Abstract
Background
The screening and diagnosis of intermittent claudication is a challenging process and often relies on the expertise of specialist vascular clinicians. We sought to investigate the diagnostic performance of the Edinburgh Claudication Questionnaire (ECQ) as a screening tool for referrals of suspected intermittent claudication from primary to secondary care.
Method
Prospectively, 100 referrals from primary care with a stated diagnosis or query regarding intermittent claudication were recruited. All participants who completed the ECQ, underwent an anklebrachial pressure index (ABPI) assessment and treadmill exercise testing. Outcomes of the ECQ were compared to clinical diagnoses of intermittent claudication.
Results
The ECQ had a sensitivity of 46.8% (95% CI: 27–65%), specificity of 63.2% (95% CI: 43–82%) and accuracy of 53.0% (95% CI: 43–63%). The diagnostic performance was not changed by combining the ECQ with a positive ABPI or post-exercise ABPI outcome for PAD.
Conclusion
The ECQ had a poor diagnostic performance in this cohort. Considering the results found here and in other recent studies, the utility of the ECQ as a screening tool and epidemiological survey tool must be questioned. Novel, low-resource diagnostic tools are needed in this population.
Introduction
Peripheral arterial disease (PAD) is characterised by atherosclerosis of the arteries supplying the lower limbs, resulting in a reduced blood supply. The prevalence of PAD is estimated to have increased by 23.5% between the years 2000 and 2010 1 with current estimates that around 237 million people are affected globally. 2 PAD is an age-associated disease with its prevalence increasing from 2.5% in those 50–59 years old to 14.5% in individuals >70 years. 3 Though a large proportion of individuals with PAD are asymptomatic, 20–25% of individuals over 60 years old experience symptoms as a result of this haemodynamic compromise. 4 The primary symptomatic manifestation of PAD is intermittent claudication (IC), which is characterised as a reproducible leg pain that occurs during physical activity, and has deleterious effects on quality of life whilst carrying an increased mortality risk.5,6
The screening and diagnosis of IC presents several challenges. At present, there exists no single gold standard test or criteria for diagnosing IC; it relies on a full history, examination, and investigations by an experienced clinician, yet even this is fallible. For the primary care physician, referral of an individual with exertional leg pain to a vascular specialist presents a convenient clinical pathway for appropriate investigations. However, the complex nature of claudication pain may lead to an unknown proportion of unnecessary referrals, presenting an increased workload to vascular services in secondary and tertiary care. This deficiency in the referral process could be ameliorated if there were easily applied IC screening tools to allow triaging of referrals. 12 Accessible PAD assessment methods include pulse palpation and measurement of the anklebrachial pressure index (ABPI). 13 However, pulse palpation has poor diagnostic accuracy 14 and ABPI measurement may not always be available in primary care due to limited equipment and/or appropriate training. The frequent co-occurrence of diabetes with PAD limits the utility of these techniques further. 15 Easy-to-apply diagnostic tools have the potential to overcome such limitations. The Edinburgh Claudication Questionnaire (ECQ) is one such tool that has demonstrated excellent diagnostic performance in the primary care setting. 16 This 6-item questionnaire was developed for epidemiological surveys and has demonstrated excellent sensitivity (91%) and specificity (99%) when compared to the diagnosis made by a primary care physician and performed similarly when compared to a vascular clinician.
Therefore, the aim of this study was to assess the diagnostic performance of the ECQ in the context of a vascular tertiary care centre to assess its suitability for stratifying claudication referrals. A secondary aim is to evaluate whether combining the ECQ with an ABPI assessment would improve its diagnostic performance.
Methods
Consecutive referrals with queries of IC from general practitioners (GP) to a single tertiary vascular centre were considered. Referrals were pre-screened by a member of the clinical team to verify that they contained GP diagnoses or queries of IC before being passed to a member of the research team. Patients were prospectively approached at outpatient vascular clinics from May 2019 to October 2019. Patients were excluded based on the grounds of prior diagnosis of IC. Referrals to eight vascular consultant surgeons were considered. Prior to clinic appointments, patients were asked to complete the ECQ. 16 Patients were excluded if they were unable to complete the questionnaire without assistance due to cognitive impairment or an inability to speak English. Following consultations, patients underwent ABPI assessment and treadmill testing.
