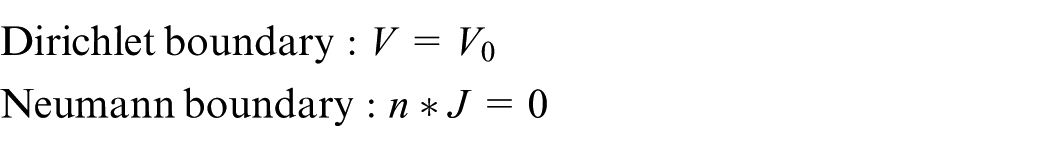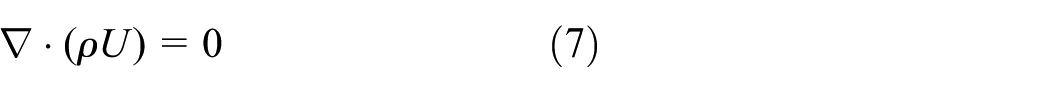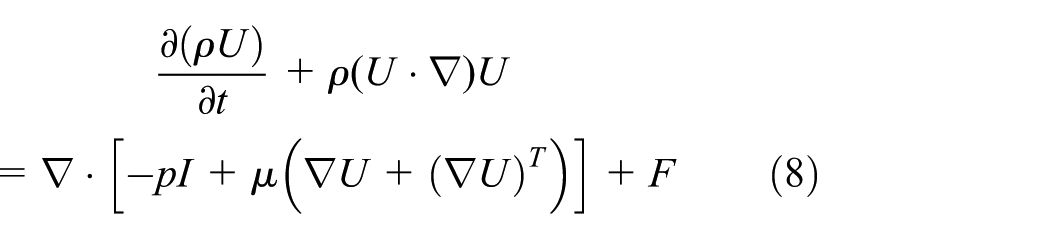Abstract
Renal sympathetic denervation is regarded as a new effective method in the treatment of resistant hypertension, but in the therapeutic temperature and complications, it has some controversy. The aim of this study was to calculate the multi-physical field for radiofrequency ablation and verify its feasibility and effectiveness with ex vivo experiments. The temperature distribution and flow field in the renal artery were analyzed by finite element method and verified by ex vivo experiments with an equivalent model. After ablating in simulation, the center temperature of the renal artery reached the therapeutic target which is 43°C (about 48°C). When increasing the time or changing the velocity, variations in the temperature ranged within 2°C. In the ex vivo experiments, the highest temperature under different conditions increased from 4.12°C to 9.61°C. Within the range of the normal velocity, that is, 8 W power output, the therapeutic purpose (ΔT ≥ 6°C) was achieved and effect on the blood and peripheral tissues was less. The temperature field has influences on the choices of treatment parameters with different degrees. The energy output is the most influential factor, while ablation time and flow velocity have less effects on the temperature field. This study preliminarily verified the safety and effectiveness of the renal sympathetic denervation.
