Abstract
In this study, 16-mm-thick S32101 duplex stainless steel were welded using hyperbaric underwater laser welding system in 0.15 MPa. The misorientation angle distribution of grain boundaries in the weld metal was analyzed using the electron backscatter diffraction technique. The ionic currents near the surface of metallography were measured by the Scanning Vibrating Electrode Technique (SVET) and performed to evaluate the intergranular corrosion (IGC) resistance based on corrosion current density. The volume fractions of ferrite and austenite and the grain boundary misorientation angle affect the IGC sensitivity of the welded joints. The relationship between the grain boundary misorientation angle of the austenite and corrosion resistance was analyzed. When the weld metal was shielded with nitrogen, and the volume fraction of austenite and the frequency of Σ3 coincidence site lattice boundary in the weld was increased. The misorientation angle distribution in the austenite and the frequency of Σ3 coincidence site lattice boundary influence the IGC resistance of the welded joints.
Keywords
Introduction
Duplex stainless steel (DSS) is one of the preferred structural materials for nuclear power plants. Compared with other stainless steels, DSS has the advantages of high strength and good corrosion resistance. Therefore, DSS is more and more applied to the structure and equipment of nuclear power plant. The service environment of nuclear power plant components is mostly high temperature and acid-base salt environment, which leads to defects such as corrosion crack and surface abrasion, which poses a serious threat to the safe operation of nuclear power plant. Underwater laser welding (ULW) can reduce the operation and maintenance costs, and has gradually become the most potential maintenance technology for underwater emergency repair in the field of nuclear power.
Laser welding technology not only has the advantages of high efficiency and clean welding process, but also has high applicability in the field of nuclear power maintenance. In DSS laser welding, the proportion of equilibrium phase is very important. However, maintaining the dual phase proportional balance of DSS has become a great challenge for underwater DSS welding. Sales et al. 1 pointed out that too high ferrite content will reduce toughness and pitting corrosion resistance, and increase hydrogen embrittlement sensitivity. Too low ferrite content will reduce the strength and stress corrosion cracking resistance of materials. According to Sakai 2 the austenite content of DSS should be higher than 50% in order to obtain high impact toughness. Jiang et al. 3 and Chen et al. 4 also proved that the lowest pitting corrosion rate is also associated with the austenite content of 50% or more. DSS is metallurgically solidified into complete ferrite, and then transformed from ferrite to austenite under the control of thermal cycle diffusion. When laser welding is carried out in underwater environment, the rapid cooling rate inhibits the formation of austenite to a great extent and limits the optimal phase equilibrium in DSS.
Westin 5 proved by experiment that nitrogen is a strong austenite stabilizer. Nitrogen protection is conducive to the loss of nitrogen in the welding process, so as to balance the content of ferrite and austenite, which not only improves the corrosion resistance of the material, but also most beneficial to the mechanical properties.
In the process of underwater laser welding, the weld metal experiences rapid heating and cooling cycle, the loss of nitrogen, and the formation of high proportion ferrite are not conducive to the corrosion resistance of the welded joint. Therefore, choosing nitrogen as shielding gas to balance the ferrite-austenite ratio is very beneficial to the corrosion resistance of DSS welded joints.
This paper mainly studies the DSS weld by underwater laser welding under the pressure of 0.15 MPa. The effects of shielding gas (nitrogen and argon) on the microstructure and Intergranular corrosion (IGC) resistance of weld metal were studied. The grain boundary orientation angle distribution in weld zone was analyzed by Electron Back-Scatter Diffraction (EBSD). The ion current in micro area of weld metal was measured by scanning vibration electrode technology (SVET), and the corrosion resistance was evaluated according to the corrosion current density, the IGC performance of DSS welds was verified according to ASTM A262-E standard.
Material and experimental procedure
Material and welding procedure
The ULW was performed using RFL-C6000 fiber laser of Raycus (specifications given in Table 1), with the maximum output power of 6 kW and wavelength of 1070 nm. As shown in Figure 1, the experimental system is mainly composed of two parts: the hyperbaric experimental chamber and the ULW mobile platform, which can meet the requirements of rapid and accurate positioning of welding and simulation of underwater pressure environment. The ULW mobile platform is shown in Figure 2(a). Figure 2 (b) and (c) are hyperbaric chamber and compressed gas inflation system respectively. During the welding process, the pressure of the chamber was kept at 0.15 MPa by filling compressed gas to simulate the condition of water depth of 15 m. The wet underwater environment was simulated by injecting water into the experimental tank, and the water depth in the tank was 30 mm. A sealed underwater laser head and local drainage device were developed. The laser beam was transmitted to the local dry space inside the local drainage device through optical fiber, and the ULW repair was realized by laser welding with filling wire technology.
Specifications of RFL-C6000 fiber laser machine.


