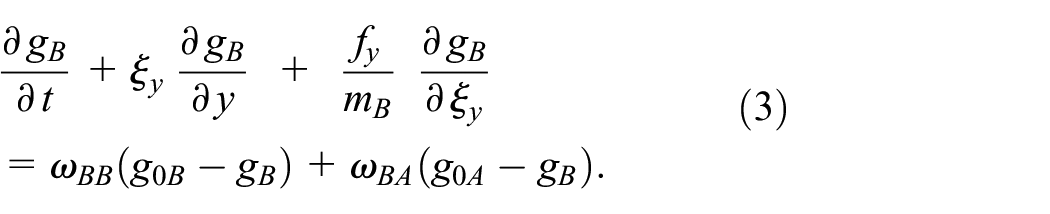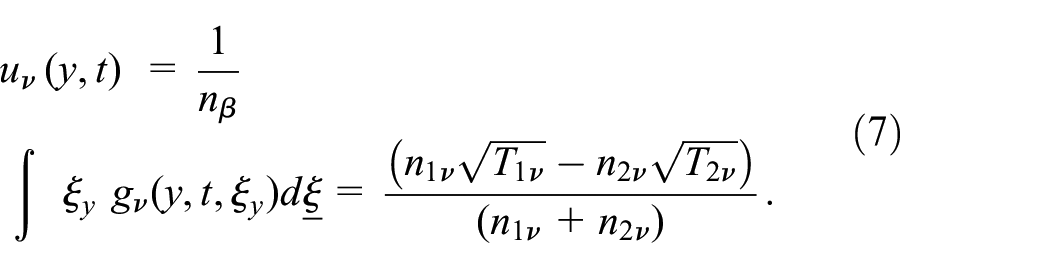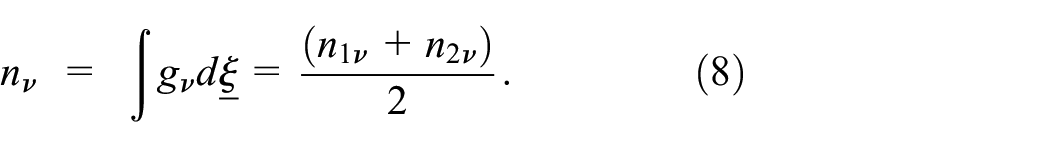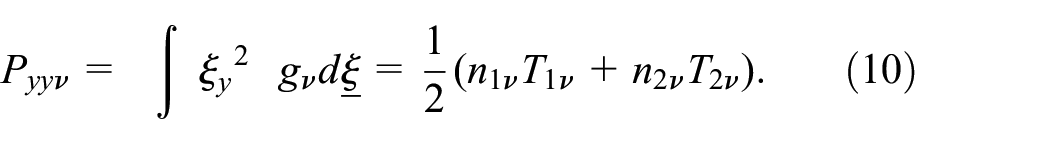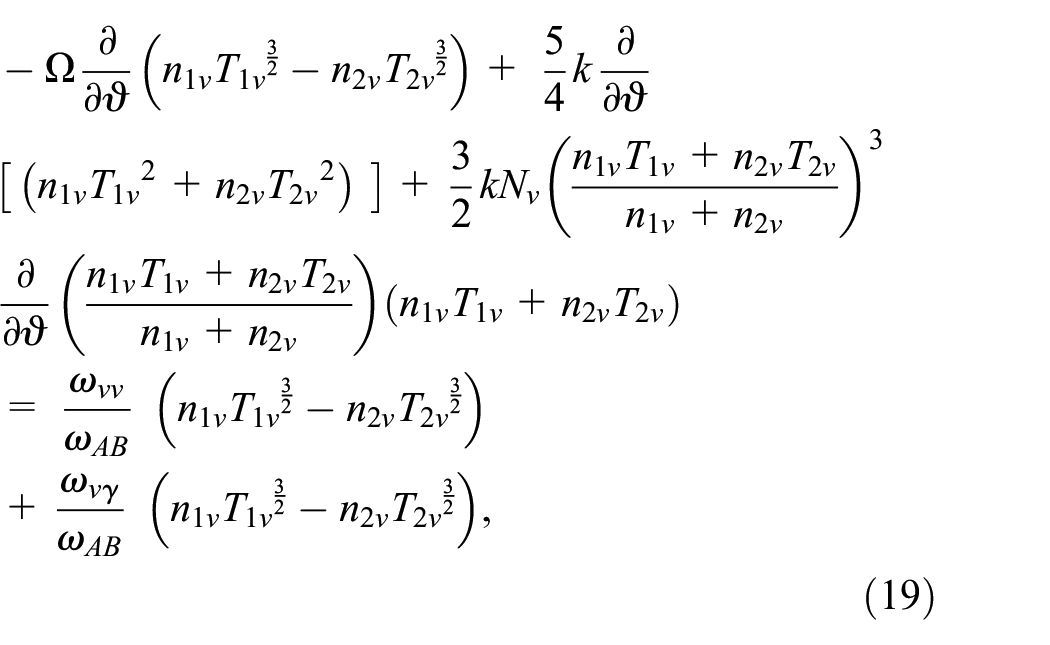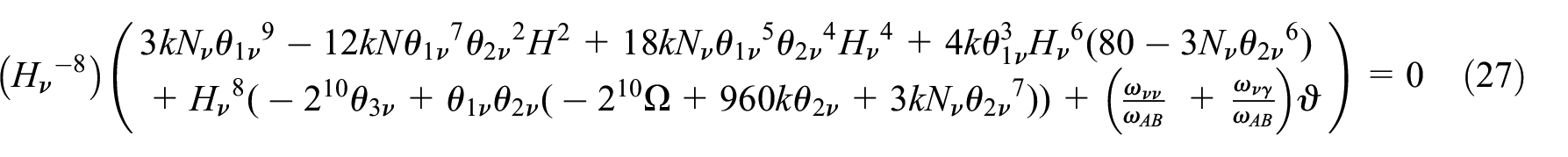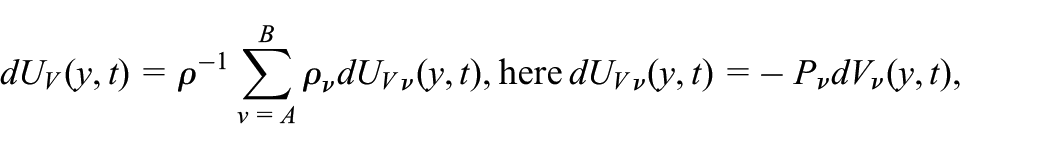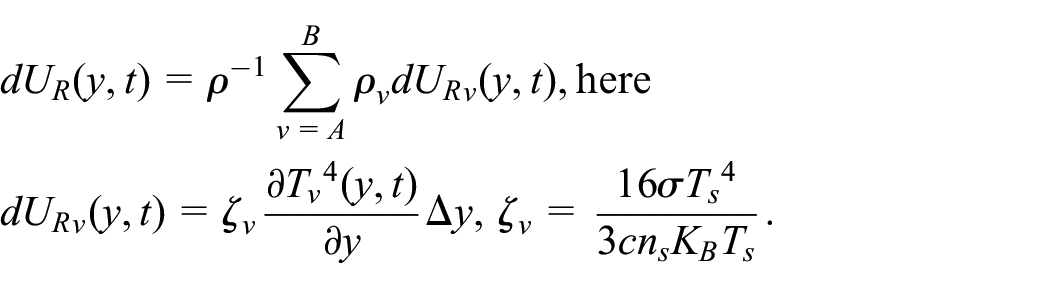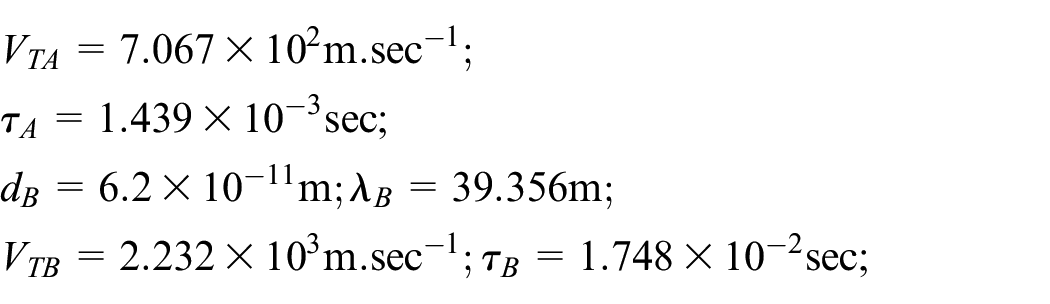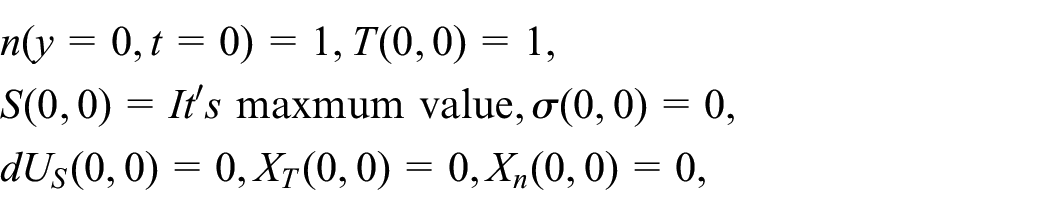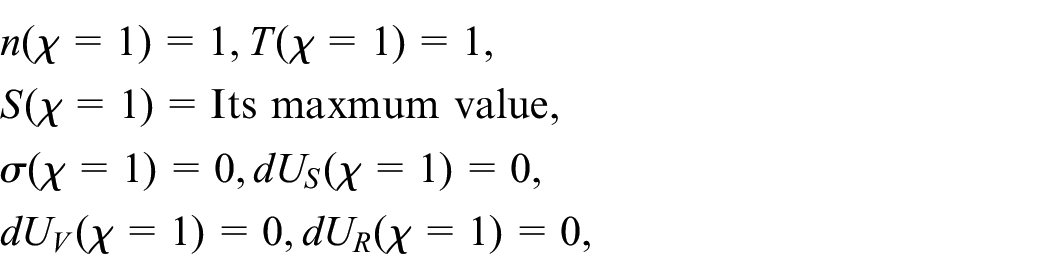Abstract
In the present paper, the effect of the nonlinear thermal radiation (TR) on the neutral gas mixture in the unsteady state is investigated for the first time. The unsteady BGK technique of the Boltzmann kinetic equation (BKE) for a non-homogenous neutral gas (NHNG) is solved. The solution of the unsteady case makes the problem more general than the stationary case. For this purpose, the moments’ method, together with the traveling wave method, is applied. The temperature and concentration are calculated for each gas component and mixture for the first time. Furthermore, the study is held for aboard range of temperatures ratio parameter and a wide range of the molar fraction. The non-equilibrium distribution function (NEDF) is calculated for each gas component and the gas mixture. The significant non-equilibrium irreversible thermodynamic characteristics the entire system is acquired analytically. That technic allows us to investigate our model consistency with Boltzmann’s H-theorem, Le Chatelier’s principle, and thermodynamics laws. Moreover, the ratios among the further participation of the internal energy change (IEC) are evaluated via the Gibbs formula of total energy. The final results are utilized to the argon-helium NHNG at different magnitudes of radiation force (RF) strength and molar fraction parameters. 3D-graphics are presented to predict the behavior of the calculated variables, and the obtained results are theoretically discussed. The significance of this study is due to its vast applications in numerous fields, such as satellites, commercial, and various industrial applications.
Keywords
Introduction
Thermal radiation is a deep-rooted part of our environment. Also, at high temperatures, the radiative heat transfer is a remarkable incomplete system analysis. Some application where TR transfer is of primary significance includes cryogenic fuel storage systems, spacecraft cooling systems, boilers and furnaces, and solar collectors.1,2 Although, the statistical-mechanical study of an NHNG in a non-equilibrium situation is a fascinating subject from theoretical as well as experimental viewpoint. Few papers deal with the gas mixture,3–7 compared with the significant number of studies in a single neutral gas.8–14
It is well known that the atoms of an NHNG radiate and absorb thermal energy. Therefore, in NHNG, the interaction with TR is remarkable, unlike solids and liquids, capable of undergoing conspicuous volume alteration. Considering this interaction makes the NHNG way more realistic. Further, radiative heat transfer in NHNG has remarkable applications from combustion processes to atmospheric modeling operations. The BKE is valid for all ranges of Knudsen number,
8
while the Navier-Stokes (NS) system is suitable to give us acceptable results for the continuum flow only, (where
