Abstract
Objectives
Various configurations of kissing stent (KS) configurations exist and patency rates vary. In response the covered endovascular reconstruction of the aortic bifurcation configuration was designed to minimize mismatch and improve outcome. The aim of the current study is to compare geometrical mismatch of kissing stent with the covered endovascular reconstruction of the aortic bifurcation configuration in vivo.
Methods
Post-operative computed tomographic data and patient demographics from 11 covered endovascular reconstruction of the aortic bifurcation and 11 matched kissing stent patients were included. A free hand region of interest and ellipse fitting method were applied to determine mismatch areas and volumes. Conformation of the stents to the vessel wall was expressed using the D-ratio.
Results
Patients were mostly treated for Rutherford category 2 and 3 (64%) with a lesion classification of TASC C and D in 82%. Radial mismatch area and volume for the covered endovascular reconstruction of the aortic bifurcation group was significantly lower compared to the kissing stent configuration (P < 0.05). The D-ratio did not significantly differ between groups. Measurements were performed with good intra-class correlation. There were no significant differences in the post-procedural aortoiliac anatomy.
Conclusions
The present study shows that radial mismatch exists in vivo and that large differences in mismatch exist, in favour of the covered endovascular reconstruction of the aortic bifurcation configuration. Future research should determine if the decreased radial mismatch results in improved local flow profiles and subsequent clinical outcome.
Keywords
Introduction
The kissing stents (KS) configuration is frequently used for aortoiliac occlusive disease (AIOD) and there is a shift towards endovascular treatment of more complex lesions (type TASC C and D). KS configurations usually protrude into the distal aorta to treat the lesions completely. Due to incomplete contact between the stent and vessel lumen, mismatch areas are created. These areas are referred to as ‘radial mismatch’ or ‘protrusion mismatch’ (Figure 1) and several studies have indicated a negative relation between mismatch and stent patency.1–6 The underlying principle is likely to be flow perturbations that, in turn, could cause low wall shear stress (WSS) and thrombus formation.
