Abstract
Objective
Profunda femoris artery aneurysms (PFAAs), which comprise true profunda femoris artery aneurysms (TPFAAs) and profunda femoris artery pseudoaneuryms (PFA PSAs), are rare but clinically significant diseases of the peripheral arterial vasculature. Our aim is to describe our institution’s 15-year experience with PFAAs (TPFAAs and PFA PSAs) to provide insight into patient characteristics, diagnostic imaging modalities, and surgical interventions that contribute to clinically important outcomes in patients with PFAAs.
Methods
We conducted a retrospective study at our institution using our radiology database.
Results
We identified six patients with PFA PSAs and four patients with TPFAAs. The clinical presentation of PFA PSAs included a triad of thigh pain, bleeding, and unexplained anemia. There was variety in the aetiologies of PFA PSAs, arising from catheterizations, upper thigh fractures, anastomotic complications, or unknown causes. Most patients with PFA PSAs had hypertension and coronary artery disease, and half of our cohort had peripheral vascular disease. All patients were imaged with duplex ultrasonography (DUS) or computed tomography (CT), the latter being more accurate. All patients with PFA PSAs underwent endovascular treatment, including glue, thrombin, or coil embolization as well as stent-graft insertions. All TPFAAs presented to our center were small and incidentally discovered, explaining the conservative management of our TPFAAs. Two of the four TPFAAs were idiopathic in nature, while one was attributed to post-stenotic dilatation, and another was found in a patient with Ehlers Danlos Syndrome. There was an association between TPFAAs and multiple synchronous or asynchronous aneurysms.
Conclusion
Pseudoaneurysms of the PFA are mostly iatrogenic in nature and can present with the triad of thigh swelling, bleeding, and unexplained anemia. If the clinical picture is suggestive of a PFA PSA but DUS does not detect a pseudoaneurysm, CT may be added as a more accurate imaging modality. Endovascular embolization is used in smaller pseudoaneurysms and in poor surgical candidates. Multiple glue, coil, or thrombin injections may be required to fully thrombose the pseudoaneurysm sac. True aneurysms of the PFA are associated with synchronous/asynchronous aneurysms and small TPFAAs should be carefully monitored, as there is a risk of enlargement and rupture.
Introduction
Profunda femoris artery aneurysms (PFAAs) comprise true profunda femoris artery aneurysms (TPFAAs) and profunda femoris artery pseudoaneurysms (PFA PSAs). These are rare but clinically significant aneurysms of the peripheral arterial vasculature, with potential for neurovascular compressive symptoms1,2 and life-threatening rupture.3,4 Pseudoaneurysms of the profunda femoris artery have an incidence ranging from 1 to 6% and are most commonly due to blunt, penetrating, orthopedic, and iatrogenic trauma. 1 The presentations of pseudoaneurysms are relatively uniform with thigh swelling and hematoma; however, time from injury to diagnosis can vary from days to years.3,4 Similar to common or superficial femoral artery pseudoaneurysms, PFA PSAs can spontaneously thrombose if small, enlarge causing compressive symptoms, or rupture. 5 However, due to the paucity of data, there is no size criterion to direct conservative, surgical, or endovascular management. True PFAAs are even rarer, accounting for 0.5% of all peripheral aneurysms and are associated with atherosclerotic wall degeneration among the aged population. 6 Most TPFAAs are asymptomatic; however, a high rupture rate of 20% requires prudent observation or intervention. 6 Both TPFAAs and PFA PSAs can be managed conservatively, surgically, or endovascularly, depending on the presentation, rupture status, aneurysm size, patient status, and patient wishes. Ultimately, the rarity of PFAAs has not allowed a conclusive study of their true natural history. The clinical presentation and management of these aneurysms has been assessed by publication of case series involving this pathology. Our aim is to describe our institution’s 15-year experience with PFAAs to provide insight into patient characteristics, diagnostic imaging modalities, and surgical interventions in patients with PFAAs.
Methods
We received approval for this retrospective descriptive study from the University Health Network Research Ethics Board. We searched our local imaging database for patients with TPFAAs or PFA PSAs using the key terms “profunda” and “pseudoaneurysm” or “aneurysm.” The search returned 213 unique imaging reports, which were ultimately filtered to 10 patients presenting to our quaternary care facility according to the CONSORT diagram (Figure 1). Inclusion necessitated true or pseudoaneurysms involving the profunda femoris artery (proximal, distal, and branches). A true aneurysm is defined as a focal dilatation to at least 1.5 times the diameter of the adjacent normal vessel and contains all three layers of the arterial wall.
