Abstract
In bone tissue engineering, three-dimensional printed biological scaffolds play an important role in the development of bone regeneration. The ideal scaffolds should have the ability to match the bone degradation rate and osteogenic ability. This article optimizes the unit cell model of the microstructure including spherical pore, gyroid, and topology to explore degradation performance of scaffolds. Boolean operation of array microstructure unit cells and selected part of a computer-aided design (CAD) femur model are adopted to create a reconstructed scaffold model. Polylactic acid/
Keywords
Introduction
Reconstruction of sophisticated bone defects continues to have significant challenges in patients with insufficient bone dimensions. Although autogenous bone grafts harvested from a healthy region of the patient are still ordinarily considered the gold standard for enhancing bone repair, their use is restricted in clinical practice due to graft resorption rates, high donor site morbidity, and circumscribed bone availability. 1 There is an urgent need for a method of improving new bone formation and reducing the amount of autologous bone grafts. Recently, additive manufacturing (AM), which includes three-dimensional (3D) printing, stereolithography (SLA), fused deposition modeling (FDM), and selective laser sintering (SLS), has received developing attention in medical devices and tissue engineering.2–5 In addition, bone tissue engineering (BTE) including 3D printed bioscaffold technology has become a promising approach to bone repair and reconstruction.6–8 Parry et al. 9 created a biocompatible polymer scaffold with 3D printing technology, capable of sustaining vascularization and tissue ingrowth. Cox et al. 10 presented a systematic characterization of bone tissue scaffolds fabricated via 3D printing from hydroxyapatite (HA) and poly(vinyl)alcohol (PVOH) composite powders.
Biocompatibility, mechanical properties, and biodegradation are crucial to tissue engineering scaffolds.
11
Abert et al.
12
used three different components in the composite material to avoid accelerated degradation and pronounced volumetric swelling. Many studies have been focused on the biomaterials, bioactivity, porosity, mechanical properties, cell proliferation, and protein differentiation of the 3D printed scaffolds.
13
Domingos et al.
14
investigated the influence of nano-hydroxyapatite and micro-hydroxyapatite (nHA and mHA, respectively) particles on the in vitro biomechanical performance of polycaprolactone/hydroxyapatite (HA) scaffolds. An ideal scaffold utilized for bone regeneration, which should be biocompatible and biodegradable and have adequate physical and mechanical properties, should mimic characteristic features of natural extracellular matrix (ECM) and synchronize the speed of scaffold degradation and new bone growth. Drost et al.
15
explored a new treatment strategy for urinary incontinence. Human bone marrow mesenchymal stem cells (MSCs) of the first in vitro passage were exposed to 5-azacytidine (AZA) to induce myogenic differentiation. Expression of stem cell surface antigens and intracellular
According to the deficiencies in the above references, this research investigated the effect of different microstructures on the degradation performance and mechanical properties of 3D printed PLA/
Materials and methods
Microstructure design of scaffolds
This research established three microstructures of 3D printed scaffolds, which consist of spherical pore, gyroid, and topology structure, to explore the relevance between the micro-architecture of the scaffolds and degradation property. The scaffolds with size of 10 × 10 × 10 mm were printed for mechanical testing. Through the uniaxial tensile test of blended materials (PLA:
1. Spherical pore microstructure
Reconstituted unit cell of spherical pore was obtained by boolean calculation through cube and sphere. The objective of optimization was to achieve the best initial mechanical performance of the scaffold at constant porosity.
Design variables, objective functions, and constraints were established according to the following equations
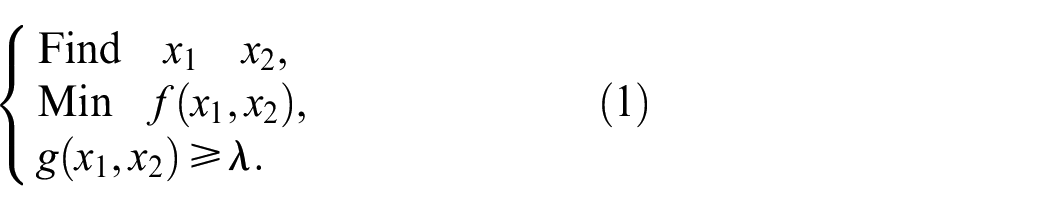
where
The size of reconstituted unit cell with spherical pore microstructure was optimized utilizing ANSYS Workbench (ANSYS, Inc. Canonsburg, PA, USA). Response surface charts obtained a more intuitive response of the two design variables to the output variables, as shown in Figure 1. By optimizing the minimum values of the maximum deformation and the maximum equivalent stress, the spherical pore radius was 1.3429 mm and the circular pore diameter was 1.1097 mm at the porosity of 70%.