7
The covered endovascular reconstruction of the aortic bifurcation (CERAB) technique was developed in an attempt to reconstruct the native aortic bifurcation in a more physiological fashion.8,9 With this technique a covered stent is expanded 15–20 mm above the aortic bifurcation and proximally adapted to the aortic wall with a larger balloon, thereby creating a cone-shaped stent. Two iliac covered stents are placed in the distal conic segment and simultaneously inflated, making a tight connection with the aortic stent thus creating a new bifurcation.
Overview of the KS technique (left) using two self-expandable stents and the CERAB technique (right) using three balloon expandable stents. For each configuration two cross sections are shown to depict the size of the in vitro radial mismatch.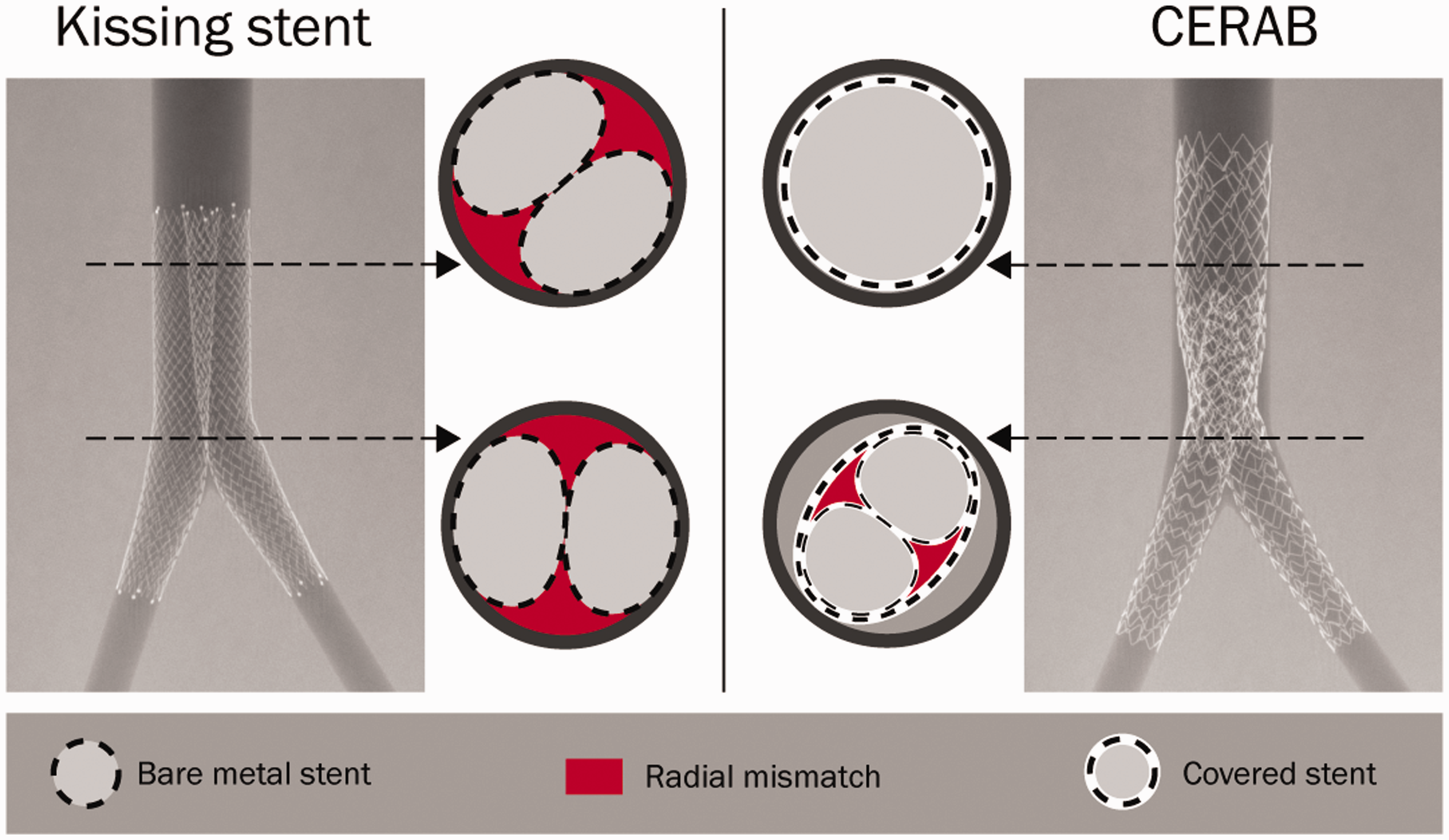
Previously, we have shown in vitro that the CERAB indeed better resembled the normal anatomy compared to KS. 10 It is questionable, however, how these results translate to the in vivo situation as local anatomy, including calcified lesions, might affect mismatch areas. In the present study, we compared the anatomy and stent geometry of two cohorts of patients treated with either the KS or CERAB technique with the primary outcome measure being radial mismatch.
Methods
Patient selection
Patients treated with the CERAB or KS configuration were identified in two hospitals. One centre solely applied the KS technique using two simultaneously deployed self-expandable, bare metal, nitinol stents (Wallstent, Schneider Inc., Minneapolis, MN, USA). These stents were deployed with the proximal end into the aorta and post-dilated. 11 The second centre solely applied the CERAB technique in case of AIOD using balloon expandable covered stents (Advanta V12, Atrium Maquet, Getinge, Hudson, MA, USA) as previously described. 9 Post-operative anticoagulation consisted of clopidogrel (3–6 months) and acetylsalicylic acid (lifetime) or marcoumar (3 months) and acetylsalicylic (lifetime), for the CERAB and KS groups, respectively. Treatment of AIOD was only indicated if the aortoiliac lesions were hemodynamically significant (>70% on computed tomographic angiography (CT-A) or Peak Systolic Velocity ratio > 2.5).