5
A pseudoaneurysm is contained by the tunica adventitia or surrounding soft tissue, rather than all three wall layers.
5
Profunda femoris arteries described as “diseased,” “enlarged,” “ectatic,” “tortuous,” “hypertrophic,” or “oozing” were excluded. Additional exclusion criteria included TPFAAs or PFA PSAs that: (1) were outside our time frame; (2) were later discovered as femoral aneurysms occurring elsewhere along the femoral vascular tree (i.e. common femoral artery and superficial femoral artery) around the time of presentation or during our search; and (3) had insufficient information for analysis. CONSORT diagram of study patients.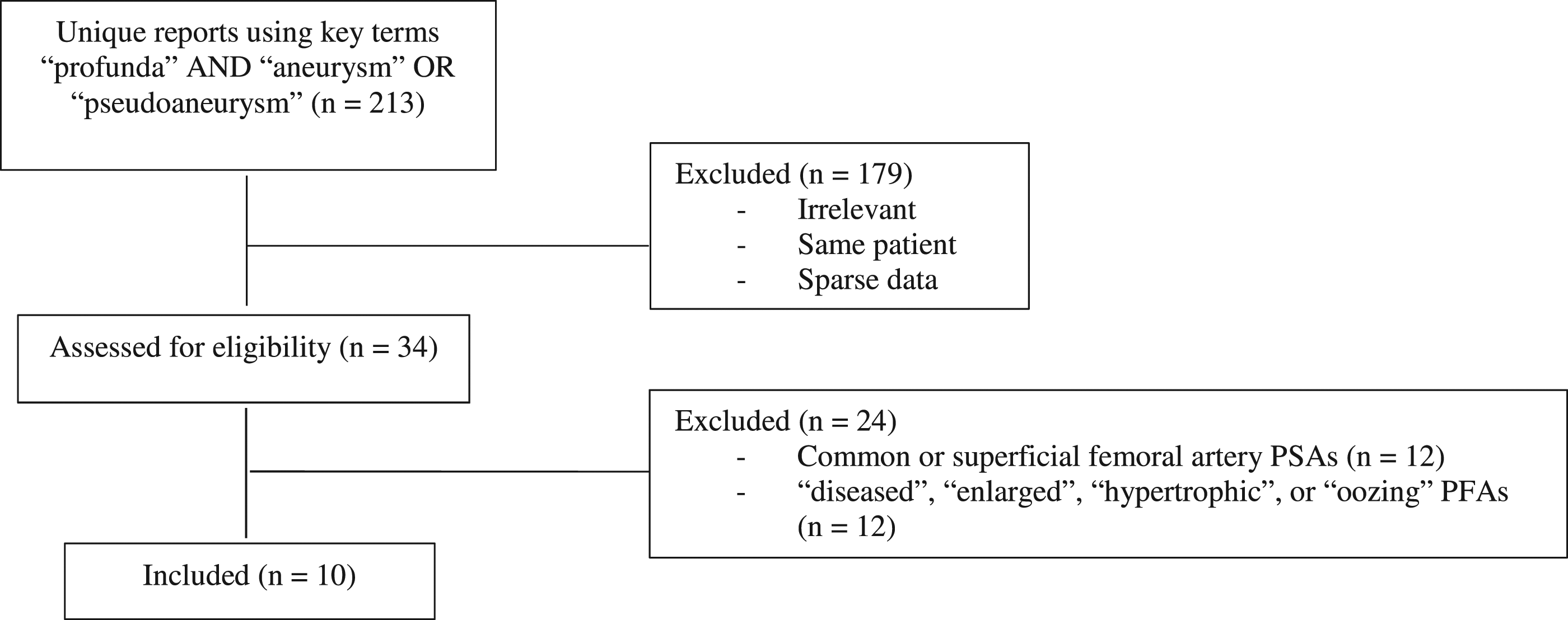
Once cases were verified by the principal investigator (GR), we used the patients’ electronic patient record to capture the following: (1) demographic data (sex, age at the time of presentation or incidental discovery); (2) signs and symptoms on presentation; (3) medical history (smoking status, hypertension, diabetes mellitus II, coronary artery disease, chronic obstructive pulmonary disease, genetic predispositions, previous or synchronous aneurysm(s), peripheral vascular disease); (4) family history (aneurysm(s) and genetic conditions); (5) imaging modalities; (6) aneurysm characteristics (etiology, size, laterality, classification, rupture status); (7) interventions; and (8) their outcomes.