ABPI assessment was performed according to the American Heart Association/American College of Cardiology guidelines. 21 Assessments of walking ability were performed using a fixed-speed treadmill test, set at an individualised speed between 1.1 and 2.6 km⋅h−1 and an incline of 10%. Patients were instructed to walk for as long as possible and make assessors aware if they experienced lower limb pain/discomfort during the test. Patients were then encouraged to continue walking until they were no longer able to tolerate the pain or until they had walked for 5 min total. The initial claudication distance (ICD), maximal walking distance (MWD), and whether participants completed the treadmill protocol or were unable to walk were recorded. Assessors were not blinded to ECQ results. Immediately following the treadmill test, participants' ABPI was re-assessed.
A clinical diagnosis was ascertained at a later date through clinical records and confirmed directly with the responsible clinician if there was any ambiguity. Clinicians were blinded to the ECQ outcome. A diagnosis of PAD was confirmed when a patient had an ABPI ≤ 0.9 and/or a post-exercise ankle pressure of < 50 mm Hg and/or a drop of ≥ 30 mm Hg compared to resting values. Additionally, PAD was also confirmed with a positive clinical diagnosis which may have been made using additional diagnostic tests such as duplex ultrasound. A diagnosis of IC was defined using clinical diagnosis alone.
Statistical analysis
Statistical analyses were performed using SPSS (SPSS version 22, Armonk, NY: IBM Corp USA). The diagnostic ability of the ECQ was compared to clinicians' diagnosis, and estimates of sensitivity, specificity, positive predictive value, negative predictive value, and accuracy were calculated. Confidence intervals (CI) for the predictive values and accuracy were calculated using the standard logit method 22 to account for disease prevalence.
This analysis was also performed to assess and compare the performance of a resting ABPI, post-exercise ABPI, and a combined ECQ and resting ABPI measurement (where both are positive) for diagnosing IC compared to a clinician.
Diagnostic performance was also assessed using the area under the curve of the receiver operator characteristic for the ECQ, resting ABPI, post-exercise ABPI and combined ECQ and resting ABPI measures. Additional exploratory analyses were also performed to explore the implications of modifying the ECQ by removal of one or more questions (see Supplementary materials for details).
Results
One hundred patients agreed to participate in the service evaluation (age: 68 ± 9 years; 60% male). All patients completed an ECQ and ABPI assessment, and 98 performed a treadmill test. The prevalence of IC in this cohort was 62%.