Inclusion criteria were:
KS or CERAB as primary treatment option. Primary patency achieved at ≥ 3 months. 3 month post-operative computed tomography (CT-A) data available. KS or CERAB configuration extending into an aneurysmatic infrarenal aorta.
Exclusion criteria were:
Three month post-operative CT-A imaging after KS or CERAB treatment is not standard care at both hospitals and, as such, the included CT-A scans were mostly indicated for other reasons. In order to have comparable groups the patients in the KS group were matched to the CERAB population, in subsequent order: TASC II classification, age (in a range of ± 5 years) and gender. No matching was performed based on the lesion types within TASC II categories. No data on patient outcome were retrieved, as the groups are too small for reliable outcome comparison. Human investigation review board approval was obtained for this study; patient informed consent did not have to be obtained.
Data analysis
Anatomical characteristics were analysed using post-operative CT-A data using the Terarecon (Aquarius, Foster City, CA, USA) software tool. A centre lumen line (CLL) was automatically constructed and manually checked in all cases. Thereafter outer to outer diameter (front to back and left to right for each slice) and length measurements were performed along the CLL. Length and diameter measurements in the aorta were performed from the lowest renal artery to the apex of the aortic bifurcation. Measurements in both iliac arteries were performed from the apex of the aortic bifurcation to the apex of the iliac bifurcation. In all cases step size along the CLL was 10 mm. Furthermore, the stent placement with respect to the aortic bifurcation was determined.
The mismatch area and stent confirmation (‘double-D’ shape) were determined using a dedicated tool designed in Matlab.
10
The mismatch area was determined using two methods, a manual segmentation (region of interest (ROI) method) and an ellipse approximation (correction was applied for overlapping ellipses). Figure 2 gives an overview of both methods. In case of a KS configuration the three ellipses were fitted on the vessel wall and stent perimeter. With the CERAB configurations the ellipses were fitted to the perimeter of the body and both legs. Stent conformation was expressed as D-ratio, calculated as the ratio between the major and minor axes of the ellipses fitted to the stent lumen (the blue circles in Figure 2(a)).
Overview of both segmentation methods, shown with the CERAB configuration. (a) Application of three ellipses to represent the configuration, the yellow ellipse defines the CERAB cuff (in case of a KS configuration this is the vessel wall), the two blue circles represent the lumina of both stents. (b) Manual segmentation of the mismatch area (ROI) between the stents and the cuff (in case of a KS configuration this is the vessel wall).
For the KS patients CT scans were made with a slice thickness between 1 and 3 mm, 120 kVp (135 kVp in one case) and mAs ranging between 45 and 235. The CT scans of the CERAB patients were made with a slice thickness between 1 and 4 mm, 110 or 120 kVp, mAs between 35 and 107. The matrix size was 512 × 512 pixels for all scans.
Statistical analysis
Data are presented as mean and standard variation, unless stated otherwise. The Shapiro–Wilk test was used to test for normality. Although data were normally distributed non-parametric testing was used due to the small sample size per group. Non-parametric testing (Mann–Whitney U test) was used to compare the patient anatomy, stent characteristics, mean radial mismatch, total mismatch volume and D-ratio. The non-parametric Chi-square test was used to compare risk factors, the Rutherford category and the TASC category. Bland–Altman plots were used to compare the ROI and ellipse method. To assess the consistency and reproducibility of the measurements the intra-class correlation (ICC) and 95% confidence interval (CI) were displayed for all measurements, and an ICC > 0.7 was considered as good agreement between the observers. All measurements were performed by two researchers (TTM and EGJ). A significance level of 5% was used. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 23 (IBM Corp, Armonk, NY, USA).
Results
Patient demographics
Patients’ demographics.