Results
Of the 10 applicable cases, six were PFA PSAs and four were TPFAAs. Due to the differences in etiology between PFA PSAs and TPFAAs, we will describe our results separately.
PFA PSAs
Demographic Data of patients with PFA PSAs and true profunda femoris artery aneurysms.

TPFAA: true profunda femoris artery aneurysm.
Summary of endovascular treatments for PFA PSAs and peri- and post-operative reinterventions.

IU: Intnational Units, GDC: Guglielmi Detachable Coils, pRBC: packed red blood cells, TXA: tranexamic acid.
All PFA PSAs were diagnosed with either computed tomography (CT) or duplex ultrasonography (DUS) (see Figure 2), with CT being more accurate in diagnosing and localizing the aneurysm site. DUS reveals a typical “to and fro” or “yin yang” pattern representing bidirectional flow in the pseudoaneurysm (Figure 2). Duplex ultrasound of a left PFA demonstrating the typical ‘to and fro’ or ‘yin yang’ sign, representing bidirectional flow in the pseudoaneurysm.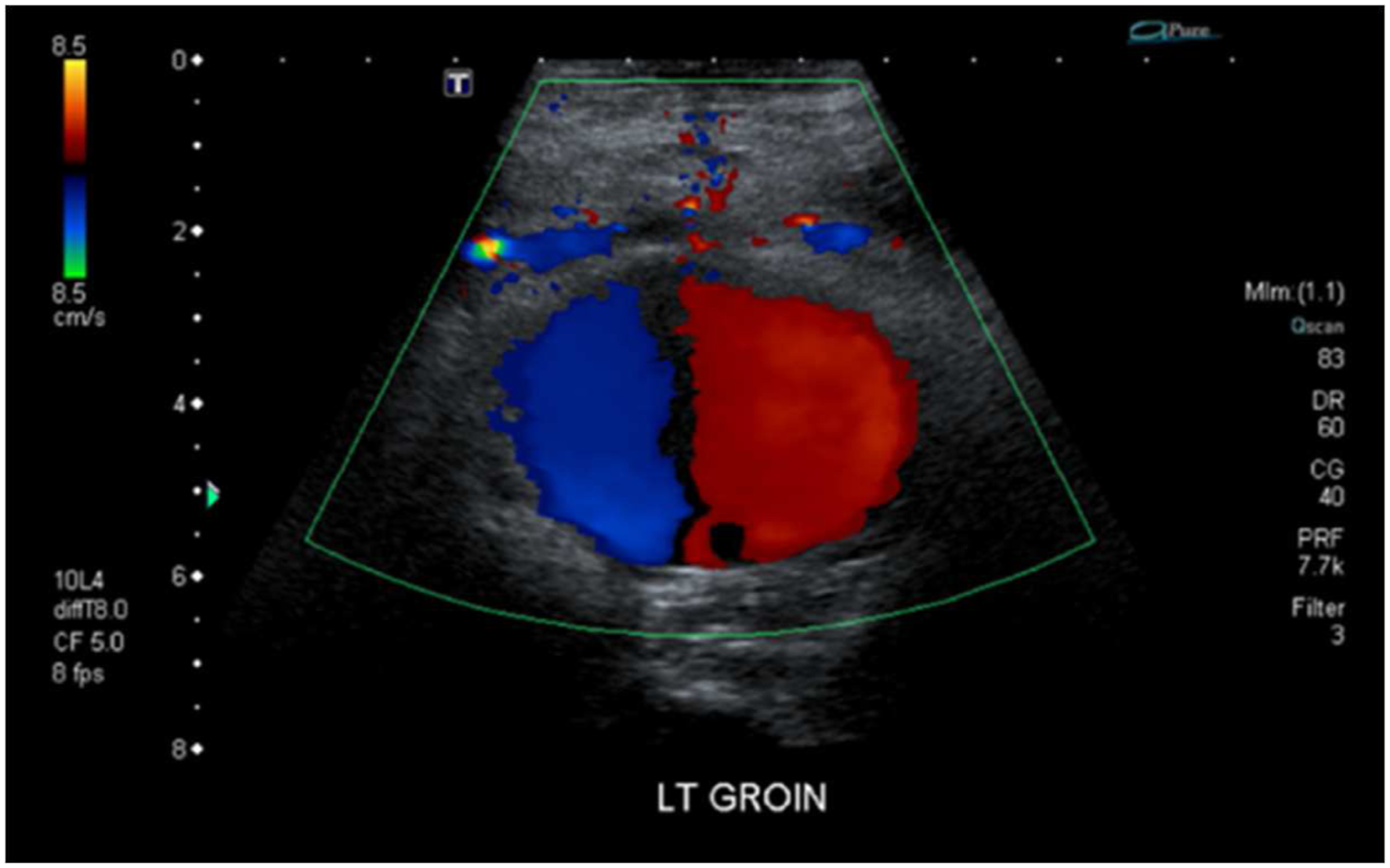
