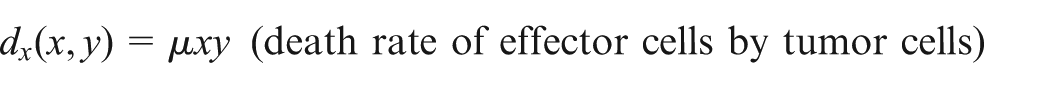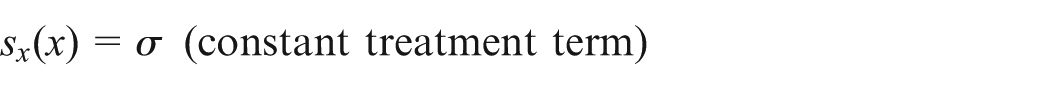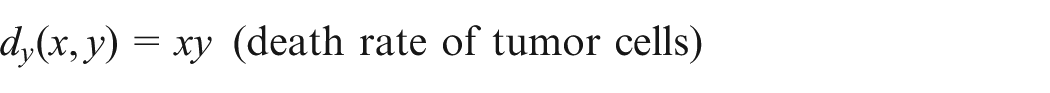Abstract
In this article, we examine the fractional order model of the cytotoxic T lymphocyte response to a growing tumor cell population. We investigate the long-term behavior of tumor growth and explore the conditions of tumor elimination analytically. We establish the conditions for the tumor-free equilibrium and tumor-infection equilibrium to be asymptotically stable and provide the expression of the basic reproduction number. Existence of physical significant tumor-infection equilibrium points is investigated analytically. We show that tumor growth rate, source rate of immune cells, and death rate of immune cells play vital role in tumor dynamics and system undergoes saddle-node and transcritical bifurcation based on these parameters. Furthermore, the effect of cancer treatment is discussed by varying the values of relevant parameters. Numerical simulations are presented to illustrate the analytical results.
Keywords
Introduction
Mathematical biology is a fast-growing subject which opens up new and exciting branches for mathematicians and biologists. Immune system has natural capacity to detect and destroy abnormal cells which may prevent the growth of many cancers. Mathematical model provides an analytical outline to address which components of the immune system play considerable role in cancer treatment. In 1825, Gompertz used mathematical model first time to explain tumor growth by considering cell replication and death. According to his model, for large populations growth is slow whereas for small populations growth is faster. After that many mathematical models have been developed to understand interactions between the different constituent of the tumor microenvironment. For overviews of simple mathematical models on this topic, see previous works.1–6 The tumor microenvironment comprises growth factors (containing cytokines and hormones), immune cells, the extracellular matrix, fibroblasts, signaling molecules (chemokines and cytokines), and other connective tissue cells. It is essential to understand these interactions in order to develop the influential cancer immunotherapies.
One of the main open problems in this area is to formulate the mathematical models that capture the dynamics of real data more accurately. In this direction, fractional calculus has made a significant impact on the dynamics of various diseases. 7 Fractional derivatives have the excellent property of capturing the memory effects which is detected in nearly all biological systems. This property cannot be covered by means of the integer order derivatives. Bolton et al. 7 have shown that fractional order Gompertz model fits the particular tumor growth data set with order 0.68 much better than the ordinary integer order Gompertz growth model. Rodrigues et al. 8 notice that dynamics of real outbreak data of dengue cannot be described by classical first-order derivative with a sufficient degree of accuracy. A fractional order model of malaria transmission among heterogeneous populations has been proposed by Pinto and Machado 9 to better approximate the real dynamics. HIV fractional complex-order model for drug resistance with three distinct growth rates for the CD4+ T helper cells is analyzed by Pinto and Carvalho. 