For this purpose, Abourabia and Abdel Wahid 9 presented a new approach for investigating the effect of the TR force on the NHNG. For steady problem, this idea was applied on a half-space filled off by an NHNG bounded by a fixed heated solid plane. The present paper is extended to investigate the non-stationary problem for an NHNG affected by a TR force. Therefore, we should solve two systems of nonlinear partial differential equations (PDEs) instead of one system of ordinary differential equations (ODEs), as presented in. 9 Abdel Wahid 2 study the behavior of a single monatomic neutral gas under the effect of nonlinear TR field in the unsteady state. He investigated the thermodynamic properties of the system and approved that the applied model is compatible with the laws of thermodynamics, Boltzmann’s H-theorem, and Le Chatelier’s principle. In, 4 Abdel Wahid presented an approximate solution of the influence of a linear RF on a neutral gas mixture using the traveling wave method and the moment method. He used the approximation form of linear radiation force. He tested the computability of his mathematical model with Boltzmann’s H-theorem, Le Chatelier’s principle, and the laws of thermodynamics and approved it. Abourabia and Abdel Wahid 5 presented an exact mathematical solution for investigating the effect of the TR force on the NHNG. For steady problem, this idea was applied on a half-space filled off by an NHNG mixture bounded by a fixed heated solid plane. The president’s paper represents a significant development of all of those studies as we utilize coupled systems of non-stationary BGK-type, one for each component of the neutral gas.
The RF effect is inserted into the force term of the BKE. These procedures are done by applying the Liu-Lees model for two-side Maxwell NEDFs using the moment method. Moreover, the manner of the macroscopic characteristics of the NHNG is estimated for different RF strength according to different fixed, rigid plate temperatures. The temperature and concentration are, in turn, substituted into the related NEDF. This approach permits us to investigate the manner of the equilibrium, non-equilibrium, and non-stationary NEDFs for different magnitudes of the molar fraction parameters. Also, the remarkable non-equilibrium thermodynamic characteristics of the entire system are calculated. Especially thermodynamic force (TF), entropy, entropy generation, entropy flux, and kinetic coefficients are investigated.
Furthermore, our model consistency with thermodynamics second law, Boltzmann H-theory, and Onsager-Casimir’s relation are illustrated. The ratios among the different participations of the IEC are obtained via the Gibbs’ formula. The results are applying to the argon-helium NHNG. Finally, the remarkable conclusions of the paper are indicated.
The physical and mathematical formulation of the problem
Consider an NHNG consisting of two species, for example, an A-Kind and B-Kind. The gas fills in the upper half-space in the system (
(I) The two velocities of the atoms (incident and reflected) are equals at the fixed plane solid plate but opposite signs. That result happens as a result of Maxwell’s formula of momentum defuses reflection. Furthermore, the exchange will be generated by the temperature difference among the atoms and the heated plane solid plate. That is taking the form of full energy accommodation. 8
(II) A TR force is acting from the heated plane solid plate on the NHNG, written in vector form2,5,8 as
Here
Here