2. Gyroid microstructure

Optimization results of spherical pore model: (a) Equivalent stress cloud. (b) Maximum deformation cloud.
Implicit surface modeling can describe the characteristics of scaffolds by using single mathematical equations.22,23 Triply periodic minimal surfaces (TPMSs) can promote cell migration and maintain high structure stiffness due to its open structure simultaneously. Here, the scaffold models were established by gyroid, which were usually used in scaffold design as a kind of TPMSs.
The gyroid surfaces were built using the following equation

The study implemented the visualization of the gyroid model and the OBJ file through K3DSurf software (Abderrahman Taha, open source), as shown in Figure 2. The OBJ file was imported into Rhinoceros software (CAD International, Avalon, NSW, Australia) to divide the surface into physical and spatial domains for creating the gyroid unit cell microstructure.
3. Topology-optimized microstructure
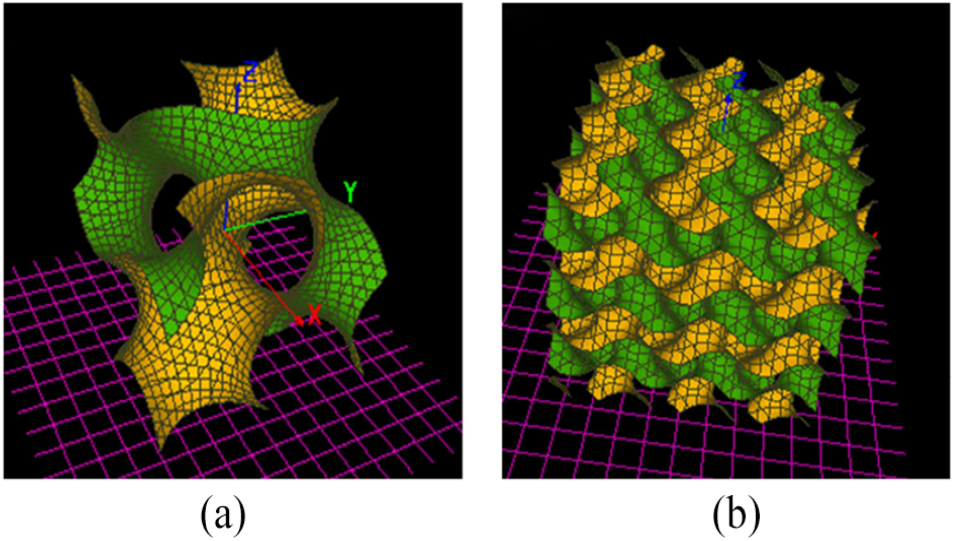
Gyroid surface structures: (a) Gyroid surface of single cycle. (b) Gyroid surface of multiple cycles.
Topology optimization as a mathematical method can optimize the material distribution to obtain desired structure according to prescribed constraints.24,25 A unit cell with three circular pores was selected as the original model, and the size of the unit scaffold was also fixed.
This research defined the objective function as strain energy, the constrain function as the volume, and controlled the porosity by volume. Topology optimization based on the principle of minimum strain energy was performed using ABAQUS software (Abaqus Inc. Palo Alto, CA, USA), as shown in Figure 3. As a result, reconstructed unit cell based on topological optimization was obtained.
