Abstract
The aim of this study is to evaluate the effects of different particle sizes colloidal nanosilica (30, 60, and 140 nm) on the early performance of Portland cement. Flowability, mechanical property, and microstructural characterization tests were conducted to investigate the macroscopic and microscopic properties. The results revealed that the flowability of the paste decreased with the reduction of the particle size of colloidal nanosilica. The main reason was that the sol may form flocculation restricting a part of free water under ion environment, and this phenomenon became more serious as the particle size decreased. The compressive strength with 60-nm sol was higher than that of the other two, and hydration activities under different situations were further proved by hydration heat analysis. The reasons causing this result were mainly because flocculation limited the original chemical activity like pozzolanic and seeding effects of nanomaterials. Through thermogravimetric analysis and X-ray diffraction, it can be concluded that the seeding and pozzolanic effects of nanomaterials dominated the hydration process at different periods. Comparing images from scanning electron microscopy, the microstructure was most compact when incorporated into 60-nm sol, and filling effects also contributed to the performance during hardening.
Introduction
The performance of cement-based material needs to be improved from the micro level, and the introduction of nanomaterials to cement science provides a new idea for the solution of this problem.1–3 Nanomaterials are the superfine materials at the nanometer level and have several particular characteristics which can bring about some special performances when added into concrete. Among these materials, nanosilica as a kind of highly effective material compared with others, mainly has three advantages including the pozzolanic reaction, the seeding effect, both of which escalate the extent of hydration, and the filling effect, which can fill the internal porosity defects; therefore, the introduction into concrete can effectively improve mechanical property and durability after hardening.4–10 Jo et al. 5 found that 6 wt% nanosilica can improve the compressive strength of mortar by 152% and 142% at 7 and 28 days, respectively. Ye et al. 6 reported that the compressive strength of the sample modified by nanosilica was more obvious at early ages. As for the durability, Tobón et al. 8 reported that mortars with 5 and 10 wt% nanosilica decreased expansion by 90% and 95%, respectively, after 2 years of immersion in sulfate environment.
When selecting materials, there are two main forms of nanosilica in its current use, that is, powder and colloidal sol. Due to the surface effect of nanosilica, surface energy of the particles is extremely high in the powder state, leading to reunite into large agglomeration, sometimes even larger than the particle size of cement.11–13 Without effective dispersion, the nanosilica will not only fail to exert its advantages but also form a weakness zone between the agglomeration and the bulk paste.10,12 Colloidal nanosilica (CNS) is another form of nanosilica, where the nanoparticles uniformly disperse and keep the initial particle size, which can better exert its nanometer effects. Since nanosilica particle in the CNS contains a large amount of water and hydroxyl groups on its surface, it also can be expressed as mSiO2·nH2O. But the sol is in the state of electrostatic stability, and different degrees of flocculation may occur in ionic solution.14,15 Nevertheless, CNS is still a relatively ideal way to use nanosilica when compared with powder state, and the comparison has been verified by Kong et al. 15 In view of the basically same modification mechanism between them, there are many similar properties in the hydration process.16–19
Because of the manufacturing process, nanosilica has different particle sizes, and it is important to select the most suitable particle size for different applications in engineering. Some researchers have studied on this aspect,20–22 Durgun and Atahan 20 used CNS with three sizes of 5, 17, and 35 nm to enhance the cohesiveness of mixture and found that the lower the average particle size, the stronger its effectiveness. Haruehansapong et al. 21 compared mortar with sizes of 12, 20, and 40 nm powder nanosilica, and the mortar containing 40 nm gave higher compressive strength. Sikora et al. 22 also found that finer nanosilica particle had a higher pozzolanic activity when 100 and 200 nm were selected as the particle sizes. Variable conclusions are existed in different particle size ranges and experiment objectives, and there are two main imperfections in their researches. On the one hand, the particle sizes they chose was too close to detect the difference between each other, especially when flocculation occurred, besides such close range was lower meaningful in cement industry. On the other hand, the impacts of sol coagulation caused by ionic solution in the early stage have not been clarified. In this study, characteristics of cement hydration in the presence of CNS at the early stage, including flowability, mechanical property, and microstructure of cement paste were investigated. Moreover, effects of particle sizes on above properties were also explored, and CNS of 30 nm (CNS-30), 60 nm (CNS-60), and 140 nm (CNS-140) with distinct size level were used in order to effectively distinguish the difference between them. All of these results are helpful to provide a comprehensive explanation for the modified improvement of CNS on cementitious materials.
Experiment
Materials
A type II Portland cement with a compressive strength of 52.8 MPa at 28 days, which met the Chinese Standard GB 175-2007 was used in this study. The basic parameters were provided by the manufacturer, and its physiochemical properties were shown in Table 1.
Physiochemical properties of cement.

