Abstract
To reduce diesel emissions while permitting the passive regeneration of the diesel particulate filter (DPF) at low temperatures, we developed three after-treatment DPF devices. These devices consisted of a ceramic body that was either bare or loaded with the catalysts CeO2 (DPF-CeO2) or Ce0.5Mn0.5O2 (DPF-Ce0.5Mn0.5O2). The effects of these units on soot, NOx, CO, and hydrocarbon emissions were assessed. On average, the DPF-Ce0.5Mn0.5O2 device outperformed the DPF-CeO2 device. In addition, increasing the engine load was found to raise the exhaust temperature while increasing the soot oxidation efficiency and reducing soot emissions. The maximum soot removal percentages of the DPF-CeO2 and DPF-Ce0.5Mn0.5O2 were 37.6% and 55.1%, respectively, under B100 working conditions. The extent of NOx removal also gradually increased as the load increased, and the average removal percentages were 8.6% and 15.0%, respectively. Both catalytic devices lowered CO emissions to a much greater extent than the bare DPF, with average removals of 45.8% and 55.6%, respectively, while the average hydrocarbon oxidation values were 39.1% and 50.9%, respectively. Notably, the hydrocarbon emissions were almost zero after Ce1-xMnxO2 catalysis under C100 working conditions.
Introduction
The primary harmful emissions from diesel engines are particulate matter (PM) and NOx, for which there is a trade-off relationship such that both cannot be decreased using internal purification methods alone.1–4 Consequently, diesel emissions control technology has become an important field of research. Diesel particulate filters (DPFs) are currently the primary means of reducing PM emissions and can provide particulate collection efficiencies in excess of 90%. However, this is purely a physical method of collecting PM and does not actually eliminate the particulates themselves, meaning that the DPF is subject to clogging. For this reason, there is an urgent demand for techniques to regenerate DPFs, especially passive systems. The passive regeneration technologies developed to date have employed chemical catalysis to reduce the activation energy associated with the oxidation of particulates, thus lowering the PM ignition temperature such that the particulates can be oxidized at the temperature of the diesel engine exhaust.5–8 Combining a DPF with an appropriate catalyst can also reduce NOx in the exhaust, and thus achieve the goal of simultaneously reducing PM and NOx. Consequently, selecting catalysts that exhibit a high level of activity is the key to optimizing passive regeneration technology.
Rare-earth-based catalysts have unique physiochemical properties because of their extremely abundant electronic energy-level structures. Among the rare earth oxides, ceric oxide (CeO2) has received considerable attention as a potential catalyst9–17 because of its special crystal structure, and because it is inexpensive and can readily and reversibly transition between Ce3+ and Ce4+. CeO2 can catalyze the oxidation of soot and also convert CO, hydrocarbons (HCS), and NOx in exhaust to CO2, H2O, and N2, respectively.18–20 In addition, CeO2 can serve as an oxygen buffer by rapidly storing oxygen when it is present in high concentrations in the exhaust and rapidly releasing it when the oxygen concentration is low. This characteristic can improve the catalytic activity and prolong the useable lifetime of the oxide.21–24 Gong et al. 23 established a model for a DPF catalytic regeneration system incorporating a Ce-based catalyst that allowed the regeneration performance to be predicted at various exhaust temperatures. The results indicated that a Ce catalyst should reduce the initial temperature required for the particulate oxidation reaction and also improve performance. Liu et al. 25 studied the effect of CeO2 on the oxidation of carbon black particles using X-ray diffraction, scanning electron microscopy, and image processing techniques. Their work demonstrated that CeO2 has desirable oxygen-storage/oxygen-release properties, high catalytic activity, and stability during the carbon black oxidation process. Daturi et al. 26 used H2 to reduce the CeO2 surface, and the resulting high concentration of surface oxygen vacancies was found to degrade NOx without the use of a reducing agent. Moreover, analyses using infrared spectroscopy and mass spectrometry determined that the NOx degradation activity was closely related to the concentration of oxygen vacancies on the CeO2 surface.
