Abstract
This article chose the new type alcohol-based fuel as the engine fuel, realized lean burn in a CA4GA5 engine, and got the lean burn limit to 20.6. Taking the gasoline as equivalent fuel, and according to 10%, 20%, and 30% volume proportion of alcohol-based fuel, three new types of alcohol-based gasoline synthetic fuel, M10, M20, and M30, were formed. A comparative study of the original engine emissions used new types of alcohol-based gasoline synthetic fuels with Compressed Natural Gas (CNG) and different blending ratios based on M0 at different stoichiometric air-to-fuel ratios was carried out. This article studied the influence of air-to-fuel ratio on cycle emissions under the lean burn condition and analyzed the characteristics of the new European driving cycle. Experimental results show that compared with the working condition of pure gasoline engine, the emissions of total hydrocarbon and carbon monoxide gradually reduce with the increase of alcohol-based fuel blending proportion, while the NOx content presents the rising trend.
Keywords
Introduction
Along with the rapid development of auto industry and the sharp increase of the vehicle holdings, the energy security and atmospheric environment are facing increasingly severe challenges. 1 From the 1960s, under the condition that the oil crisis put great pressure on European and American countries’ energy securities, alcohol fuel came into sight.2–5 With the development of social economy, the Chinese oil import has gotten a rapid growth,6–11 and the external dependence on oil has also increased. China became a net importer of oil since the early 1990s. As a fuel, methanol is an oxygen-rich content which can be full combustion. With the deepening of methanol research, it was found that up to 50% oxygen content was conducive to the complete combustion of fuels and then reduced the exhaust emission of hydrocarbons of vehicles.12–16 And it had a great substituted ratio and its sources of raw materials were abundant, such as coal, natural gas, coke-oven gas, and coal bed methane. China has abundant coal resources, and the technology of producing methanol by coal is mature with low cost. So the methanol can be one of the most effective alternatives to petroleum resources in China. Since the 21 st century, although the international research and application of methanol fuel have stalled and become a controversial issue in China, there are still some researchers continuing to carry out relevant researches.17–19 Some studies used 3%–5% methanol and gasoline blending. 20 This mixing effectively increased oxygen content in gasoline with a little agent and has been widely used in Europe and China. The result showed that it had no effect on the dynamic properties of gasoline and increased the octane number and reduced the exhaust pollution. Some studies used 85% methanol and 15% of additive. 21 In spite of this, the methanol gasoline research is still not mature enough. The current studies mostly focus on alcohol-based fuel which contains only one kind of alcohol, such as methanol gasoline. This article is designed to study the emission characteristics of new fuels. In order to achieve this goal, the new types of alcohol-based were used as the engine fuel. A comparative study of the original engine emissions used new types of alcohol-based gasoline synthetic fuels with CNG and different blending ratios based on M0 at different stoichiometric air-to-fuel ratios (AFRs) was carried out. And then the influence of the different alcohol-based fuel blending ratios on the new European driving cycle (NEDC) cycle emission performances such as the cold-start emission performance was analyzed.
The new types of alcohol-based gasoline synthetic fuel
The blending technology of alcohol-based fuel
The two-step method was adopted to deal with the methanol. The methanol was blended by incomplete dehydration method and distilled by adding effective additives simultaneously. The molecular structure was combined by catalysis again and the alcohol-based fuel was obtained. In addition, in order to reduce the vapor pressure of alcohol-based fuel and improve its distillation range and calorific value, a certain proportional additives was still needed to be added to the obtained alcohol-based fuel. And we should dehydrate the industrial methanol and remove the impurities at atmospheric pressure.
The blending of new types of alcohol-based gasoline synthetic fuel
The new type of alcohol-based gasoline synthetic fuel was blended by adding a certain proportion of the alcohol-based fuel into the gasoline. The fuel was composed of low-carbon alcohol, multi-carbon alcohol, aromatic hydrocarbon, solvent naphtha, additives, and others. Among them, the low-carbon alcohol was the main constituent of the methyl alcohol, and the solvent naphtha was gasoline. The general volume fraction is shown in Table 1.
New vehicle alcohol-based gasoline fuel composition.