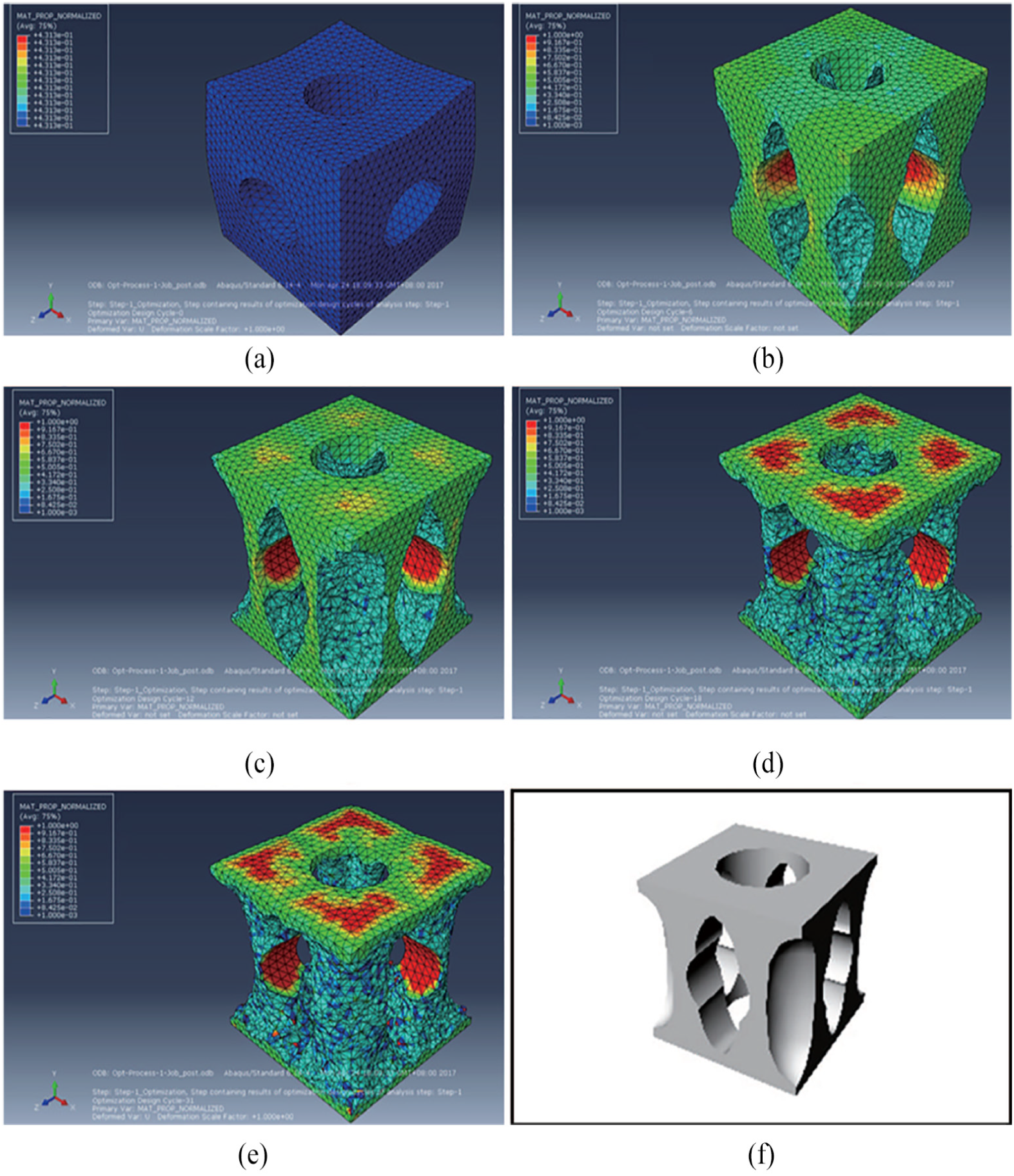
Process of topology optimization. (a) Initial model. (b) 6th topology optimization model. (c) 12th topology optimization model. (d) 18th topology optimization model. (e) 31st topology optimization model. (f) Reconstructed unit cell based on topological optimization.
Preparation of scaffolds
Digital representation (CAD model) of porous scaffold was established by 3D reconstruction of an average adult human femur, which converts computed tomography (CT) images to stereostructure by Mimics (Materialise Inc., Leuven, Belgium) and Geomagic Studio (Geomagic Inc., North Carolina, USA). Boolean operation of array reconstructed unit cells and selected part of the CAD femur model was adopted to create reconstructed scaffold model. As shown in Figure 4, a reconstruction model with spherical pore structure was introduced.
