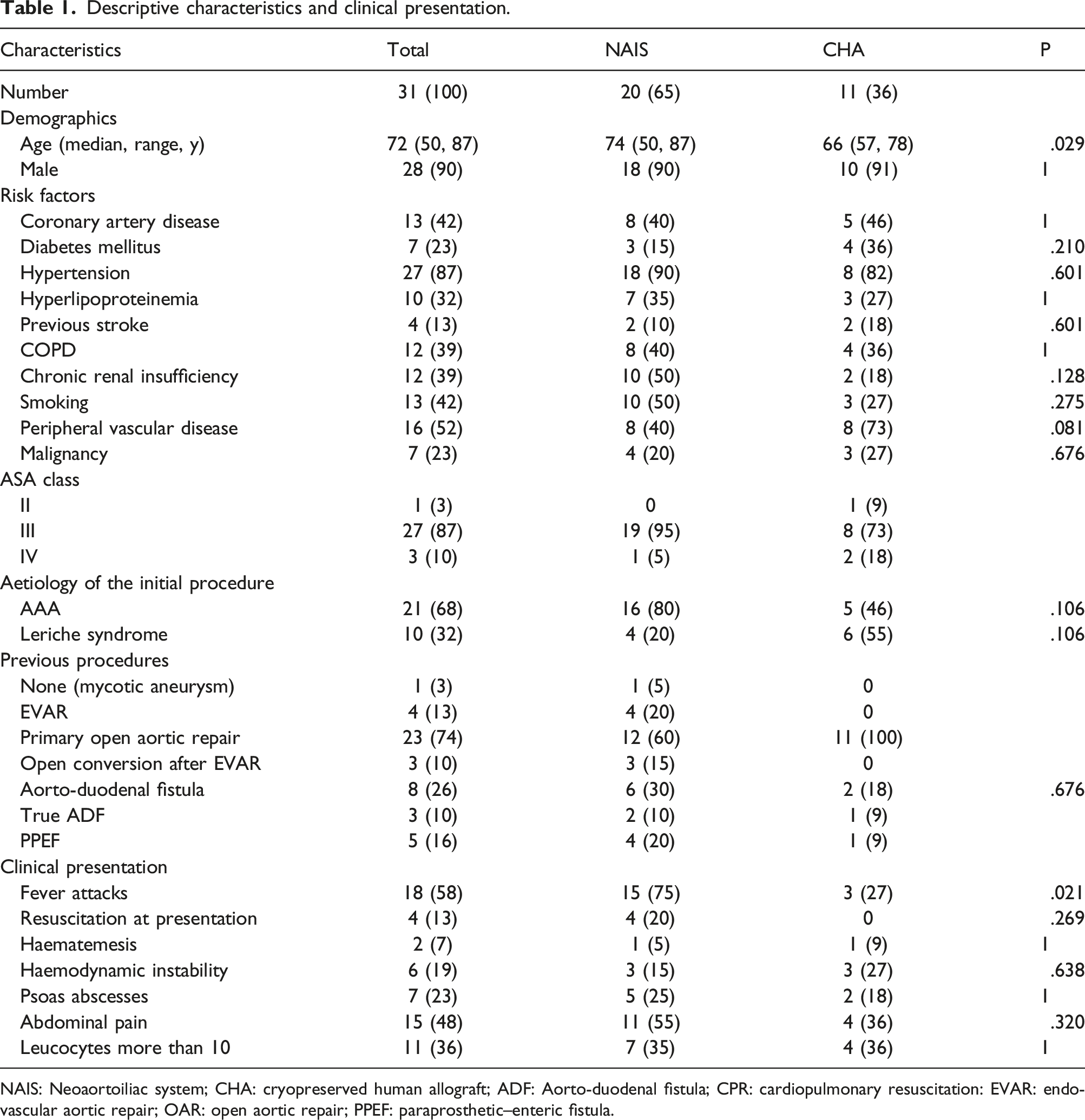
NAIS: Neoaortoiliac system; CHA: cryopreserved human allograft; ADF: Aorto-duodenal fistula; CPR: cardiopulmonary resuscitation: EVAR: endovascular aortic repair; OAR: open aortic repair; PPEF: paraprosthetic–enteric fistula.