Experimental apparatus and equipment
Experimental engine
The engine (CA4GA5) was a four-cylinder electronic control 16 valves, Variable Cam Timing-Intake (VCT—i) port fuel injection gasoline engine produced by China FAW Group Corporation. Before operation and performance testing, the engine operation specification has been completed and the transformation of lean burn has been completed. The main parameters are shown in Table 2.
Main parameters of CA4GA5 engine.
Based on the differences in the volume fraction of the alcohol-based fuel, the new types of vehicle alcohol-based gasoline synthetic fuel were divided into M10, M20, and M30. In order to analyze the original emission characteristics of M10, M20, and M30, the three-way catalytic converter installed in the original machine was replaced by an empty substrate without precious metal coating.
Vehicle drum test bed
Vehicle drum test bed mainly included environment simulation module, chassis dynamometer system (including driver auxiliary equipment), exhaust analysis system, and main control center:
Environment simulation module. Using the Electronic Throttle Control (ETC) environment simulation module produced by the United States ETC company, temperature ranges from −40°C to 60°C; control accuracy of ±1°C; humidity ranges from 5% relative humidity (RH) to 95% RH; control accuracy of ±5 RH; the maximum wind speed of 200 km/h; the maximum illumination intensity of 1200 W/m2; control accuracy of ±2.5%; thermal simulation system ranges from +20°C to +75°C.
Chassis dynamometer system. The main parameters of the selected dynamometer were as follows: maximum measurement speed of 200 km/h; speed accuracy of 0.08 km/h; maximum range of 5840 N; full-scale accuracy of ±0.1%.
Experimental vehicle
The vehicle used here is a self-brand gasoline/CNG dual-fuel economy sedan. The main parameters are shown in Table 3.
Parameters of testing vehicle.

Analysis of AFRs impact on emission performance
Figures 1–3 demonstrate the relationship between the emission of total hydrocarbon (THC), carbon monoxide (CO), or NOx and AFR with pure gasoline M0 and new types of alcohol-based gasoline synthetic fuels M10, M20, and M30 under the conditions of Pme is 0.3 MPa or 0.7 MPa and the engine speed is 2000 r/min or n = 3200 r/min. According to these figures, when the engine is under working condition with the same load and different speeds, the AFR has a certain effect on the emission performance of these four fuels.

Comparisons of THC emission at different AFRs:(a) on Pme (0.3 MPa) and (b) on Pme (0.7 MPa).
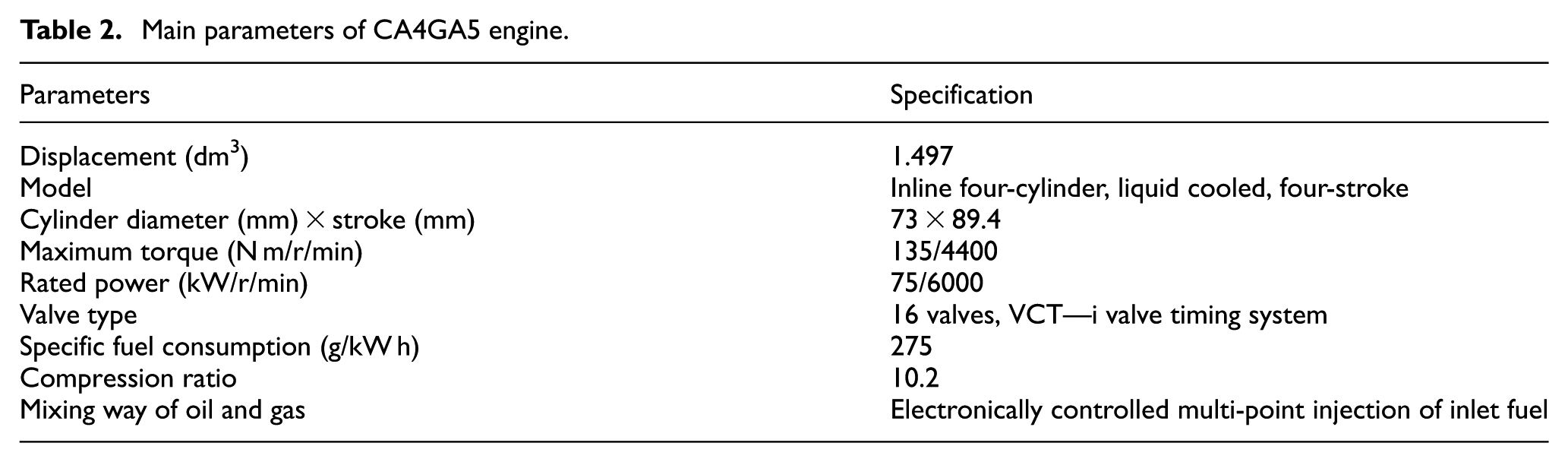
Comparisons of CO emission at different AFRs:(a) on Pme (0.3 MPa) and (b) on Pme (0.7 MPa).

