Abstract
Acid leaching is extensively employed for extractive metallurgy, but its use on reclamation of shot blasting media has not been extensively studied. The parameters of acid leaching process such as rate of stirring, the leached material grain size, the solid (material) to liquid (acid) ratio, type of acid, concentration of acid, leaching temperature, and leaching duration govern the effectiveness of this process. The goal of this article is to investigate the effectiveness of acid leaching to recover the iron from used shot blasting media. Influence of varying leaching durations on the recovery percentage of iron from the used shot blasting media is investigated, and it is found that acid leaching can remove impurities from used shot blasting media and it can successfully recover 73.6% of iron on the particle surface level and 44.3% of iron overall. Hence, it is a feasible process for recovery of iron from used shot blasting media.
Introduction
Shot blasting is a process of impinging target surface with a stream of metallic blasting media (shots) under immense pressure. This process utilizes pressurized fluids from centrifugal wheel to propel the blasting media. 1 It is used in common applications such as smoothening the surfaces, removing rust or old paint from the surfaces, reclamation of metals from e-waste, and cleaning of heat exchanger fouling. The cleaned contaminants reported to incur cost as much as 0.25% of gross domestic product (GDP) and 2.5% additional release of CO2. 2 Shot blasting process generates a mixture of used blasting media and waste from the cleaned surface which needs to be suitably disposed. 3 Waste from the blasting media contains valuable metals such as iron, and its disposal without recovery brings adverse economic and environmental impact. 4 Disposal recycling of used media not only involves additional processing energy but also destroys primary energy employed in the manufacturing of the shots. Thus, a systematic procedure is required for the separation and recovery of iron (Fe). Recovered iron from the used steel shots can be reused for various industrial applications and processes. The recovery not only serves as significant benefit but also minimizes industrial waste. Various techniques including physical, chemical, and pyrometallurgical methods and leaching are employed for recovery of valuable constituents from the blasting waste. Physical, pyrometallurgical, and chemical processes are cost-ineffective and have secondary affluent issues, and recycling of the produce of such treatments destroys the primary energy. Leaching process is therefore a popular method in extractive metallurgy, and its merits may be fruitfully extended to recover shot blasting media. 5
The leaching of metals with acids falls under the category of hydrometallurgical processing, which in itself is a branch of extractive metallurgy. The hydrometallurgical processes are accomplished in three steps: leaching, purification, and recovery. Leaching dissolves the metal in a suitable aqueous solvent such as water, acid solutions, alkaline solutions, oxidizing solutions, and so on, and purification refers to the removal of impurities that contaminate the metals to be extracted. The surface of the used media covered with impurities initially dissolves less in the leaching media. As the dissolution progresses, more active subsurface is exposed which dissolves more with time. But, at the same time, progress of dissolution reduces the potency of the leaching media. 6 Thus, appropriate leaching time is an important aspect of recovery which not only affects process time but also the amount of leaching media and the amount of final waste. Finally, in the recovery step, the reduced metal or its compound is precipitated from the leached and purified solution. 7
Several researches have investigated recovery of a large number of metallic elements from different types of industrial waste. Sobianowska-Turek et al. 8 investigated leaching for recovery of Mn and Zn from zinc-manganese dioxide and zinc-carbon waste batteries. Elements such as Yttrium, Europium, Zn, Fe, Ti, Si, and Al are also extracted by leaching of cathode-ray tubes (CRT). 9 The effectiveness with which leaching can be performed depends largely on factors such as mixing/stirring speeds, grain-size, ratio of amount of solid (stock) and solvent (i.e. liquid) (S/L), type and concentration of acid, leaching temperature, and leaching time. 10 Various acids are used in the leaching process including phosphoric acid (H3PO4), 9 nitric acid (HNO3), 11 sulphuric acid (H2SO4), 12 and hydrochloric acid (HCl). 13 Each acid elicits different chemical reaction on various components present in the material being leached.
During the initial condition of the stock such as size of pellets or particles, S/L has substantial influence on leaching. Furthermore, during leaching, the stock needs to be homogeneously mixed with acids and should be continuously stirred at a specified rate depending on the stock being leached. During stirring, acid reacts with the pellets and releases reaction products and the metal being recovered through diffusion. The dissolution rate in the process of diffusion-controlled dissolution is described by equation (1) 14