Regarding the diagnostic instruments, Abdominal/pelvic CT scans were performed on all patients. Leading diagnostic CT findings included periprosthetic fluid collection in 29 (94%) patients, trapped air in 13 (42%), a proximal false anastomotic aneurysm in 2 (6%) and psoas abscess in 7 (23%) patients. Esophagogastroduodenoscopy was obtained in nine (29%) patients with suspected aorto-duodenal fistula. Blood in the duodenum was shown in four patients. However, a fistula could not be identified by using EGD in any patient.
Micro-organisms cultured.
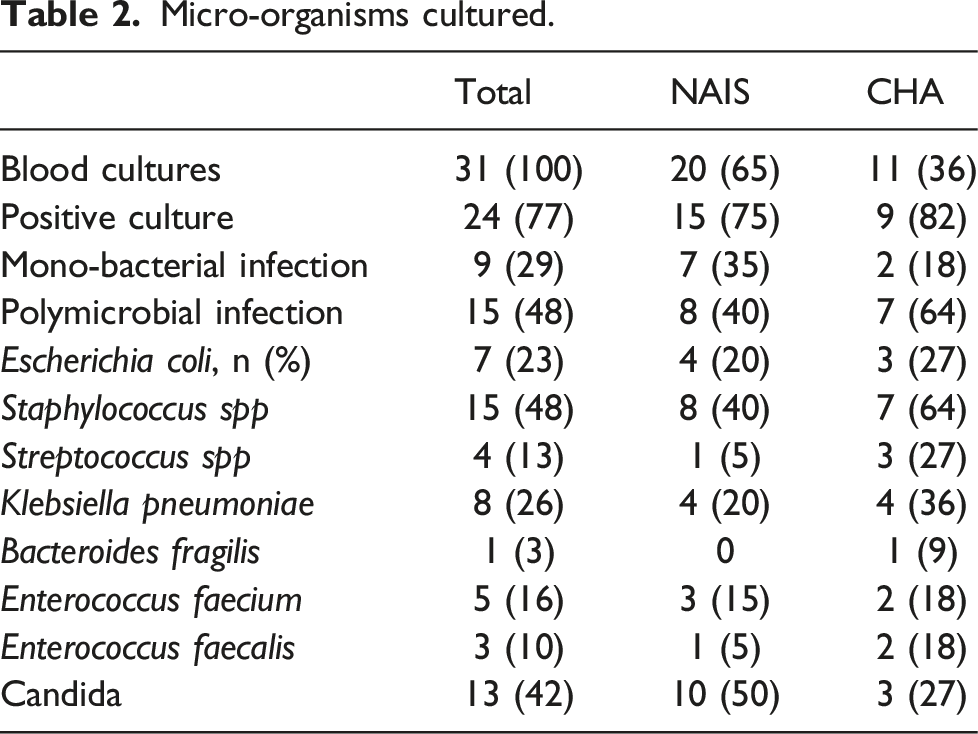
The most common bacteria isolated were staphylococci strains in 15 (48%) of those, methicillin-resistant Staphylococcus aureus (MRSA) in 3 (10%) patients. In 7 (23%) cases, bacterial growth remained negative, 9 (29%) patients had a mono-bacterial infection, and 15 (48%) had a polymicrobial infection. In 13 (36%) patients, candida was positive, including Candida albicans in nine patients, Candida glabrata in three patients and Candida parapsilosis in one patient.
Details of the surgical treatment, complications and outcome.