Lee’s moment method15–18 is employed here to gain the solution of the BGK kinetic equation. By adding heat to a NHNG, allowing it to expand, it is made rarer than the non-homogeneous gas neighbor sections. It continues to create an upward stream of the heated NHNG, which is usually followed by a flow in the reverse direction by the more distant parts of the NHNG. The fresh portions of NHNG are carried into the heat source neighborhood, taking their weather along with them to other regions.4,7 We assume that the temperature of the NHNG atoms rising upward is T1
v
while the temperature of the NHNG atoms going downward is
By multiplying the BGK-type by a velocity function
Atoms of any NHNG component v with the second species
where
The obtained equation (5) are known as the general equations of transfer. 16 Applying the non-dimensional forms by assuming:
here
The bulk velocity of the gas:
The number density:
The temperature:
The static pressure:
The y-component heat flux:
The functions
Now, let
The y-direction component of the heat flux has the form:
The equations of state complement the above equations:
Thus, we acquire the fourth equation in the form:
Traveling wave solution method17,18 is utilized to solve the problem, by assuming
That will replace dependent variables from functions in
where n is a positive integer. Substituting from equations (16) and (17) into equations (12) and (13) we acquire:
Know, we intend to solve equations (14, 15, 18, and 19) with the boundary value problem to evaluate
Substituting from equations (14) and (20) into equation (18), taking into consideration equation (10), we get:
Integrating equation (21), for
Here we assume
Moreover,
To make our calculation simpler, and make better use of the equation (20), let us introduce the function
From equations (23) and (24) we can obtain:
Now, we can integrate equation (19) w.r.t.
here
After solving this equation, we obtain eight roots for
The magnitudes of the integrated constants can be evaluated with the initial and boundary condition
Moreover, the temperature of the incident atoms is T2. The heat of the reflected atoms from the solid plane plate is represented by temperature T1, which can be expressed, as mentioned in:4,5
here
The obtained formulae represent the initial and boundary conditions of the problem.
Substituting the calculated quantities
We can acquire the sought NEDFs, which enable us to investigate the manner of the NHNG atoms. That was not possible if we utilized the NS equations system instead of the BGK-type.
The nonequilibrium thermodynamic characteristics of the system
The entropy S per unit mass of the NHNG has the form:20–23
while the Boltzmann’s entropy generation in the non-stationary state
According to the theory of thermodynamics;4,7,13 the TFs can be evaluated:
The first is
The second TF is
The third TF is
After calculating the entropy generation and the TFs, the kinetic coefficients Lij can be obtained from the following relation:20–24
The Gibbs relation for the IEC
The pressure and modification in the volume are
Results and discussion
The manner of the NHNG was examined under the influence of a TR force in the non-stationary state of a plane heat transfer problem in the entire system (NHNG + heated solid plane plate). Moreover, the TR is presented in the force term of the BKE for an NHNG subject to the inequality
1- Wherein the equilibrium as
2- Wherein the equilibrium as