where
The acid leaching is thus fast and cost-effective, significantly saves energy, and reduces affluent. Post-leaching suitability for reuse of the leached media for industrial applications can be of significant benefit. But, a number of factors control the effectiveness of leaching processes. This article presents an investigation on isolating, separating, and recovering iron from used blast cleaning media through acid leaching process. The recovered shots after blasting were found to be good enough to be utile for other processing applications such as powder metallurgy.
Materials and methods
The conventional methodology (please refer Figure 1) involves tedious handling of the waste, disposal of the waste, and scraping of the used shot blasting media. This is both environmentally invasive and not economic. The methodology adopted in this work, not the valuable blast media, is recovered in such a form in which it is suitable to be reused as a raw material for powder metallurgy application.
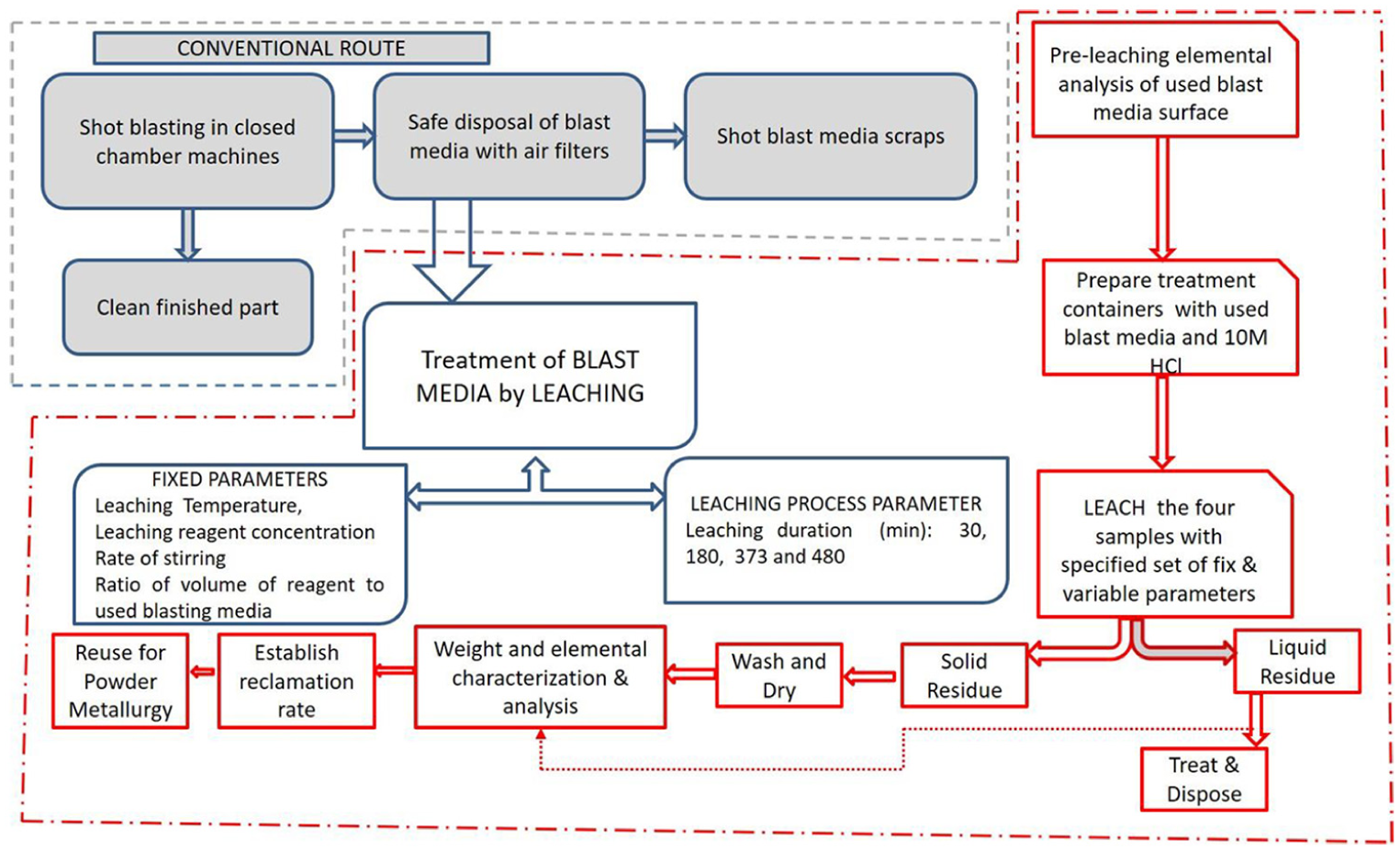
Methodology adopted in the present work.
The virgin steel shots (Siam IKK Co., Ltd., Malaysia), used as blasting media, were retrieved after shot blasting and were leached to reclaim Fe metal. The virgin shots were characterized to establish initial elemental composition (as given in Table 1).
Composition of virgin steel shot blasting media.