The ECQ classified four patients as having atypical leg pain, 3 as having typical “grade 1” claudication, where IC only occurs when walking uphill or quickly, and 36 as having typical “grade 2” claudication which occurs at a normal walking pace on flat terrain.
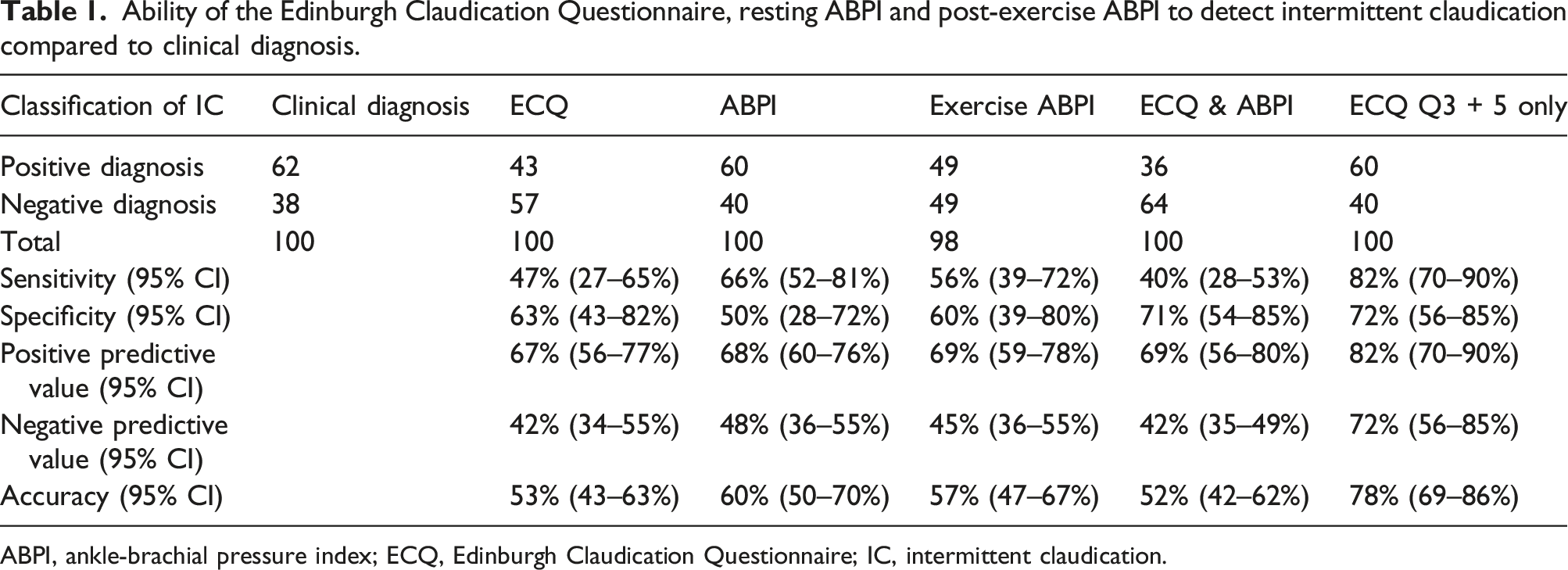
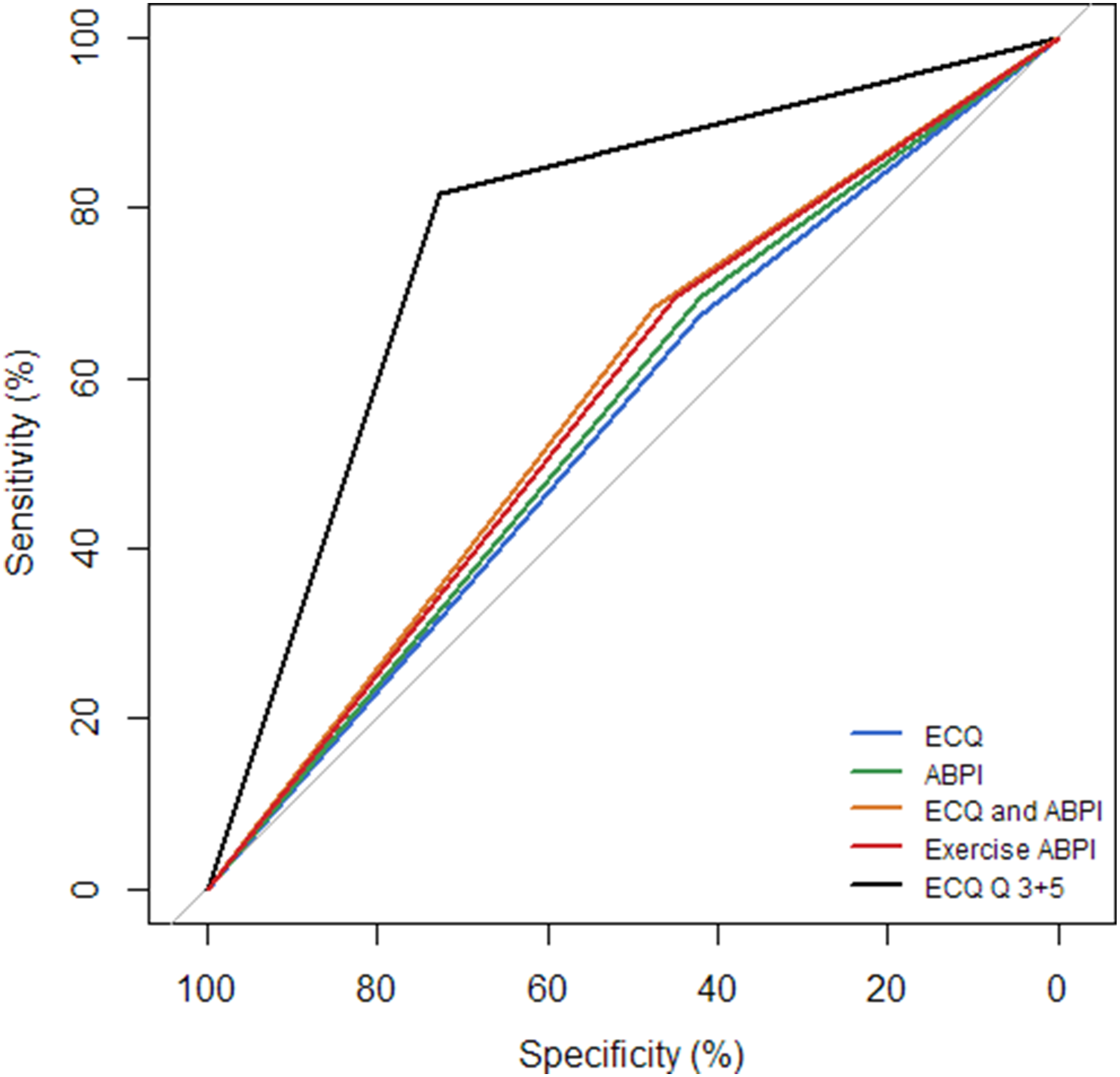
The ECQ correctly classified 29 individuals as having IC and 24 as not having IC and incorrectly classified 14 as having IC and 33 as not having IC (Figure 1). The diagnostic performance of the ECQ, resting ABPI, post-exercise ABPI and combined ECQ and resting ABPI measures against clinical diagnoses of IC are presented in Table 1. All measures presented overlap in the 95% CIs, indicating no clear differences in performance. Combining the ECQ with an ABPI measurement had no observed effect on diagnostic performance. Receiver operating characteristic curves for the ECQ, ABPI, ECQ and ABPI combined, and post-exercise ABPI for detecting intermittent claudication are displayed in Figure 2. The area under the curve for each were as follows: ECQ, 0.55 (95% CI [0.43–0.67]); resting ABPI, 0.59 (95% CI [0.47–0.70]); post-exercise ABPI was 0.58 (95% CI [0.46–0.69]); and combined ECQ and resting ABPI, 0.56 (95% CI [0.44–0.67]). No differences were observed between these outcomes. Exploratory analysis revealed that a more accurate diagnosis was achieved in this