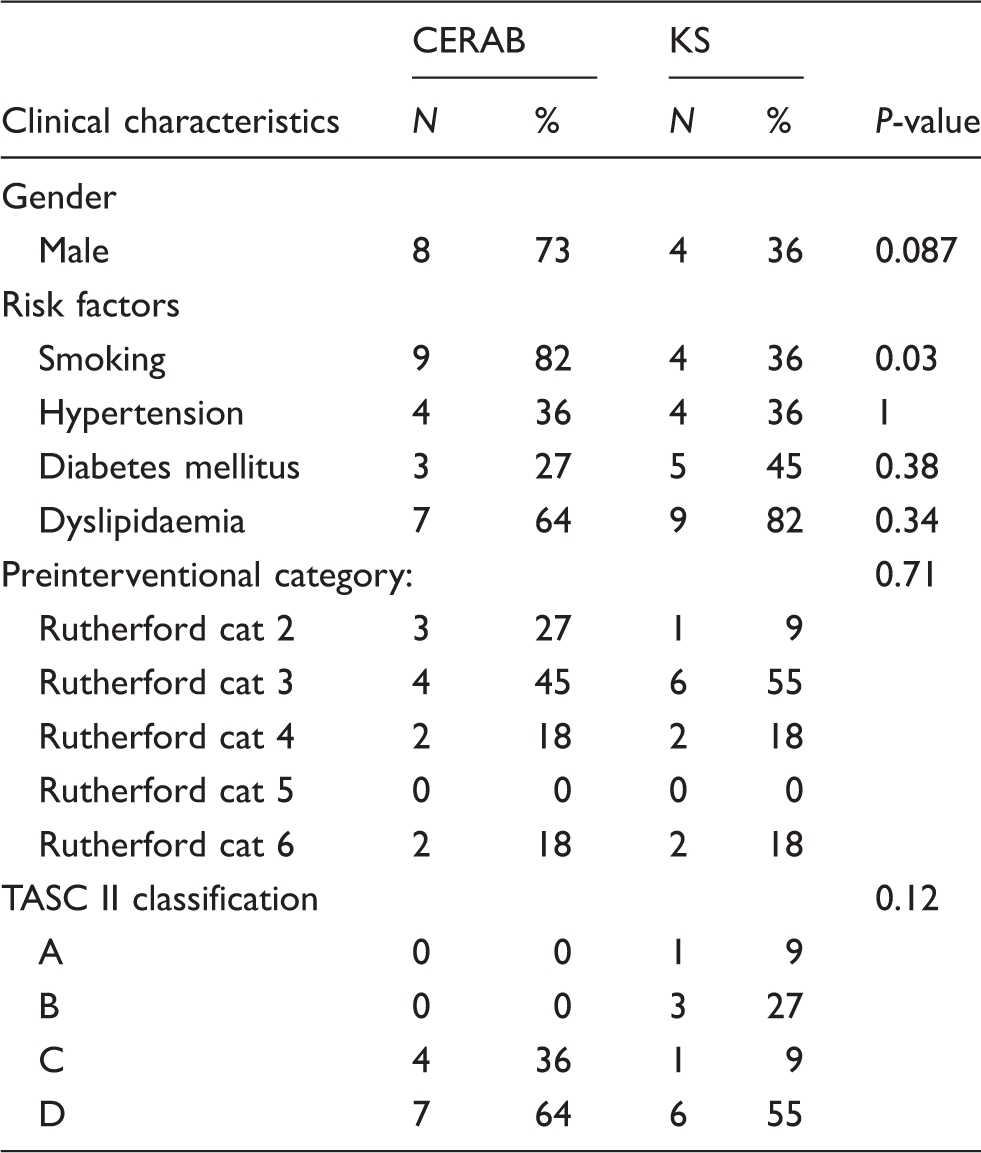
Demographic data of CERAB and KS patients. Risk factor definitions: smoking = active smoker or former smoker that stopped smoking less than five years preintervention; hypertension = patients on antihypertensive medication or BP >140/90; diabetes = patients on an antidiabetic medication (insulin and/or oral medication).