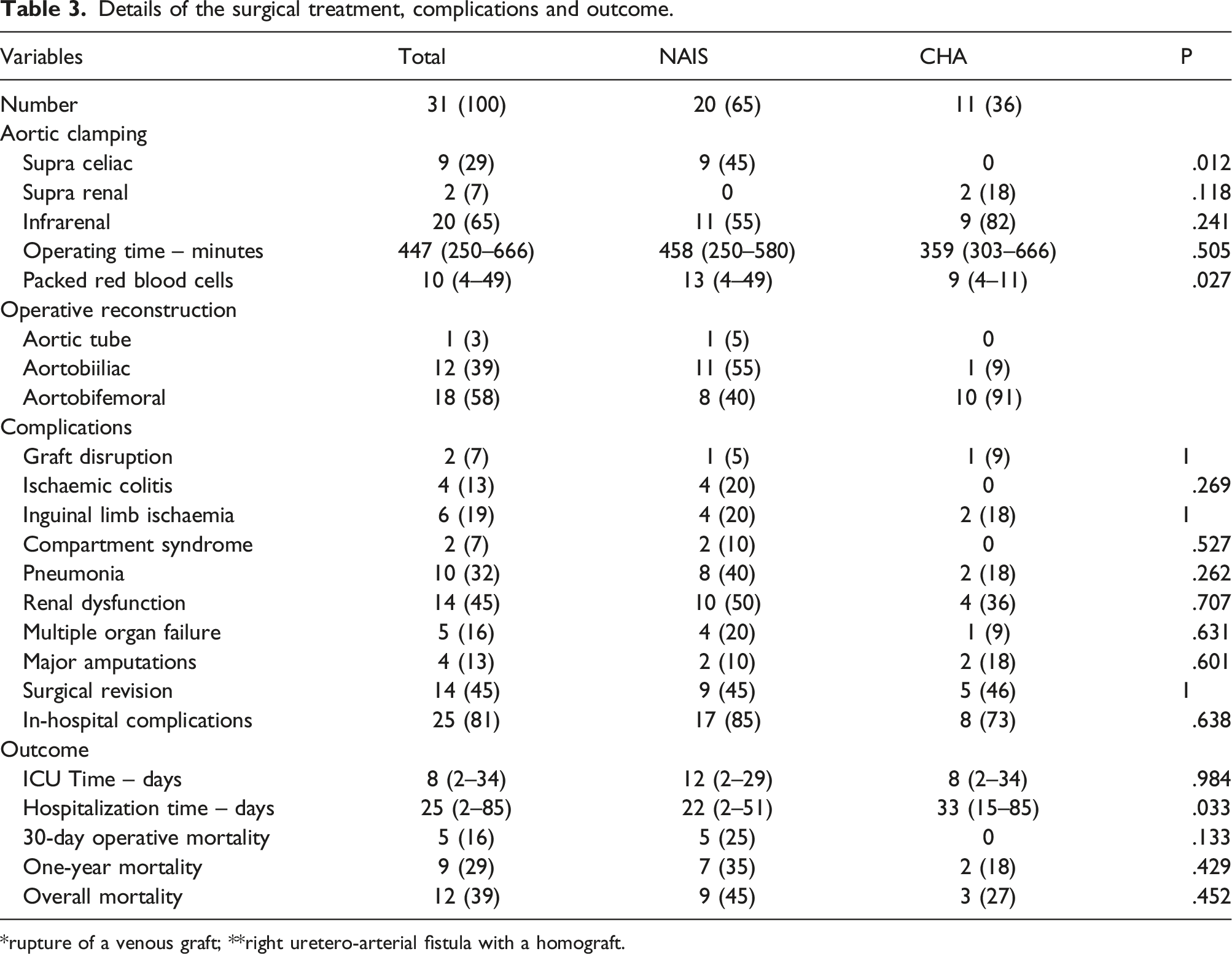
*rupture of a venous graft; **right uretero-arterial fistula with a homograft.
The median operative time of the NAIS was longer than CHA without a statistically significant difference (458 min vs. 359 min, p = .505). The primary technical success was achieved in all cases.
The postoperative morbidity for all patients was 81%, with no significant difference between NAIS and CHA groups (85% vs. 73%, p = .638). There was no limb thrombosis of the new reconstructions. However, six (19%) patients developed complications with distal embolization and needed embolectomy of the peripheral arteries. Of those two (10%) NAIS patients developed compartment syndrome with the need for fasciotomy. Limb loss occurred in 4 (13%) patients, including 2 (10%) NAIS patients and 2 (18%) CHA patients. They all had a chronic peripheral arterial disease, and three of them had irreversible ischaemia on presentation with occlusion of the femoral artery. In addition, one NAIS patient developed complications with distal (femoral) disruption of the vein 15 days after surgery and died with hemorrhagic shock. Another CHA patient developed bleeding from a degenerated side branch of the CHA graft 18 days after surgery. The patient underwent a stent-graft to stop the bleeding and survived with otherwise unremarkable follow-up.
There were no significant differences between NAIS and CHA groups in ICU stay (12 vs 8 days, .984) but one in hospitalization (22 vs 33, p = .033).