In the present study, the performance of a specially designed catalytic DPF (CDPF) device was assessed, based on the removal of pollutants from the exhaust of a 4DB1-40 diesel engine. Both CeO2 and Ce0.5Mn0.5O2 were selected for use in this system based on their high catalytic activities, and a wall-flow honeycomb ceramic filter was used as the support for these materials by coating the ceramic with the catalysts via an impregnation method.27,28 The CDPF units were installed in the diesel exhaust system to carry out an experimental study. Trials were also performed using a blank DPF without the catalyst coating for comparison purposes, and the effects of the two catalysts (CeO2 and Ce0.5Mn0.5O2) on carbon, NOx, CO, and HC emissions were investigated. The potential disadvantages of this new device were assessed, and possible improvements were identified, with the aim of demonstrating new concepts for DPF catalytic regeneration technology.
Experimental
Experimental apparatus
The test engine was a four-cylinder, turbocharged, intercooled, electronic control common-rail diesel engine, for which the primary technical parameters are provided in Table 1. No changes were made to the original structure or operating parameters of the diesel engine before the experiment, and only the original after-treatment device was uninstalled.
Specifications of the test engine.

A schematic diagram of the test bench is shown in Figure 1. In this apparatus, sampling tubes were installed at the front and rear of the DPF to monitor the pollutant concentrations and allow assessments of the effect of the DPF on NOx, CO, HC, soot, and PM emissions. During these trials, a CAC250 electric dynamometer was used to control the revolution rate and torque of the engine, and the speed and torque measurement accuracies were 0.05% and 0.1%, respectively. An FC2005 monitoring system was utilized to measure the performance indexes of the engine (power, torque, revolution rate, fuel consumption, soot output, flow, and temperature) in real-time with a high level of precision. The emissions data were automatically and simultaneously recorded. Gaseous pollutants, such as NOx, HCs, and CO, were determined using a Horiba MEXA-7200D emission analyzer. Among these, NOx was analyzed via a chemiluminiscence method, while CO was detected using non-dispersive infrared spectroscopy and HCs were monitored using a flame ionization detector. The measurement accuracies for NOx, HCs, and CO were 2 ppm, 2 ppm, and 1 ppm, respectively. The fuel consumption rate was measured using a 735 S instantaneous fuel consumption meter (AVL Co.), with a measurement accuracy of 0.12%. The air intake flow was determined with a Sensyflow FMT700 -P mass flow meter. The soot output was measured using a 415 S filter-type smoke meter, with a measurement accuracy of 0.01 FSN.
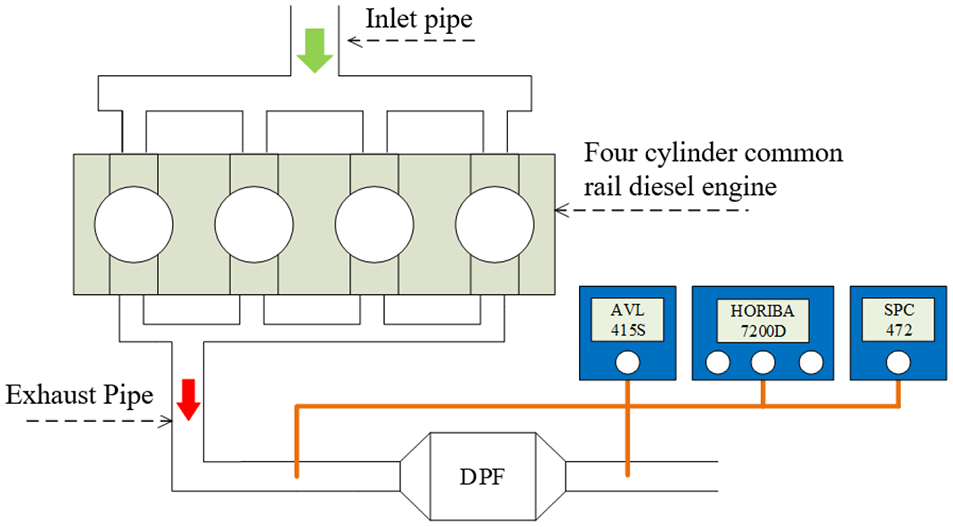
Schematic diagram of the engine test apparatus.