Keywords
Introduction
Resistant hypertension (RH) refers to use three or more than three types of antihypertensive drugs, including a diuretic, but blood pressure still cannot be reduced to the target lower than 140/90 mmHg (1 mmHg = 0.133 kPa). 1 RH, vasodilation and vasoconstriction in the body factor secretion disorder, mental stimulation, and the change in external environment factors may cause the central neural system to influence the long-term control of the renal sympathetic nerve in patients. Studies have shown that the sympathetic nervous system can not only adjust blood pressure but also play an important role in long-term hypertension regulation. 2 Kidney plays a key role in the pathogenesis of hypertension by adjusting the cortex manifold sodium water absorption and keeping the balance of water and salt.3,4 The over-activation of the sympathetic nervous system is one of the main factors of RH.5,6 Esler 7 confirmed that the renal sympathetic nerves release neurotransmitters norepinephrine. There are two categories of renal sympathetic nerve: the afferent nerve and the efferent nerve. These two types of nerves are located in the outer membrane of renal artery. Renal sympathetic over-activation increasing catecholamine material release is a key reason of the formation of high blood pressure. Sympathetic nerve excitement of hypertensive patients causes the excessive secretion of norepinephrine. Relevant animal experiments showed that removal of the renal sympathetic nerve could decrease the blood pressure.6,8
Renal sympathetic nerve resection was first applied to the associated RH and then for the treatment of hemodialysis patients with end-stage renal disease. Blood pressure has been decreased significantly after the resection of bilateral renal sympathetic nerve and can be controlled more easily than before.9–11 According to recent studies, the increased impulse frequency of renal sympathetic nerve and the activation of efferent nerve lead to the secretion of norepinephrine, the increase in blood pressure, and the enlargement of left ventricular. Hence, the incidence of arrhythmia and the risk of cardiovascular death increase. 12 Renal sympathetic nerve resection can avoid a series of pathological cycles which benefits more than drugs. However, the complete removal of sympathetic nerve is complicated, which highly requires qualified skills of surgery doctors. It is often along with cutting of the internal organs. However, the surgery always causes trauma and needs longer recovery time, which seriously influences the patients’ quality of life and causes more complications, such as palpitations and intestinal disorders.
Radiofrequency ablation (RFA) is an intervention treatment through thermal energy to eliminate the pathological tissues. The molecules under the action of high-frequency electromagnetic field can generate heat by friction so as to rise temperature to kill cells. This technology has become a preferred method of tachyarrhythmia with less trauma and higher operative successful rate.13–15 Catheter-based renal sympathetic denervation (RSD) for RH, also called renal denervation (RDN), is a new method applied in the treatment of RH in recent years. It works through the catheter with insertion of renal artery, releasing radiofrequency energy and damaging renal sympathetic nerve fibers on the outer membrane, blocking its afferent and efferent signals to reduce the renal sympathetic nerve activity. Inserting catheter in renal artery releases radiofrequency energy to damage renal sympathetic nerve fibers on the outer membrane and blocks its afferent and efferent signals so as to reduce the renal sympathetic nerve activity. By avoiding excessive secretion of norepinephrine, RH is able to be cured with less adverse reactions and patients will rely less on high blood pressure drugs. 16
Krum et al. 10 combined renal arteriography with cardiac RFA to treat the RH by catheter-based RSD. The results of Simplicity HTN-1 were published in 2009, which aroused widespread attention around the world. RF electrodes were inserted into each renal artery by a guiding catheter. The ablations were performed in both renal arteries under a standardized treatment protocol and algorithm. Four to six points ablation was able to be finished within 2 min. Catheter tip impedance and temperature were monitored constantly. Radiofrequency energy was delivered with 8 W as peak power from the first distal main renal artery bifurcation to the ostium proximally, and electrodes were placed longitudinally and rotationally under fluoroscopic guidance. Among all the 45 patients who had taken the surgery, only one case reported renal artery dissection before the release of radiofrequency. No perforation and thromboembolic events were reported in the 1-year follow-up observation, and the blood pressure of patients enrolled has decreased. The results showed that the catheter-based sympathetic denervation maybe a safe and effective method for non-drug treatment, with the gradual decrease in blood pressure levels and no serious adverse events.
In 2010, a larger sample size of multichannel, randomized, and controlled clinical trials were conducted (Symplicity HTN-2) in 24 hypertension treatment centers of Europe, Australia, and New Zealand. Symplicity HTN-1 and Symplicity HTN-2 have shown efficacy and safety of the RSD treatment of RH. However, there was a prospective, single-blind, randomized, sham-controlled study on Symplicity HTN-3 which did not achieve a positive result in 2014. 17 In 2015, the world’s first case of cryoablation for RDN was conducted by professor Ge et al. However, there were still some problems which were unsolved both domestic and abroad for this surgical method. For example, whether RF energy can successfully penetrate the entire renal artery wall; the extent of contact to the renal artery wall; ablation depth; and location and ablation energy output level are also needed further explanation, since different ablation sites also cause different ablation effect 18 and the progress of RF equipment (consider percutaneous transluminal renal angioplasty and multi-electrode technology). The main purpose of this study was to verify the feasibility and effectiveness of RSD treatment through methodological perspective by the calculation of multi-field coupling, as well as to investigate the influence of temperature field on voltage, power, ablation time, and velocity.