CERAB: covered endovascular reconstruction of the aortic bifurcation; KS: kissing stent.
Anatomic comparison.
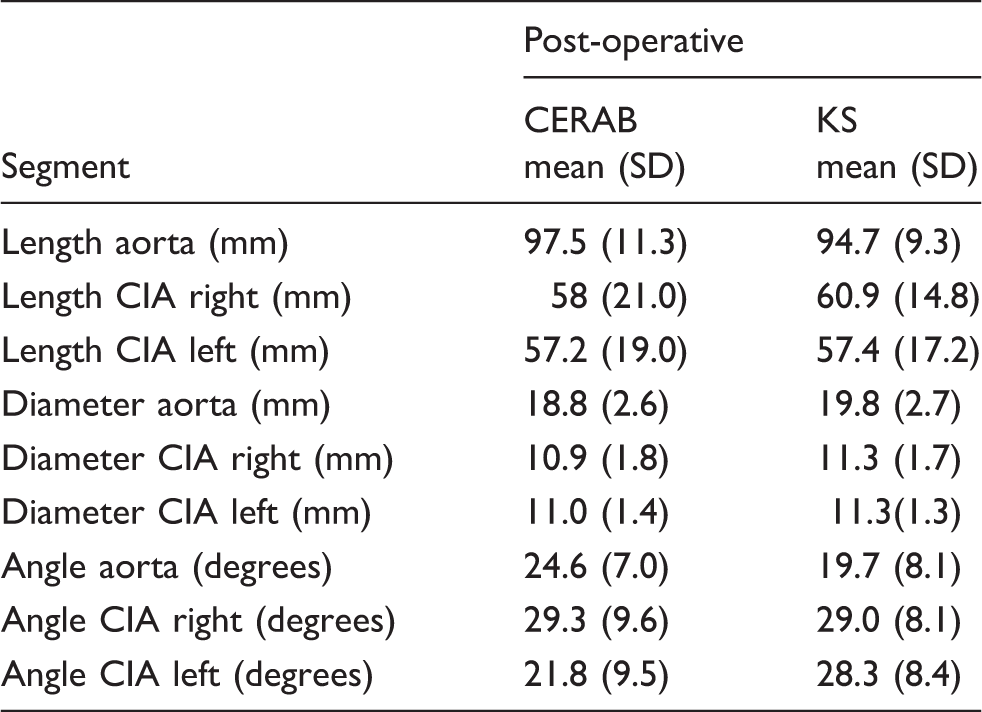
P < 0.05.
CERAB: covered endovascular reconstruction of the aortic bifurcation; CIA: common iliac artery; KS: kissing stent.
Stent placement.
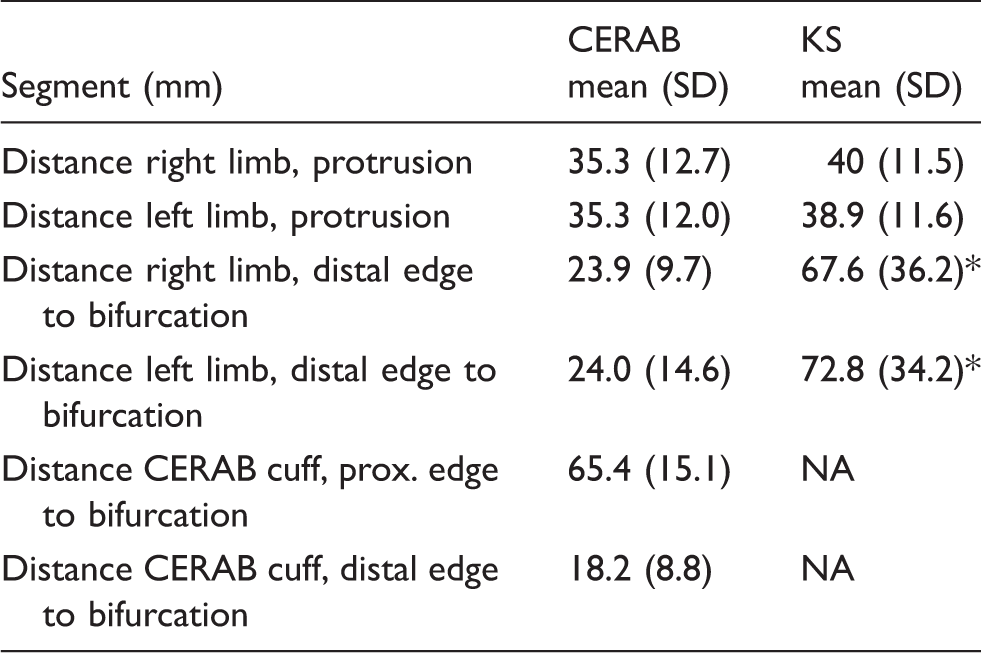
CERAB: covered endovascular reconstruction of the aortic bifurcation; KS: kissing stent; NA: not applicable.