10 A mathematical model of dengue fever outbreak is proposed by Diethelm 2 using fractional derivative. Recently, Huo et al. 11 studied a fractional order HIV model to assess the impact of vaccines in a homosexual community. They have shown that the vaccinated reproduction below unity is not threshold of HIV eradication when effectiveness and the dosage of the vaccines are low. A new critical threshold is derived in order to eradicate the HIV. Numerous fractional dynamical models have been given by Magin12–14 for biological systems, for example, membrane charging, vestibular ocular models, one-dimensional cable model for nerve axon, viscoelastic models of cells, and tissues. Stability analysis and bifurcation techniques can help to determine qualitative dynamics of infectious disease models. Recent papers15,16 provide a good survey of the methods available to analyze the stability of fractional differential equations. In El-Saka et al., 17 stability, persistence, and Hopf bifurcation of some continuous time fractional order differential equations are studied. Equilibrium points, existence, uniqueness, stability, and numerical solution of fractional order Lotka-Volterra predator prey and rabies system have been studied by Ahmed et al. 18 Theory of fractional differential equations can be found in Kilbas et al. 19 and Miller and Ross. 20
The immune reaction to a tumor is generally adjudicated by cells. It is difficult to understand dynamics of the antitumor immune reaction in vivo. Cytotoxic T lymphocytes (CTL) and natural killer (NK) cells perform a leading role in this process. The aim of this article is to investigate fractional order mathematical model of the CTL effect to a flourishing tumor cell population. We examine the long-term behavior of tumor model by varying the values of parameters that might be changed as a result of cancer treatment. Fractional dynamics also exhibit the mechanisms of tumor dormancy. Tumor dormancy is the state when tumor cells persist for long time duration with minor or no change in population of tumor cells. 21 Dormant states can emerge after cancer treatment. Dormant states also observe at initial stages of tumor progression. After a long duration, small dormant tumors may escape from immune system and show uncontrolled growth by sneaking through mechanism. 21
This article is organized as follows. Formulation of fractional order immune tumor model is given in section “Generalized tumor-immune model.” Existence of physically significant equilibrium points and their stability are investigated in section “Equilibrium points and stability analysis.” Section “The Grünwald–Letnikov Approximation” describes the Grünwald–Letnikov scheme for tumor model. Section “Bifurcation analysis” contains the bifurcation analysis of tumor model. Numerical simulations for possible cases of bifurcation parameters are presented in section “Numerical simulations.” Discussions are given in the last section.
Generalized tumor-immune model
Immune system eliminates the very small tumors before they develop into clinically apparent by reducing their intensification on initial stages. Therefore, mathematical models are very helpful to investigate the tumor dynamics based on interactions between tumor-immune cells. Immune cells are also called effector cells. Mathematical model for the interactions among effector cells and a thriving immunogenic tumor in vivo is given by Kuznetsov et al. 22 using the following coupled system of differential equations
where x corresponds to the density of the effector cells and y indicates the density of the tumor cells. First equation (1) describes the dynamics of immune cells where
both of these terms are based on connection between the effector tumor cells
The tumor cells include
Our aim is to examine the dynamics of the following fractional order system
together with the initial conditions
The first term on the right-hand side represents the source of immune-effector cells. The second term is the recruitment of tumor-specific effector cells which follow the Michaelis–Menten factor to specify the saturated effect of immune response. On the other hand, the effector cells are being eradicated by tumor cells which follow the law of mass action at a proportional rate
Parameters values.

