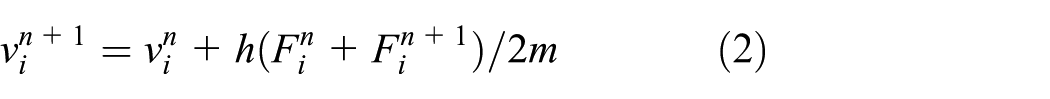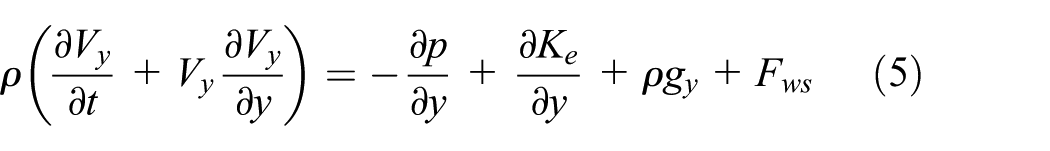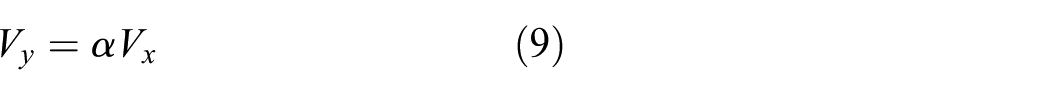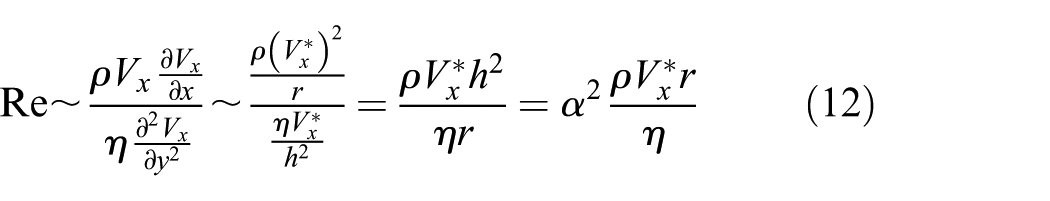Abstract
Investigation on the mechanism of impact-contact occurred at multiphase interface is of great importance in technique control of mechanical polishing as it is the basic dynamic process connected with mass transfer and interfacial pressure. Classical continuum mechanics is not fit for study the physical essence of complex dynamic behavior in the impact-spreading at nano length scale because of the small thickness of fluid film and the discrete property of surface morphology. Molecular dynamics method has already been proved to be one of the most efficient toolkit on atomic scale discrete phenomenon and thus being employed in this research to study the complex mechanism of nano-scale impact-spreading. The study shows that the liquid film behaves like a stretched membrane under the unbalanced forces and the real traverse spreading is an anisotropic process resulted by the anisotropic surface structure which also influences the nonuniform distribution of film. The result justifies that boundary lubrication at the interface is resulted by poor spreading behavior on rough surface and will affect the transportation of abrasive particle and materials removal rate. The results also justify that the mechanical similarity can be difficult to hold because of the complexity of surface texture (rough surface) and the different contour profiles resulted by random movement of molecule. Furthermore, energy distribution shows that physical adsorption plays an important role in the impact-contact process which is also justified by the adsorption structure of water. With the increasing of impact velocity, part of outer molecules breaks away the constraints generated by the surface tension and forming a free-state water layer.