Characterization was done through energy-dispersive X-ray (EDX) and X-ray fluorescence (XRF) analyses. Average grain size of shots was also obtained through particle size analyzer. Leaching was performed in an acidic medium comprising of HCl in 10 M concentration. HCl was employed because most chlorides are soluble in water; hence, chlorides formed after leaching will get dissolved in the acid solution and can be easily removed.
The samples of used shot media were leached in HCl for varied durations. Four samples of 200 g of used shot blasting media were prepared by mixing 150 mL of 10 M HCl and placed in treatment containers. During leaching, the treatment mixtures were stirred at the rate of 200 rpm and the temperature was maintained at room temperature. Each of the four samples was leached for different durations: 30, 180, 373, and 420 min, respectively. After leaching, the liquid and solid residues were separated through filtration, and solid residue was washed, dried, and subsequently characterized to identify post-treatment elemental composition. A comparison of pre-treatment and post-treatment elemental composition was made to establish recovery rate, and total recovery percentage of iron was determined. Effect of leaching duration on the recovery percentage of iron was subsequently analyzed through measured results.
Results and discussion
The characterization of used shot was done post blasting in pre-leaching-treatment condition to analyze element pickup via XRF, SEM (scanning electron microscope), and EDX. Through a comparison of the elemental composition of virgin shots with that of the used shots, contamination of the surface during blasting was analyzed. The used shots were subsequently leached and the analysis of the residues was done to obtain the recovery rate.
Pre-leaching characterization and analysis of the used shots
The results of surface XRF spectroscopy, SEM, and EDX analysis on the surface of the used blast media in pre-leaching-treatment condition are given in Tables 2 and 3, and Figure 2, respectively.
Pre-treatment elemental composition by XRF.

XRF: X-ray fluorescence.
Pre-treatment EDX results.

EDX: energy-dispersive X-ray.

Pre-experimental EDX analysis electron micrograph.
The results of XRF (given Table 2) reveal that the element of interest (i.e. Fe) comprises of only 0.65%. On comparing the elemental composition (Table 1) of the virgin steel shots with that of used shots (Table 2), it is evident that the shots have picked up significant amount of elements such as Ti and Zn after blasting (in concentration of 26.12% and 10.5%, respectively). These elements were picked-up from the surface of the substrate which was blasted, or from the impurities removed during shot blasting.
Morphological analysis was performed by SEM (Figure 2) and EDX (Table 3) for the elemental analysis of the steel shot. Three points considered for EDX are labeled as 1, 2, and 3 in the SEM micrograph shown in Figure 2 (region 4 depicts the marked area). Overall composition of entire area (labeled as 4) was determined by averaging the composition obtained from each of the three spots.
Results of elemental concentration (wt%) from EDX (Table 3) obtained at three spots (identified in Figure 2) indicate that the Fe has overall weight percentage of 4.3, and at all the three spots, Fe appears to be scarce on surface. Similar to results of XRF (Table 2), it is inferred that a large portion of surface is contaminated by impurities. However, unlike results of XRF characterization (Table 2), the element with highest wt% is oxygen (O) instead of Ti and Zn. This indicates that a large portion of sample surface possessed oxide impurities. Presence of oxygen on the surface is most likely caused by oxidation of iron shots during exposure to air. The heating during plastic deformation of the shots during blasting would have facilitated the oxidation.
Post leaching characterization and analysis of the used shots
During leaching, the metallic and non-metallic materials present on the surface of the shots being leached are dissolved in the leaching reagent. The amount of the material being dissolved with the increase in leaching duration is high. But, at the same time, as the residue dissolves in the leaching medium, its dissolution potency is adversely affected. Thus, the analysis of effect of leaching duration on the recovery rate, dissolution amount, and the particle size of the remnant shots becomes involved.
Elemental characterization of used shots post leaching
During leaching, four samples of 200 g each were leached in 10 M HCl for four different leaching durations. For the analysis of recovery of elements, XRF analysis was performed. The recovery data obtained from XRF were used to obtain trend of percentage recovery. The results of XRF analysis for each of the four samples are given in Table 4. Elements that were initially present in the shot blasting media (Fe, Si, P, S, Cr, and Mn) were considered for obtaining the recovery trend. The XRF data of the elements initially present in the shots have been segregated and presented in Table 5.
XRF elemental concentrations for various leaching durations.