Schematic diagram of underwater laser welding test system in hyperbaric environment.

Hyperbaric underwater laser welding test system: (a) experimental platform, (b) hyperbaric chamber, and (c) hyperbaric inflation system..
S32101 DSS were used as the base metal (BM), and the dimensions of test plates were prepared with 600 mm × 300 mm × 16 mm. A U-groove was performed on the surface of test plate, the welding parameters shown in Table 2, and the design of filling process for U-groove welded joint is shown in Figure 3(a). Figure 3(b) is the weld cross-section. The chemical compositions of both the BM and filler metal are listed in Table 3.
Welding parameters.


U-groove design and cross-sectional appearance of (a) the schematic diagram and (b) the welded joint.
Chemical composition of base metal and filler metals (wt.%).

Microstructural investigation
The microstructure of weld samples were characterized by optical microscopy (OM) and scanning electron microscopy (SEM) combined with energy dispersive spectroscopy (EDS). The microstructure, ferrite and austenite phase fractions and Coincident Site Lattice (CSL) boundaries were characterized and analyzed by electron backscatter diffraction (EBSD) technique. This technique uses the field emission SEM. The electron beam had an accelerating voltage of 20 kV, the surface of the samples was electro-polished at 20 V direct current (DC) with a solution that consisted of 5 g CrO3, 65 mL phosphoric acid, 20 mL sulfuric acid, and 15 mL distilled water.
Scanning vibrating electrode technique (SVET)
The SVET test were performed in 0.05 M NaCl solution at room temperature, using a SEVT from Princeton micro area electrochemical system (Figure 4, AMETEK, versascan, USA). The corrosion current density of two weld samples was measured, the microelectrode has a black platinum probe with a diameter of about 10 μm, vibrates with an amplitude of 20 μm, and is about 200 μm away from the surface. Before each experiment, the electrode surface was ground to 2000 particle size, polished with diamond gypsum, washed with distilled water, and dried in hot air. The sample was adhered to the epoxy sleeve, and the tape around the epoxy was used as a solution reservoir. The vibration frequency of the probe was 200 Hz and the test step was 20 μm.

Princeton micro area electrochemical system.
Intergranular corrosion (IGC) test
IGC performance test shall be conducted according to ASTM A262 Standard Practices E – copper–copper sulfate sulfuric acid test. 6 Sensitization at 700°C for 1 h before test. The specimen was covered with copper shot and grindings and immersed in a solution of 16 wt% sulfuric acid with 6 wt% copper sulfate. The solution was then heated to its boiling point and maintained at this temperature for 20 h. The bending angle shall not be less than 90°. After bending, observe whether there are cracks in the macro morphology of the welded joint to determine whether the welded joint meets the requirements of IGC performance.
Results and discussion
Microstructure and austenite volume fraction
Figure 5(a) to (e) shows the microstructure of weld metal (WM), heat affected zone (HAZ), base metal (BM) of S32101 DSS welded joint with different shielding gas. The BM exhibited a typically balanced microstructure after rolling and solution treatment. According to the quality-control report of Anshan Iron and Steel Co., Ltd., the austenite content in BM was 56%. Ferrite, grain boundary austenite (GBA), intragranular austenite (IGA), and widmanstatten (WA) are the main constituent phases in the weld metal. During the process of laser welding, the molten pool metal cools and crystallizes rapidly, austenite transformation from high-temperature ferrite occurred in the form of GBA, WA, and IGA. The austenite began precipitating from the ferrite grain boundaries to form WA and IGA. Some austenite surrounding the ferrite formed the GBA. Zhang et al. 7 also obtained similar results during the experiment.