cohort with the ECQ if only the responses to questions three and five were acted upon (Supplementary Table 1); hence, the diagnostic performance of this simplified version is also presented in Table 1. The area under the curve of the receiver operator characteristics of this model was 0.77 (95% CI [0.69–0.87]). Diagnostic performance of the Edinburgh Claudication Questionnaire for detecting intermittent claudication. Ability of the Edinburgh Claudication Questionnaire, resting ABPI and post-exercise ABPI to detect intermittent claudication compared to clinical diagnosis. ABPI, ankle-brachial pressure index; ECQ, Edinburgh Claudication Questionnaire; IC, intermittent claudication. Receiver operating characteristic curves are presented for detecting intermittent claudication for the Edinburgh Claudication Questionnaire (ECQ), ankle-brachial pressure index (ABPI), ECQ and ABPI combined, post-exercise ABPI, and the ECQ when only questions 3 and 5 were considered. The area under the curve for each was as follows: ECQ, 0.55 (95% CI [0.43–0.67]); resting ABPI, 0.59 (95% CI [0.47–0.70]); post-exercise ABPI was 0.58 (95% CI [0.46–0.69]); combined ECQ and resting ABPI, 0.56 (95% CI [0.44–0.67]); and the ECQ when only questions 3 and 5 were considered, 0.77 (95% CI [0.69–0.87]).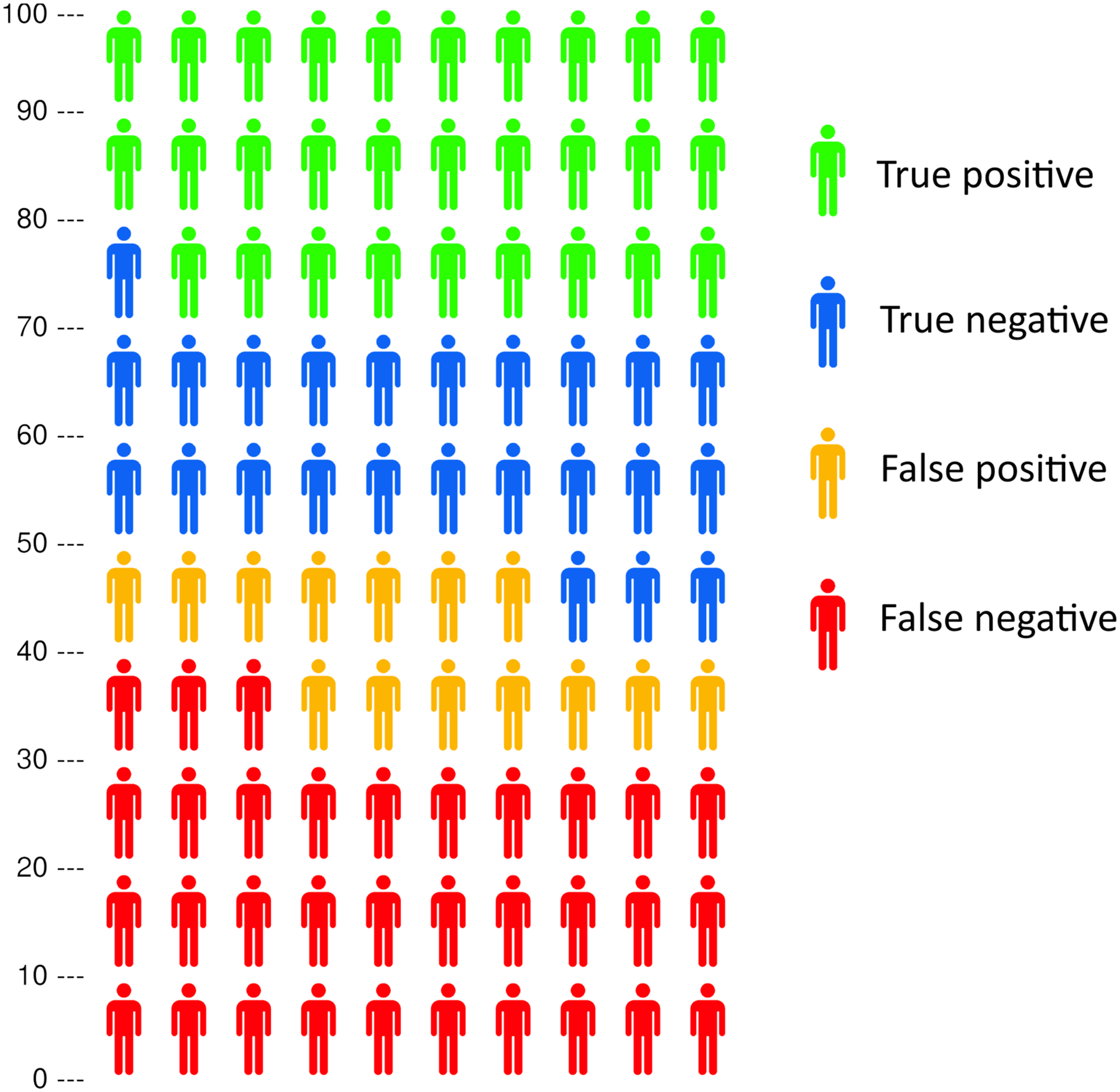
Ability of the Edinburgh Claudication Questionnaire to detect PAD.