This study adopted the finite element method to calculate the multi-physical field, including temperature field, flow field, and electromagnetic field for RFA of RSD, and applied ex vivo experiment to verify its feasibility and effectiveness. This study could provide a theoretical reference for clinical operation.
Methodology
Finite element simulation
We used a single electrode with temperature control model to produce an alternating current at 460 kHz into renal tissue. The treated part of kidney model had a length approximately of 2 cm, a maximum width of 2 cm, and a height of 4 cm. The kidney model was generated as parallelepiped as shown in Figure 1. The mesh arises from the finite element method employed for solving the system equations. Blood vessels (the renal artery) were modeled as a vertical pipe of 0.5 cm diameter with 0.1 cm thick blood vessel walls being positioned in the kidney.19–21 Renal tissue was considered to be an isotropic material with inhomogeneous characteristics regarding its dielectric and thermal parameters. For simplicity, most parameters describing the model in Figure 1 were assumed constant. Although they could be slowly time variant or even depend on specific tissue effects on a molecular scale. Regarding these dependencies, we implemented a mathematical formulation, which Pop et al. 22 obtained from the dielectric parameters of porcine renal cortex with a radiofrequency of 460 kHz. 23

Model of kidney with blood vessel.
All radiofrequency heating models were based on thermoelectric coupling problem in the time domain analysis. Temperature distribution in biological tissues can be obtained by solving biological heat equation, an improvement of Pennes bioheat transfer equation 24 as in the following
where ρ is the tissue density (kg/m3), c is the tissue-specific heat capacity (J/(kg °C)), k is the thermal conductivity of tissue (J/(m s °C)), T is the temperature of tissue (°C), t is the time (s), cb is the specific heat capacity of blood (J/(kg °C)), ωb is the blood perfusion rate (kg/(m3 s)), Tb is the blood temperature in the heating zone (°C), Qm is the metabolic heat generation (J/(m3 s)), and Qr is the electromagnetic heat source.
For biological tissues, ωb generally has small difference. However, in some rare cases, the value of the ωb varies and increases with heating time because of the accumulation of heat inside the vessels or blood coagulation. The accumulation of heat in the blood vessels is due to the constant expansion of blood vessels 25 and the accumulation of heat capacity of blood, or a more serious situation, that is, when the heat is enough, the tissue cells will reach necrosis which can lead to blood coagulation. 26
Qp is the heat perfusion item. As the electrode was closely attached to the artery wall in RSD, the main ablation energy was almost concentrated in the renal arteries which adhered by sympathetic nerve. Blood flow can take away most of the energy, so this item was basically negligible. However, the cooling effect cannot be ignored. We considered this part in Qr.
In theory, the Qm exists, but in this study, it was ignored. Since RFA of RSD, the ablation area was very small, typically in millimeters or short of centimeter level. Therefore, the metabolic heat generated in this area was totally negligible relative to the heat generated by RFA. 27
Under the frequency of RFA, the displacement current can be neglected. Therefore, the tissue cling to electrode can be considered as pure resistance. Based on this, one type of quasi-static method was used to solve this kind of electrical problem.28,29 Therefore, the energy of external heat quantity Qr can be expressed as follows
Due to the lack of RF power source in the tissue, we can find out the distribution of electric potential V by solving the Laplace equation (4)
The boundaries of electrode tip and insulating shaft are regarded as Dirichlet boundary and Neumann boundary, respectively. Constant voltage is applied on the active part of the electrode, and the ground is acted as the other boundary
where V is the voltage (V), n is the unit normal vector, and J is the current density (A/m2).
In the process of solving, the potential was assigned and then calculation was started. This is the parameterized scanning method. The potential V is set to 10 V/12 V. Electrode surface boundary should be covered. All peripheral tissue made the grounding. The potential value was 0.