Experimental DPF device
Because of the significant exhaust output from a diesel engine and the high strength required of filter materials, honeycomb carriers are typically used to ensure sufficiently low resistance to flow together with the appropriate level of strength. A cordierite ceramic was used as the support in this study, and the physical parameters of this material are summarized in Table 2. Typically, the preparation of the sizing agent is an important step in fabricating such ceramics, and the compatibility and stability of this agent must be considered, because these factors directly affect the eventual catalytic performance. The primary components of the sizing agent used in these experiments were pseudoboehmite, acetic acid, and γ-Al2O3. Stoichiometric quantities of pseudoboehmite, acetic acid, and deionized water were initially weighed out and mixed to form a suspension to enhance the specific surface areas of the catalysts. Subsequently, the catalysts were loaded onto the DPF using an impregnation method, after which the impregnated DPF was heated.
Physical properties of the cordierite support.

Experimental program
The European steady-state test cycle (ESC), which has been in effect in the European Union since 2001, includes a test cycle for the Euro III standard and above that includes 13 operating conditions (four load rates at A, B, and C rotational rates and idle). In the present study, these test procedures were initially employed to derive the external characteristic curve of the diesel test engine. During these trials, the throttle position was fixed at 100% and the rotational rate of the engine (as controlled by an electric dynamometer) was increased from 800 to 2800 r/min. The resulting curves showed that the A, B, and C rotational rates were 1787, 2230, and 2673 r/min, respectively, while the corresponding loads were 25%, 50%, 75%, and 100% and the engine idle speed was 800 r/min. In this work, the ESC13 procedure was used to provide the operating parameters, in conjunction with the specific operating sequence and times provided in Table 3.
Thirteen operating conditions of the ESC cycle.

ESC: European steady-state test cycle.
Three after-treatment devices were assessed: a DPF ceramic support loaded with the CeO2 catalyst (DPF-CeO2), a DPF loaded with the Ce0.5Mn0.5O2 catalyst (DPF-Ce0.5Mn0.5O2), and a bare ceramic with no catalyst. The soot, NOx, HC, and CO emissions obtained with each of these systems were determined under the ESC13 working conditions and compared to explore the effects of the catalysts on pollutant removal.
Direct, continuous sampling of the exhaust after preheating was performed under each set of operating conditions during the test cycle and the average values were calculated. This exhaust gas sampling, as well as measurements of the engine exhaust flow and power, were performed in real-time during operation of the engine, and the resulting data were weighted. Gaseous pollutants were monitored during the last 30 s of each distinct set of working conditions, and the average concentrations of NOx, HC, and CO emissions associated with each set of conditions are reported. Soot emissions were also measured under the ESC13 working conditions. Throughout the test process, the flow of exhaust particulates was diluted with compressed air and the particulates were collected on filter paper. The sampling time under each set of working conditions was a weighting coefficient of each 0.01 for 4 s.
Results and discussion
Exhaust temperatures and oxygen levels during the ESC cycle
Both the exhaust temperature and oxygen concentration are critical factors determining the effectiveness of the after-treatment device. In this work, trials were performed using the same engine conditions with the various DPF permutations to compare the effects of the different catalysts. Figure 2(a) and (b) shows the oxygen concentrations and exhaust temperatures at the entry to the DPF during the ESC13 test cycle. It is evident from Figure 2(a) that the oxygen concentration in the exhaust decreased as the engine load was increased and increased (although to a lesser extent) with the revolution rate. The maximum oxygen concentration of 18.58% was obtained under idling conditions. This occurred because the fuel feed increases along with the load such that the air–fuel ratio in the combustion chamber of the cylinder decreases and oxygen consumption increases. Because the fuel feed is lowest while idling, the highest oxygen surplus is present.

O2 concentrations and exhaust temperatures during the ESC test cycle: (a) O2 concentration; (b) exhaust temperature.
Figure 2(b) demonstrates that the diesel exhaust temperature increased with both the load and the revolution rate, with values of 489°C, 544°C, and 571°C for a full load at revolution rates A, B, and C, respectively. This correlation is attributed to increases in the fuel feed at higher loads, which raise the thermal energy output during the combustion process. In addition, at a constant load, increasing the revolution rate will increase the number of work cycles per unit time and the gas flow field in the cylinder, leading to more intense combustion that raises the exhaust temperature.