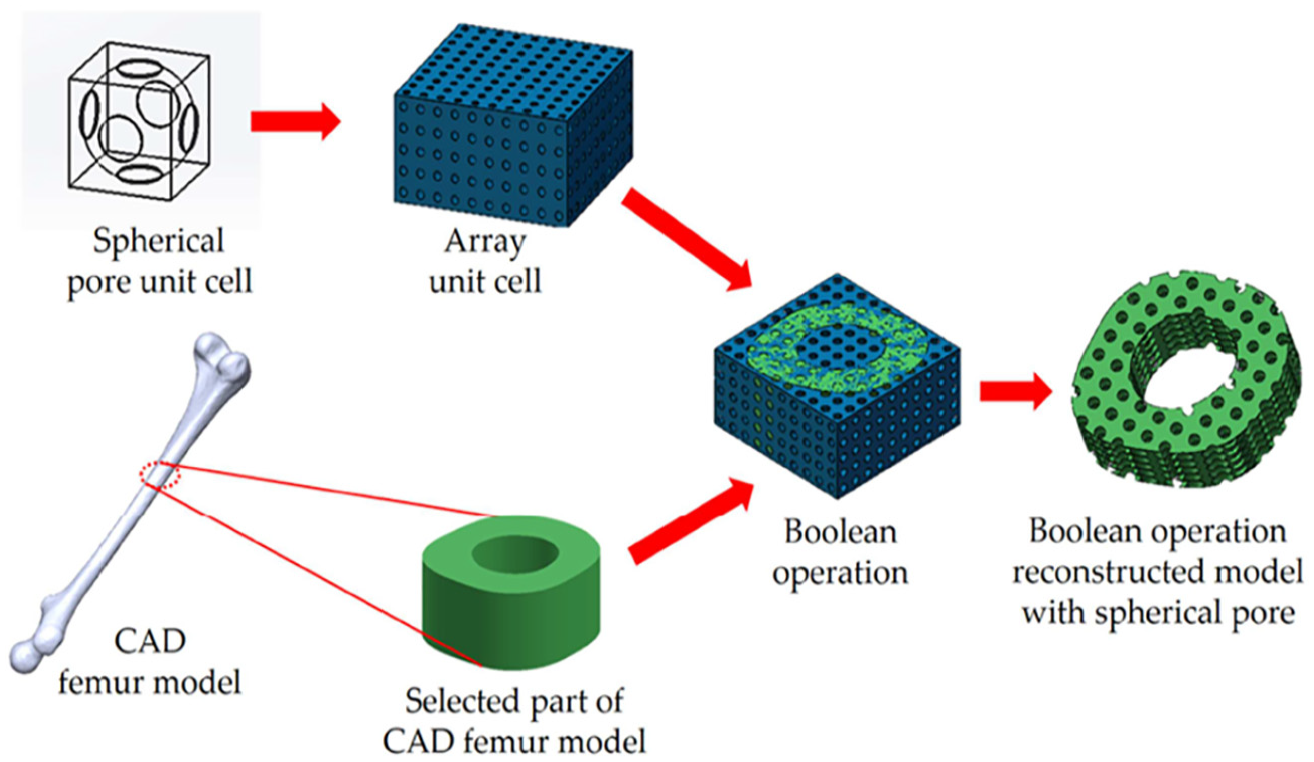
Boolean operation of array unit cells with spherical pore structure and selected part of CAD femur model.
Regarding material composition, HA,
The bio-inks for 3D printing of the scaffolds are composed of PLA,
The composite materials can be obtained with PLA:
Three-dimensional bio-printer (Regenovo Biotechnology Corp.) was then used to fabricate all PLA/

The designed and built scaffolds with spherical pore, gyroid, and topology architecture.