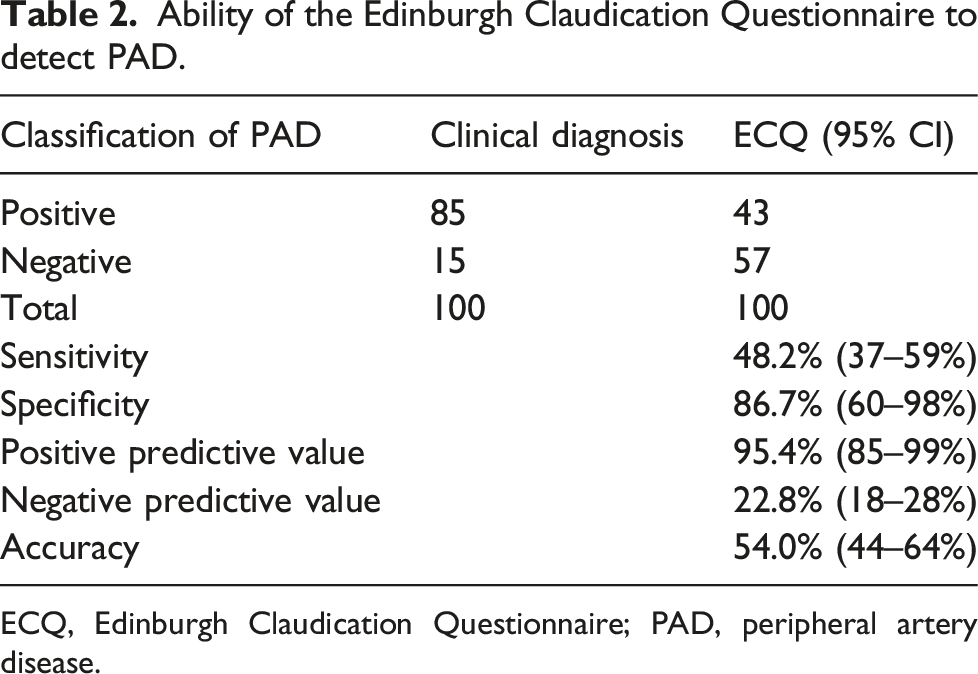
ECQ, Edinburgh Claudication Questionnaire; PAD, peripheral artery disease.
Discussion
The diagnostic performance of the ECQ to diagnose or exclude IC in a cohort of referrals to a tertiary vascular centre is poor, in contrast with previous research. Combining the ECQ with an ABPI assessment to confirm IC had no effect upon diagnostic accuracy. In light of these findings, the use of the ECQ in its present form as a clinical tool to stratify patient referrals, as an inclusion criterion for research trials or as a data collection tool in epidemiology, appears inappropriate. Its use in such contexts could have profoundly negative effects resulting in misleading research findings and the potential for wasteful resource allocation. There may however be potential to optimise the ECQ by removal or modification of existing questions.
All indicators of diagnostic performance were lower in this study than those of the original study of Leng et al. 16 The prevalence of IC in this study cohort was similar to Leng et al.’s cohort of ‘clinic patients’ (62% vs 58%) albeit using half the sample size of present study. This is the first study to question the ECQ’s diagnostic performance in IC. However, the present study does not replicate the original study design, and as such, there are several possible explanations for why our results differ.
One key methodological difference is in the type of clinician making a diagnosis in each study. Leng et al. 16 validated the ECQ using the diagnosis of IC from GPs in the absence of other diagnostic tests in one cohort and using the diagnosis of a vascular specialist clinician (non-consultant) with access to ABPI and exercise tests in a separate cohort of clinic patients. In comparison, diagnoses in the present study were made by consultant vascular surgeons with access to additional diagnostic tests such as duplex ultrasound and angiography where necessary. The extent the diagnostic ability of the clinicians may have differed between the studies is uncertain. It is reasonable to assume that having greater access to advanced diagnostic imaging tests that were less available 25 years ago may predispose the clinicians in the current study to a greater diagnostic accuracy. Should this be true, it is appropriate to evaluate tools such as the ECQ against this modern standard of care. An accuracy of 60% in GP referrals demonstrated here suggests that GP diagnoses should not be used to validate a tool such as the ECQ and that there is a need for tools to improve diagnostic accuracy in primary care. Another factor which may explain the different findings in this study is the variation in patients being assessed, derived from asynchronous cohorts from small geographical areas. It cannot be ascertained to what extent factors such as co-morbid load, education and socioeconomic status may have contributed to the lower accuracy of the ECQ in this modern cohort.