(a) Concentrations nA versus y and t for fixed χ = 0.5, and CA = 0.5, (b) concentrations nB versus y and t for fixed χ = 0.5, and CA = 0.5, and (c) concentrations n versus y and t for fixed χ = 0.5, and CA = 0.5.

(a) Temperature TA versus y and t for fixed χ = 0.5, and CA = 0.5, (b) temperature TB versus y and t for fixed χ = 0.5, and CA = 0.5, and (c) temperature T versus y and t for fixed χ = 0.5, and CA = 0.5.

Entropy S versus y and t for fixed χ = 0.5, and CA = 0.5.
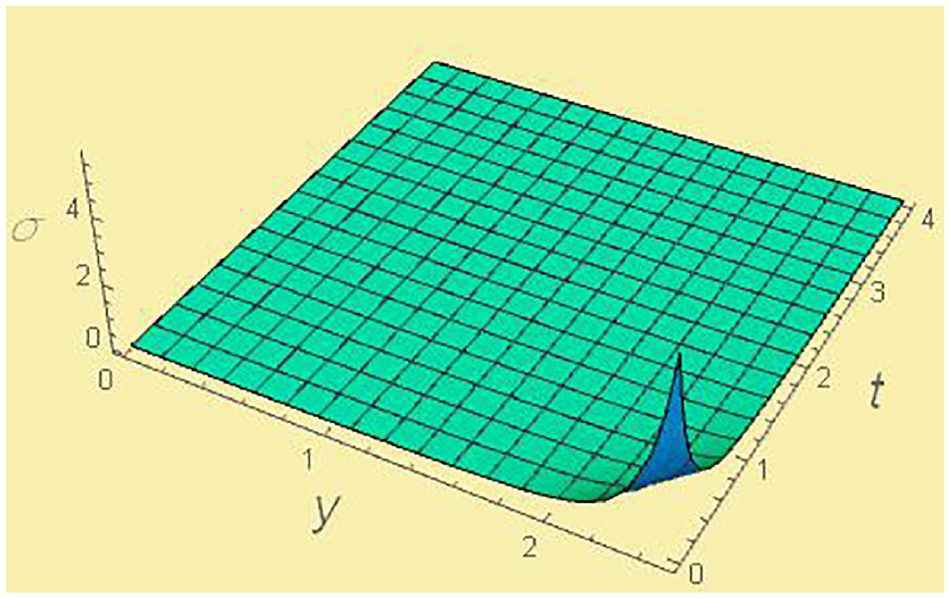
Entropy production σ s versus y and t for fixed χ = 0.5, and CA = 0.5.

Kinetic Inequality versus y and t for fixed χ = 0.5, and CA = 0.5.

dUS versus y and t for fixed χ = 0.5, and CA = 0.5.

dUv versus y and t for fixed χ = 0.5, and CA = 0.5.

dUR versus y and t for fixed χ = 0.5, and CA = 0.5.

Thermodynamic force XT versus y and t for fixed χ = 0.5, and CA = 0.5.

Thermodynamic force Xn versus y and t for fixed χ = 0.5, and CA = 0.5.

Thermodynamic force XR versus y and t for fixed χ = 0.5, and CA = 0.5.

(a) Concentrations nA versus t and χ for a fixed y = 0.4, and CA = 0.5, (b) concentrations nB versus t and χ for a fixed y = 0.4, and CA = 0.5, and (c) concentrations n versus t and χ for a fixed y = 0.4, and CA = 0.5.

(a) Temperature TA versus t and χ for a fixed y = 0.4, and CA = 0.5, (b) temperature TB versus t and χ for a fixed y = 0.4, and CA = 0.5, and (c) temperature T versus t and χ for a fixed y = 0.4, and CA = 0.5.

Entropy S versus t and χ for a fixed y = 0.4, and CA = 0.5.

Entropy production σ s versus t and χ for a fixed y = 0.4, and CA = 0.5.

Kinetic Inequality versus t and χ for a fixed y = 0.4, and CA = 0.5.

dUS versus t and χ for a fixed y = 0.4, and CA = 0.5.

dUv versus t and χ for a fixed y = 0.4, and CA = 0.5.

dUR versus t and χ for a fixed y = 0.4, and CA = 0.5.

Thermodynamic force XT versus t and χ for a fixed y = 0.4, and CA = 0.5.

Thermodynamic force Xn versus t and χ for a fixed y = 0.4, and CA = 0.5.

Thermodynamic force XR versus t and χ for a fixed y = 0.4, and CA = 0.5.
Now, we will discuss the manner of the NHNG atoms in the non-equilibrium state:
Figure 1(a)–(c) clarify the meaning that the gas concentration is an extensive parameter, while Figure 2(a)–(c) elucidate the implication that the gas concentration is an intensive parameter. A similar manner was introduced by Figures 23 and 24(a)–(c) and Figures 12 and 13(a)–(c), while the number density n decreases, with time, the temperature T growing, these happen for all magnitudes of t. That is, according to the fact of the uniform pressure, check Figures 1 and 2(c). Similarly, while the number density n decreases, with time, the temperature T growing, these happen for all magnitudes of

(a) Concentrations nA versus t and CA for fixed χ = 0.5, and y = 0.4, (b) concentrations nB versus t and CA for fixed χ = 0.5, and y = 0.4, and (c) concentrations n versus t and CA for fixed χ = 0.5, and y = 0.4.