Comparisons of NOx emission at different AFRs: (a) on Pme (0.3 MPa) and (b) on Pme (0.7 MPa).
From Figure 1, as the AFR increases from 14.7 to 20.6, the THC emission reduces first and then increases. In the process of AFR increase from 14.7 to 17.6, the THC emission (Pme = 0.3 MPa, n = 2000 r/min) of M0, M10, M20, or M30 reduces by 52.14%, 57.73%, 51.61%, and 67.03%, respectively. When AFR = 17.6, the emission of THC reaches the minimum. And the THC emission (Pme = 0.3 MPa, n = 2000 r/min) of M0, M10, M20, or M30 is 2357, 1942, 1845, and 1143 ppm, respectively. During these processes, the increase tendency of THC emissions is obvious. When AFR = 20.6, the emission of THC reaches the maximum. And the THC emission (Pme = 0.3 MPa, n = 2000 r/min) of M0, M10, M20, or M30 reaches 21,750, 16,305, 14,246, and 11,861 ppm, respectively, correspondingly increasing by 822.34%, 739.22%, 672.74%, and 937.10%, compared with those in the same working condition with AFR = 17.6. The trend of increase is greater than that of decrease. This is mainly because that with the increase of AFR, the in-cylinder mixture is more dilute. And the fuel is burnt more fully, which is in favor of the reduction of THC emission. When AFR = 17.6, the combustion of in-cylinder mixture is the most abundant and THC emission reduces to the minimum. With the further increase of AFR, the combustible mixture is too thin and results in slowing the burn rate, aggravating surface quenching phenomena, thickening the chilled layer, increasing incomplete combustion of mixture, and causing a large number of unexploded THC emission.
From Figure 1, it can be seen that in the increasing process of AFR, with the increase of the mixing proportion of alcohol-based fuel under the same operating conditions, the THC emission is on a declining curve. This is mainly because that the alcohol-based fuel is oxygenated fuel, the percentage of oxygen in fuel gradually increases with the increase of blending ratio, which is conducive to the oxygenation of THC. The THC emission of M0 or M10 is similar with each other, whose change of average rate is 0.05%, while the THC emission of M20 is close to that of M30 (the change of average rate is 0.17%). Compared with the THC emission changes of M20, the average reduction rate of M10 is about 16.86%. This is largely because that although the oxygen content of M10 increases, mixing proportion is still low and its influence on M10 is not obvious, while the mixing proportion of M20 or M30 is relatively high, and their influence on THC emission is more apparent.
From Figure 2, it can be seen that with the AFR gradually increasing from 14.7 to 20.6, the CO emissions of these four types of engine fuel are on a declining curve. When AFR is 14.7, the CO emission reaches the maximum, and with the increase of AFR, the CO emission rapidly reduces. When AFR is 20.6, the CO emission reaches the minimum. And when AFR changes from 19.8 to 20.6, the change of CO emission becomes unobvious. This is mainly because that with the increase of AFR, the oxygen content of combustible mixture also increases, which offers a favorable condition for the further oxidization of CO. Therefore, with the gradually increase of AFR, the CO emission rapidly reduces. After AFR increases to 19.8, the increase of AFR has little influence on CO emission since oxygen content has been excessive.