The in-hospital 30-day and 1-year mortality for all patients was 16% (5/31) and 29% (9/31), with no significant differences between NAIS and CHA at 30 days (25% vs. 0, p = .133) or 1 year (35% vs. 18%, .429). The cumulative survivals of the NAIS and CHA patients were (65% vs. 82%) at 1 year and (65% vs. 61%) at 2 years, including the perioperative deaths (Figure 1). Five NAIS patients died during the hospital stay; three of them had end-of-life decisions. After a median follow-up of 19 months (1–66 months), 12 (39%) patients died, including nine patients with NAIS and 3 with CHA reconstructions. Kaplan-Meier survival estimates of the patients treated with Neoaortoiliac system and cryopreserved human allograft (Kaplan–Meier log-rank test, p = .595).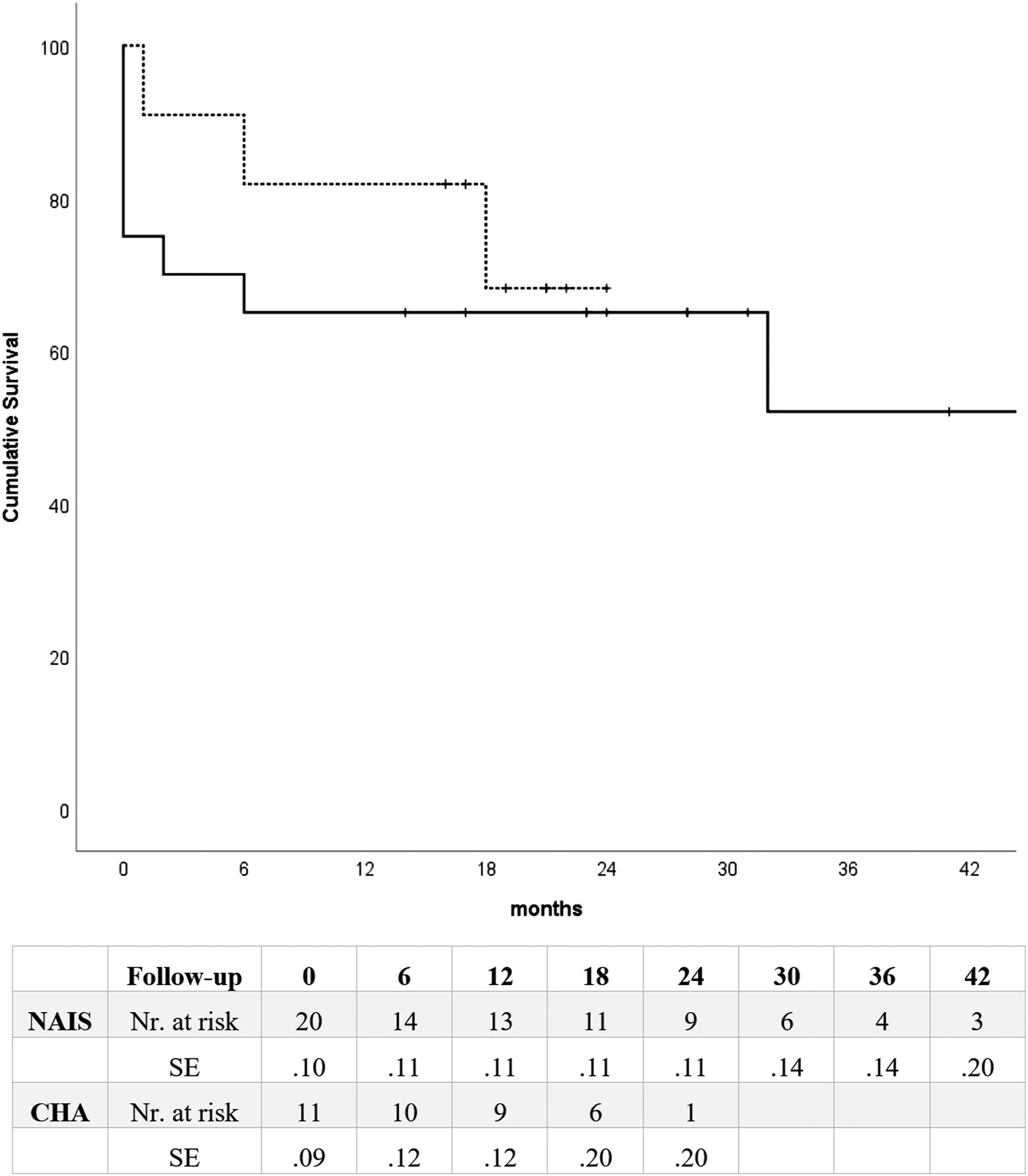
The causes of death included overwhelming sepsis in 5 (42%) patients, graft disruption in one (8%) NAIS patient, non–small cell lung cancer in one (8%) patient, COVID-19 in one (8%) patient and unknown (8%) in one.