Introduction
The impact-contact dynamic behavior of single and clouds of drops on solid surface has been studied for over a century not only because of the great scientific interest but also due to its relevance in industrial applications as diverse as spray coating, spray painting, inkjet printing, fuel injection, soil erosion due to rain drop impact: F Heslot et al. 1 found that the final stage of spreading is not a pancake but rather is a two-dimensional gas; PJ Smith et al. 2 investigated the track of organometallic ink spreading at room temperature; T Mao et al. 3 studied the spread and rebound of liquid droplets on flat surface using CCD camera and found that the maximum spread has intense relationship with liquid viscosity; YY Yan 4 and M Voue and J De Coninck 5 studied the spreading problems at different scales; H Gau et al. 6 studied the liquid morphology on different structured surfaces; H Dong et al. 7 developed an apparatus for visualizing the drop formation and impaction on substrate; P Lenz and R Lipowsky. 8 found that the morphology of wetting layers on imprinted surface was determined by the geometry of the underlying surface domain; F Heslot et al. 9 studied the influence of surface energy on the profiles of drop on the substrate; the multibody contact-sliding and the dynamic evolvement mechanism at the interface were investigated by the author.10,11
The behavior of droplets on substrate, and the subsequent dots, lines, and films that can be formed by depositing and overlapping droplets is also a critical consideration in nanofabrication process such as mechanical polishing technique (Figure 1) as this strongly influences the evenly distributed of slurry and thus the surface global flatness and surface damage. One of the most important research topic in polishing is the relation of the droplet diameter and its contact angle with spreading dynamics on rough surface. The fully spreading of droplet is helpful on transferring the abrasive grain to interface and carry off the removal materials. The important characterization parameters are droplet volume and droplet speed. Several events such as deposition, rebound, receding breakup, and rim instability can happen when a droplet impacts on surface, which depend on the velocity of the impact, and properties of the fluid and surface. The high-velocity spherical drop will produce a shock rim in the liquid drop which expands radially. Liquid rim either continues to spread but at a reduced rate (wetting surface) at the maximum diameter or keeps at rest and then retracts toward the point of impact (partially wetting and nonwetting surfaces). 12 The liquid will retract toward the equilibrium position and stop after some oscillations in the case of partially wetting surface. 11 On the other hand, the liquid retracts beyond the equilibrium position and rises on the surface in case of nonwetting surfaces. If the surface energy is low enough, rebounding occurs where the liquid separates from the surface. Therefore, the spreading of liquid droplet on solid surface is a more complex phenomenon than generally considered. Many experiments on the dynamic spreading of droplets at small scale revealed even more complex and fascinating phenomena, including a variety of droplet shapes. 12
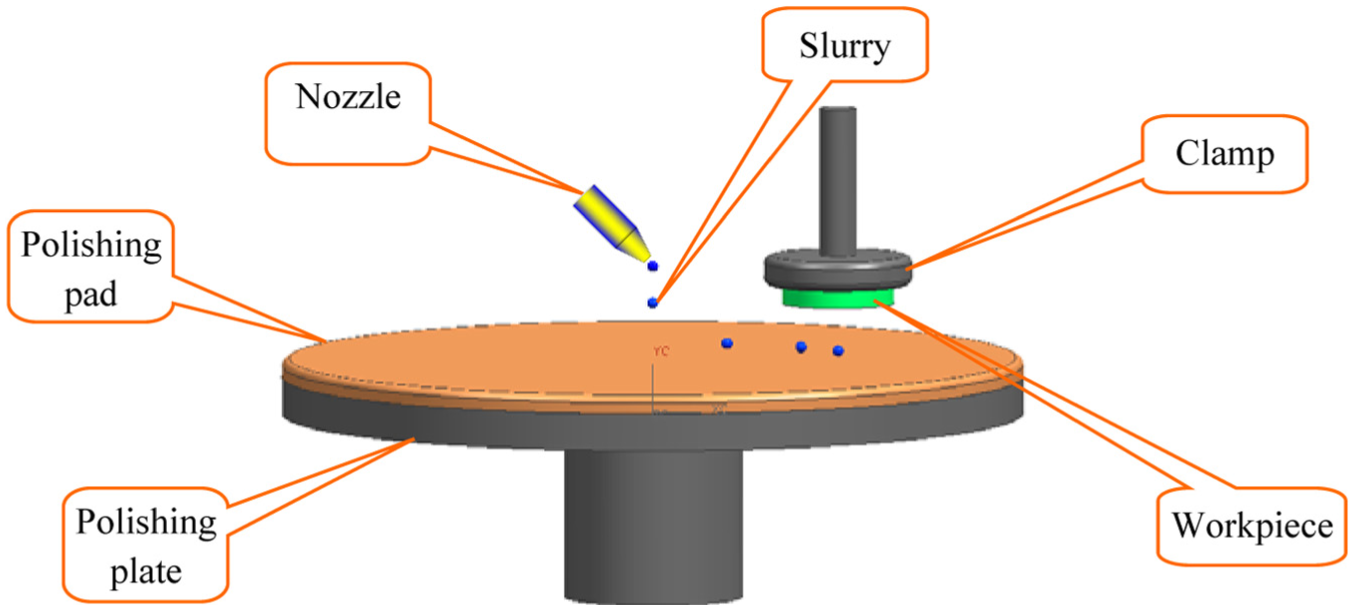
Scheme of mechanical polishing technology.