Form Figure 2, under the same operating condition, with the increase of blending ratio of alcohol-based fuel, the CO emission presents a downward trend. This is mainly because that the alcohol-based fuel is oxygenated fuel. The gradual increase of the percentage of oxygen in fuel with the increase of blending ratio provides an appropriate condition for further oxidation of CO. At the same AFR, the CO emission of M0 is close to that of M10, and that of M20 is close to that of M30. Compared with M10, the CO emission of M20 decreases more obviously at the beginning. This is because the blending ratio of M10 is lower and the influence on CO emission is not obvious compared with gasoline. The blending ratio of M20 or M30 is higher, and the oxygen promotes the further oxidation of CO. It can be concluded from the above that at the early stage, the CO emission of different alcohol-based fuel rapidly declines with the increase of AFR and then decreases slowly. Until the AFR reaches 19.8, the change trend of CO emission tends to be flat. Besides, along with the increase of blending ratio of alcohol, the CO emission under the same AFR presents a downward trend.
As the AFR increases gradually from 14.7 to 20.6 in Figure 3, the NOx emissions of these four types of fuel increase first and then show a trend of decline. What is more, it reaches the maximum value when the AFR is in the range of 17.5–19.5. This is mainly because that with the increase of AFR from 14.7, the oxygen concentration in the mixture gradually increases, the combustion tends to be more completely with higher combustion temperature and the NOx emission increases. When the AFR is in the range of 17.5–19.5, the engine is in the economic fuel consumption area with the best combustion status and highest combustion temperature. At this time, the mixture in cylinder is under the oxygen-enriched condition. The concurrence of high temperature of the engine, oxygen-enriched condition, and the duration of high temperature make the NOx emission reaches the peak value, and the NOx emission of M0, M10, M20, or M30 (Pme = 0.7 MPa, n = 3200 r/min)is 2950, 3014, 3141, and 3183 ppm, respectively. With the further increase of AFR, the mixture becomes rarer and rarer, resulting in a decrease of the maximum combustion temperature of mixture and thus making the NOx emission decrease. When the AFR is 20.6, the NOx emission reaches the minimum value, and the NOx emission of M0, M10, M20, or M30 (Pme = 0.7 MPa, n = 3200 r/min)is 407, 428, 513, and 609 ppm, respectively.
From Figure 3, it can be seen that under the same operating conditions, the NOx emission shows a trend of increasing with the increase of blending ratio of alcohol-based fuel in the alcohol-based gasoline synthetic fuel. This is mainly because that the alcohol-based fuel is an oxygenated fuel. And the ratio of oxygen, which is conducive to the generation of NOx, increases gradually with the increase of blending ratio. Although the latent heat of vaporization of alcohol-based fuel reduces the combustion temperature and suppresses the formation of NOx, the promoting effect of oxygen content in alcohol-based fuel is greater than the inhibiting effect of latent heat of vaporization. At the same AFR, the NOx emission of M0 or M10 is very close, and the average changing rate of that is 3.17% (Pme = 0.7 MPa, n = 2000 r/min). While the NOx emission of M20 is close to that of M30, the average change rate of that is 13.32% (Pme = 0.7 MPa, n = 2000 r/min). This is mainly because that when the blending ratio of alcohol-based fuel is higher than 20%, the oxygen atom from the combustion of the mixture will magnify the oxygen concentration of part of the cylinder, which is conducive to the generation of NOx.
Experimental study on NEDC emission characteristic
Experimental program
The experiment was carried out in accordance with the “Emission Test of Exhaust Pollutants After Cold Start at Normal Temperature (Type I Test)” formulated in GB18352.3-2005 “Limits and Measurement Methods for Emission of Light Pollutants for Vehicles (III, IV),” including Urban Driving Cycle and Extra Urban Driving Cycle, as shown in Figure 4. The “Urban Driving Cycle” is divided into six parts according to the operation, as shown in Table 4. The “Extra Urban Driving Cycle” is divided into six parts according to the operation, as shown in Table 5.