(a) Temperature TA versus t and CA for a fixed y = 0.4, and χ = 0.5, (b) temperature TB versus t and CA for a fixed y = 0.4, and χ = 0.5, and (c) temperature T versus t and CA for a fixed y = 0.4, and χ = 0.5.
Figure 25(a)–(c) shed light upon an increment in
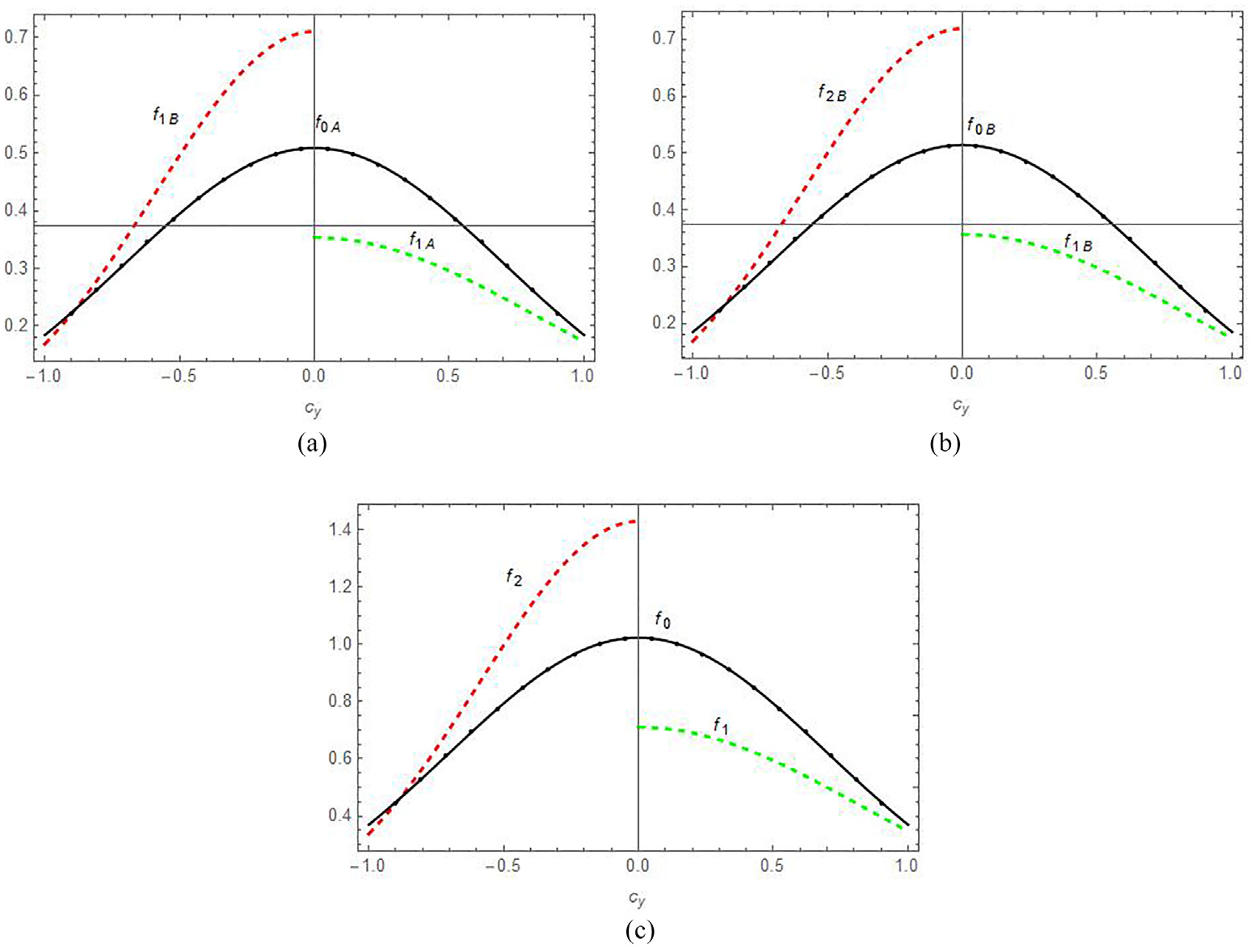
(a) The comparison between the combined perturbed velocity distribution functions fA [f1,f2] and equilibrium velocity distribution function foA at (χ = 0.5) for a fixed y value (y = 0.4), (b) the comparison between the combined perturbed velocity distribution functions fB [f1,f2] and equilibrium velocity distribution function foB at (χ = 0.5) for a fixed y value (y = 0.4), and (c) the comparison between the combined perturbed velocity distribution functions f [f1,f2] and equilibrium velocity distribution function fo at (χ = 0.5) for a fixed y value (y = 0.4).
The entropy S always growing with time, and the entropy production σ has non-negative magnitudes for all values of time t, temperature ratio x, and the molar fraction
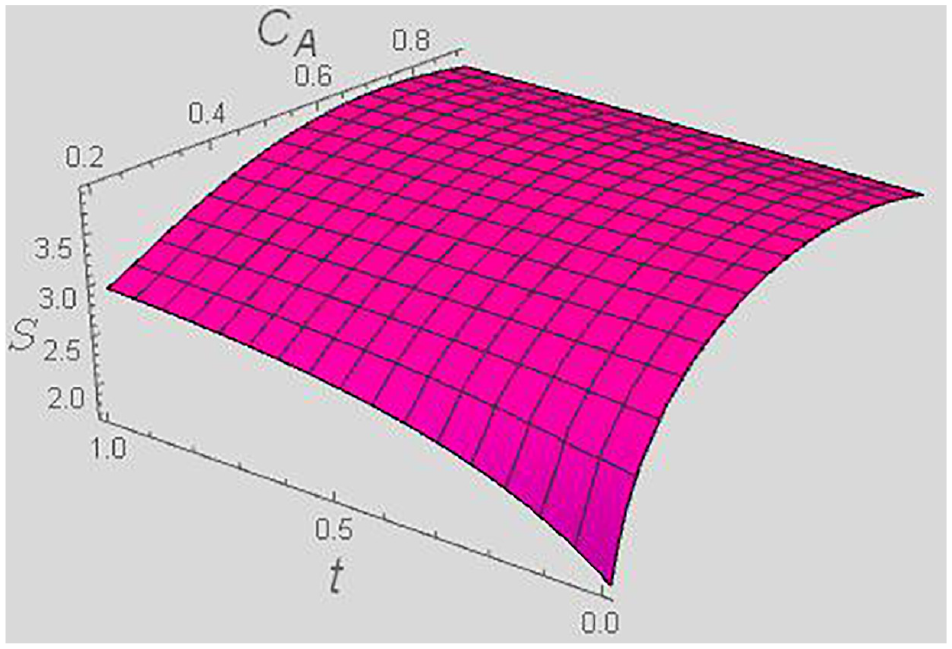
Entropy S versus t and CA for a fixed y = 0.4, and χ = 0.5.

Entropy production σs versus t and CA for a fixed y = 0.4, and χ = 0.5.
The manner of the various participants of the IEC can be clarified in Figures 7–9, 17–19, and 28–30). The Gibbs formula derives the numerical ratios among the diverse involvement of the IEC based upon extensive parameters total derivatives. Bear in mind their different tendencies, the maximum numerical magnitudes of the all contribution at different RF strengths, are in quantity ordered as follows:
For fixed values (
; see Figures 7–9. The participation of
For a fixed value (
; see Figures 28–30. The participation of
For a steady value

dUS versus t and CA for a fixed y = 0.4, and χ = 0.5.

dUv versus t and CA for a fixed y = 0.4, and χ = 0.5.
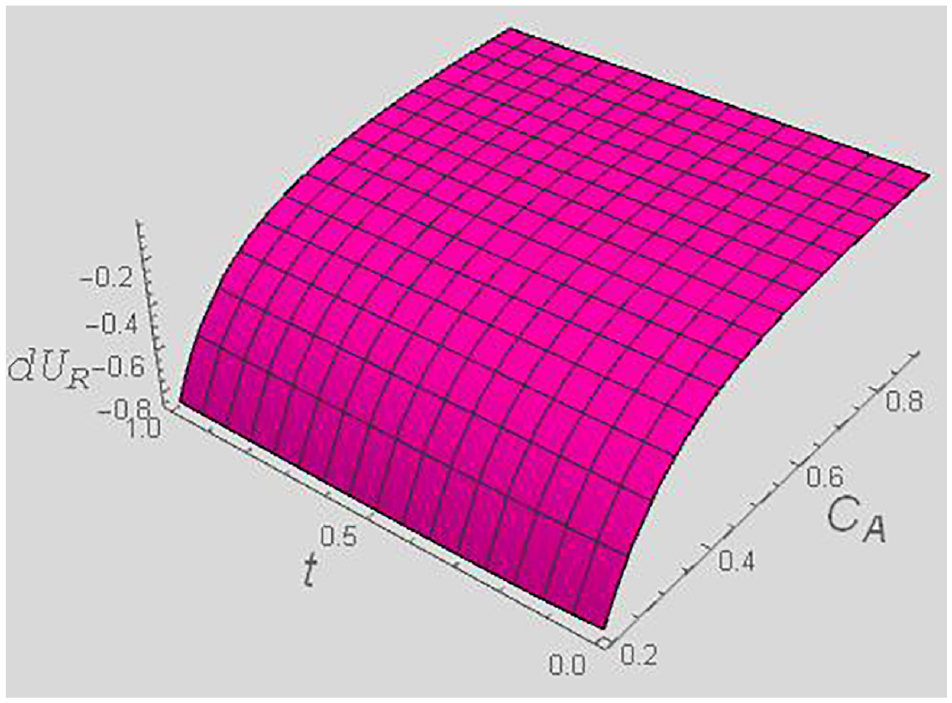
dUR versus t and CA for a fixed y = 0.4, and χ = 0.5.