Then, we substitute the distribution of electric potential V into equations (5) and (6), and electric field intensity and current density can be obtained
where V is the voltage (V),
Here, we directly considered the blood motion in the model instead of using convective boundary conditions. We considered a laminar fluid flow, plus the high- and low-flow velocities as mentioned above.
30
In this case, there is a bidirectional thermal-flow coupled problem, and the incompressible Navier–Stokes equation from fluid dynamics works together with the heat transfer equation. Therefore, the so-called advection term
31
was included on the right of equation (1), which represents the heat loss due to blood flow. Where
where U is the blood velocity vector (m/s), ρ is the tissue density (kg/m3), and t is time (s)
where P is the pressure (Pa), I is the normal stress (N), μ is the dynamic viscosity (Pa s), and F is the normal volume of forces (N/m3)
where K is the heat conductivity coefficient (W/m °C), c is the blood specific heat capacity (J/kg °C), and ST is the volume heat source (J).
A no-slip condition (nonpermeable surface; the fluid at the wall was not moving) was applied on the upper surfaces of the fluid volume, at the symmetry plane, and at the tissue–blood and electrode–blood interfaces, respectively. An inlet boundary condition of velocity type was applied to the laminar flow on the top surface of the fluid, while an outlet boundary condition of zero pressure/no viscous stress was fixed on the bottom surface of the fluid volume.
Under the high-frequency electric field, the electrolyte and ion in biological tissues usually moves at high speed, thus causes joule heat due to the high-speed friction. The tissue temperature rose immediately near the electrode. Due to the heat conduction effect, damage scope expanded. Blood flow was associated with surgery, and the simulation took into account the blood perfusion.32–34 Under the influence of electric field, thermal process includes radiofrequency current heating, thermal conductivity, and biological metabolism of tissues.
For the real tissues in certain environment, the corresponding material properties were set in the model, including the relative dielectric constant and conductivity, the density, specific heat, thermal conductivity, and the dynamic viscosity. The thermal initial conditions were set to 37°C for the whole area. On the outer boundaries, the temperature was also set to 37°C and the potential was set to 0 V. The potential of the electrode was set to the values which have been determined by voltage metering during experiments. Table 1 shows an overview for different material parameters.
All parameters are measured under 37°C.
As temperature distribution usually changes with time, the solver settings were completely transient solution. In addition, in order to facilitate the analysis of post-processing for different time, the solve time step was set to 0.1 s and solving time was set for 60 s. We can get the temperature field in different time and the change trend in the whole process. Relative tolerance is set as 0.1, the initial step length is set as 0.01, the backward difference method was used, and solver with free step method was used. Maximum backward differentiation formula (BDF) had an order of 5 and the minimum BDF had an order of 1. Because the model involved in problems with the distribution of temperature field, it is essentially a transient problem. For transient solver, we adopt PARDISO solving method and pre-order algorithm for nested subdivision method. For solving process termination techniques, we used the tolerance method with maximum number of iterations of 10 and tolerance factor of 1. The transient solution of iterations was conducted with incomplete LU decomposition. Relationships between the temperature and time were analyzed.
Anatomy shows that renal artery is separated by the aorta located in the medial side of the spine. Most of the renal arteries have no contact with other tissues. Thus, the model simulates the blood vessels of smooth muscle tissues. Due to the maximum diameter of 5 mm of the renal artery and the treatment requires catheter which has influence on the blood flow, the electrode diameter of 0.5 mm was used in this model. The temperature distribution under different voltage value was simulated. Studies have shown that tissue degeneration occurs when the maximum temperature of vascular and nerve tissue exceeds 43°C–45°C. 40 Therefore, in order to ensure that the ablation zone temperature is above 43°C, we should seek a more feasible treatment plan without affecting the blood flow of the blood vessel.
Ex vivo experiment
We adopted FYJS85-IV RFA experimental instrument, including a single radiofrequency electrode, Agilent34972A Data collector, ZJ-LCD-M digital display flow meter, and thermocouple.