Postoperative intravenous antibiotics and antifungals were administered in our patients for at least 6 weeks, based on the isolated strains. The administration of oral suppressive antibiotics depended on the clinical and laboratory parameters of infection.
Discussion
Infections after EVAR and open aortic repair are rare and represent about 0.5–2% of aortic surgeries.5–8 Many surgical techniques have been described for treating aortic graft infections, including in situ and extra-anatomic reconstructions. However, in situ reconstructions with neoaortoiliac system or cryopreserved human allografts have the lowest infection rates (0–7%) and have been considered the best option for replacing infected aortic grafts in many studies.9,2.
Many studies reported excellent results using NAIS with reported 30-days mortalities of 0–15%.10–14 Nevertheless, our results show that not all patients with aortic graft infections are suitable for such complex procedures as NAIS. Therefore, the patient’s overall clinical status, cardiorespiratory capacity, imaging, and laboratory findings should be carefully evaluated preoperatively. 15
There were very complex cases in the current study, including four patients who had needed cardiac resuscitation at presentation because of bleeding or septic shock; four patients had developed graft infection after open conversion of EVAR, and eight cases aorto-duodenal fistulas. These factors influence the operating time as well as the surgical preparation and aortic reconstruction. Another critical factor is the end-of-life decision of the patients and their relatives after operations. The treatment of these patients needs extended ICU stay and rehabilitation, which induces patients and relatives to decide to stop the suffering in some cases.
NAIS procedures have many advantages including, excellent patency, better limb salvage rate and lack of recurrent infection. On the other hand, these complex and prolonged procedures show many impediments, including extensive operating times, significant blood loss, difficulty in controlling body core temperature and venous complications. 16