Three types of CNS (CNS-30, CNS-60, and CNS-140) were prepared by sol–gel method by the hydrolysis process of tetraethoxysilane (TEOS), and their sizes were controlled by adjusting the pH value of solution and reaction time during reaction process. The main steps include: (1) the TEOS, anhydrous ethanol and ammonia water were added into an airtight container according to a certain proportion, (2) mixing for 1.5–3.0 h depending on the demand with magnetic stirrer, (3) the solution was treated for 10 min under ultrasound until the solution was transparent. The basic properties of CNS were listed in Table 2.
Basic properties of CNS.

CNS: colloidal nanosilica.
Mixing and test methods
Cement was mixed with CNS with a mass fraction of 5% of the cement, and water binder ratio was constant at 0.5. The prepared samples were distinguished by particle size of sol, for example, CNS-30 represents the sample with addition of 30 nm CNS and no other chemical additives were used during whole experiment. Cubic cement paste specimens of 40 mm × 40 mm × 40 mm were casted, demoulded after curing for 1 day, and then placed in saturated limewater solution at 20.0 ± 2.0°C until tests began. The compressive test was conducted by universal test machine with a loading rate of 2.4 kN/s, and the results were the average of three experiment values.
Flowability was tested by fluidity and rheometer immediately after mixing. In addition to fluidity, the use of rheology into cement-based materials was more helpful to quantify the flowability.23,24 Rheological test was implemented by a rheometer (Kinexus Lab+, Malvern, England), its procedure included three parts: pre-shear stage (30 s) at a shear rate of 50 s−1 in order to ensure all samples have a same intial state, rest stage for 30 s, and test stage containing a upward ramp of shear rate from 0 to 100 s−1 within 1 min and an analogous downward ramp from 100 s−1 to stop. Finally, the data in descending part were analyzed, since it conformed steady state and showed a more regular behavior. 25
To analyze the whole hydration process, a microcalorimeter (TAM Air, TA Instruments, USA) was used to monitor the impacts of CNS on cement hydration. All the samples were mixed for 2 min, and then injected into an ampoule which was then placed into the isothermal calorimeter. The experiment was set to a reaction temperature of 20°C and lasted 3 days. Besides, to compare the internal phase composition in the hardened paste at early stage, thermogravimetric analysis (TGA; TGA 2, METTLER TOLEDO, Switzerland) and X-ray diffraction (XRD; D8 ADVANCE, Bruker, Germany) were employed on hydration products at 1 and 3 days. The hydration reaction was terminated by alcohol, and the specimens were dried in a vacuum drying chamber until constant weight before test started. During TGA measurement, samples ground into powder were heated in argon atmosphere from 30°C to 900°C at a heating rate of 10°C/min. The XRD measurement was performed in the scanning range from 5° to 60°, and scanning speed of 2°/min. A small piece sample was chosen from the hardened paste, and scanning electron microscopy (SEM; EVO-LS15, Zeiss, Germany) was implemented to compare the real microstructure of samples.
Results and discussions
Flowability of the paste
As we know, flowability is drawing more people’s attention, especially with the wide use of high-performance concrete (HPC), which is of great significance not only to the difficulty level in cast progress but also to the mechanical property and durability after hardening.26,27 The flowability was tested by fluidity and rheometer, and the results were shown in Figures 1 and 2.
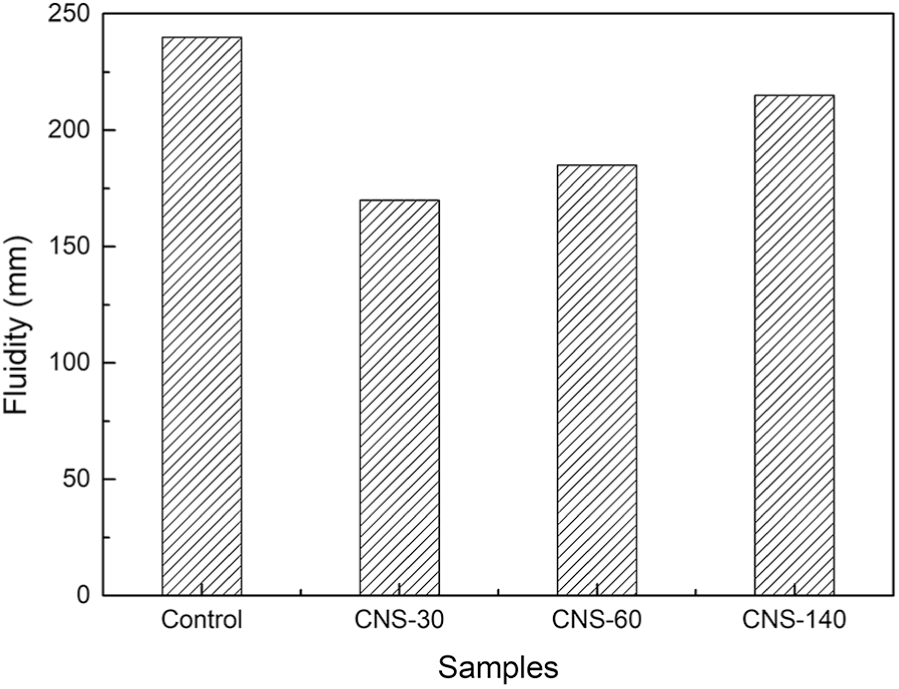
Effects of CNS on fluidity.