Considering the thermal parameters of the real human body, the experiment used a silicone pipe with a diameter of 1.5 cm to ensure easy operations of RFA; 50% concentration of glycerol solution was used as the blood. The peripheral tissues were filled with phantom41,42 to simulate renal parenchyma, which has a certain visibility and advantages of good thermal conductivity. The main thermal property parameters were shown in Table 2. Reynolds number was calculated by equation (10) for human renal artery, and the equivalent velocity was calculated by changing the velocity range of the fluid in the pipe through the circulating pump. According to the position of RF electrode, the corresponding measurement points of temperature are arranged
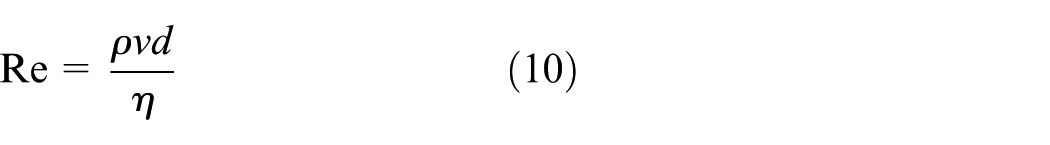
Material parameters’ comparison.

where Re is the Reynolds number, ρ is the liquid density (g/cm3), v is the flow velocity (cm/s), d is the pipe diameter (cm), and η is the coefficient of viscosity (Pa s).
Physiological parameters of renal artery velocity range for normal people needed in the experiment were 0.18–0.32 m/s. The electronic flow meter and control valves were installed on the pipe wall before inlet with glycerol solution to measure inlet velocity and show real-time display through the cross section of flow. Real-time velocity was obtained by equation (10) of Reynolds-equivalent principle to ensure that the experimental operation of Reynolds calculated values was in the range of human renal artery. The key problems were discussed on the thermal-equivalent model. We can control the velocity through the flow meter ball valve within 1.9–3.36 L/min. The positive pole was linked together with radiofrequency electrode, and the negative plate was pasted on the phantom. After connecting the experimental apparatus, we set the parameters of ablation power (8 and 5 W) and time (8 and 5 min). Specific experimental conditions and comparisons with the simulation are shown in Table 3.
Comparisons of the experimental conditions.

The electrode was inserted into the pipe from the liquid inlet to ensure the tip was against the pipe wall, with six thermocouples (Figure 2(a)). The first and sixth thermocouples measured the temperature change in the ablation site and the opposite side of the electrode location, respectively. The remaining four thermocouples (2–5) were inserted into the liquid to measure the temperature change of water flow, with 1 cm interval of thermocouple 1.

Ex vivo experimental model.
Temperature data were collected with temperature variation and the influence of the liquid flow was analyzed. Because the material property of the experiment is different with real tissues, there were still some restrictions and deficiencies.
Results
Simulation calculation
First, we obtained the temperature, flow velocity, and pressure data through finite element simulation. The temperature more directly reflects the effect of ablation. Figure 3 shows the temperature field distribution after ablation. It can be seen that the temperature at the center reached 48°C, exceeding the target value of 43°C. It has achieved the purpose of the renal sympathetic nerve necrosis on the outer membrane of vessels, and the change in the whole temperature field had little influence on the blood temperature.

Temperature field distribution.
Figure 4 shows the ablation area of above 43°C around the blood vessel walls. The maximum thermal diffusion area was above the electrode, reaching the lateral wall of the vessel at 2 mm with less tissue damage.

Effective isosurface.
The temperature variation curves of three measurement points were shown in Figure 6, for electrode location on the outer membrane (a) and for the opposite position of electrode and the center temperature of blood flow which was in horizontal position with electrode by 1 cm distance (b). Figure 5(a) shows that the RF energy can make tissues quickly reach the target temperature (about 48°C), but the highest temperature is below 100°C without producing carbonization. As ablation time increases, the accumulation of heat dose decreased gradually. Figure 5(b) shows that the influence on the contralateral tissue and the blood center was very slight, and the temperature increased was less than 0.5°C. With arterial blood flow during RFA, blood flow can take away part of the heat to make the outer membrane temperature reach the target value, as well with minor damage to the surrounding tissues and blood. The heat accumulation increased with ablation time, and at only a certain extent, the increasing trend tends to slow.