The average size of the PFA PSAs was 58 mm in its largest dimension, ranging from 28 mm to 120 mm. Half of all PFA PSAs were in the proximal PFA and the other half involved branches of the PFA. Half (n = 3) of all PFA PSAs were contained and the other half were actively bleeding. One contained PFA PSA ruptured during a CT angiogram (Table 2, patient 2). The actively bleeding PFA PSAs differed in the time from injury to diagnosis. The pseudoaneurysm that arose due to anastomotic leakage was found to be bleeding by virtue of its etiology. Another bleeding pseudoaneurysm developed secondary to cardiac catheterization and took 21 days for a diagnosis. The patient had groin pain post-catheterization, but DUS did not reveal a PSA. The patient was discharged and ultimately visited the emergency room to address the pain where CT revealed their pseudoaneurysm. The final bleeding pseudoaneurysm took 35 days from injury to diagnosis. The patient underwent repair of a hip fracture where an osseous fragment punctured the PFA. Shortly after, the patient required 1 unit of blood to address low hemoglobin and fluid resuscitation for their acute kidney injury. Later, the patient lost consciousness and upon waking, the patient’s thigh pain increased which sent them to the emergency room for imaging which revealed their pseudoaneurysm. The delay in diagnosis underscores the importance of a high index of suspicion using clinical presentation (painful and swollen mass with anemia) and knowledge of PFA PSA aetiologies.
All PFA PSAs were treated endovascularly, as illustrated in Table 2. Coil embolization of a PSA arising from a PFA branch is depicted in Figure 3 A and B. Importantly, half of our endovascular therapies required a reintervention, evidenced by visualized flow in aneurysm sacs postoperatively by DUS or by presentation to an emergency room with recurrence of symptoms leading to DUS imaging. (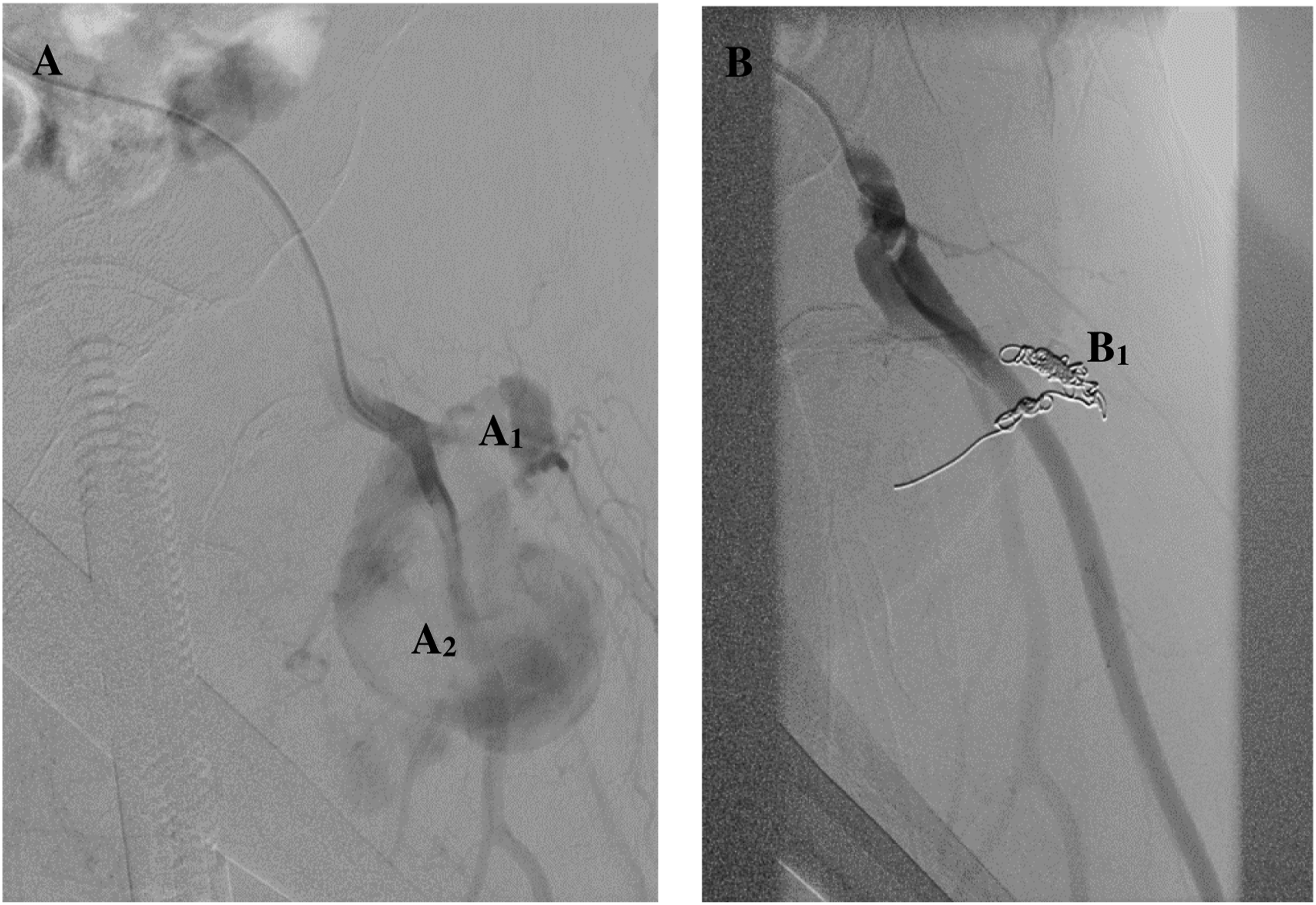
True profunda femoris artery aneurysms
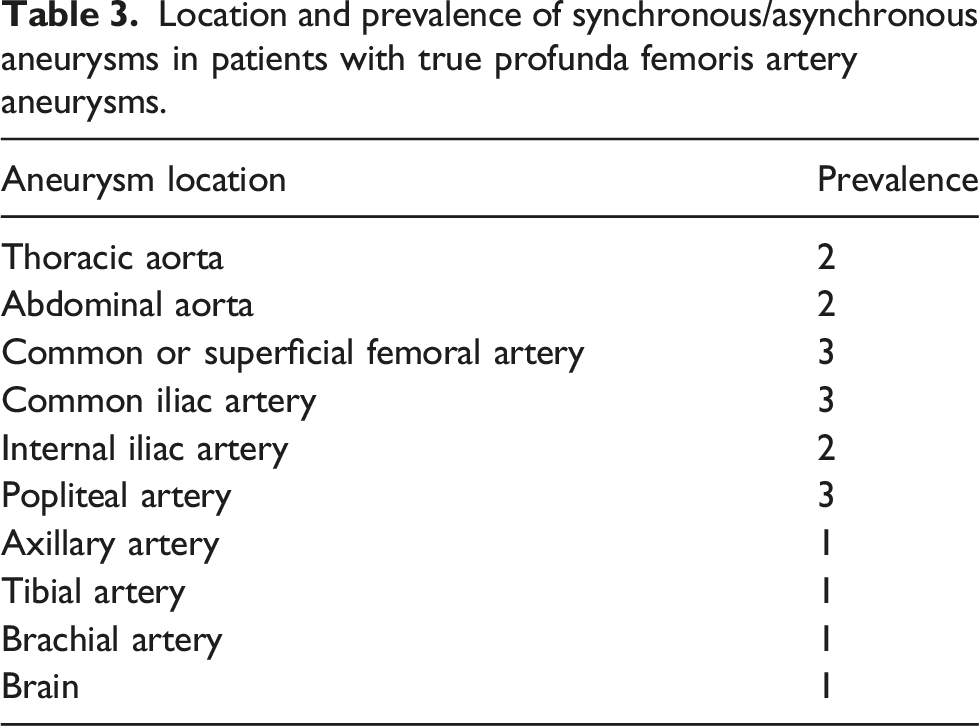
Location and prevalence of synchronous/asynchronous aneurysms in patients with true profunda femoris artery aneurysms.