Presently, most research on the dynamics of spontaneous drop spreading have been done by assuming ideal cases because of the complexity of influence of rough surface. The fine structure in the thin film is usually discussed in terms of precursor films driven by Van der Waals forces can only be appropriate for films much thicker than the size of their constituent molecules, which can be treated hydrodynamically. However, different behaviors are seen in ultrathin films with only several molecular diameters thickness. Experiments justify that the spreading liquid consists of distinct monomolecular layers which advance across the solid with different velocities but not varying smoothly. Such films are too thin to be described by lubrication equations. The complexity of solid/liquid interfaces makes it difficult to accurately predict the behavior of liquid droplets on solid surfaces. Droplet spreading on surface represents a multi-scale phenomenon with scales ranging from continuum to molecule. We have been motivated thereby to conduct molecular dynamics (MD) simulations to investigate the spreading characteristics of nano-scale water-droplet on rough surface. While we are restricted to small drop whose initial radius is only at atomic sizes, the results complement the experiments through detailed three-dimensional information during the spreading process.
Today, the dynamical behavior of the macroscopic part of the drop far from the substrate is well understood while the behavior near the substrate is not fully uncovered. The Langevin theory is not fit for all of the conditions because of the rather closely packed arrangement of the molecules. Therefore, we focus on the development of correlations and scaling laws to examine the effect of droplet size on the spreading characters by means of MD simulation of water spreading on rough surface. MD simulation 13 has already been recognized as an ideal approach to gain insights into the atomic scale process and understand their mechanisms because of its high temporal and spatial resolution. Therefore, the large-scale classical MD method is adopted in this work to investigate the dynamic process and gain insights into the atomistic scale multiphase process.
Methodology
The dimension of substrate is 250 Å × 5 Å × 250 Å along x,y, and z direction (Figure 2). The water-drop is sphere and the diameter of which is 60 Å. The downward feeding speed is 30 m/s, the environment temperature is 293 K, and the computation time step is

MD simulation.
MD is a computer simulation method for studying the physical property of ensemble composed of atoms and molecules; the atoms and molecules are allowed to interact for a fixed period of time, giving a view of the dynamical evolution of the ensemble. The trajectories of atoms and molecules are determined by numerically solving Newton’s equations of motion for a system of interacting particles, where forces between the particles and their potential energies are calculated using interatomic potentials or molecular force fields. The method was originally developed within the field of theoretical physics in the late 1950s, but is applied today mostly in chemical physics, materials science, and the modeling of biomolecules.
The classical water models treat the water molecule as rigid and rely only on nonbonded interactions. The electrostatic interaction is modeled using Coulomb’s law and the dispersion and repulsion forces is characterized by the Lennard–Jones potential. The potential models TIP4P can give more reasonable description of water molecule and represented by
where kC is the electrostatic constant, has a value of 332.1 Å·kcal/mol in the units commonly used in molecular modeling; qi is the partial charges relative to the charge of the electron; rij is the distance between two atoms or charged sites; and A and B are the Lennard–Jones parameters. The charged sites may be on the atoms or on dummy sites (such as lone pairs). In most water models, the Lennard–Jones term applies only to the interaction between the oxygen atoms.
The TIP4P model (Figure 3), first published in 1983, is widely implemented in computational chemistry software packages and often used for the simulation of biomolecular systems. The four-site models place the negative charge on a dummy atom (labeled M in the Figure 3) placed near the oxygen along the bisector of the HOH angle. This improves the electrostatic distribution around the water molecule. Four-site models were investigated and found to be superior. In this case, the negative charge is moved off the oxygen and toward the hydrogen at a point (M) on the bisector of the HOH angle. Equation (1) still applies with little increase in complexity: ten distances are now required to evaluate the function instead of nine for the three-site model. This four-site form was first proposed by Bernal and Fowler along with a set of parameters based on calculations for properties of the monomer, dimer, and ice. This work adopted four-site TIP4P model and study the atomic impact-spreading dynamic process.
