Abstract
Objectives
Thoracic endovascular aortic repair (TEVAR) is the standard of care for descending thoracic aortic aneurysms (DTAA), and newer generation stent grafts have significant design improvements compared to earlier generation devices.
Methods
We report the first commercial use of the Medtronic Valiant Navion stent graft for treatment of an 85-year-old woman with a 5.8 cm DTAA and a highly tortuous thoracic aorta.
Results
A percutaneous TEVAR was performed using a two-piece combination of the Valiant Navion FreeFlo and CoveredSeal stent graft configurations for zones 2–5 coverage. The devices were successfully delievered through highly tortuous anatomy and deployed, excluding the entire length of the aneurysm with precise landing, excellent apposition and no evidence of endoleak. The patient tolerated the procedure well and has had no stent graft-related complications through one-year follow-up.
Conclusions
Design enhancements such as a lower profile delivery system, better conformability, and a shorter tapered tip are some of the improvements to this third-generation TEVAR device. Coupled with the multiple configuration options available, this gives physicians a better tool to treat thoracic aortic pathologies in patients with challenging anatomy. The early results are encouraging, and evaluation of long-term outcomes will continue.
Introduction
Thoracic endovascular aortic repair (TEVAR) is the current standard of care for descending thoracic aortic aneurysms (DTAA) repair in patients with suitable anatomy, as there is a significant morbidity and mortality benefit when compared to open surgical repair according to clinical trials and recent guidelines.1–3 First-generation stent grafts were associated with higher rates of complications and reinterventions, especially in patients with unfavorable anatomy. These anatomical challenges inspired the innovation of newer generation devices. The Valiant Navion stent graft system (Medtronic Inc., Santa Rosa, CA) is a third-generation TEVAR device with favorable 30-day outcomes reported in its recently published pivotal clinical trial. 4