An exploratory analysis of the implications of using different permutations of the questions included in the ECQ revealed that in this cohort, a superior diagnostic performance was observed using versions of the survey that omitted question 2: ‘Does this pain ever begin when you are sitting or standing?’ Very similar diagnostic performance was observed when only including question 1 and/or question 3 with question 5 (Supplementary Table 1). Such a modification to the ECQ appeared to transform the performance of the tool from useless to potentially useful. It is important to consider why this alteration in performance was observed. There are numerous unmeasured factors relating to the cohort in the present study and Leng’s original cohort that may have led to differing comprehension of and responses to question 2. One possible explanation for this discrepancy could be that patients’ perceptions of pain varied between studies. Ischaemic pain, secondary to PAD, is complex and may have nociceptive, inflammatory and neuropathic elements 8 and as such, perceptions of claudication pain vary considerably. 9 Only 10–35% of individuals with PAD present with leg pain that is ‘typical’ for IC, that is, originating in the calf, only commencing upon exertion and quickly being relieved by rest. 7 Approximately 20% of elderly people report leg pain whilst walking 10 and there is a range of painful pathologies which are associated with age such as knee and hip osteoarthritis, diabetic neuropathy, muscle strains and compartment syndrome. 11 Arguably, question 2 from the ECQ may not adequately distinguish between true claudication pain and many other pathologies.
IC is caused by PAD. PAD, in the form of a stenosis of ≥ 50%, is detected by APBI with a high degree of reliability and accuracy.23–25 It might be expected that applying a criterion that required both a positive ECQ and ABPI to classify IC would improve the specificity of classifications. However, only marginal effects were observed compared to the ECQ alone (Table 1). This is possibly due to the high prevalence of PAD in this cohort and the large proportion of false negatives classified by the ECQ. In this study, the ECQ was good at ruling out PAD (specificity: 87%) but not detecting it (sensitivity: 48%). Two other UK studies have found similar performance for detecting PAD in individuals with leg pain using the ECQ, with Boylan et al. 26 finding a sensitivity of 53% and a specificity of 87% and Poots et al. 27 finding a sensitivity of 57% and specificity of 82%. Criqui et al. 14 found worse results using the Rose Claudication Questionnaire with a sensitivity of 9.2% and specificity of 99% for detecting large-vessel PAD. At present, it appears that questionnaires are inadequate substitutes for ABPI assessments for triaging individuals with PAD.
The ECQ is widely used in epidemiological surveys and as a screening tool for inclusion in research trials in populations with IC in secondary/tertiary care settings.17–20 The ECQ is the only survey for claudication which has been validated against the diagnoses of GPs and vascular specialist clinicians. Similarly designed widely used surveys such as the WHO/Rose Questionnaire 7 and San Diego Claudication Questionnaire 28 were not validated against a gold standard before implementation. In the case of IC, the current gold standard assessment is an experienced vascular clinician’s diagnosis. Despite this, the Rose and San Diego Claudication questionnaires are responsible for most of the epidemiological estimates of the prevalence of IC to date. Leng et al. 16 estimate that the Rose Questionnaire only has a sensitivity of 60% (95% CI [56–64%]) and specificity of 91% (95% CI [85–99%]) in identifying IC diagnosed by a consultant. The San Diego Claudication Questionnaire’s validity and accuracy is assumed to be the same as the ECQ. 9 This study has demonstrated that the ECQ is not informative. Thus, the ECQ and by extension the San Diego Claudication Questionnaire should not be recommended as a data collection tool, and previous research adopting these tools or the Rose Questionnaire should be interpreted with caution.
Many epidemiological studies have used the ECQ and ABPI measurement to assess the prevalence of IC and PAD, respectively, and suggest a greater relative risk of IC in individuals with PAD. In a cohort of 30,025 Chinese adults > 35 years, Wang et al. 29 found a prevalence of IC of 0.3% using the ECQ and a prevalence of PAD of 5.8%. Si et al. 30 found in a population of 2489 Australian adults (∼72 years) that the prevalence of IC was 10.9% according to the ECQ. Davies et al. 31 report a 3% prevalence of IC using the ECQ in a UK population of 1101 adults > 45 years with an elevated CVD risk. The prevalence of ECQ defined IC in individuals with an ABPI < 0.9 was greater than those with an ABPI of > 0.9 with relative risks of 10.4 (95% CI [8.0–13.6]), 1.6 (95% CI [1.3–1.8]) and 13.9 (95% CI [5.9–32.7]), respectively, confirming that individuals with a positive ABPI are more likely to have a positive ECQ. The differences in IC prevalence between these studies are likely related to different demographics but may also be explained in part by a bias caused by nurses assisting with the completion of the questionnaire by Davies et al., 31 whereas Si et al. 30 had patients complete the ECQ unaided. It is unclear whether the participants in Wang et al.’s 29 study received assistance completing the questionnaire. Basgoz et al. 32 found that completion of the questionnaire led by a trained interviewer rather than self-administration resulted in a sevenfold higher rate of individuals receiving a positive ECQ diagnosis. Whether this assistance improves the accuracy of the ECQ is not known, and it is unclear whether any assistance was given to the original Leng et al. 16 cohort.