Although the simultaneous work of two surgical teams can potentially reduce the procedure time, the difficulties in controlling the temperature and fluids of the patients stay challenging in the patients after non-staged NAIS (Figure 2). Therefore, staging the NAIS procedure over 2 days, with harvesting the femoral vein 1 day earlier than the revascularization procedure, was advocated in many studies.17,18 On the other hand, staged procedures run the risk of the vein not being used if the proximal anastomosis with the aortic neck is technically impossible.
15
(a) Contrast computed tomography scan demonstrating fluid and air bubbles around the stent-grafts (arrow), (b) PET-CT with F18-FDG revealing intense uptake in the infrarenal aorta around the stent-graft (arrow). (c) Intraoperative imaging showing in situ surgical repair using the femoral vein (Pantaloon configuration of neoaortoiliac segment) (arrow), (d): Postoperative computed tomography scan with a 3D reconstruction of the aortic repair.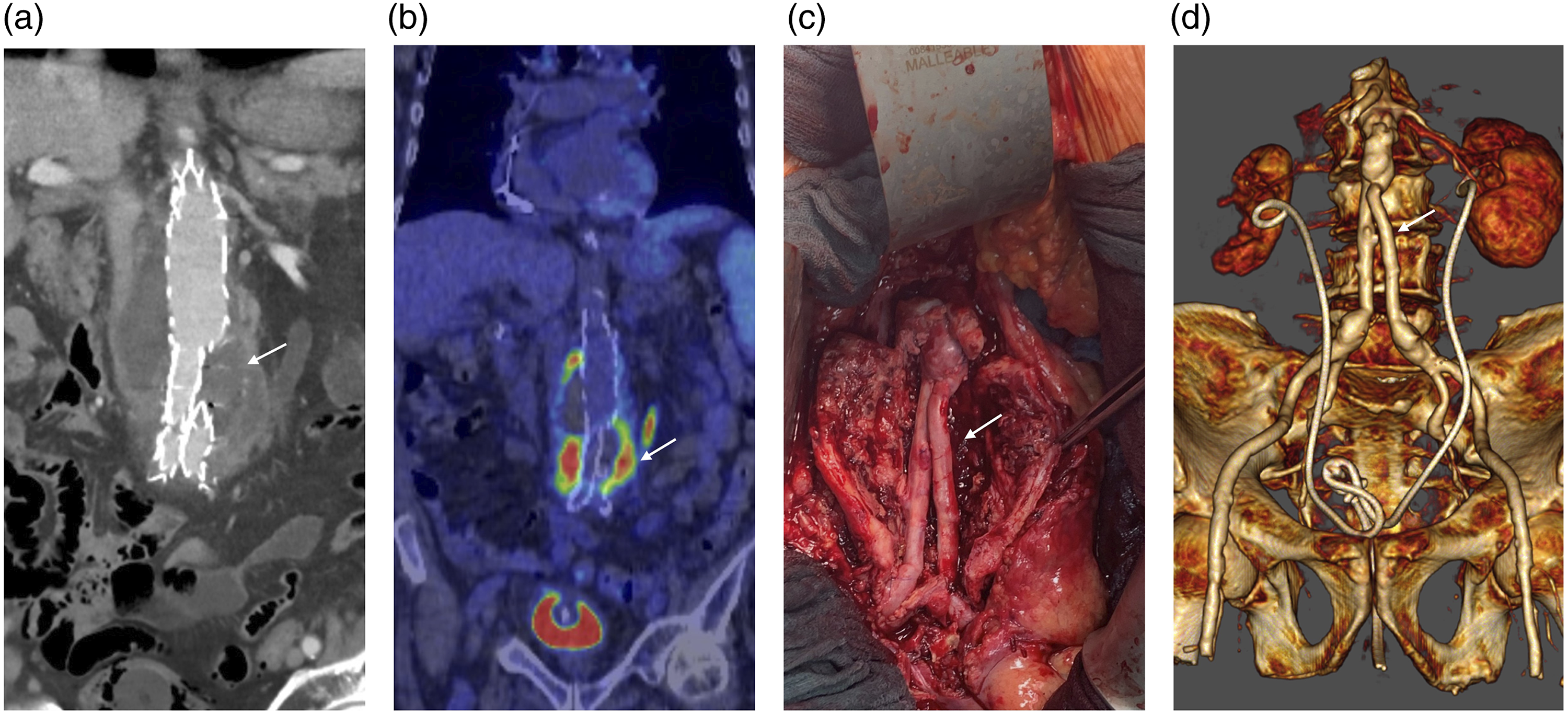
Although CHA reconstruction has no in-hospital mortalities compared to NAIS in the current study, both have comparable high complications and morbidity rates. These complications included major amputations, limb ischaemia, sepsis, cardiopulmonary complications, compartment syndrome requiring fasciotomy and wound infections.
Some studies reported a fasciotomy rate of 25% after NAIS procedures. 19 However, in the current study, 10% of the NAIS patients and none of the CHA patients developed compartment syndromes with the need for fasciotomy.
In situ reconstruction with NAIS or CHAs is the first-line therapy in our institution. However, NAIS and CHAs stay demanding procedures for the patient and the vascular team and are not appropriate for very sick patients, severe sepsis or rapid active bleeding control. On the other hand, conservative approaches, such as percutaneous periaortic drainage, graft lavage or omental flap, are contraindicated in cases of aorto-enteric fistula. Additionally, NAIS is contraindicated in patients with a history of deep venous thrombosis.