We denote

Lemma 2.1
Assume that the function
Then, the initial value problem

has a unique solution.
Theorem 2.2
There is a unique solution for the initial value problem (2) and the solution remains in
Proof
From Lemma 2.1, we obtain the unique solution on

on each line bounding the nonnegative octant, the vector field
Equilibrium points and stability analysis
To understand the dynamics of (2), first we will find the equilibrium points of the system. Nullclins, that is, the curves along which
Letting

Letting

The classification of equilibrium points is as follows:
Tumor-free equilibrium:
Tumor-infection equilibrium:

here y is nonnegative solution of

Depending on the parameter values, there may be from zero to three tumor-infection equilibrium points.
To discuss the local asymptotic stability, we have the following linearized system

where

The associated transcendental characteristic equation of system (2) is given by
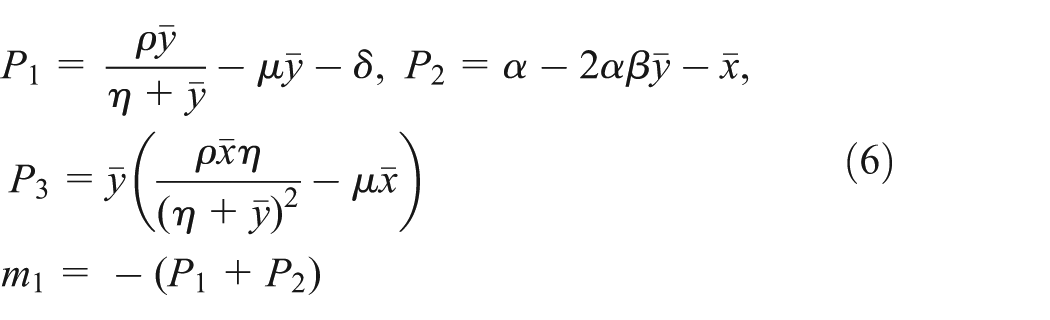
and

Then, we get

Hence, eigenvalues of the Jacobian matrix are given by

Local stability of tumor-free equilibrium point
In the absence of tumor (i.e.

This gives

Clearly,

Here,
Theorem 3.2
The tumor-free equilibrium point of the system (2) is locally asymptotically stable if
Local stability of tumor-infection equilibrium point
The tumor-infection equilibrium corresponds to the case where tumor infection persists

where




The infection equilibrium point of the model (2) can be obtained by substituting the solutions of (8) into (4). Let
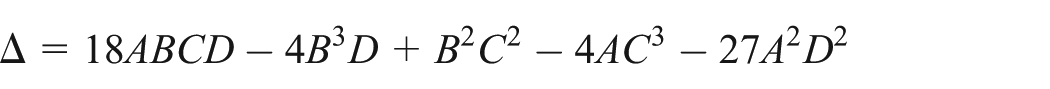
Notice that



Due to
Theorem 3.4
If
If
If
If two distinct positive tumor-infection equilibrium points if and only if one positive tumor-infection equilibrium points if and only if
If one positive tumor-infection equilibrium points if and only if one positive tumor-infection equilibrium points if and only if two multiple positive tumor-infection equilibrium points if and only if
If
If one positive tumor-infection equilibrium points if and only if three distinct positive tumor-infection equilibrium points if and only if one positive tumor-infection equilibrium points if and only if
If one positive tumor-infection equilibrium points if and only if two multiple positive tumor-infection equilibrium points if and only if one positive tumor-infection equilibrium points if and only if
If
Using Proposition 1 given by Ahmed and Elgazzar, 25 we can propose the following theorem regarding stability of tumor-infection equilibrium point.
Theorem 3.5
The tumor-infection equilibrium point of the system (2) is locally asymptotically stable if one of the following conditions is satisfied
where
The Grünwald–Letnikov approximation
Let

with

where

and

These binomial coefficients are recursively connected 26 by

where the first coefficient is
Assume that there exists a unique solution

Let
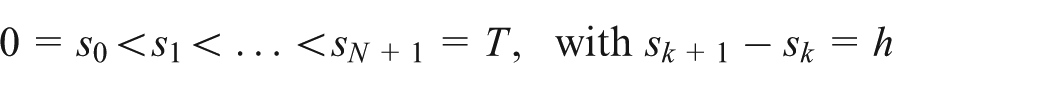
Then, the explicit Grünwald–Letnikov method is given by 26