We present the first commercial use of the Valiant Navion stent graft system in an 85-year-old female with a 5.8 cm proximal DTAA and a highly tortuous thoracic aorta. This case report follows the guidelines of the SCARE criteria. 5
Presentation of case
An 85-year-old female with history of hypertension, hyperlipidemia, coronary artery disease and former tobacco use presented with an asymptomatic 5.8 cm proximal DTAA. A computerized tomography (CT) scan revealed severe tortuosity and angulation of her thoracic aorta. The proximal extent of the DTAA abutted the origin of the left subclavian artery (LSA) with an average proximal neck diameter of 32.8 mm (Figure 1). Treatment options were discussed with the patient including medical therapy with follow-up surveillance vs. intervention, and the patient’s decision was to proceed with immediate repair.
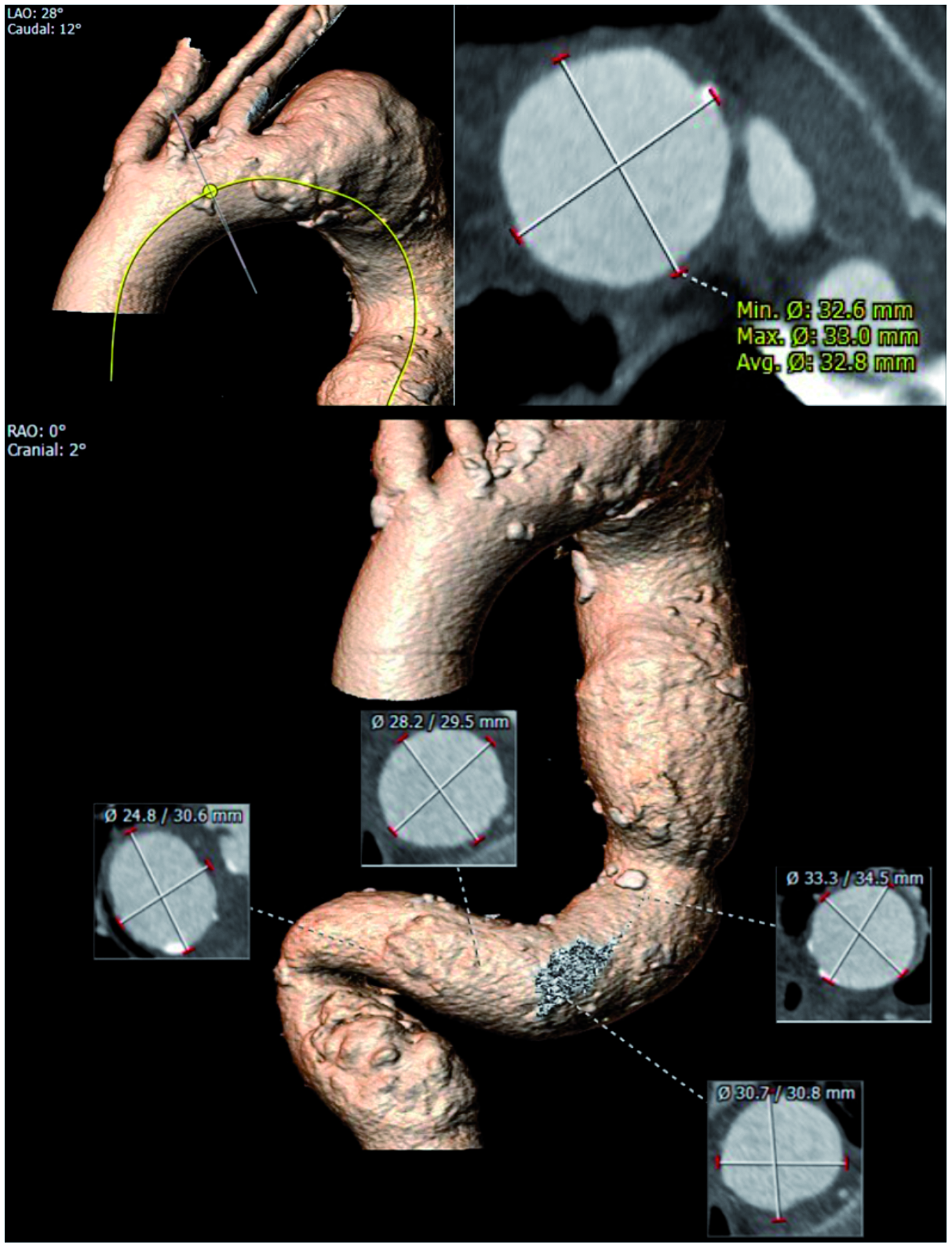
Computerized tomography 3D reconstruction showing the extreme angulation and tortuosity of the descending thoracic aorta in the first commercial patient treated with the Valiant Navion stent graft system.
Based on the patient’s anatomy, TEVAR with zones 2–5 coverage was planned in staged fashion. A LSA to common carotid artery transposition was successfully performed one month prior to TEVAR. The patient received general anesthesia, and a cerebral spinal fluid lumbar drain was placed. Bilateral percutaneous femoral access was obtained. A Medtronic Valiant Navion 37 mm × 229 mm FreeFlo stent graft was used as the proximal device (Figure 2(a)) with a 43 mm × 218 mm CoveredSeal graft extending distally to zone 5, excluding the entire aneurysm (Figure 2(b)). Completion angiography (Figure 2(c)) revealed complete exclusion of the DTAA with excellent apposition and no evidence of endoleak.
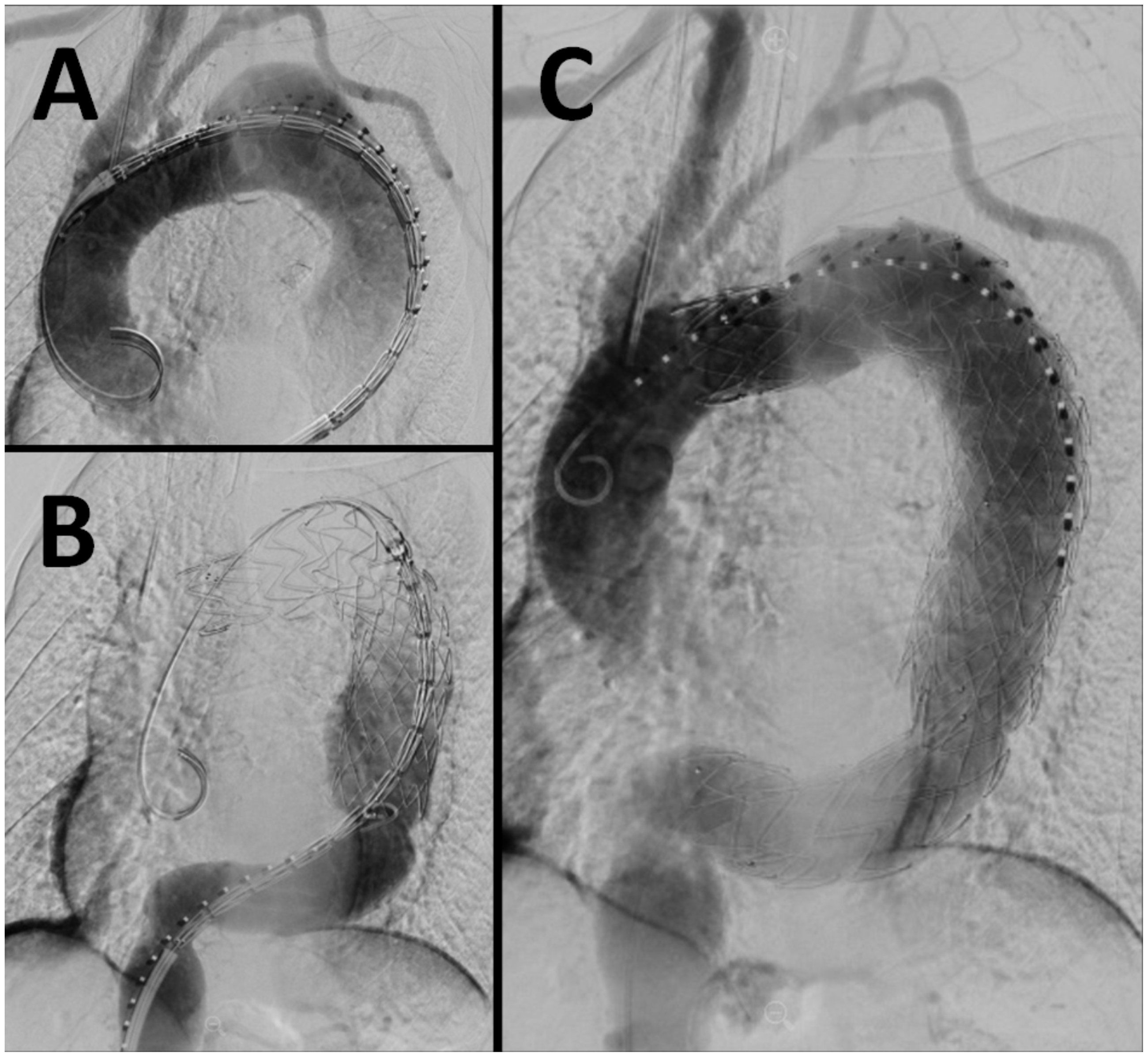
(a) accurate positioning of a proximal FreeFlo graft was achieved with good apposition to outer wall, (b) a distal CoveredSeal graft was deployed to cover the entire length of aneurysm, (c) completion angiogram showing good placement of both grafts and no evidence of endoleaks. The patient tolerated the procedure well.