P < 0.05.
Mean mismatch area
Geometric analysis.
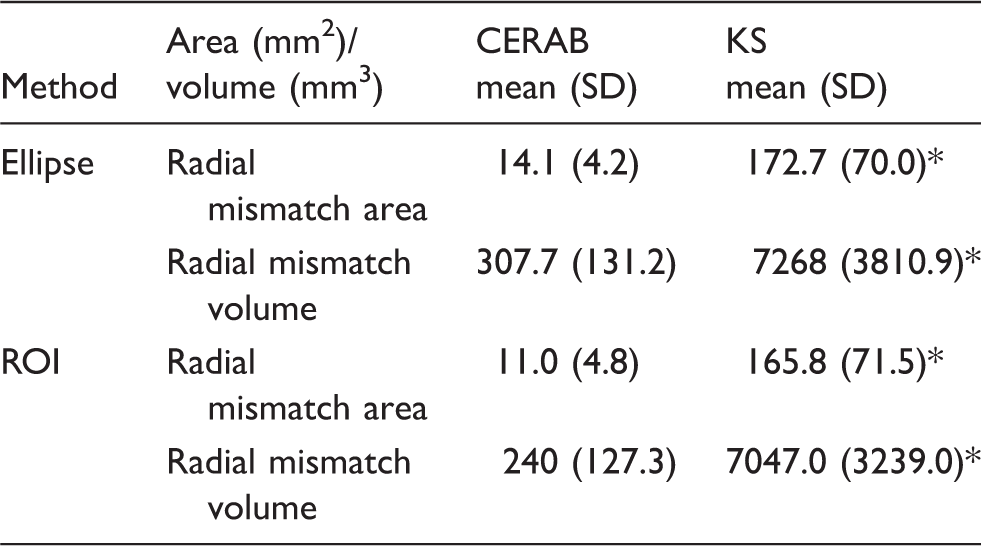
P < 0.05.
CERAB: covered endovascular reconstruction of the aortic bifurcation; KS: kissing stent; ROI: region of interest.
Total mismatch volume
Total mismatch volume was higher in the KS group for both the ellipse and ROI method (P < 0.05). With the ellipse method a 24-fold increase was observed for the KS cohort (P < 0.05) while the ROI method depicted a 30-fold increase (P < 0.05).
Stent conformation
The obtained D-ratio was not significantly different between groups (CERAB 1.20 and KS 1.15, respectively, P > 0.05). The mean area difference between the left and right stent area for the CERAB was 4.3 mm2, for the KS a difference of 16.8 mm2 was noted (P < 0.05).
Comparison of methods and researchers
Bland–Altman plots were created to compare the ellipse and ROI method (Figure 3) without significant difference between the two methods. For the ellipse method the ICC was 78% (CI 9.6–94.3 %) for the CERAB and 80 % (−0.4 to 96%) for the KS measurements. For the ROI method the ICC was 87.5 % (CI 40–97%) for the CERAB and 80% (CI 22–95%) of the KS measurements.