where

Let

Bifurcation analysis
In this section, we explore the long-term outcome of the system by using bifurcation analysis. We will investigate the effect of parameters on the tumor dynamics, that is, how the number of equilibria and their stability change by varying the different parameters.
For the parameter values given in Table 1,
If
If
This implies that tumor-free equilibrium point is stable on
If
If
Hence, according to Theorem 3.4 part (9), system (2) has a unique positive tumor-infection equilibrium point when
For
For
From Figure 1, we can see that the tumor-free equilibrium is unstable for
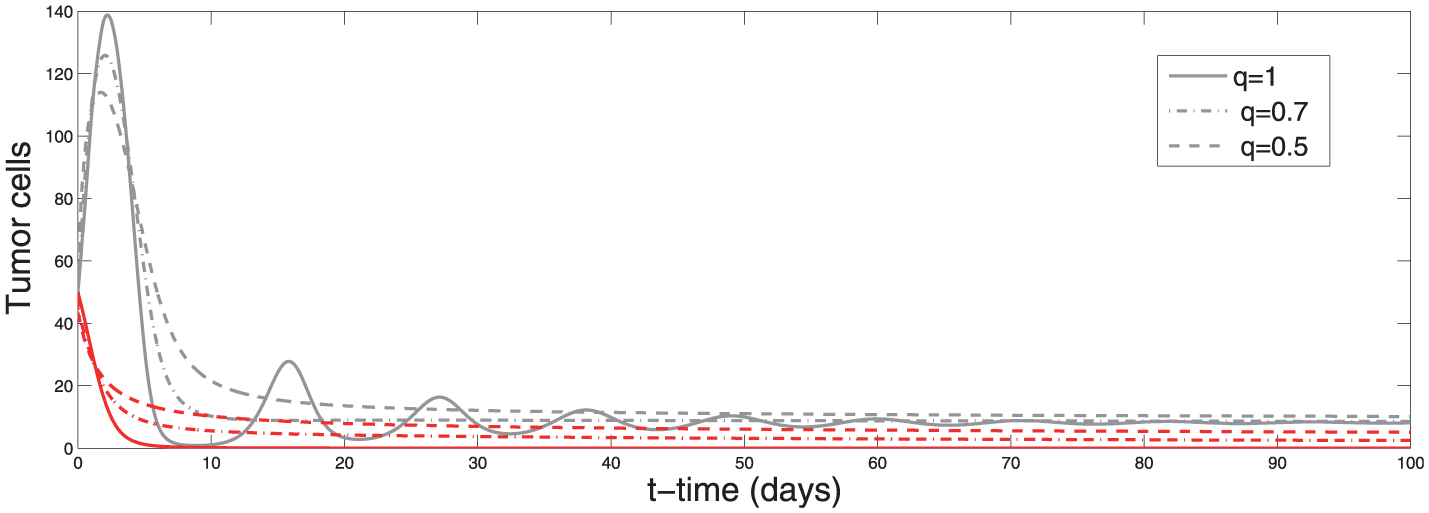
Effect of parameter
With the help of bone marrow transplant, parameter
Using same argument as in previous discussion, we conclude that there is saddle-node bifurcation at
If
If
This yields that change in stability of tumor-free equilibrium occurs as
For
For
We can see with raise in
The value of
For
For
This implies that with the increment in
Numerical simulations
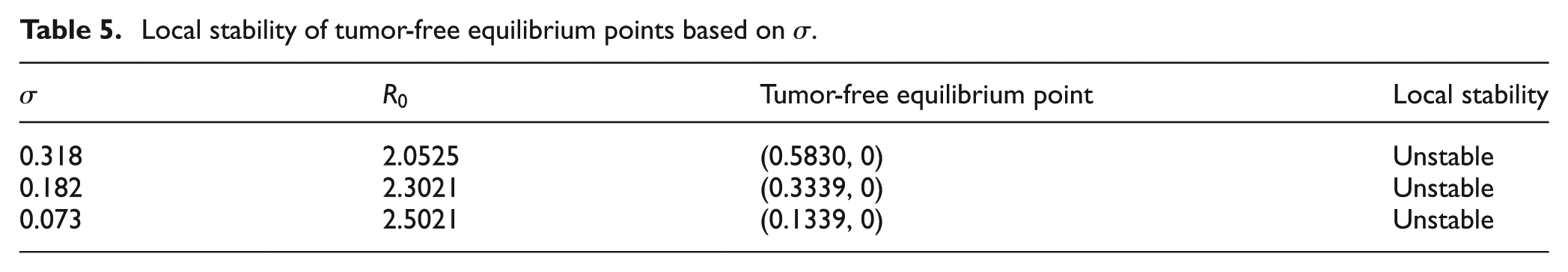
In this section, we employ the Grünwald–Letnikov method to fractional model (2). In order to verify the analytical results with numerical simulations, first, we will compute the local stability of tumor-free and tumor-infection equilibrium points of fractional order tumor model (2) using Theorem 3.2 and Theorem 3.4, respectively, for selected values of bifurcation parameter
Local stability of tumor-free equilibrium points based on
Local stability of tumor-infection equilibrium points based on

Figure 2 shows the solution of the system (2) for the parameter values given in Table 1. We obtained convergence in a shorter time for fractional order