Kinetic coefficient
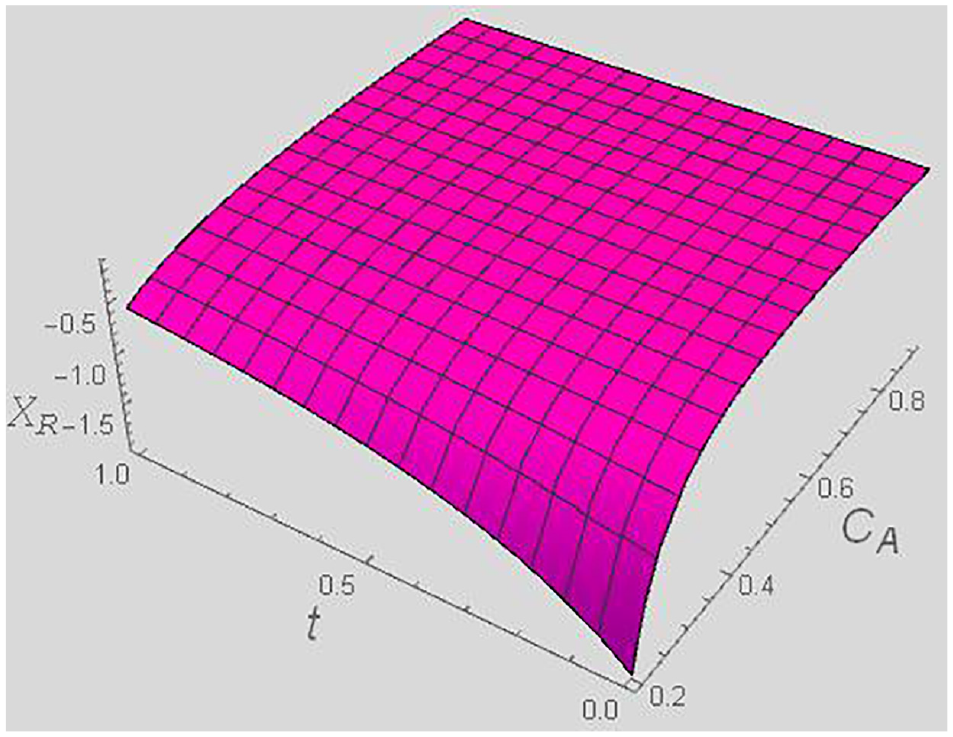
Thermodynamic force XR versus t and CA for a fixed y = 0.4, and χ = 0.5.

Thermodynamic force XT versus t and CA for a fixed y = 0.4, and χ = 0.5.
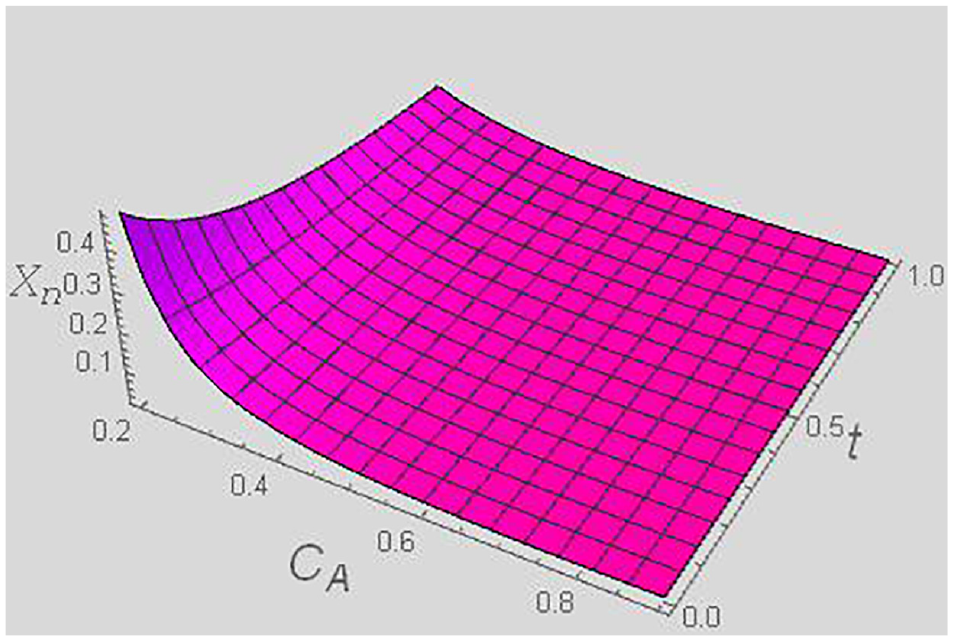
Thermodynamic force Xn versus t and CA for a fixed y = 0.4, and χ = 0.5.
Figure 21 indicated that,
From our investigation, the constraints imposed on the kinetic coefficients