The operating team can also select the appropriate CHAs for the reconstruction based on their calibre and length (Figure 3). In contrast, the surgeon may encounter numerous difficulties in obtaining the appropriate length and diameter of deep femoral and popliteal veins, especially when aorto-bifemoral reconstruction is necessary. (a, b) Contrast computed tomography scan demonstrating fluid around the aortic graft and abscess extending to the left psoas muscle (arrows), (c) Intraoperative imaging showing aortobifemoral bypass using a homograft (arrow), (d): Postoperative computed tomography scan with a 3D reconstruction of the aortic repair.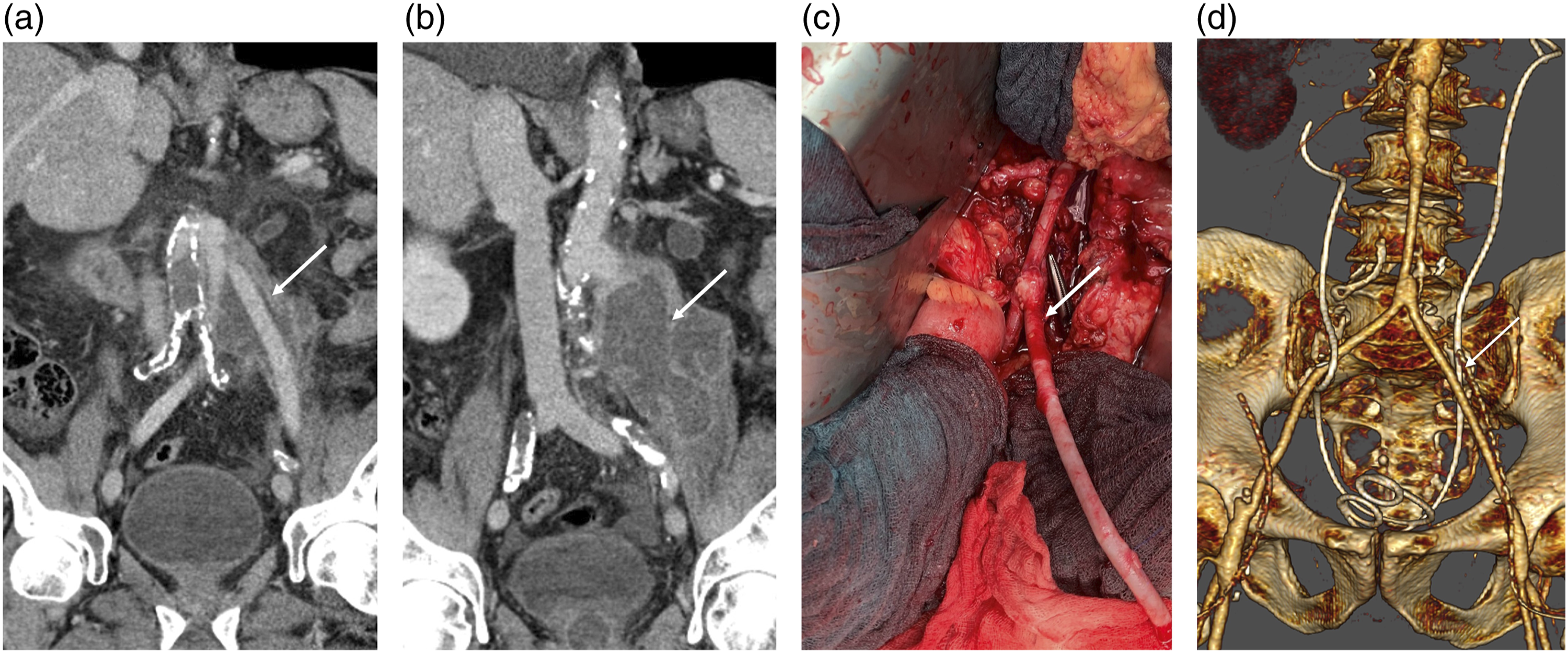
Although rare, anastomotic disruption has been described with the use of NAIS and CHA.14,20 For example, one patient with NAIS developed graft degeneration of the femoral anastomosis in our series because of persistent sepsis. Additionally, one CHA patient developed degeneration and bleeding from one of the allograft side branches but was treated successfully with a stent-graft.
Other problems related to the use of NAIS and CHA include intimal hyperplasia at the anastomotic sites, kinks and reinfection. 16
Our results showed that not every patient with an aortic graft infection is suitable for NAIS reconstruction. However, patients with a suitable general situation have excellent results after NAIS or CHA compared to traditional single-stage or multistage extra-anatomic bypass followed by resection of infected aortic grafts.2,9
Microorganisms can be identified in 75–98% of the cases. 2 In the current study, specimens were positive in 77% of the cases, and staphylococci strains were the most common bacteria isolated.
This study is limited by its retrospective nature and the relatively small number of patients. Additionally, there is a selection bias because most patients needed individual surgical decisions for the treatment. Additionally, selecting NAIS in the emergent situation where CHA is impossible may represent a treatment bias. However, the high mortality rate of these patients may point to the fact that further evaluation of the NAIS as an option in emergent situations is needed.
Conclusion
NAIS and CHA procedures show comparable short- and midterm results with high morbidity and mortality rates. Both procedures are demanding and may not be appropriate for very sick patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Ethics Committee of the Charité – Universitätsmedizin Berlin approved this study (Number: EA4/017/19).