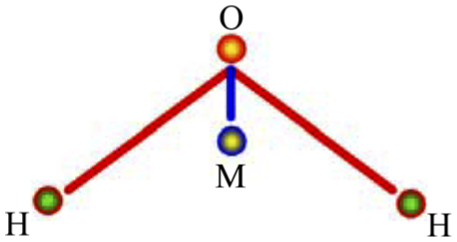
Four-site water molecule model.
The computation of atom trajectory requires numerical integration of the differential equations from the initial state which the water-drop approaches substrate but has not yet reached the final state which the water-drop spreading totally on the substrate surface. Many kinds of methods are verified to be suitable for performing the numerical integration which include the fourth-order Runge–Kutta method, the Leap-Frog method, the Verlet method, the Velocity-Verlet method, and so on. This article adopted the Velocity-Verlet method because it is a symplectic algorithm which can prevent the energy dissipation and have high computation efficiency in the long-term numerical simulation
where
Results and analysis
Numerical analyzing the process
Figures 4–7 shows different stages of nano-scale impact-contact process in mechanical polishing process. It has already been justified that the dynamic process happened at solid–liquid interface has intense relationship with the competition between solid–liquid and liquid–liquid interaction forces. The liquid surface under the effect of unbalanced forces behaves like a stretched membrane and can be characterized by the surface or interfacial tension force

Impact-spreading of droplet on the rough surface: (a) time step: 3000, (b) time step: 6000, (c) time step: 9000, (d) time step: 12,000, and (e) time step: 15,000.

Variation tendency of interaction force: (a) interaction force component along x-axis, (b) interaction force component along y-axis, and (c) interaction force component along z-axis.
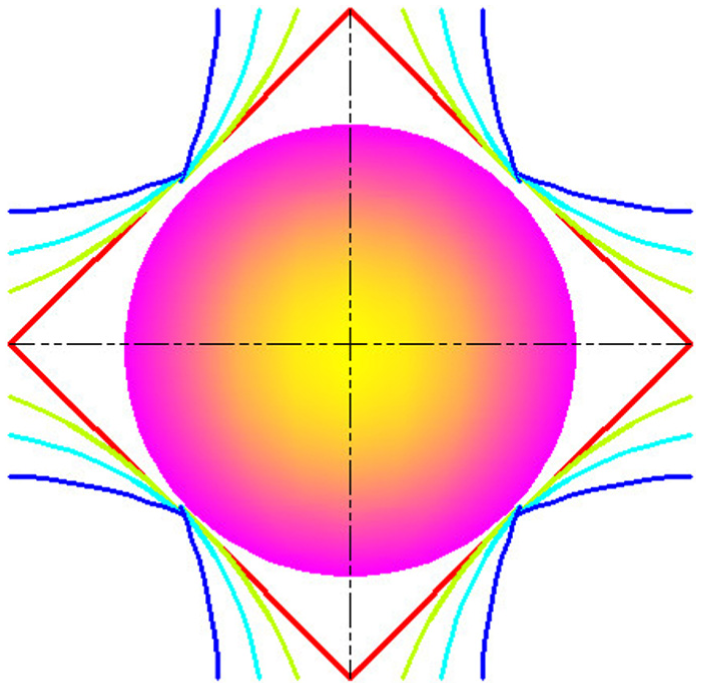
Spreading mode (from red line to blue line).