Kinetic coefficient

Kinetic coefficient

Kinetic coefficient

Kinetic coefficient
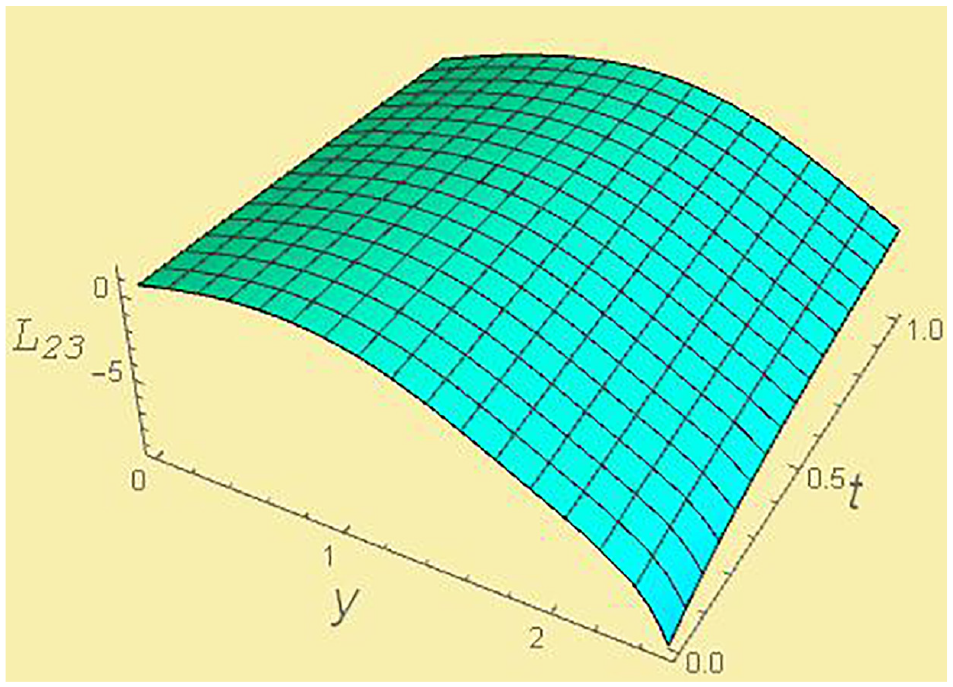
Kinetic coefficient

Kinetic coefficient

Kinetic coefficient
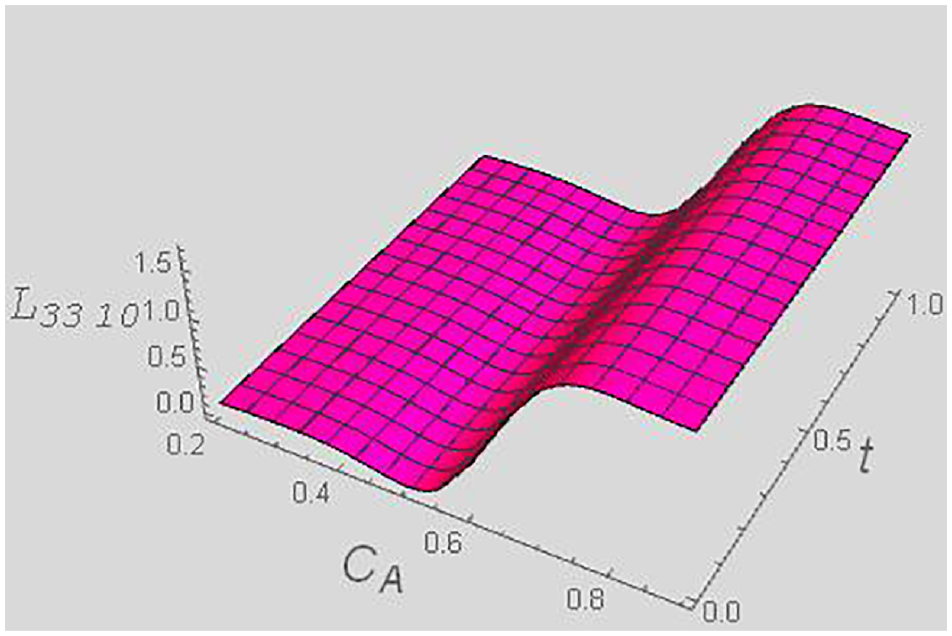
Kinetic coefficient

Kinetic coefficient

Kinetic coefficient
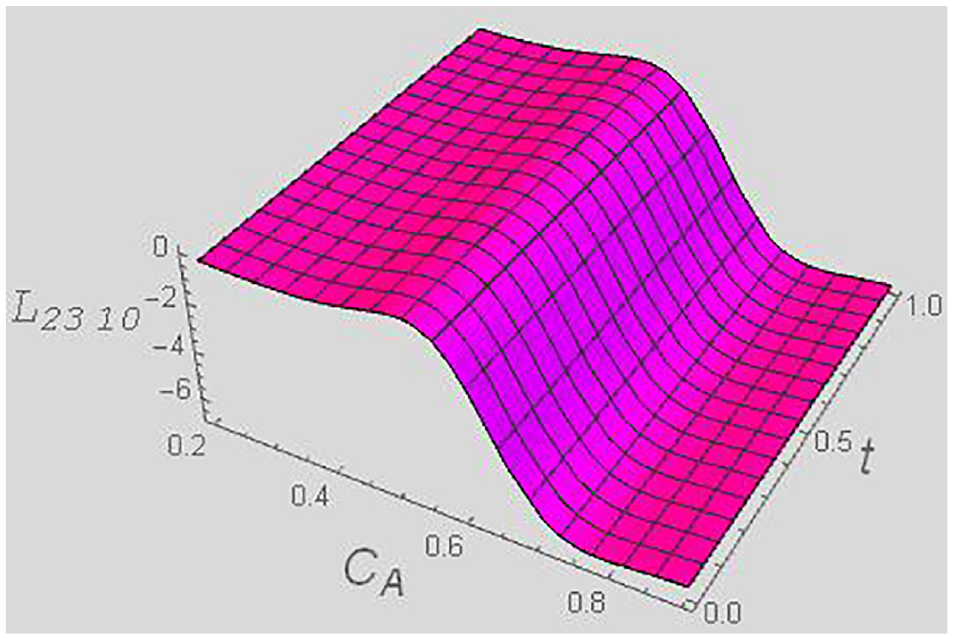
Kinetic coefficient
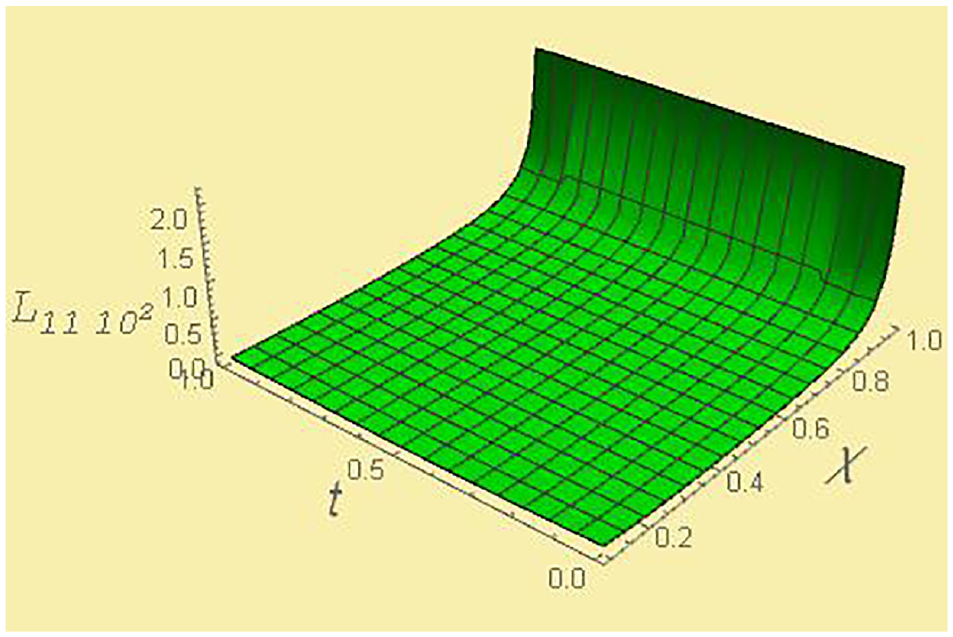
Kinetic coefficient