The patient tolerated the procedure well and was discharged on post-operative day three. No stent graft-related complications and no reinterventions were reported through one-year follow-up.
Discussion
This patient with a 5.8 cm proximal descending TAA and complex anatomy was successfully treated with the Medtronic Valiant Navion stent graft with no complications or reinterventions after one year. The lower profile delivery system, conformability of the stent graft, easy controlled deployment and proximal configuration options are the design aspects that increase the likelihood of improved clinical outcomes in more complicated TEVAR cases. Results from the recent pivotal trial for the Valiant Navion system demonstrated benefits in all-cause mortality, aneurysm-related mortality, and major adverse rates when compared to the earlier generation Captivia device.2,4
This third-generation TEVAR device has two proximal configuration options (FreeFlo proximal bare stent or CoveredSeal) and is available in various sizes, tapered and straight, allowing for treatment tailored to the patient’s anatomy and pathology. Although severe access and aortic tortuosity was reported in 70.9% and 85.7% of patients, respectively, there were no access or deployment failures, and only one Type Ia endoleak was reported, which did not require intervention. 4 While there is some concern with the use of a bare stent device at the proximal landing zone due to potentially higher risk of retrograde type A dissections, this event is uncommon (1.1% of trial population) and current literature is ambiguous as to the effect of bare stent designs.6,7 The Valiant Navion has shown promising early results and we look forward to continued follow-up to assess the long-term performance of this stent graft system.
Footnotes
Acknowledgements
The authors acknowledge Ming-Jay Chow, Medtronic Inc., for his assistance in the preparation of the article. Consent was obtained from the patient.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA is on the speaker’s bureau for Medtronic Inc. All other authors have no conflicts of interest related to this article to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