Effect of Ce1-xMnxO2 on diesel engine emissions
Effect of Ce1-xMnxO2 on soot emissions
The stability and properties of slurry are directly related to the performance of catalyst. The main components of the paste used in this study were pseudo-thin bauxite, acetic acid, and γ-Al2O3. Three DPF supports were coated with the same slurry such that changes in the emission concentrations were primarily caused by the catalytic activity of the CeO2 or Ce1-xMnxO2. Figure 3 summarizes the soot emissions from the diesel engine downstream of the three DPF after-treatment devices during the ESC test cycle. The soot emissions after the various DPF devices all show a similar trend: the emissions increase along with both the load and the revolution rate and are lowest while idling. Under the same working conditions, the Ce0.5Mn0.5O2 removed soot more efficiently than the CeO2, presumably because the Mn-loaded nano-CeO2 possessed superior catalytic activity and a wider temperature window, and so more effectively promoted soot oxidation. Under a 25% load at rotational rates A, B, and C, the catalysts do not exhibit a suitable level of activity because of the low exhaust temperatures, and so the after-treatment soot emissions of all three DPF units are basically the same. The exhaust temperature increased along with the load, which in turn enhanced the activity of the Ce1-xMnxO2 and the soot oxidation efficiency to reduce soot emissions. Typically, the optimal soot removal by catalysis is achieved under B100 working conditions, and the soot removal efficiencies of the CeO2 and Ce0.5Mn0.5O2 under these conditions were found to be 37.6% and 55.1%, respectively. This greater efficiency is due to the higher exhaust temperature produced under B100 working conditions as being higher than the exhaust temperature under A100 conditions. The higher temperature increases the catalytic activity, improves the oxidation efficiency of soot, and reduces the emission of soot. However, the revolution rate associated with the B100 conditions is lower than that under C100 conditions, meaning that the exhaust will flow through the DPF over a longer time span, which could also promote the catalytic oxidation of soot particles.
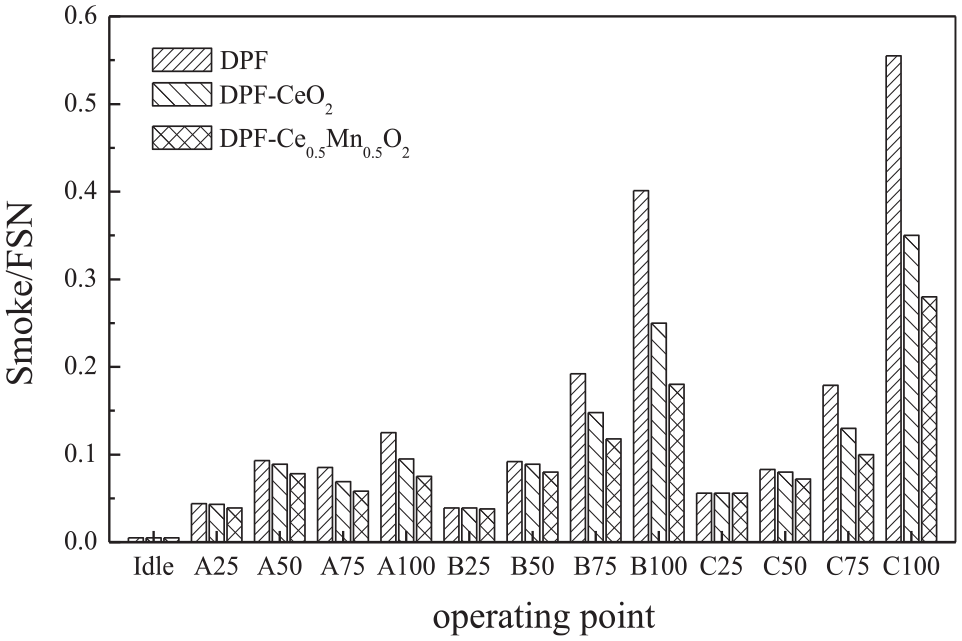
Smoke emissions during the ESC test cycle after the DPF.