Microstructure of the weld zone (a and b) with argon and nitrogen shielded gas respectively and (c and d) of the HAZ; (e) of the base metal.
In addition, ferrite-austenite transformation also occurred in HAZ near the fusion line. Due to fact that the temperature of HAZ near the fusion line can increase to approximately 1000°C, the α/γ phases of the original duplex microstructures will also further evolve. In the subsequent cooling process, some ferrite transforms into austenite, but the growth of the austenite grains was inhibited due to the decrease in the high temperature residence time in the underwater environment. On the other hand, the formation of WA and GBA requires a relatively smaller driving force to occur at a higher temperature with little undercooling. Therefore, WA and GBA austenite structures were not observed, the HAZ consists of a large number of coarse polygonal ferrite and irregular austenite. The width of the polygonal ferrite band was approximately 50 μm. This region is called carbon poor region or ferrite stable region in the literature of Celik and Alsaran 8 , Kurt et al. 9 , and Wang et al. 10 For the convenience of discussion, it is called transition zone (TZ) in this paper. The microstructure of TZ under nitrogen protection was quite different from that under argon protection. The joint protected by nitrogen has more austenite content and the austenite distribution was more uniform in TZ.
Figure 6 shows the austenite volume fraction of weld, HAZ, and base metal with different shielding gas. When argon was used as shielding gas, the volume fraction of austenite in weld zone and heat affected zone was 38.87% and 47.13% respectively. When nitrogen was used, the volume fraction of austenite was 41.4% and 47.53%. It can be found that when nitrogen was used as the shielding gas, the austenite content increases. The austenite content in weld zone is lower than that in heat affected zone. The reasons for this phenomenon can be attributed to two factors: (1) Insufficient time was available for austenite transformation from ferrite due to the fast cooling rate and a larger temperature gradient than HAZ in the weld zone; (2) Nitrogen protection reduces nitrogen loss during welding and improves austenite transformation efficiency. 11

Austenite volume fractions in the WM and HAZ of the welded joints with different shielding gas.
Grain boundaries misorientation angle distribution
According to the orientation characteristics of adjacent grains, grain boundaries can be classified into two groups: ordinary grain boundary and coincidence lattice grain boundary (CSL). In the research of Eghlimi et al. 12 reported that ordinary grain boundaries can be divided into low-angle grain boundaries (LAGB < 5°), moderate grain boundaries (5° <MAGB < 15°), and high-angle grain boundaries (HAGB > 10°). In the case of LAGB, the misorientation angle between the two adjoining grains is low and the interface is stable enough to accommodate the misorientation by an array of dislocations. With increasing misorientation angle, the dislocation spacing decreases, and the stable dislocation structure of grain boundary gradually fails. This is because individual dislocations are no longer distinguishable and overlap one another, which is the case of HAGB. 13 It was also found in Randle 14 and Eghlimi et al.’s 12 research that increasing the proportion of LAGBs and optimizing its distribution can improve the IGC and other properties of materials.
Figure 7(a) to (d) shows the distributions of the ferrite and austenite grain boundaries in the two typical welds of the DSS. Figure 7(a) and (b) respectively show the distribution of different angle grain boundaries in ferrite and austenite with argon protection, and Figure 7(c) and (d) respectively show the distribution of different angle grain boundaries in ferrite and austenite with nitrogen protection.

The distributions of the grain boundaries for the ferrite (a and c) and austenite (b and d).
Figure 8(a) and (b) present the relative frequencies of the different grain boundaries for the ferrite and austenite in the weld metals, the ferrite grains were mainly constituted of LAGBs, a relatively small amounts of HAGBs and a few MAGBs. Furthermore, there were significantly more HAGBs in austenite than ferrite in each zone, while the LAGBs in each austenite were obviously fewer in number than the LAGBs. As reported by Eghlimi et al., 15 the weld metal mainly contained a large number of LAGBs, whether for ferritic or austenitic grains. MAGBs were primarily formed between the ferrite grains. There are relatively few of MAGBs between austenite grains in each zone. The research conclusion is consistent with the experimental results.

The relative frequencies of the low-angle, moderate-angle, and high-angle grain boundaries for (a) the austenite and (b) ferrite in the different welding shielded gas.
Grain boundary studies have revealed that grain boundary phenomena (such as grain boundary diffusion, sliding, precipitation, cracking, and corrosion) are closely related to the crystallographic structure of grain boundaries, as reported by Lehockey et al. 16 It mainly depends on the dislocation energy in the grain boundary (also known as grain boundary energy). The dislocation energy depends on the phase difference between grains. So grain boundary energy γ of the variation relationship with orientation difference conforms to the formula, as mentioned by Read and Shockley 17 :