XRF: X-ray fluorescence.
XRF concentrations initially present elements after various leaching durations.

XRF: X-ray fluorescence.
A graph of wt% of the elements versus leaching time based on the data in Table 5 was plotted to analyze the trend of recovery of specific elements over leaching time and is given in Figure 3.

Graph of element percentage versus leaching duration.
The trend for rate of recovery of Fe is obtained and is represented through a dotted line in the plot shown in Figure 3. It is evident that the amount of Fe increases by 0.16% as leaching duration is increased from 0 (initial) to 180 min resulting in an average recovery of 0.0009% per minute, indicating a slow rate of removal of impurities and recovery of iron. However, with increase of leaching duration from 180 to 373 min, there is a sharp increase of 37.97%, that is, an average recovery rate of 0.20% per minute. Finally, when the leaching duration is further increased from 373 to 420 min, amount of iron witnessed an increase of 5%, that is, the rate of increase in Fe slows down to 0.11% per minute. When compared to the original composition of the shot blasting media (Table 1), which comprises of 97% Fe, the acid leaching process managed to recover 44.3% of Fe.
Similar to the pre-experimental EDX characterization of initial shots, spectra for treated shots were also obtained. The SEM micrograph of treated sample is depicted in Figure 4. Four distinct points on particle’s surface (labeled as 1 to 4, and average area of rectangle is labeled as “5”) were considered for EDX spectrum. For each point, the elemental compositions were obtained from a set of spectrum patterns and wt% of individual elements were considered for analysis.

Post-experimental EDX analysis scanning electron micrograph.
Elements present on the surface of preprocessed sample were O, Na, Ti, Ca, C, Fe, Si, Zn, and Mg (please refer Table 3). However, the EDX spectrum of treated samples showed that only two elements were present and they are Fe and O. Absence of other elements from the surface implies that the leaching procedure has successfully removed the impurities, leaving only Fe and possibly its oxides on surface of the particles. Overall elemental composition of entire area (i.e. area 5) was obtained by combining and averaging the composition obtained from each of the four spots and the same is given in Table 6.
Elemental concentrations of post-leached samples obtained through EDX.

EDX: energy-dispersive X-ray.
Across area 5, wt% of Fe is 73.6%, while the rest of it is O. These results are in contrast with the pre-treated EDX findings, in which only 4.3% of iron was present on the surface of particle, while a large portion was taken up by foreign elements, predominantly O, Ti, and Zn. This indicates that the leaching process has effectively removed the impurities from the surface with the exception of O. Presence of 26.4% of oxygen implies that the surface has been oxidized during shot blasting process and due to exposure to air for prolonged periods of time while the used shot blasting media is stored.
Post leaching particle morphology and size distribution of used shots
Particle size distribution of the virgin steel shots is given in Figure 5. The particle size distribution of the reclaimed media after leaching was also plotted and the same is given Figure 6.