Effect of Ce1-xMnxO2 on NOx emissions
Figure 4 compares the NOx emissions in the diesel exhaust after passing through the three after-treatment devices (DPF, DPF-CeO2, and DPF-Ce0.5Mn0.5O2) under the 13 working conditions. These data show that compared with the blank DPF, the NOx emissions under each set of working conditions were decreased to varying degrees by the Ce1-xMnxO2 catalyst. The removal efficiency increased with increases in the load, and the performance of the Ce0.5Mn0.5O2 was also higher than that of the CeO2. Removal efficiencies were 2.1% and 8.9% at a 25% load and rotational rate A for CeO2 and Ce0.5Mn0.5O2, respectively, and 21.2% and 37.1%, respectively, at a 100% load. The blank DPF showed poor performance because it had no catalyst. In addition, a greater concentration of PM would presumably be gathered with increases in the load and the temperature would also increase, both of which are conducive to the removal of NOx. The NOx removal efficiency of the Ce1-xMnxO2 tended to plateau at a 25% load at each revolution rate because the low temperatures under these conditions did not promote the reactions of PM and NOx. Notably, the temperature was only 180°C under idling conditions, which is below the catalyst activation temperature, and so there was no NOx removal effect. By contrast, the temperature under C100 working conditions was higher than the optimal catalyst activation temperature, resulting in a lower NOx removal efficiency than at revolution rates A and B.
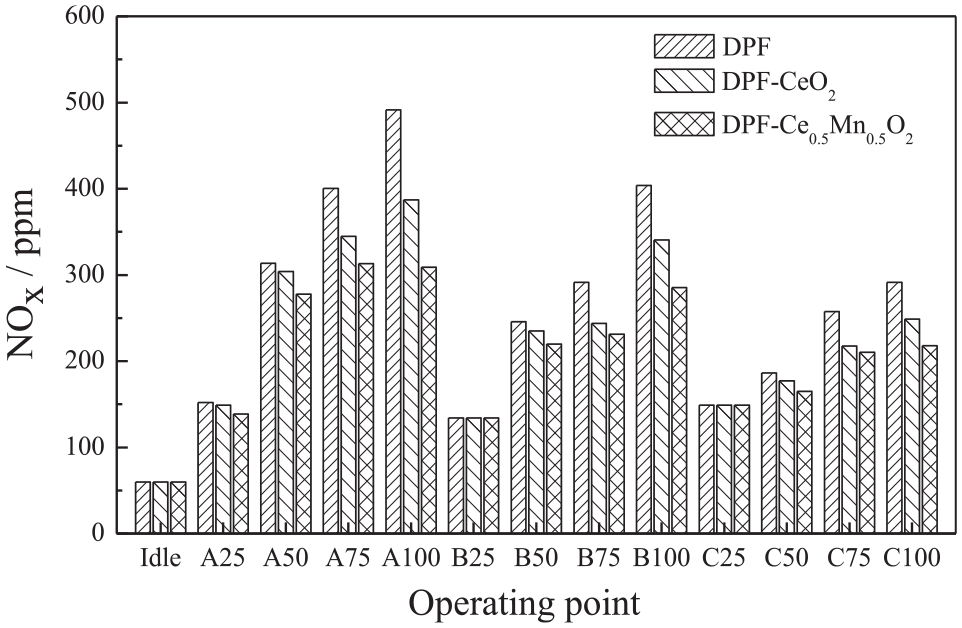
NOx emissions during the ESC test cycle after the DPF.
The NOx emission concentrations obtained from all three DPF units increased with increases in load when the revolution rate was held constant. This effect can be attributed to increases in the average effective pressure, flame temperature, and average temperature in the cylinder as the load increases, all of which are associated with higher NOx emissions. At the same load, faster revolution rates provided lower NOx emissions. When the diesel engine combustion system is unchanged, the injection advance angle is fixed. At higher rotational speeds, the physical mixing time of the oil bundle and the air is shorter. Less mixture is formed during the combustion delay period, and the premix heat release rate is lower, so less NOx is created. In other words, higher engine speed leads to shorter reaction time or residence time at high temperature, causing the level of NOx emissions to decreases with engine speed. 29 The average NOx removal efficiencies of the CeO2 and Ce0.5Mn0.5O2 were determined to be 8.6% and 15.0% under the ESC13 working conditions. These data are consistent with (although not identical to) the results obtained from catalytic activity simulations.30,31
Effect of Ce1-xMnxO2 on CO emissions
CO is the primary intermediate product during the combustion of diesel fuel and is at least partly converted to CO2 in the cylinder. This transformation proceeds via the following reaction.