Where
It can be seen from the formula that the interface energy of small angle grain boundary is relatively low and stable. Therefore, it is generally accepted that low-energy grain boundaries have higher resistance to grain boundary deterioration than high-energy grain boundaries. Similarly, the concept of Grain Boundary Engineering (GBE) was proposed in 1984, which primarily intends to prevent grain boundary degradation by introducing high-frequency low-energy grain boundaries into the grain boundary network in materials. Generally, the high frequency of CSL boundary will lead to the discontinuity of random boundary, so as to improve the resistance to grain boundary degradation. Michiuchi et al. 18 reported that an appropriate deformation and annealing processing of austenitic stainless steel produced a high frequency of CSL boundaries and significantly improved resistance to IGC.
Figure 9 shows the distributions and the frequencies of the special and random grain boundaries for the ferrite and austenite grains in the weld metal. In both weld metals with argon and nitrogen shielded gas, austenite twins (Σ3) constitute the largest proportion of special boundaries. Similar conclusions were obtained in Lehockey et al.’s 19 research. However, the consistency between random pairs and theoretical values in nitrogen-shielded welds is higher, indicating that (Σ3) special boundary distribution is more uniform, showing better corrosion, cracking, and segregation resistance.

Grain boundary misorientation angle distribution of the DSS WM with different shielding gas: (a) the ferrite (b) the austenite.
Evaluation of intergranular corrosion (IGC)
Scanning vibration electrode technology (SVET) is an advanced technology to measure the corrosion potential and corrosion current density in small areas by using vibration microprobe without contacting the sample surface. Scanning electrochemical microscope (SECM) can make the change of electrochemical activity of austenite-ferrite dual phase microstructure into a visual state, accurately determine the corrosion of tiny areas, and has strong spatial detection ability. In this paper, SVET was applied to detect the IGC behavior of duplex stainless steel weld structure in micro state. Combined with CSL grain boundary theory, the IGC mechanism of austenite grain boundary in chloride solution is determined.
Figure 10 is the SVET mapping results of two kinds of welds. It is found that the corrosion degree of austenite and ferrite grain boundary is significantly different, and the corrosion degree of argon protected weld is more serious than that of nitrogen protected weld. As shown in Figure 10 that the corrosion current plane graph presents a strip interphase distribution structure, which is related to the shape and distribution of ferrite and austenite grains. According to the three-dimensional morphology of corrosion current, the maximum corrosion current density of nitrogen-shielded weld is about 3.9 μA/cm2, the corrosion current density of argon-shielded weld is about 5.6 μA/cm2, the corrosion current density of the base metal is about 3.7 μA /cm2.

Maps of ionic currents for the two welds with biaxial deformation of (a and b) for BM, (c and d) for argon, and (e and f) for nitrogen; scanned area: 2000 μm × 2000 μm.
By comparing the two groups of data, it can be concluded that the corrosion resistance of the weld protected by nitrogen is higher than that protected by argon, which is close to the corrosion resistance level of the base metal.
After SEVT test, the corrosion morphology was observed by scanning electron microscope. Figure 11(a), (c) and (d) shows the corrosion morphology of base metal, argon-shielded weld, and nitrogen-shielded weld respectively. As shown in Figure 11(a), preferential corrosion was found at the boundary interface between austenite and ferrite of BM. Figure 11(b) is a schematic diagram of the IGC of duplex stainless steel. Due to the high levels of Cr and Ni in duplex stainless steel, a passive film with a thickness of 2–4 nm can be naturally established in corrosive (oxidation) environment. However, under some specific conditions, in the defects area such as non-metallic inclusions on the metal surface and precipitate grain boundaries, permanent breakdown occurs locally in the passivation layer in a specific corrosive medium, resulting in corrosion on the unprotected metal surface. The initiation stage of IGC usually occurs in places with chloride ion concentration, resulting in local breakdown of passivation layer.