Left: Bland–Altman plot for the ROI and ellipse method, calculated area. Right: Bland–Altman plot for the ROI and ellipse method calculated volume. The black lines indicate mean difference and the dashed line indicates 2 SD difference from the mean.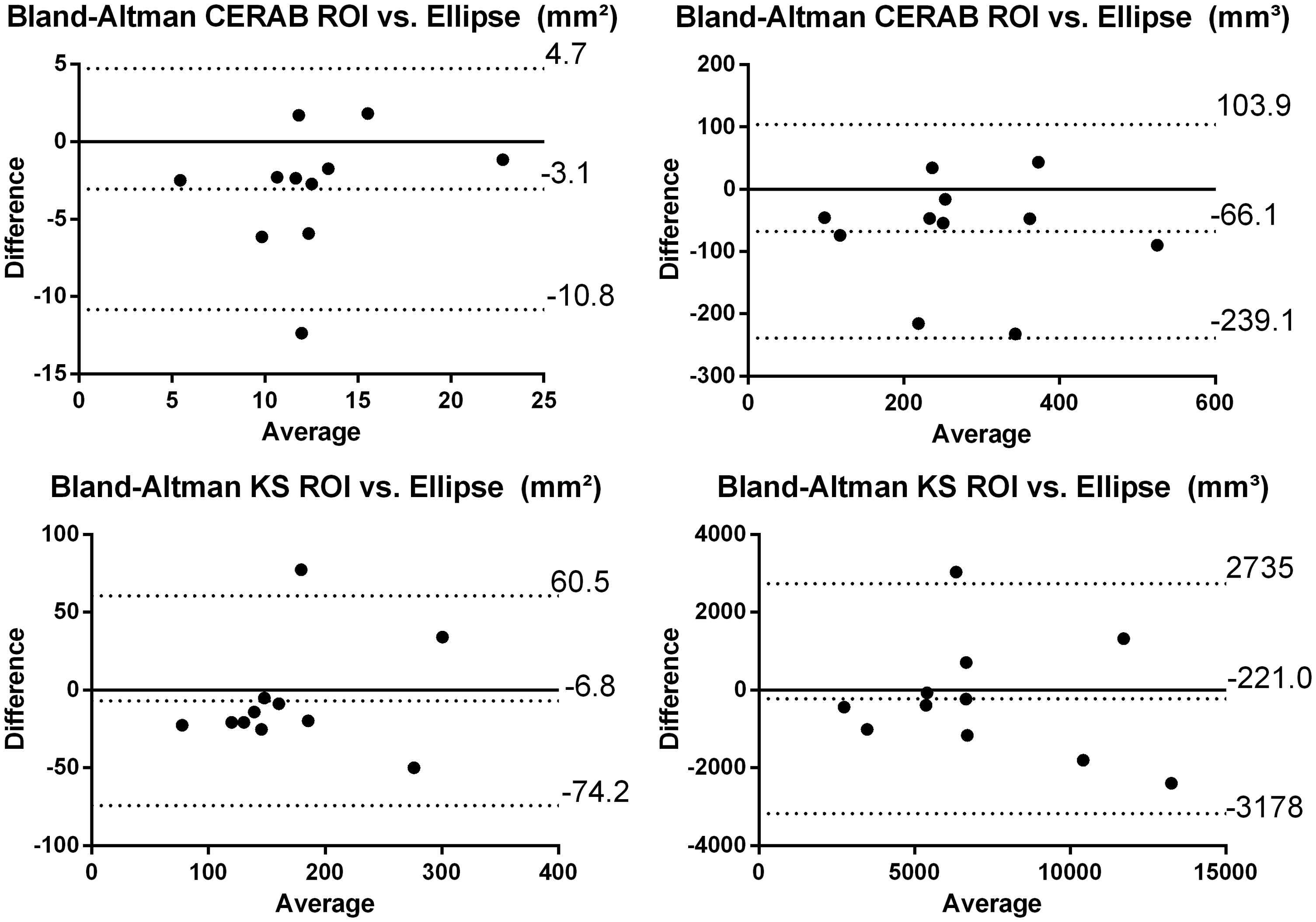
Discussion
In the present study, we have shown that CERAB is an improved reconstruction of the aortic bifurcation, in terms of mismatch, compared to KS. Others previously showed a relation between protrusion (i.e. mismatch) and patency.3,5 Significant differences exist (up to 16-fold area increase) between the mismatch areas of KS and CERAB, in favour of the latter. Mismatch volumes showed even larger differences, however, this could be due to the fact that protrusion length of the KS configuration exceeds that of the CERAB in all cases.