All TPFAAs were asymptomatic and incidentally discovered using CT imaging (n = 3, 75%) or DUS (n = 1, 25%). Two of the TPFAAs were idiopathic in nature, one TPFAA was present in a patient with Ehlers Danlos Syndrome who had a history of multiple aneurysms, and one TPFAA was determined to arise secondary to post-stenotic dilatation.
Growth and changing characteristics of true profunda femoris artery aneurysms over the duration of surveillance.

TPFAA: true profunda femoris artery aneurysm.
Discussion
The profunda femoris artery (PFA) is the chief supplier of blood to the thigh. Usually, the PFA emerges from the posterolateral aspect of the common femoral artery and runs deep to the adductor longus muscle, tracking the linea aspera of the femur. 12 This deep anatomical location provides the benefit of muscular protection from trauma but can obscure signs and symptoms for timely diagnoses and render surgical access difficult, if an injury involves the PFA. 12
Profunda femoris artery pseudoaneurysms
Femoral artery pseudoaneurysms (involving the common femoral artery, superficial femoral artery, and profunda femoris artery) are rare occurrences that arise most commonly secondary to penetrative iatrogenic injuries. 12 The most common iatrogenic aetiologies of PFA PSAs are catheterizations, orthopedic procedures involving the proximal femur (penetration from either surgical equipment or osseous fragments), and anastomotic pseudoaneurysms.9,12 Non-iatrogenic penetrating injuries include gunshot wounds and stab wounds.13,14 Other causes of PFA PSAs include blunt trauma, infections, hip torsion, and IV drug use.15–18
Our study highlighted the rarity of PFA PSAs, with six cases presented to our quaternary center over the past 15 years. Prevalence among men and women were roughly equal, reflecting the current literature.19–22 Regarding clinical presentation, we observed the triad of thigh swelling, hematoma, and unexplained anemia in all patients, as described by Chong and colleagues. 23 The diversity in aetiologies of PFA PSAs was also congruent with the literature,7–9 as our patients developed PFA PSAs secondary to catheterizations, hip fractures, anastomotic pseudoaneurysms, or without a clear cause. As such, a strong index of suspicion for a PSA PFA should be formulated when integrating the clinical picture of: (1) documented aetiologies; (2) signs and symptoms such as the triad of thigh pain, bleeding, and unexplained anemia 23 ; and 3) diagnostic imaging modalities such as CT, DUS, and angiography. 13
The anatomical locations of our PFA PSAs were localized to the proximal PFA or its branches, with none occurring in the distal PFA. Although DUS has been shown to be over 90% accurate in diagnosing pseudoaneurysms, 26 we found differences in the use of DUS or CT for PFA PSA detection. When DUS was used as an initial investigation, it failed to visualize one PFA PSA adjacent to a large thigh hematoma resulting in a delayed diagnosis, was incorrect regarding the location of two PFA PSAs, and was accurate regarding the presence and location of one PFA PSA. In our study, CT was always accurate in detecting and localizing PFA PSAs. In a patient with a clinical picture suggestive of a PSA, but with no aneurysm detected using DUS, CT can be added as a more accurate imaging modality.