Surface morphology of SVET corrosion of base metal, argon-shielded and nitrogen-shielded welds, (a) base metal, (c) argon-shielded weld, (d) nitrogen-shielded weld, and (b) corrosion diagram.
Generally, there is a high chloride concentration gradient at the ferrite and austenite grain boundaries, and corrosion first occurs here and extends into ferrite. Once formed, it will rapidly expand to ferrite and finally to austenite. However, the actual damage of the passivation layer preferred to starts from the ferrite phase. This means that austenite grains are more resistant to chloride environment than iron matrix. The reason is that austenite contains higher CSL boundary, which shows better IGC resistance and segregation than iron matrix.
Pitting resistance equivalent number (PREN) is a traditional method to evaluate the local corrosion sensitivity of DSS in chlorine-containing solutions. It is generally believed that the local corrosion behavior of ferrite, austenite, or precipitate in grain boundary is the main factor inducing IGC. Therefore, the effect of shielding gas on the transformation mechanism of ferrite-austenite and its correlation with IGC can be further determined by PREN value.
The following equation is widely used, for example, in the literature of Garfias-Mesias et al. 20 , Perren et al. 21 , and Hu et al. 22 :

Table 4 shows the average composition of the major alloying elements in austenite allotrope and ferrite phases in the weld metal, together with the PREN values calculated with equation (1). The alloy elements of each phase shall be measured at least three times at each weld metal to ensure reliability. Figure 12 shows EDS spectra of each phase. The higher the value of PREN was, the better the pitting corrosion resistance could be. It can be seen that the PREN equivalent of ferrite was lower than that of austenite (GBA, WA, IGA) and the difference attributes to the fact that the high content of Cr and Mo in austenite phase, which indicates that local pitting earliest occurs in the lower pitting equivalent stage, and gradually extended to the direction of high PREN value position. In other words, the continuous austenite grid structure of GBA and WA is an obstacle to the propagation of corrosion, which verifies the conclusion that austenite has better corrosion and segregation resistance than ferrite. The same conclusion is obtained in the literature of Gideon et al. 23 , Kim et al. 24 , and Ruiz et al. 25
Chemical compositions of different phases in welds.

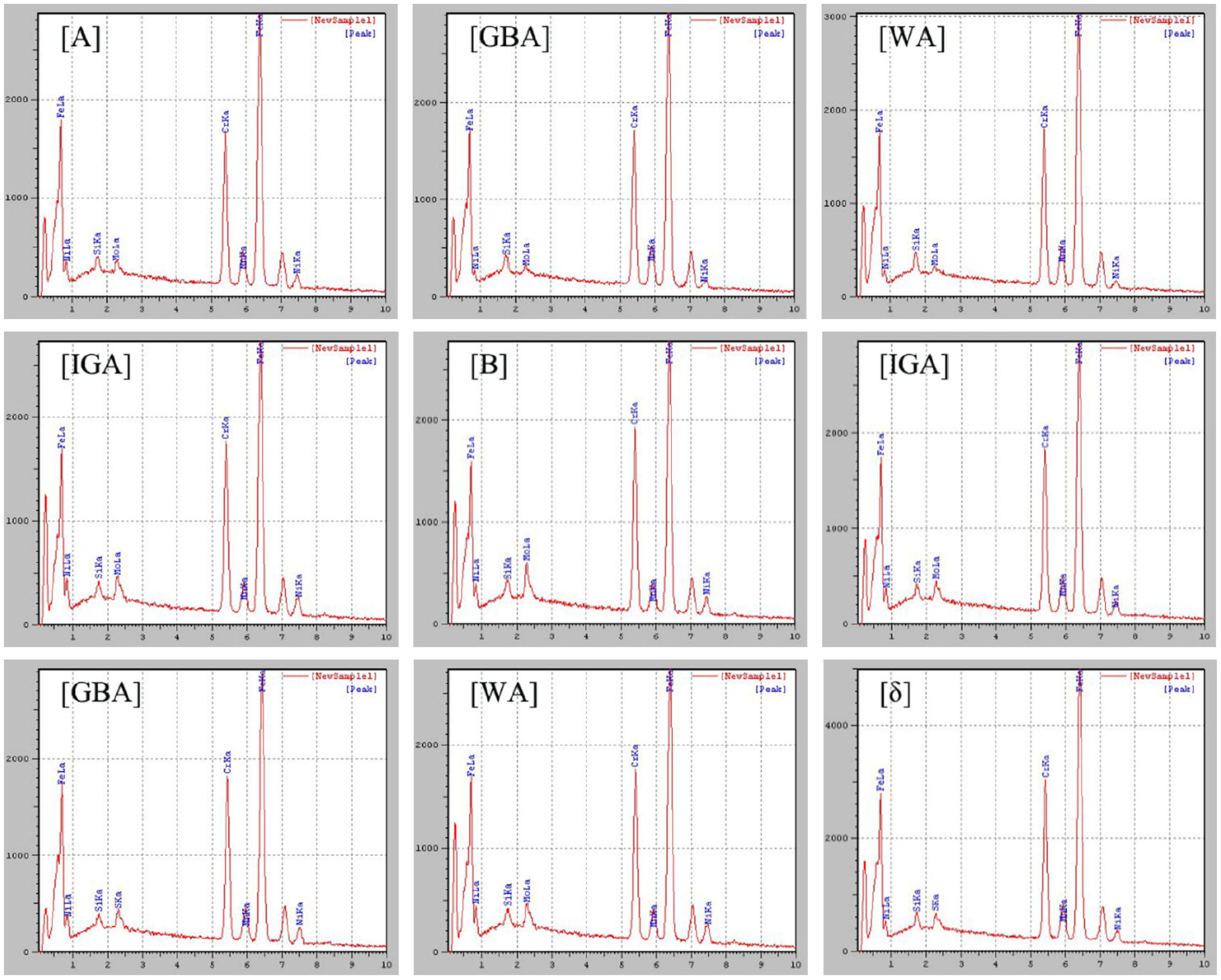
EDS spectra and major alloy contents measured from the marked position in Figure 11(c) and (d): GBA, WA, IGA, and ferrite.
In addition, the nitrogen-shielded weld has a higher PREN value, indicating that nitrogen as protective gas is conducive to supplement the loss of nitrogen element in the welding process, balance the ferrite-austenite phase ratio, and improve the ability of weld to resist IGC corrosion.
Bending tests
The IGC test results for A262 Standard Practice E test are shown in the Figure 13. Figure 13(a) shows the intergranular corrosion test device, Figure 13(b) shows the process of bending test after intergranular corrosion test, and the bending angle is not less than 90°. Figure 13(c) and (d) show the morphology of the joints protected by argon and nitrogen before and after the bending test, respectively. It is observed that cracks appear on the joint surface in the argon-shielded weld, while no obvious cracks appear on the joint surface in the nitrogen-shielded weld, which proves that the nitrogen-shielded weld meets the intergranular corrosion requirements in ASTM A262 Standard for stainless steel intergranular corrosion sensitivity test.