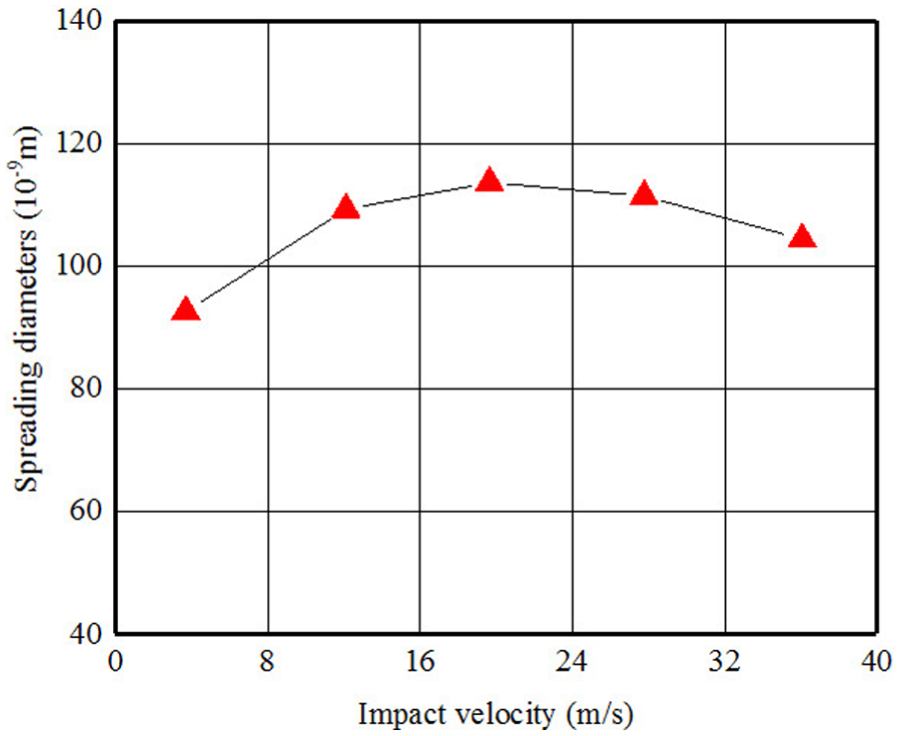
The relationship between impact velocity and spreading diameter.
The interface is gradually generated with the spreading which result in the increasing of contact area. The potential energy (result by the variation of center of mass in y direction) and the kinetic energy of water-drop together provide the energy of new interface. It is different from daily experience that the water-drop will cover all substrate surfaces because the spreading is activated by the work which depends on the kinetic energy of the drop. As the water-drop spreading is a complex dynamic behavior, some unknown mechanism dominates this process, namely, the initialization and the suspending ought to be some kind of energy transformation process.
Materials surface is in general the place where powerful electrostatic field will exert larger attractive force on polar molecules and result in physical adsorption. Theoretically, the structured surface ought to be composed of many potential wells act as adsorption sites for capturing water molecule. In fact, the water layer will not stably stayed on the surface if there are no physical adsorption. Figure 8 shows the percentage of sites on the substrate surface with energy. The most negative the energy values, the stronger the site is. The site energy characterizes the energy of interaction between the water molecule and the surface and taken to be zero if the water molecule lies in infinite distance from the surface.

The energy distribution of substrate surface.
It is shown that about 20% of the surface site have a water interaction energy of ∼90 kJ/mol or less which justifies the important role of physical adsorption at atomic scale two-phase interface. The physical adsorption is helpful about generation stable two-phase interface but it will hinder the spreading process.
Water-drop with different sizes and different velocities can cover different parts of the substrate surface by means of different spreading behaviors. Qualitative relationship ought to be existed between cover area, impacting velocity, volume of water-drop (determine the capability of doing work), and interface interaction. The equilibrium of thin liquid films on the solid surface is the result of the combination of above factors.
Figure 9 shows some typical adsorption structure of water on substrate surface. Some water molecules are observed being physically adsorbed on the rough surface and their polar axis oriented about 14° with the surface plane. Comparing with free water molecule, the OH bond and the HOH angle are both increased. The adsorption of water clusters is observed which justifies that both H-bonding and water-surface interactions are involved in the surface adsorption. The trimer is least stable with the adsorption energy of only 359 meV. The adsorption energy of the dimer is 433 meV and thus being more stable than trimer. This energy difference reflects the variation in the number of water-surface bonds and the H-bonds formed in the adsorbed clusters. Furthermore, one-dimensional water chain is observed on the surface. This chain is stable because it favors intermolecular H-bonding and the dipole–dipole interactions.

The adsorption of water molecule on substrate surface.