Kinetic coefficient

Kinetic coefficient

Kinetic coefficient
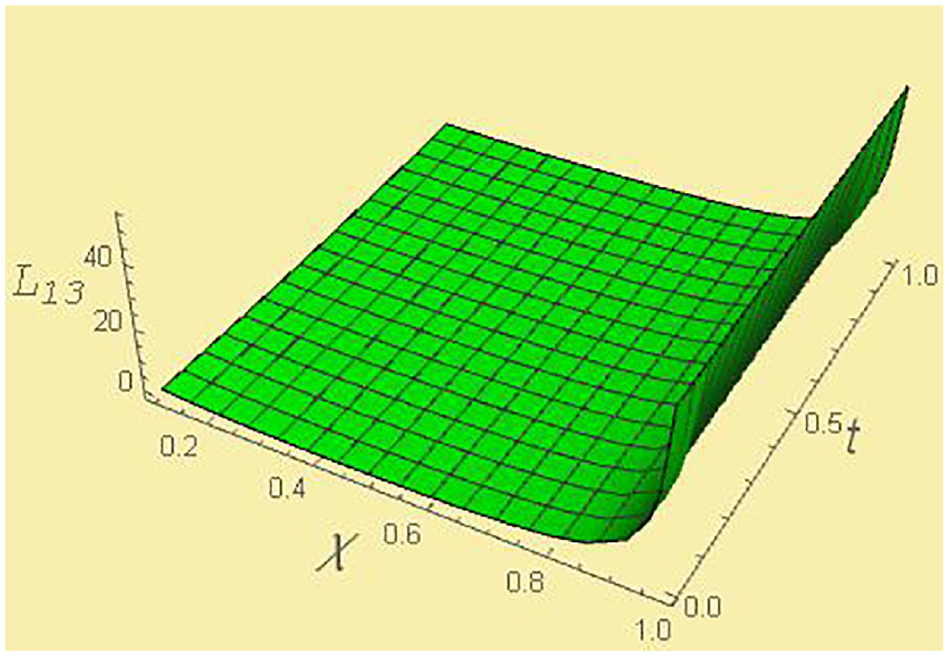
Kinetic coefficient

Kinetic coefficient
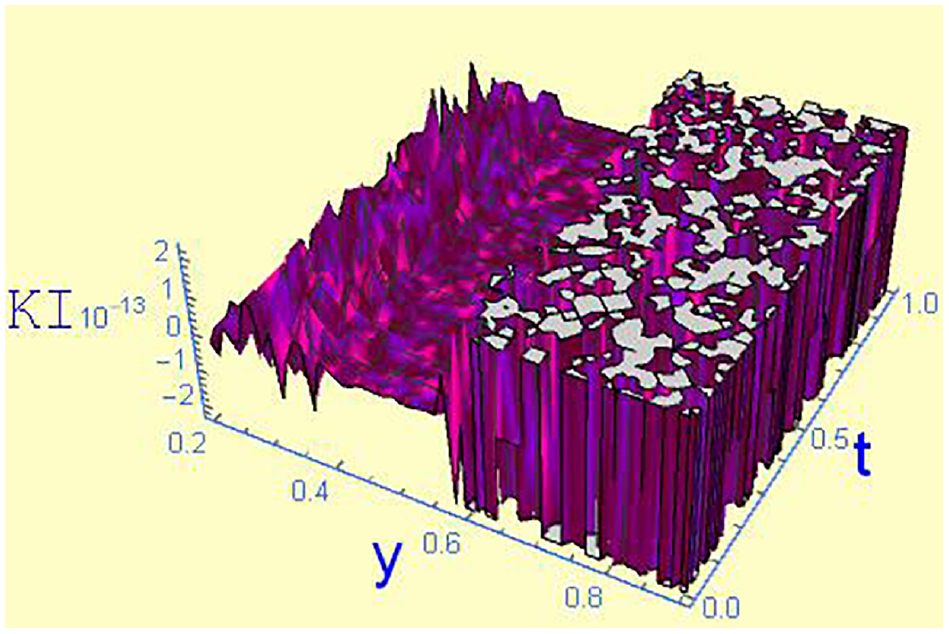
Kinetic Inequality versus t and CA for a fixed y = 0.4, and χ = 0.5.
Conclusion
The calculated results were demonstrated to conclude the following:
(a) The predominant vital factors that are directly proportional to the efficiency of the radiation energy in the total energy modifications of the entire system are:
(i) The ratio between the temperature of the plane solid plate surface and the NHNG atoms. (ii) The ratio between the mass of each component of the NHNG. (iii) The molar fraction of each component of the NHNG.
(b) At a temperature (
(c) The lighter NHNG component, helium, is influenced by a TR force more than the more massive gas component, argon.
(d) The first laws of thermodynamics (notably, the second law), the Le-Chatelier’s rule, the Boltzmann H-theorem, and the Onsager-Casimir reciprocal relations are all consistent with our new model.
(e) The negative sign at specific kinetic coefficients, related to cross effects, implies in such cases that according to the imposed TF (gradient), there is a heat flux in the reverse direction to the main flux. For instance, the negative sign in front of
Footnotes
Nomenclature
C The velocity of the gas particles.
d Effective collisions sphere diameter
fy The thermal radiation force component along y-axis direction.
Jy The entropy flux component along y-axis direction.
KB Boltzmann constant.
Kn Hydrodynamic Knudsen number.
Lij The kinetic coefficients.
P The pressure.
P s The pressure at the plate surface.
R The gas constant.
S Entropy per unit mass.
T The mean temperature.
Ts The temperature at the plate surface.
T1 The temperature related to g1.
T2 The temperature related to g2.
u The flow(bulk) velocity
U The internal energy of the gas.
dUR The radiation energy change.
dUS The IEC due to variation of entropy.
dUV The IEC due to variation of concentration.
V The gas volume.
VT Thermal gas velocity.
X1 The TF corresponding to variation of concentration.
X2 The TF corresponding to variation of temperature.
X3 The TF corresponding to variation of radiation field energy.
c The velocity of the light.
g The DF.
g1 DF for upward going particles
g2 DF for downward going particles
m The particle mass.
n The mean density.
ns The concentration at the plate surface.
n1 The concentration related to g1.
n2 The concentration related to g2.
y displacement variable.
Acknowledgements
I would like to thank Dr. Taha Abdel-Karim for helpful discussion, support, and encouragement.
Handling Editor: James Baldwin
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Egyptian Academy of Scientific Research and Technology by the associated grant number (No.6508), under the program of ScienceUP Faculties of Science.
Data Availability
No data, models, or code were generated or used during the study.