The process of intergranular corrosion test: (a) the intergranular corrosion test device, (b) the process of bending test, (c) the morphology of the joints before bending test, (d) the IGC test results of the bending test.
Discussion
In this paper, the microstructure and corrosion characteristics of S32101 welded joints by underwater laser welding in 0.15 MPa hyperbaric environment were studied. The effect of welding shielding gas on the microstructure and corrosion characteristics of DSS weld was further studied. The conclusions are as follows:
When nitrogen was used as shielding gas, the volume fraction of austenite was increased, and the microstructure of weld metal was still mainly composed of ferrite, GBA, IGA, and WA. The HAZ structure near the fusion line experienced an austenite transformation similar to weld metal, which is composed of a band of coarse polygonal ferrite and irregular austenite. The width of the polygonal ferrite band is approximately 50 μm.
In the process of DSS underwater laser welding, whether argon or nitrogen was used as shielding gas, the austenite twin (Σ3) constitutes the largest proportion of special boundary, and the distribution of Σ3 special boundary in nitrogen protected weld is more uniform, showing better corrosion, cracking, and segregation resistance.
In the electrochemical experiment of SVET, the austenitic ferrite phase has obvious corrosion difference. The current density of weld protected by argon is about 5.6 μA/cm2, while protected by nitrogen is about 3.9 μA/cm2, the base metal is about 3.7 μA/cm2, indicating that the corrosion resistance of the weld protected by nitrogen is higher than that protected by argon, which is close to the base metal.
After the micro electrochemical test of SVET, the corrosion area was observed by scanning electron microscope and detected by EDS. The test results show that the weld protected by nitrogen has higher pitting resistance equivalent (PREN). Local pitting first occurs at the lower pitting equivalent stage, and the corrosion pit obviously expands to the ferrite phase. In other words, the continuous austenitic grid structure of GBA and WA is an obstacle to the spread of corrosion.
The intergranular corrosion sensitivity of argon-shielded and nitrogen-shielded welded joints were tested according to the A262 Standard Practice E. The nitrogen-shielded welds meet the intergranular corrosion performance requirements in the inspection standard.
Footnotes
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Award Cultivation Foundation from Beijing Institute of Petrochemical Technology (Project No.BIPTACF-010) and National Science and Technology Major Project (2018ZX06002006).