In the case of oxygen-enriched combustion, the CO level in the exhaust will increase with increases in the stoichiometric excess of fuel. However, during the combustion of a dilute fuel mixture, the production of CO will remain essentially unchanged with changes in the equivalence ratio. Typically, if R represents the carbon hydroxyl, the reaction that generates CO during the combustion of a hydrocarbon can be expressed as below.
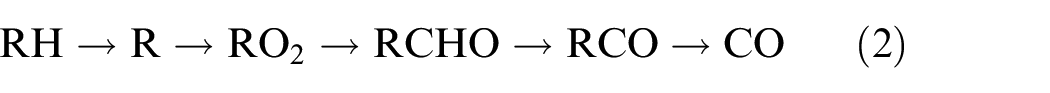
Thus, the primary source of CO during combustion is the pyrolysis and oxidation of RCO to form CO.
Figure 5 compares the CO emissions after the exhaust has passed through the three after-treatment devices (DPF, DPF-CeO2, and DPF-Ce0.5Mn0.5O2) under all 13 working conditions. The CO emissions after the DPF-CeO2 and DPF-Ce0.5Mn0.5O2 were significantly lower than the value obtained using the bare DPF, indicating that both catalysts had an effect. The catalytic efficiency of the Ce1-xMnxO2 was greater than 93% at a 100% load at each revolution rate, while the oxidation efficiency was almost 100% under the C100 working conditions, giving close to zero CO emissions. In comparison, under idling and low load conditions, the exhaust temperatures were below the activation temperatures for the catalysts, which led to low CO oxidation efficiencies. The average catalytic efficiencies of the CeO2 and Ce0.5Mn0.5O2 for CO oxidation were 45.8% and 55.6%, respectively, under the ESC13 working conditions. This is attributed to an increase in the number of oxygen vacancies that promote CO oxidation in the Ce0.5Mn0.5O2. The addition of Mn likely increases the concentration of lattice defects in the CeO2, which is conducive to the formation of oxygen species and so contributes to oxygen adsorption, migration, and transformation. As a result, the CO oxidation efficiency is markedly improved.
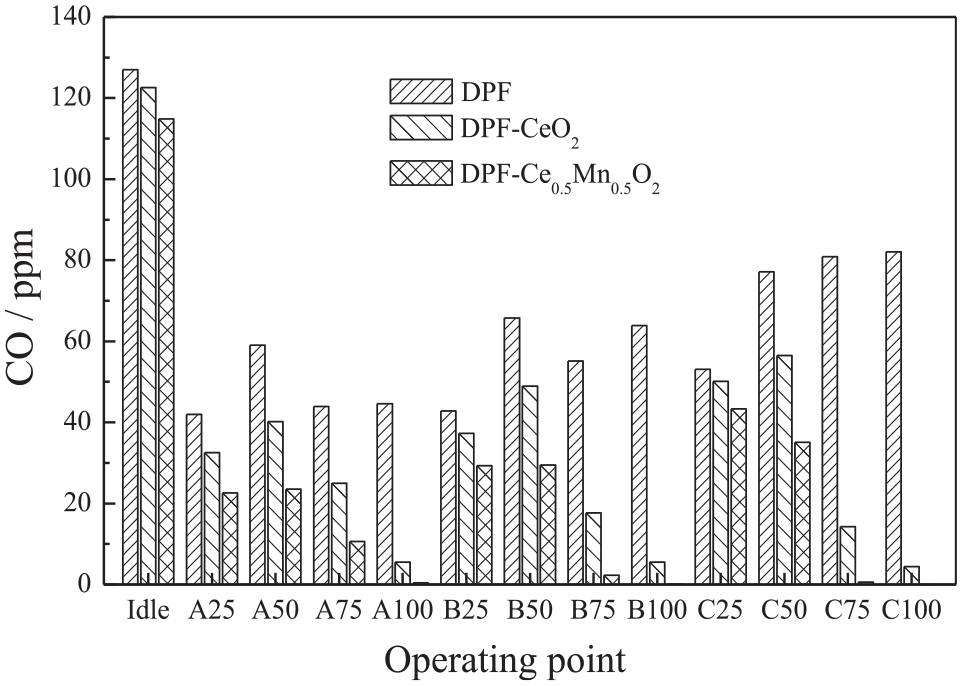
CO emissions during the ESC test cycle after the DPF.