Degradation model of scaffolds
To establish the degradation model, a mathematical expression that can indicate the degradation rate and other factors is needed. Due to many factors affecting the degradation of materials, there is not a recognized degradation model. The present degradation models can be roughly divided into three categories: phenomenon model, probability model, and empirical model. Phenomenal models are models based on diffusion, dissolution, and reactions that can be applied to a wide range of polymer types. The probability model calculates the degradation probability according to the degradation time by assuming that the degradation of bone biological scaffolds is a random process. The empirical model is mainly based on a large number of experimental data to establish the regression equation, easy to determine the importance of certain factors.
PLA was used as the main material in this article. As the hydrolysis of PLA was dominated by bulk degradation, the degradation model was selected. First, the change of water content is simulated by Fick’s second law

where
The change of molecular weight is caused by hydrolysis and is related to water content

where
The change of elastic modulus is considered to have a linear relationship with molecular weight

where
Since the water molecules are diffused from the outer scaffold, the water content at the boundary is 1. According to its boundary conditions, the solution of Fick’s second law in the finite diffusion volume is

where
The average concentration of the commonly used volume in engineering is

where
The above equations can observe the real time change of elastic modulus of bone biological scaffolds in the degradation process so that further simulation analysis can be carried out.
Mechanical testing of scaffolds
The surface appearance of the scaffolds was observed by SEM (S-570; Hitachi, Japan) after the scaffolds were coated with gold by a sputter coater. The morphology and mechanical properties of the scaffolds were analyzed in the process of degradation. Compressive mechanical properties were analyzed by a mechanical tester (Instron 5569; Instron Corp., USA). Compressive force was applied in the Z direction of 3D printed scaffold with a 2.5-kN load cell and a cross-head speed of 1.5 mm/min.