Current estimates of the prevalence of IC based on ECQ data may be inaccurate. With the ECQ, we observed a false positive rate of 14% and false negative rate of 33% in a population with a 62% prevalence of IC. Crudely, our data suggests that the true prevalence of IC in studies using the ECQ may be around 44% (95% CI [24–75%]) higher than previously thought. A more precise revision of previous estimates of the true prevalence of IC from epidemiological survey using the ECQ is desirable, though the poor accuracy of the ECQ precludes the use the statistical techniques necessary to achieve this. 33 An alternative approach would be to make inferences about IC prevalence using epidemiological data derived from healthcare utilisation; however, this is also likely to underestimate the prevalence of IC. It is supposed that 10–50% of individuals who suffer with this treatable condition never consult a doctor 4 which may be as result of misappropriation of the symptoms of IC as a normal part of the ageing process or a lack of physical fitness. 31 Knowledge and awareness of PAD by the public and non-specialist healthcare practitioners is poor. Less than 2% of people with PAD are aware they have it, 29 and less than 20% who have received a diagnosis of PAD are able to identify IC. 34 As such, IC is most likely underdiagnosed and undertreated. Notwithstanding their current shortcomings, survey-based methods of estimating the prevalence of IC remain favourable due to their low-resource use and potential for wide distribution. Clearly, superior research tools are needed to produce accurate estimates of the prevalence of IC.
Further implications of our findings are that the ECQ is not an appropriate tool for stratifying patient referrals to vascular services nor for use as an inclusion criterion for research trials. The accuracy of the ECQ is not sufficient for us to recommend its use, even in conjunction with a positive ABPI. This is particularly true of clinical trials performed in secondary and tertiary care settings. The poor diagnostic performance of the ECQ in this cohort was rectified by modification of the questions included; however whether this improved performance occurred due to characteristics that were unique to this cohort cannot be ascertained without verification of this observation in other cohort studies. As such, the use of a modified version of the ECQ cannot be recommended at present.
Limitations
Our study had a number of limitations. Our data reflect patients at a single UK vascular unit, and results may not be generalisable to patients referred for IC at other institutions where clinical diagnostic processes or patient characteristics may vary. It is possible, though unlikely, that ‘clinic patients’ in the original ECQ study 16 represented a different demographic, with a different disease severity possibly due to variations in the referral pathway compared to our study population. However, this cannot be readily ascertained from the data collected in both studies.
We are unable to assess whether there is a bias in referrals to vascular services for complaints of lower limb pain due to greater accessibility of outpatient services compared to other relevant specialities, though the reasonable proportion of accurate referrals would refute this. Exclusion of non–English-speaking patients and individuals with cognitive impairment may limit the generalisability of our findings.
Conclusion
This study has found that the diagnostic performance of the ECQ is poor in leg pain referrals to a tertiary care setting. This leads to questions about the utility of this questionnaire and the implications of its use in epidemiological and experimental research. Specifically, the findings presented in this study suggest that estimates of the prevalence of IC based upon the ECQ may not be accurate. We recommend against the use of the ECQ in both a routine clinical and research-based setting. There is a clear need for more accurate questionnaires to accurately diagnose IC. Preliminary data suggests that it may be possible to achieve this with minor amendments to the ECQ.
Supplemental Material
sj-pdf-1-vas-10.1177_17085381211059665 - Supplemental material for The Edinburgh Claudication Questionnaire has poor diagnostic accuracy in people with intermittent claudication
Supplemental material, sj-pdf-1-vas-10.1177_17085381211059665 for The Edinburgh Claudication Questionnaire has poor diagnostic accuracy in people with intermittent claudication by Saïd Ibeggazene, Andrew Stirrup, Sean Pymer, Joanne Palmer, Paris L Cai, George E Smith and Ian C Chetter in Vascular
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this service evaluation project was provided by Hull University Teaching Hospitals NHS Trust.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