New European driving cycle (NEDC).
Operation of urban driving cycle decomposition.

Operation of extra urban driving cycle decomposition.

Effect of alcohol-based fuel on the THC emission of driving cycle
It can be seen from Figure 5(a) that there are more THC emission of M0, CNG, M10, M20, or M30 in 195 s’ urban cycle case 1. This is because that the AFR is under the stoichiometric value due to the enrichment for fuel in the cold-start stage. In addition, cold shock effect is strong, mixed gas atomization is not good, and combustion is not stability due to the low temperature of cylinder and piston under the cold-start condition. THC emission of M0, CNG, M10, M20, or M30 in urban cycle cases 2, 3, and 4 is steady relatively because of the increasing of temperature and complete combustion. Figure 5(c) shows THC emission under Extra Urban Driving Cycle. The THC emission of M10, M20, or M30 is greater than M0 and CNG, and the fluctuation frequency is larger. That is because the enrichment for fuel leads to the incomplete combustion.

Comparisons of THC emission: (a) on NEDC (1–195 s), (b) on NEDC (196–780 s), and (c) on NEDC (781–1180 s).
Effect of alcohol-based fuel on the CO emission of driving cycle
As it is shown in Figure 6(a), the peaks of the CO emissions of these five fuels occur in urban cycle case 1 because the non-uniform fuel atomization and low mixture gas temperature at the beginning of the engine operation. And the CO emission gradually reduces and then stabilizes in urban cycle cases 2, 3, or 4 because of the higher mixture gas temperature, uniform fuel atomization, and enough CO oxidation under engine stable operation according to Figure 6(b). However, the CO emission is relatively high under 400 s suburban driving cycles because the fuel delivery increases and the O2 decreases relatively under heavy load operating conditions.

Comparisons of THC emission: (a) on NEDC (1–195 s), (b) on NEDC (196–780 s), and (c) on NEDC (781–1180 s).
Effect of alcohol-based fuel on the NOx emission of driving cycle
By comparing the statistics of Figure 7(a) and (b), the NOx emission under urban driving conditions 2, 3, or 4 is higher than that of urban driving condition 1. That is mainly because the production of NOx is lower with the non-uniform fuel atomization and low burning temperature at the beginning of the engine operation. By comparing the statistics of Figure 7(b) and (c), the NOx emission under suburban driving conditions is relatively high. That is mainly because the engine speed is higher under suburban conditions, which causes the increase of in-cylinder fuel mass per unit time and the increase of NOx emission.

Comparisons of NOx emission: (a) on NEDC (1–195 s), (b) on NEDC (196–780 s), and (c) on NEDC (781–1180 s).
Conclusion
This article took gasoline as an equivalent fuel and formed other three new types of alcohol-based gasoline synthetic fuel, M10, M20, and M30, according to 10%, 20%, and 30% volume proportion of alcohol-based fuel. And the effect of AFR on the original engine emissions of three conventional emissions, THC, CO, and NOx, of new types of vehicle alcohol-based gasoline synthetic fuel under the lean burn condition were studied. And then the test that used pure gasoline M0, CNG, and new alcohol-based gasoline synthetic fuels M10, M20, M30 was carried out on the drum test bed under NEDC. The following conclusions are drawn:
First, under certain engine speed and load, the fuel consumption ratio of M0, M10, M20, and M30 reduces first and then increases with the increase of AFR. The minimum value appears when AFR is in the range of 16.5–18.5.
Second, with the increase of AFR, the THC emission of M0, M10, M20, or M30 reduces first and then increases. The minimum value appears when AFR = 17.6. And when AFR is less than 17.6, the decreasing trend is smaller than the increasing trend when AFR is greater than 17.6. Under the same operating conditions, with the increase of blending ratio of alcohol-based fuel, the THC emission reduces. With the increase of AFR, the CO emission gradually decreases. And under the same operating conditions, with the increase of blending ratio of alcohol-based fuel, the CO emission reduces. With the increase of AFR, the NOx emission increases first and then reduces, and the minimum value of that appears when AFR changes from 17.5 to 19.5. Under the same operating conditions, with the increase of blending ratio of alcohol-based fuels, the NOx emission presents an increasing trend. And the emissions have the similar trend on the drum test bed under NEDC.
Third, in the first urban driving cycles, due to cold start of the engine, the temperature of combustible mixture is low and the narrow gap effect is obvious. These bring out a large amount of unburned THC and combustion intermediate CO, which reach peak under this driving condition. And the low combustion temperature and anoxic environment lead to the minimum NOx emission peak during the whole experiment process.
Footnotes
Handling Editor: Haiping Du
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to express their deep gratitude to National Science Foundation of China (approval no. 51576140), National High Technology Research and Development Program (“863” Program) of China (approval no. 2008AA06Z322).