Scaffold degradation testing in vitro
Ideal tissue engineering scaffold could gradually degrade when it was implanted into the human body. The scaffold rate of degradation in vivo should match with the growth rate of repair tissue. In vitro accelerated degradation experiments were performed to explore how scaffolds changed during degradation in a short time. PBS was chosen as the degradation liquid. The scaffolds are placed in a calorstat of 70°C after the scaffolds were put into the PBS. Due to the long cycle of in vitro degradation experiments, it usually takes several months for many polymer and calcium phosphate materials to change. To observe the mechanical properties of the scaffold in a short time, an in vitro accelerated degradation experiment was carried out at a temperature of 70°C based on the standard of accelerated degradation experiments. (70°C is the recommended temperature for accelerated degradation of PLA.) The timing method of accelerated degradation experiment is the same as that of non-accelerated degradation experiment, which is measured in days. In vitro accelerated degradation experiment is helpful to quickly observe how the mechanical properties of scaffolds change with degradation. But more accurate results still need to be obtained through in vivo degradation experiments to be studied in the future.
Statistical analysis
All data obtained at least in triplicate (
Results and discussion
Mechanical testing of scaffolds
The mechanical properties of the 3D printed scaffolds were shown in Figure 6. The results demonstrated that the mechanical properties (Young’s modulus and compressive stress) of the 3D printed scaffold with topology-optimized structure were better than the structures of gyroid and spherical pore before degradation. However, Young’s modulus reduction of the scaffolds with topology structure presented significantly higher than the scaffolds with spherical pore structure after degradation (
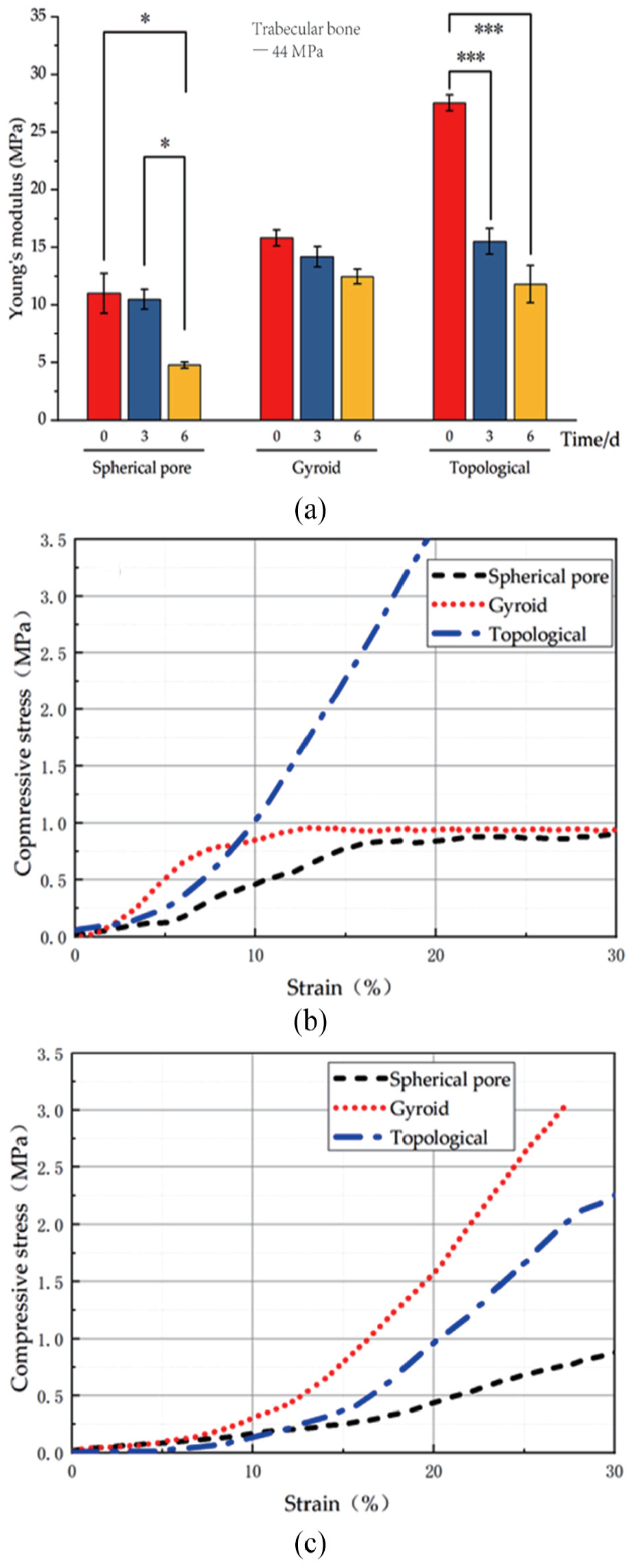
Mechanical testing of 3D printed PLA/
Microstructure analysis of scaffolds
The cell adhesion on the surface of scaffolds is affected by protein adsorption. After the scaffolds are implanted in the body, the protein is quickly adsorbed on the surface. The adsorbed proteins act as ligands to bind receptors on cell membranes, leading to cell adhesion. The microstructure of the scaffold surface plays an important role in the regeneration of BTE.