Mathematical analyzing of process
Generally, in the three-dimension spreading of water-drop, the capillary regime is an initial stage of spreading of small drops, whereas the gravitation-regime is the final stage of spreading of small drops or big drops. Transition from the capillary regime of spreading to the gravitational regime takes place at the moment tc
where r(t) is the radius of drop base at time t and is referred to below as the radius of spreading; γ and ρ are the interfacial tension and density of the liquid, respectively; g is the gravity acceleration. However, the gravitational effect is minimal compared to the surface force as surface force is the dominate factor at nano-scale. Therefore, the g in equation (4) ought to be substituted by acceleration resulted by the dynamic interaction force. The variation tendency of the three component of interaction force is shown in Figure 5. There are three stages in this impacting–spreading process considering Figures 4 and 5, namely, the compression along negative direction of y-axis (deformation accumulative), traverse spreading, and sputtering along the direction of y-axis (Figure 10). The contour profile of the water-drop remains unchanged except part of water molecules at the bottom of sphere are compressed into the sphere and result in a densified layer which corresponding to the left part of Figure 5(b) (before 3000 time steps) and no traverse spreading is happened. The real spreading process is initialized after 3000 time steps which result in the decreasing of impacting force between water-drop and substrate. Furthermore, the real traverse spreading is anisotropic process because of anisotropic surface structure at atomic scale. Figure 7 gives the relationship between spreading diameter and impact velocity. The spreading area increases initially when the impact velocity less than critical value and after that the it shrinks with the increasing of impact velocity. This phenomenon ought to be attributed to more and more high-velocity molecules break away from the constraint exerted by surface force.

Simplified shape profile in the impact-spreading process: (a) green: impacting region and (b) blue: spreading region.
This atomic scale spreading process can be analyzed using the surface diffusion model as follows
where
If set
The water-drop has a relative high slope in the impacting stage, then
and hence, we can estimate the Reynolds number of impact stage as follows
The expression (equation (12)) justifies that the Reynolds number is proportional to
Comparing with the results acquired from the spreading on smooth surface, larger force along traverse direction (along x-axis and z-axis) is generated in spreading along rough surface. If the size of drop is less than some critical value, it will spread along randomly distributed surface structure which results in the open-channel flow. The substrate is finally being covered by equilibrium liquid layer. The thickness (h) of the film is determined by the potential of action of surface forces which has intense relationship with surface roughness. The investigation justifies that boundary lubrication ought to be existed at interface of polishing process because of poor spreading behavior on rough surface and it will affect the transportation of abrasive particle and materials removal rate.
Conclusion
The spreading of water-drop over rough solid substrates is a challenging problem with technical significance for many industries such as mechanical polishing, inkjet printing. There is no widely accepted explanation about the impact-contact at nanometer scale near the substrate, although the dynamical behavior of the macroscopic part far from the substrate is well understood. In this article, MD simulation of impact-contact phenomena on rough surface associated with nano-sized water-droplet has been carried out and some novel phenomena are observed:
The liquid surface under the effect of unbalanced forces behaves like stretched membrane determined by the surface force or interfacial tension force. The real traverse spreading is anisotropic process because of anisotropic surface structure.
Although spreading geometrically similar for similar geometries (geometrically similar channels, geometrically similar bodies in a flow) at microscopic scale, the mechanical similarity can be difficult to hold because of the complexity of surface texture (rough surface) and the random movement of water molecule. Therefore, the general contact angle cannot be an efficient characterization parameter for nano-scale impact-spreading process.
The local thickness of the film (spreading area) is a function of the coordinate because of the rough surface; the mean value of the thickness of the film on rough surface must differ from the thickness of a flat film on ideally smooth surface.
Part of high-velocity molecules will break away the constraint generated by the surface tension in the case of high impact velocity. On the other hand, the water-drop will stay on the substrate surface with small deformation and almost no spreading will be activated when the impact velocity is smaller than some definite value.
In mechanical polishing, boundary lubrication ought to be existed at the interface because of poor spreading behavior on rough surface which can deteriorate the abrasive particle transportation and materials removal rate.
The structured surface sites greatly influence the behavior of water molecule justifies the important role of physical adsorption at two-phase interface in the atomic scale. The physical adsorption is helpful about generation of stable two-phase interface but it will hinder the spreading process.
Footnotes
Academic Editor: Xiaotun Qiu
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the following foundation: China National Nature Foundation (grant no. U1430122), Tianjin Natural Science Foundation (grant no. 15JCYBJC19900), and Aeronautical Science Foundation of China (grant no. CALT201509), The National Natural Science Foundation of Guangdong province people’s government joint fund—supercomputing science application research funding (grant no. nsfc2015_296), and State Key Laboratory of Smart Manufacturing for Special Vehicles and Transmission System Open Fund (GZ2016KF006).