Effect of Ce1-xMnxO2 on HC emissions
Figure 6 compares the HC emissions after the three after-treatment devices (DPF, DPF-CeO2, and DPF-Ce0.5Mn0.5O2) under the ESC13 working conditions. The HC emissions obtained from all three devices decreased as the load increased at all revolution rates, and the HC emissions were highest under idling conditions. This occurred because a low load produces a very dilute mixture in the cylinder such that the combustion becomes less intense and extremely low temperatures are present in the cylinder. Under such conditions, there is also a weak mixture in the vicinity of the injection site, meaning that the fuel is not completely combusted, which is the primary source of unburned HCs. Therefore, the HC emissions from a diesel engine under idling and low load working conditions are higher than those at high loads. The data in this figure also demonstrate that the Ce1-xMnxO2 significantly lowered the HC emissions. Typically, the catalytic efficiencies of the CeO2 and Ce0.5Mn0.5O2 with regard to HC reduction were 92.3% and 98.7%, respectively, under the C100 working conditions, due to the extremely high exhaust temperatures during these trials. Importantly, almost zero HC emissions were observed after Ce1-xMnxO2 catalysis. In addition, the average HC oxidation efficiencies of the CeO2 and Ce0.5Mn0.5O2 under the ESC13 working conditions were 39.1% and 50.9%, respectively. The improved performance of the latter is primarily attributed to changes in lattice defects and a higher concentration of oxygen species following the addition of Mn to the original CeO2, indicating that lattice oxygen is also directly related to the catalytic activity. These results also demonstrate that the presence of oxygen species enhances HC oxidation efficiency. Specifically, the activity of the original oxide was increased by approximately 50% after Mn doping.
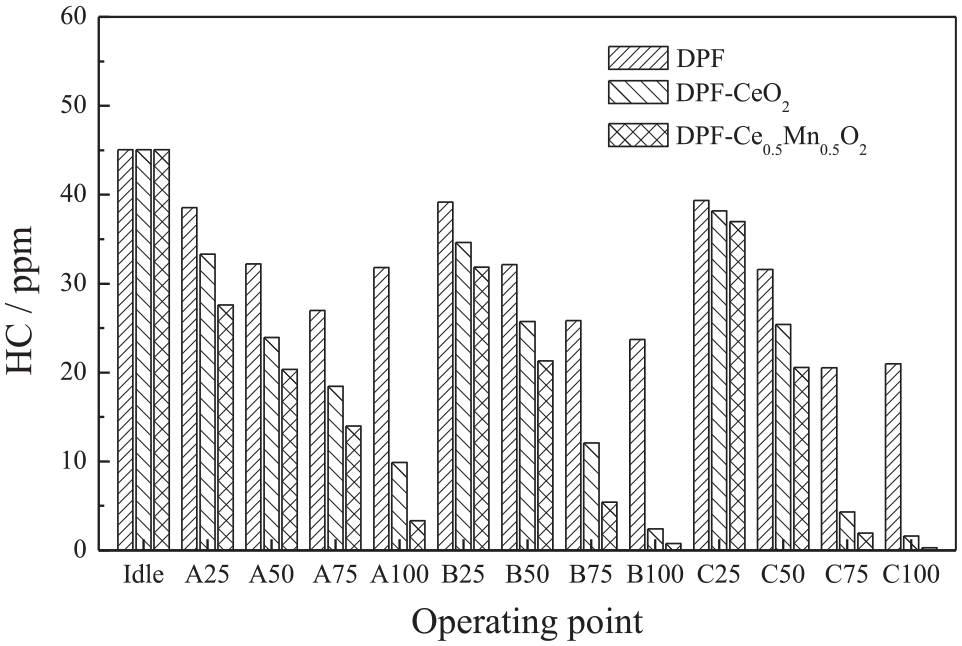
HC emissions during the ESC test cycle after the DPF.
Emission characteristics
Soot is typically formed as a result of the thermal decomposition of fuel at high temperatures under oxygen-deficient conditions. The transition of Ce from the CeO2 (+4) valence state to the Ce2O3 (+3) state occurs via a relatively low-energy process. Thus, CeO2 can provide the oxygen necessary for the reduction of soot while being converted to Ce2O3 as in equation (3). 30 While acting as an oxidation catalyst, CeO2 also lowers the activation temperature for carbon combustion and thus enhances soot oxidation and promotes complete combustion.