Effects of CNS on rheological property.
From Figure 1, the decrease of fluidity is apparent when CNS is added and larger reduction occurs when smaller nanoparticle is used, which is consistent with analysis of rheological curves in Figure 2. The relationship between shear rate
In colloidal state, a large amount of water is absorbed on the particle’s surface and remains stable. However, according to Derjaguin, Landau, Vervey, and Overbeek (DLVO) theory, the stability of sol is affected by the concentration of ions in the solvent.29,30 In the CNS, colloid particles are negatively charged and susceptible to positive ion, such as Ca2+ that accounts for the largest proportion in the hydration process.14,15 With the decrease of particle size, higher specific surface area will be more susceptible to the surrounding environment, forming loose flocculation and coating layer around cement particles that restricts a certain amount of water inside, and the free water content is proportional to the flowability. 31 Therefore, ion in solvent is the main cause affecting the flowability, and this effect is more obvious as particle size decreases.
Mechanical property at early ages
The introduction of nanosilica increases the mechanical strength of concrete based on current research; however, it may have variable performances under different particle sizes. Mechanical properties were measured at different early ages and the results were shown in Figure 3.

Compressive strength of hardened cement paste.
It can be seen from the development of compressive strength that the difference in compressive strength at day 1, 3, and 7 is not obvious, but the sample mixed with CNS-60 is slightly higher than other samples. At 14 and 28 days, the differences between strength become more pronounced. The strength of the sample mixed with CNS is higher than that of the reference, and the order of strength from more to less are the group of CNS-60, CNS-30, and CNS-140. The modification mechanism mainly includes three parts: pozzolanic, seeding, and filling effects. Nanosilica can react with calcium hydroxide (CH), refining crystal distribution in the interfacial transition zone (ITZ) and producing extra hydrated calcium silicate (C-S-H) gel, act as nucleation sites to benefit the hydration progress and fill the internal porosity defects as a filler.10,17,32,33
Indeed, the smaller particle size of nanomaterial has a higher chemical activity when they are evenly distributed in cementitious materials; however, at the same time, it is more susceptible to other factors, such as the environment in which the chemical reaction occurs. During cement hydration, flocculation will reduce inherent chemical property like pozzolanic and seeding effects of nanosilica due to the reduction of total specific surface area.12,15 This condition is most obvious when CNS-30 is added due to its largest specific surface area, resulting in a lower compressive strength, and moreover the chemical activity of CNS-140 may be relatively low compared with the other two. In contrast, the CNS-60 has the highest chemical activity and most effective improvement of mechanical property.
Microstructural investigations
Hydration heat measurement
In order to further explain the influence of CNS on hydration from the micro-level, various experimental methods were used to analyze its hydration and hardened properties. By measuring the hydration heat of cement within a certain period of time, the impacts of CNS on hydration can be obtained, and the experimental results were shown in Figure 4, where the rate of heat evolution and total heat were calculated on the basis of a unit weigh cement.