The temperature rise curve of different sites: (a) temperature of electrode site and (b) temperature of contralateral tissue and the blood center.
From a physical perspective, the radiofrequency energy rises the temperature of the surrounding tissue, and it depends on electromagnetic radiation and heat transfer of blood vessel walls. For blood flow, the temperature barely increased, and there is no use to adjust the blood flow to take away or increase the heat transfer of the electrode. When the temperature is too high (more than 50°C), 40 vascular endothelial cell loss or blood coagulation could occur. Because of the scouring, blood flow reached certain cooling effect, and temperature field and flow field had a process of mutual influence.
Figure 6 shows the average changes in flow velocity after ablation; as can be observed from Figure 6, the top surface was set as entrance with a velocity of 0.5 m/s in the simulation, and the underside for the export of flow boundary. It can be seen the temperature field has less effects on flow field. There was a slow flow velocity of small amplitude in the downstream part. According to the clinical situation, some patients may have mild thrombosis after operation. This is due to the renal artery endothelial cell damage caused by thermal effect where blood cells are stagnant. In that sense, even if the ideal model of the simulation result is good, the technology in the application for clinical situation is necessary to make strict rules on patient selection, such as secondary hypertension, type 1 diabetes, and renal vascular anomalies (including severe renal artery stenosis, renal angioplasty, or renal artery carotid stenting). In addition, on the basis of the simulation calculation, the subsequent study should explore the optimum composition method on ablation time, power, and the position of the RF electrode in order to obtain the ablation effect with little damage.

The situation of flow field.
When the boundary conditions were set, the influence on temperature field was discussed by changing the energy output, ablation time, and the blood flow velocity. The curves of the Figures 7 and 8 were the electrode center temperature curves. The electrode center coordinates were (10, 6.5, 21). Figure 7 shows that when increasing the output energy of RF electrode, the highest temperature on electrode rise significantly. While the current field of potential V0 increased from 10 to 12 V, the central highest temperature was increased from 48.11° C to 52.99°C, and the effective temperature range increased at the same time. The overall trend of temperature increase is consistent, and the ablation temperature tends to be slow in the later stage. When the ablation time increased, the electrode temperature has little effect (Figure 8). The temperature may rise rapidly in a short time, and then, it gradually increases in a certain range. Therefore, the time is not a decisive factor for ablation effect. Changing the average velocity of the entrance, temperature rise data were shown in Figure 9. We can see that when the flow velocity increased from 0.4 to 0.7 m/s, it has little influence on the overall temperature trends.

The highest temperature in different potential.
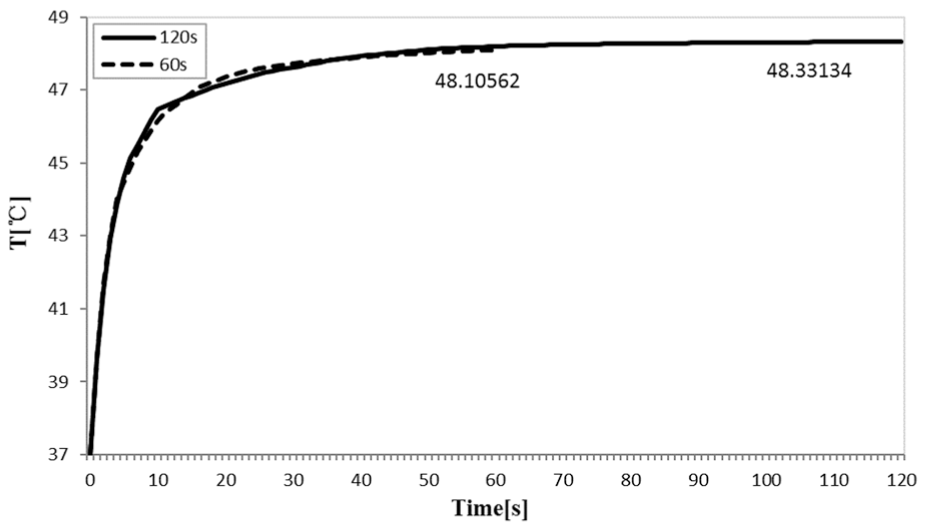
The highest temperature in different time.