SEM image analysis of 3D printed PLA/
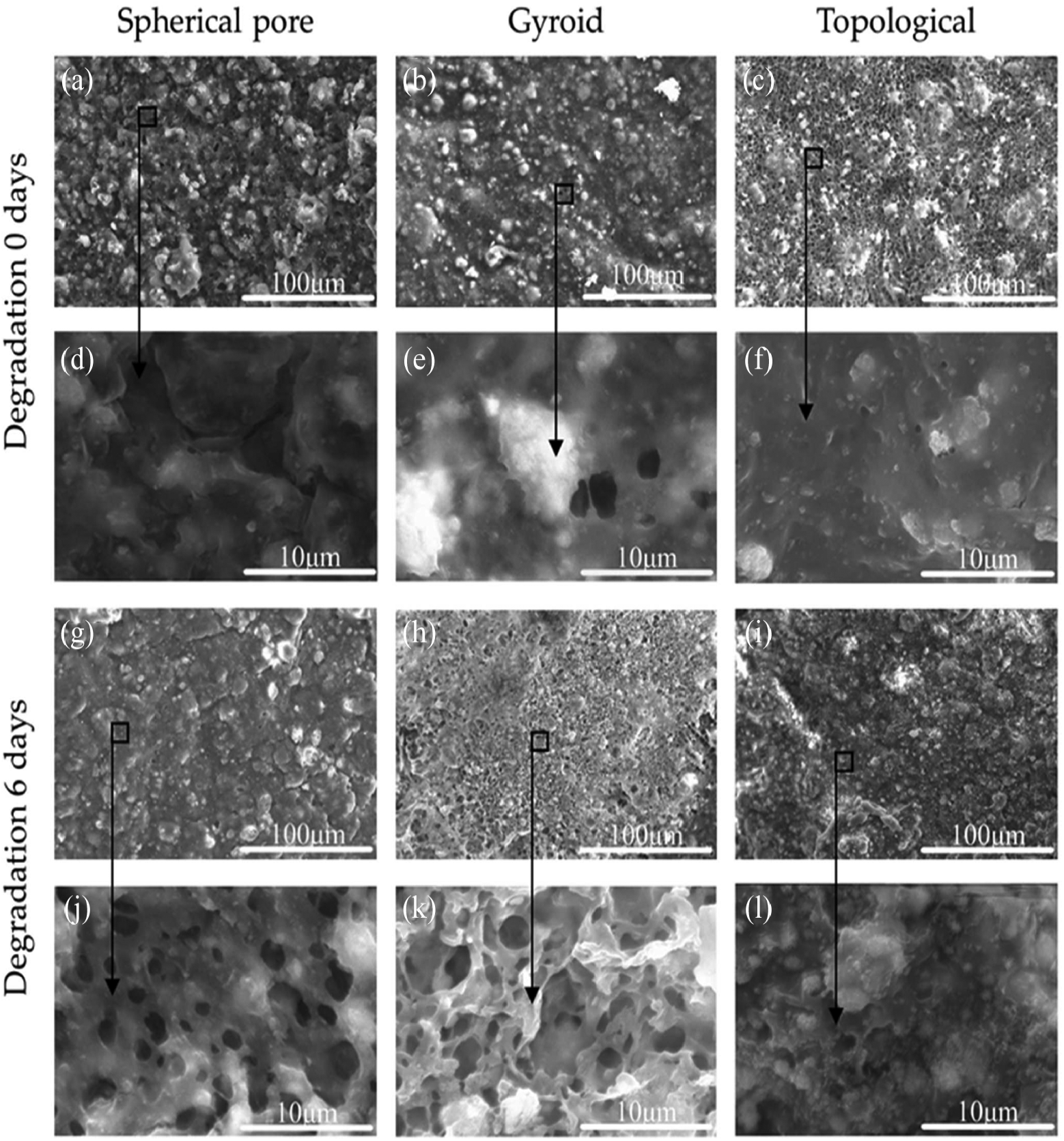
SEM image analysis of 3D printed PLA/
Mass analysis of scaffolds
In order to explore the mass changes of three bone scaffold structures in PBS simulated body fluids, the mass changes before and after scaffold degradation were analyzed, as shown in Figure 8. At day 3 and day 6, three samples for each type of scaffolds were taken out and put on a filter paper for seconds to remove the PBS from the scaffolds and then weighed. The results showed that the mass reduction of the 3D printed scaffold with topology structure was the best, followed by gyroid before degradation.

Mass of 3D printed PLA/
Based on the above analysis, the mechanical properties and degradation properties of the scaffolds with gyroid microstructure are better, and they are more suitable to be biological scaffolds for human bone tissue repair.
Discussion of scaffold degradation
Scaffold degradation is the key factor of bone tissue regeneration in BTE. As a kind of temporary scaffolds, bone biological scaffolds will degrade continuously over time. The degradation rate of ideal biological scaffolds should match the tissue regeneration rate and provide early mechanical support. As for the degradation of biological scaffolds in vivo, due to the fluid flow of human tissue and bone mineralization, the degradation rate of scaffolds is predicted to be faster than that of in vitro, and the regeneration rate of bone tissue is also faster. Further studies are needed to determine the mechanical properties of biological scaffolds in the process of degradation in vivo, as well as the actual efficiency of scaffold degradation and tissue regeneration and biocompatibility.
Conclusion
The scaffolds must not only have mechanical strength but also achieve time-controlled bio-absorption. The PLA/
Footnotes
Acknowledgements
The authors wish to thank the anonymous reviewers for their comments which led to improvements of this paper.
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (81704054), and the Fundamental Research Funds for the Central Universities (Grant No.HIT NSRIF. 2020032).