Current therapies for PFA PSAs include US-guided compression, US-guided thrombin or glue injection, endovascular coil embolization, endovascular stent-graft insertion, and open surgical repair. 13 Our management modality depended not only on pseudoaneurysm size, but also on patient status and wishes. For example, open repair was avoided in several poor surgical candidates, while stent-graft insertion was recommended but avoided in a relatively young and active patient based on their fears and concerns. The literature reports successful initial thrombin injections for femoral artery pseudoaneurysms,27,28 although some large series reported a significant proportion of patients who required two or three injections for thrombosis, which depended on aneurysm size. 29 We experienced the need for glue/thrombin reinterventions in half of our patients. The need for our reinterventions did not depend on the aneurysm size, as we had one patient with a 3 cm aneurysm requiring two thrombin injections and another patient with a 5 cm aneurysm who fully thrombosed on the first attempt. Additional reasons for multiple injections include occult vascular injury 30 and technical insufficiencies. 31
True profunda femoris artery aneurysms
The occurrence of TPFAAs is even rarer, accounting for 0.5% of all peripheral artery aneurysms. 19 Furthermore, the most recent systematic review on TPFAA management reported a total of 82 cases in 73 patients reported to date in the literature. 6 Most TPFAAs are due to atherosclerotic wall degeneration among the aged population. 6 As with other arterial aneurysms, a high collagen to elastin ratio as well as decreased smooth muscle cells in the tunica media are evident in TPFAAs. 24 These histological findings mirror the high risk of aneurysm rupture which is significant in TPFAAs (around 18%) 6 and can explain the association of TPFAAs with connective tissue diseases and concurrent or asynchronous aneurysms. 25 Slightly more TPFAAs are symptomatic (painful and pulsatile masses) while the remaining TPFAAs are asymptomatic, most often diagnosed through other aneurysms. 6
There were 4 cases of TPFAAs presented to our institution over the past 15 years. The mean diameter in this study (2.1 cm) is smaller than the mean diameter (5.4 cm) in a large systematic review, 6 which is sensible given that all our cases were asymptomatic and incidentally discovered, while only 30% of TPFAAs in the systematic review were asymptomatic. 6 As the literature suggests, TPFAAs are strongly associated with multiple synchronous or asynchronous aneurysms 25 which we found in our study (Table 3). Importantly, there was a TPFAA accompanied by several synchronous aneurysms, including a brain aneurysm, in a patient with Ehlers Danlos Syndrome. This raises the question of whether cerebral imaging among those with several peripheral arterial aneurysms is necessary to prevent significant morbidity and mortality through early detection; however, the association between peripheral arterial aneurysms and brain aneurysms warrants elucidation before any recommendation can be made. When considering surgical or endovascular management of TPFAAs, one should consider the size of the aneurysm as well as synchronous aneurysms that may take priority in terms of management. We recommend effective triaging of TPFAA and synchronous aneurysm management through a cost-benefit analysis. Otherwise, small (using the smaller size threshold of 2.0 cm) and asymptomatic TPFAAs should not be ignored but carefully monitored annually using DUS, as the rupture rate of TPFAAs is quite high at roughly 20%. 6 There are several therapeutic procedures described for true profunda aneurysms. 7 Surgical repair through ligation, resection, and revascularization remains the most common therapeutic procedure. Surgical techniques include simple ligation, resection of the affected segment with interposition of the autologous vein or prosthetic graft. Endovascular techniques include covered stent graft implantation and endovascular embolization which can be considered for aneurysms in which surgery is not tenable because of patient and aneurysm factors.32,33
Conclusion
Profunda femoris artery aneurysms are indeed rare, with 10 cases presented to our quaternary facility over 15 years. Pseudoaneurysms arising from the PFA are primarily iatrogenic and can present with the triad of thigh swelling, bleeding, and unexplained anemia. The use of multiple imaging modalities (e.g. DUS and CT) is warranted when there is a high index of suspicion for PFA PSAs, but no aneurysm seen on initial imaging. Endovascular embolization is the mainstay treatment for smaller aneurysms and in patients who are poor surgical candidates. As demonstrated in our study and in a large series, 29 endovascular reinterventions (i.e. thrombin, coil, glue) are not uncommon and primary therapy may require multiple injections before complete thrombosis. With regards to small TPFAAs incidentally discovered (usually due to imaging of asynchronous/synchronous aneurysms), careful surveillance is prudent as the rupture rate of TPFAAs is higher than other peripheral arterial aneurysms. 6
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