The highest temperature in different flow velocity.
The calculation results showed that the biggest factor on the temperature field is the RF energy. The maximum temperature can rise above 48°C, compared with the normal body temperature of 37°C before ablation. The change in temperature ΔT can reach at least 11°C.
Ex vivo experimental results
As shown in Figure 2, temperature data were obtained from ex vivo experiments. Because there were certain differences in initial temperature of peripheral tissue, liquid, and each thermocouple, the mean values of temperature were collected in Table 4. Normal human body temperature is 37°C, while the target temperature of completely renal sympathetic nervous necrosis is 43°C. The ΔT should be reached for 6°C. It can be seen that temperature of thermocouple 1 under different conditions increased from 4.12°C to 9.61°C. The experimental results were not fully achieved; the expected values only in the case of high power can reach the target. One of the biggest controlling factors is the magnitude of output energy. The RFA instrument used in experiments has certain differences in clinical situation. The adjustable range of actual power is very small. Figure 10 shows the trend of temperature rise in six thermocouples of one group. We can see that thermocouple 1 rose significantly and can achieve the goal of ΔT≥ 6°C or higher. Thermocouple 6 rose slowly and the final temperature gap was less than 2°C. Thermocouples 2–5 with the highest temperature rise was roughly less than 0.5°C. These measuring points located at the liquid in the pipe. Although RF electrode released most of the heat, the liquid washed away largely part of it. Therefore, the ΔT here was very small. It also demonstrated that the temperature field change produced by RFA had a relatively small impact on flow field.
Ex vivo experiment data.

Figure 10 shows that under the same conditions, thermocouples 2–5 are almost in the same growth trend.

The trend of temperature rise of six thermocouples.
Similarly, the ex vivo experiment system also set up three corresponding variable parameters to compare with the simulation. When the flow velocity was 2.9 L/min, for example, Figures 11 and 12 show increase in ablation power and time and the change in △T on thermocouple 1. It can be seen that the increase in ablation power can largely influence the temperature field, whereas the heating time did not have any impact. The results were similar to the trend of simulation, but certain deviation exists in the actual values of △T. This may be due to the shortcomings of experiment device itself, and there were some differences in thermal property parameters of the experimental materials.
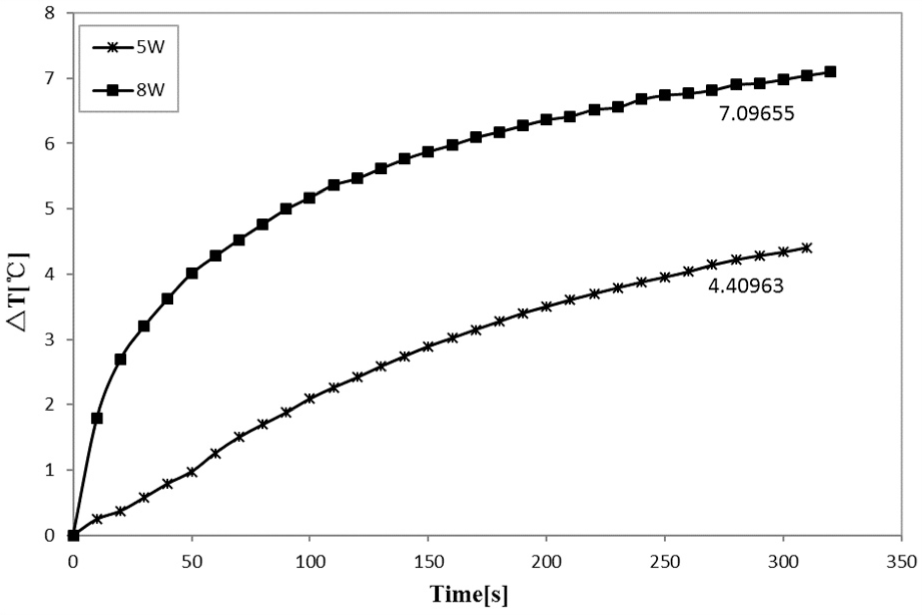
The △T of power increased.