Particle size distribution of initial steel shots.
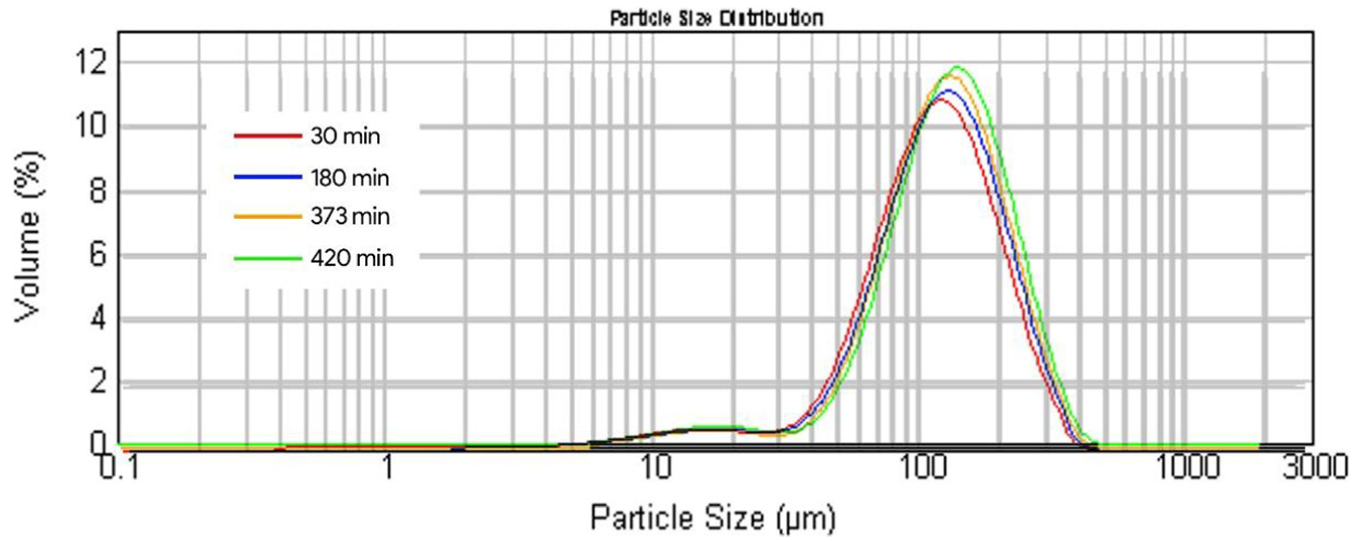
Combined particle size distribution for all four samples.
Results of Figure 5 illustrate that median particle size of the samples being 110.749 μm is well within suitable range for powder metallurgy applications, which is 38–250 μm. 15 Meanwhile, the particle size distribution of the sample ranges from a minimum of 35.974 μm for the 10th percentile, d(0.1), to a maximum of 217.217 μm for the 90th percentile, d(0.9). In essence, the average particle size in pre-experiment samples is of suitable size to be utilized as metal powder for powder metallurgy applications.
Four sets of particle size distribution plots for different leaching times were obtained and are given in Figure 6. Figure 6 shows that the particle size distribution across all four samples remains relatively close without much deviation. Furthermore, Figure 7 indicates that the overall size of particles gradually increases with increase in leaching duration. The acid leaching process does not significantly influence the particle size distribution of the used shot blasting media. It is evident from Figure 7 that the variation in the particle size is attributed to two attributes, that is, percentile (d(0.1), d(0.5), and d(0.9)) and leaching time (30, 180, 373, and 420 min). However, whether the two attributes are independent or not for affecting the particle size needs to be tested. For this purpose, chi-square test for significance was employed at a significance level (α) of 5%. The following null (Ho) and alternative (HA) hypotheses were formulated:

Graph of particle size distribution according to percentiles.
The calculated chi-square value was found to be 2.181, whereas the critical value of the chi-square was obtained from the standard chi-square table at α = 0.05 and this value was found to be 12.592. On comparing the calculated and critical values of the chi-square, it is found that critical value is much greater than the calculated value indicating that the null hypothesis is not rejected. Thus, it may be concluded that the two attributes are significantly independent for affecting the particle size recovered from the acid leaching process. It is also found that the median size of the particles of all leaching durations, considered in this study, is in the range (38–25 μm) which is suitable for direct powder metallurgy applications. 15
Furthermore, the employed acid concentration and leaching duration imply that through suitable expanse of range of concentration and duration, better recovery of the Fe metal may be possible. The scope of the study can be further extended to explore the effects of other factors in the acid leaching process that can enhance the rate of Fe recovery. It may be suggested based on the present experimental campaign that other factors that can be considered for future studies are: leaching temperature, type of acid, acid concentration, rate of stirring etc. Multi-stage leaching and combination of alternative techniques (such as sintering) may also be considered to further enhance the efficiency of acid leaching with the use of process optimization techniques. Successful complete recovery of Fe from shot blasting media may contribute greatly in reducing raw material costs for powder metallurgy, as well as in the preservation and conservation of the environment through recycling of used material.
Conclusion
This article presented an experimental investigation to reveal the effectiveness of the acid leaching process in removing impurities and recovering iron from used shot blasting media. Based on the results of the present investigation, following conclusions are drawn:
Acid leaching is an efficient and effective process for recovering iron from used shot blasting media as the percentage of recovery from the surface of the shot blasting media is found to be as high as 73.6%.
Acid leaching process does not affect the particle size significantly, and therefore, the particle size is suitable for direct use in powder metallurgy applications.
In the acid leaching process, initially, the percentage of iron recovery increases with increase in the leaching duration, the recovery reaches to a peak, and subsequently it drops with further increase in the duration.
Thus, based on the results of the present investigation, it may be finally concluded that the acid leaching process should be used not only to remove impurities but also to recover iron from the surface of the shot blasting media with a particle size appropriate for direct use in powder metallurgy applications. Future work includes more comprehensive investigation including more factors under study.
Footnotes
Acknowledgements
The authors are grateful to the Raytheon Chair for Systems Engineering for funding.
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work is financially supported by Raytheon Chair for Systems Engineering.