Impacts of CNS on cement hydration: (a) differential curves and (b) integral curves.
From the curves, the addition of CNS does not remove or add additional peaks but only changes the intensity or time of these peaks. Two exothermic peaks can be observed in the Figure 4(a), which are labeled with capital letter A and B. Peak A may represent the formation of C-S-H from C3S and C3A hydration, while peak B is caused by the transformation of ettringite (AFt) to AFm phase after the gypsum has been consumed.34,35 Combined with Figure 4(b), the introduction of CNS increases the hydration degree at different levels, and it is obvious that the group of CNS-60 has the most effective improvement on hydration reaction, followed by the group of CNS-30 and CNS-140. Higher hydration degree means more hydration product, leading to a more compact internal structure. The explanation in section “Mechanical property at early ages” can be used to illuminate the cause of this situation, flocculation of CNS will reduce nanoparticle chemical activity, and the analysis results of hydration reaction are same as the development of compressive strength.
TGA
TGA was used to further determine the microstructure composition changes, the TGA curves of samples with different particle sizes CNS at 1 and 3 days curing time were presented in Figure 5, and analysis results were listed in Table 3.

TGA curves of samples with different CNS: (a) 1 day (b) 3 days.
Mass loss of samples at 1 and 3 days.

CH: calcium hydroxide; CNS: colloidal nanosilica.
The mass loss within 30°C–900°C is due to the combined thermal effects and can be divided into two parts. The first part is the dehydration or decompositon of C-S-H gel, AFt, or AFm at 135°C–150°C. It is difficult to quantitatively analyze what is the content of a substance because these hydration products have similar temperature to change its orginal state. 36 The second part is mainly the decomposition of CH at 400°C–500°C and CaCO3 carbonized by CH at around 650°C–750°C, and the total content of CH can be used to analyze the role of CNS in cement hydration.
As shown in Table 3, total (CH) or corresponding percentage calculated by chemical reaction reduces with the introduction of CNS, which is consistent with other studies.6,9,10 However, the formation of CH under different particle sizes and test periods is distinct. For the samples at 1 day, the sum of CH reduces with the increases of the particle size, and the mass loss reaches to the relative maximum when the particle size is 140 nm. But at 3 days, the above trend is just the opposite, where more mass loss appears with the decrease of particle size. The main reason for this phenomenon is the relative magnitude between pozzolanic activity that consumes CH and seeding effect that promotes the generation of CH. During 1 day curing, seeding effect is more obvious than pozzolanic activity, so the amount of CH generated by seeding effect is greater than that consumed by pozzolanic activity, and this phenomenon increases as the particle size decreases. At 3 days, increasing intensity of pozzolanic activity leads to a significant reduction in the CH content, which is more pronounced as the particle size decreases.
XRD
The XRD diagrams of the samples with different particle sizes CNS cured at 1 and 3 days were presented in Figure 6. The main peaks including unhydrated content (C3S and C2S) and hydration products (AFt, CH, and CaCO3) were marked in the diagrams.