Because of its high thermal stability, the Ce2O3 formed while reacting with the soot remains active after being generated in the initial combustion process and is subsequently reoxidized to CeO2 through the reduction of NO, 31 via the reaction in equation (4). 31 Emissions of NO are also thus at least partly inhibited as the CeO2 is regenerated.

As shown in Figure 5, increased CO emissions were observed when using the blank DPF. When using this device, there was insufficient oxygen to convert all the carbon into CO2, with the result that some of the fuel did not fully combust and CO was generated as an intermediate product during combustion. However, because of its useful oxygen storage and release capabilities, the Ce1-xMnxO2 can act as an oxygen buffer and provide oxygen to promote the conversion of CO to CO2. The corresponding reaction is presented in equation (5).

Cerium ions can lower the energy barrier associated with breaking H—C bonds and so accelerate high temperature dehydrogenation reactions. The corresponding reaction is presented in equation (6).

The catalytic mechanism of the Ce1-xMnxO2 is illustrated in Figure 7, which shows that the presence of cerium oxide enhances oxygen mobility during the reaction cycle. In addition, smaller particle sizes facilitate the migration of oxygen from the interiors of nano-crystallites to their surfaces. The interactions in Ce-Mn oxides can be regarded as a transition of lattice oxygen (Olatt) and chemisorbed oxygen (Oads) to active O* on the active sites of the catalyst surface, which favors the soot oxidation process. During the redox cycle, the Ce4+/Ce3+ redox couple permits oxygen storage, because the CeO2 acts as an oxygen buffer by storing/releasing oxygen species in association with the couple. This, in turn, facilitates the mobility of oxygen in the redox cycle. In addition, partial doping with Mn inhibits the growth of catalyst grains and increases the specific surface area of the catalyst. The concentration of lattice defects in Ce1-xMnxO2 increases and oxygen species are formed more abundantly after Mn enters CeO2, because these defects are conducive to the adsorption, migration, and transformation of oxygen. Moreover, the concentration of lattice oxygen, which has a strong adsorption and activation effect, increases with increasing Mn doping concentration.

A schematic summarizing the catalytic effect of Ce1-xMnxO2.
Conclusion
In this study, both CeO2 and Ce1-xMnxO2 catalysts were loaded onto DPF supports. The abilities of these materials to simultaneously catalyze the removal of soot, NOx, CO, and HCs from diesel exhaust were examined and several conclusions were drawn.
First, the soot removal efficiency of the Ce0.5Mn0.5O2 was superior to that of the CeO2 under the same working conditions. The diesel exhaust temperature increased with the load, which enhanced the Ce1-xMnxO2 activity and therefore the soot oxidation efficiency, thus reducing soot emissions. The highest degree of soot removal was achieved under B100 working conditions. The purification efficiencies of the CeO2 and Ce0.5Mn0.5O2 under these conditions were 37.6% and 55.1%, respectively.
Second, the Ce1-xMnxO2 catalyst caused varying degrees of reduction in NOx emissions (depending on the working conditions) compared with the results obtained using the blank DPF. The NOx removal efficiency increased with the load, and the Ce0.5Mn0.5O2 catalyst outperformed the CeO2 catalyst. The average NOx removal percentages exhibited by the CeO2 and Ce0.5Mn0.5O2 were 8.6% and 15.0%, respectively, under the ESC13 working conditions.
Third, the average catalytic efficiency of the Ce0.5Mn0.5O2 for CO elimination was higher than that of the CeO2 under the ESC13 working conditions, with the two materials showing average efficiencies of 45.8% and 55.6%, respectively. Finally, the HC removal percentages obtained from the CeO2 and Ce0.5Mn0.5O2 were 92.3% and 98.7%, respectively, under the C100 working conditions because of the extremely high exhaust temperatures associated with those conditions. Importantly, almost zero HC emissions were obtained when using the Ce1-xMnxO2. The average HC oxidation efficiencies of the CeO2 and Ce0.5Mn0.5O2 under the ESC13 working conditions were 39.1% and 50.9%, respectively.
Footnotes
Acknowledgements
The authors acknowledge support from the University Natural Science Research Project in Jiangsu Province (19KJD470003) and Research Foundation of NIIT (YK18-04-04, YK18-04-03).
Handling Editor: David Chalet
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