These results confirm earlier in vitro data, 10 but that study largely underestimated the mismatch size for both the KS (21.4 mm2 vs. 165.8 mm2) and CERAB (3.1 mm2 vs. 11.0 mm2). A better agreement between the in vivo and in vitro mismatch measurements could have been anticipated for CERAB, as mismatch between cuff and limbs is less depending on anatomy than the mismatch between the KS and the vessel wall. In case of a suboptimal placed CERAB in vitro, with the limbs protruding in the already flared proximal part of the aortic cuff, the in vitro results better match the observed in vivo results (11 mm2 vs 12.6 mm2), suggesting that there is room for improvement in clinical practice. Precise placement of the limbs inside the non-tapered part of the cuff is important to minimize the mismatch area. To discard radial mismatch as a parameter of interest, the use of the AFX stent (Endologix Inc., Irvine, CA, USA) might be a good solution when treating AIOD, as this is a one piece bifurcation graft. 12
The high D-ratios (1.6 and 1.4) observed in vitro for the KS and CERAB configuration, respectively, were not observed in the current study (1.15 and 1.2). Possibly the rigid walls of the vessel phantom, used in the in vitro studies, forced the stents more into a double D-shape or the in vitro oversizing was larger compared to the stents used in the present series.
The demonstrated mismatch in vivo is an important parameter for research. Previous in vitro research showed flow stagnation between the neobifurcation and anatomical bifurcation in the KS configuration. A covered KS configuration showed large vortices at the mismatch areas at the neobifurcation, causing pathologic proximal WSS and oscillatory shear index (OSI) values. 7 The CERAB showed only small flow recirculation in the cuff, without affecting WSS and OSI values. Basic knowledge on this subject can aid the choice of the optimal stent configuration for AOID treatment.
Limitations of the current study are the fact that post-operative CT-A data were only available for a limited number of patients treated. However, given the highly statistical significance of the differences it is debatable whether a larger sample size would affect the outcome. Scoring of calcifications was not applied to investigate the possible influence on mismatch area. Furthermore no follow-up data was used in this study to verify if the large difference in mismatch had any effects on patency. The sample sizes were considered too small to reliably observe any difference in clinical outcome and would render any conclusion on clinical impact unreliable. Data from two sites was included for analysis, in order to reduce selection and performance bias. However, this could also have introduced differences in pre, per and post-operative care and in imaging protocols, which might have influenced the observed differences. Nevertheless the observed differences in geometry were highly significant so that this potential confounder was not considered to be likely. The current study only included self-expanding bare metal KS as a comparison for the CERAB configuration. The results could be different with balloon expandable stents, but also in different configurations like crossing or non-crossing positions. The self-expanding bare metal configuration is, however, a very relevant configuration as observed in a recent review which showed that 50% of cases were treated with self-expanding bare metal KS. 13
Conclusion
We demonstrated that radial mismatch is present and that large differences are present between KS and CERAB, with a decreased geometrical mismatch in favour of the latter. In vitro measurements have underestimated the average mismatch area and volume in comparison with the in vivo situation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