The △T of ablation time increased.
Under the condition of ablation power of 5 W and time for 5 min, we compared the △T in thermocouple 1 when flow velocity changes. Figure 13 shows that the faster the flow velocity, the smaller the △T. When the minimum flow velocity was 1.9 L/min, ablation time for 5 min to achieve maximum average △T was 5.3239°C, and the maximum velocity of 3.36 L/min can achieve the largest average △T of 4.1222°C. With the increase in flow velocity, the average △T decreased by 1.2017°C. The faster the flow velocity, the more to take away the heat. The flow field has certain cooling effect on temperature field. Ex vivo experimental results showed that the influence of velocity on the temperature field with the results of the simulation is significantly different. The simulation of the model was based on the real geometry parameters of the human body. As the arterial space is limited and the blood flow has small variation, the heat change is not obvious. For the experiment device, the same thermal-equivalent principle based on the Reynolds number, we increased the pipe diameter and length and reduced the velocity of liquid. In this way, the space is large enough to observe the change in the temperature field. In other words, the velocity still has a certain influence on RFA, but the influence rate is still determined by the geometric and physical parameters of the specific circumstances. In the simulation, the highest temperature (corresponding with the thermocouple 1) can rise quickly. But in the experiment, it rise slowly. While ex vivo experiments were always being slowly rising, the inconsistent heat accumulation was manifested. This effect should be taken into account in subsequent experiments with the results of the whole process of liquid flow.

The △T when the flow velocity changes.
From the experimental results, power for 8 W can reach the target temperature, while the ablation time was not as long as necessary to renal sympathetic nerve damage in clinic. But the best time should be analyzed by further data. Flow velocity has a certain influence on temperature field and should be considered on the personalized therapy. The subsequent experiments should study the hemodynamic parameters to determine an optimal ablation combination.
Physiological parameters used in the simulation and the actual situation may have some individual differences. There is currently no consideration for the properties varying with temperature. In the future work, we will consider this condition. As the model and the experiment device are idealized, the results may have relative error. However, from the view point of methodology, the feasibility and effectiveness can be verified.
Conclusion
This article studied the multi-physical field of RDN by RFA, based on the finite element simulation and ex vivo experiment. The results showed that the temperature field has some influence on the selection of treatment parameters with different degrees of effect. The energy output was a major factor. With the increase in power, the temperature rise increases largely. While ablation time and flow velocity had less effect on the temperature field. When increasing the time or changing the velocity, temperature variation values were controlled with about 2°C. Within the scope of the normal velocity of 8 W power output, it achieved the therapeutic purposes (△T ≥ 6°C) and had less effect on the blood and peripheral tissue. This study preliminary verified the safety and effectiveness of RSD. Flow velocity difference had a smaller effect on the performance. But if we want to get a better curative effect, the patient’s individual therapy needs to be considered.
Footnotes
Acknowledgements
T.D. was responsible for the design, data collection, and overall investigation. T.D., Q.N., and Z.T. were responsible for computational modeling and data analysis part. T.D. and Y.C. were responsible for the experimental statistical analysis part. All authors (1) have made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; (2) have been involved in drafting the manuscript or revising it critically for important intellectual content; and (3) have given final approval of the version to be published. Each author has participated sufficiently in the work to take public responsibility for appropriate portions of the content. The authors thank professor Haisheng Yang for his guidance on improving the language of this article.
Handling Editor: Assunta Andreozzi
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by National Natural Science Foundation of China (31771021) and Natural Science Foundation of Beijing Municipality (3162006).