XRD diagrams of samples with different CNS: (a) 1 day and (b) 3 days.
In the early stage, nanosilica has a higher modification activity.9,37 From Figure 6, by introducing CNS of different particle sizes, the peaks of C3S, C2S, and CH decrease at 1 and 3 days, and the changes of AFt and CaCO3 are not obvious. The reduction of C3S and C2S indicates the acceleration of cement hydration due to CNS, and the decreasing CH content is caused by the pozzolanic activity, which leads to an increase in the amount of C-S-H gel.6,10,16 However, due to the different chemical reactivity of nanomaterials, the specific amount of the hydration products vary. Among the diffraction peaks of substances, the CH has the most obvious change, and its trend corresponds to the result of TGA, which further supports the explanation in section “TGA.” The seeding effect and pozzolanic activity of nanosilica dominate the hydration process in turn, but both improve the hydration degree of cement. It is necessary to note that as a negative substance, the variation of the CH in TGA and XRD is just to explain the role of CNS at different hydration times, and certainly cannot represent the development of its mechanical strength.
SEM
Figure 7 shows the morphological characteristics of the samples’ microstructure with and without CNS obtained by SEM at 3 days. The main hydration products AFt, CH, and C-S-H gel can be clearly distinguished in these images.

SEM images of hydration products at 3 days: (a) control, (b) CNS-30, (c) CNS-60, and (d) CNS-140.
Compared with the control group, the amount of CH and AFt that have negative influences on the strength development decreases when CNS is used and C-S-H gel significantly increases, which makes the connection between internal materials closer and microstructure more compact, just like the interpretations in literatures.6,10,16,17 According to Figure 7, in the group of CNS-60, the number of hydration products is the largest and the internal structure is the closest, followed by CNS-30 and CNS-140, which correspond to the development of mechanical strength. It should be noticed that the improvement of microstructure in hardened paste is not only due to the chemical properties of CNS but also influenced by its filling effect that is determined by the size of flocculation structure of the CNS. As mentioned above, in the colloidal state, the smaller the particle size leads to an easier form of flocculation under ion environment, and only the suitable size of flocculation structure can effectively fill the internal porosity defects in the hardened paste. Under the combined effects of the above factors, applying CNS-60 is the most ideal way to improve the performance of concrete.
Conclusion
In this work, influences of three particle sizes of CNS on the properties of paste at different stages were studied and the following conclusions can be drawn:
The flowability of the fresh paste reduces as the particle size decreases, and when fitting with Bingham model, the effect of particle size on yield stress is more pronounced than that on plastic viscosity. This phenomenon is due to varying degrees of CNS flocculation which restricts a part of free water under ion environment and becomes more serious with the decrease of particle size.
Through the comparison of compressive strength, the addition of CNS-60 has more contribution to the development of compressive strength at any test period, followed by the group of CNS-30 and CNS-140. In addition, the results of compressive strength are consistent with the order of hydration rate.
Based on the curves of cement hydration, the sample of CNS-60 exhibits higher hydration activity than the other two, leading to the different hydration degrees at the same time. The flocculation, which limits the original chemical activity like pozzolanic and seeding effects of CNS, is the main cause.
CH values are changing with particle sizes and hydration times through TGA, which can reveal that CNS has variable performances at different stages, and the phenomenon is further confirmed by XRD test results. The SEM investigations show that the sample with CNS-60 has more reasonable hydration products and a higher degree of compactness than the other two, resulting in higher compressive strength.
Footnotes
Handling Editor: Grzegorz Golewski
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (grant nos.: 51778269 and 51672106), and by the 111 Project of International Corporation on Advanced Cement-based Materials (grant no.: D17001).