Solution of the system (2). Upper panel represents simulation results of effector cells x with initial condition

Phase portraits of effector cells x and tumor cells y with
The approximate solutions of system (2) for fractional order
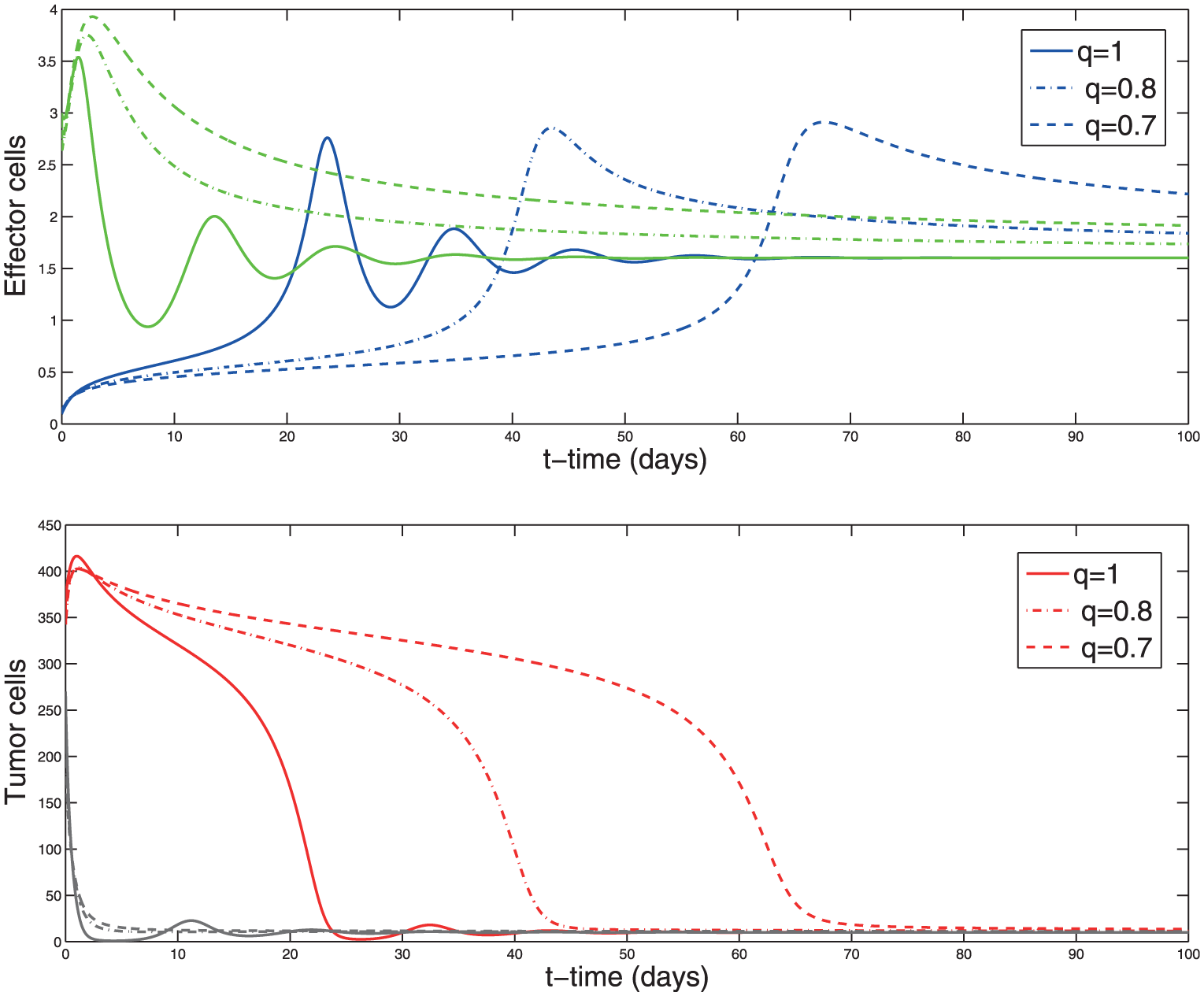
Solution of the system (2) with

Solution of the system (2) with
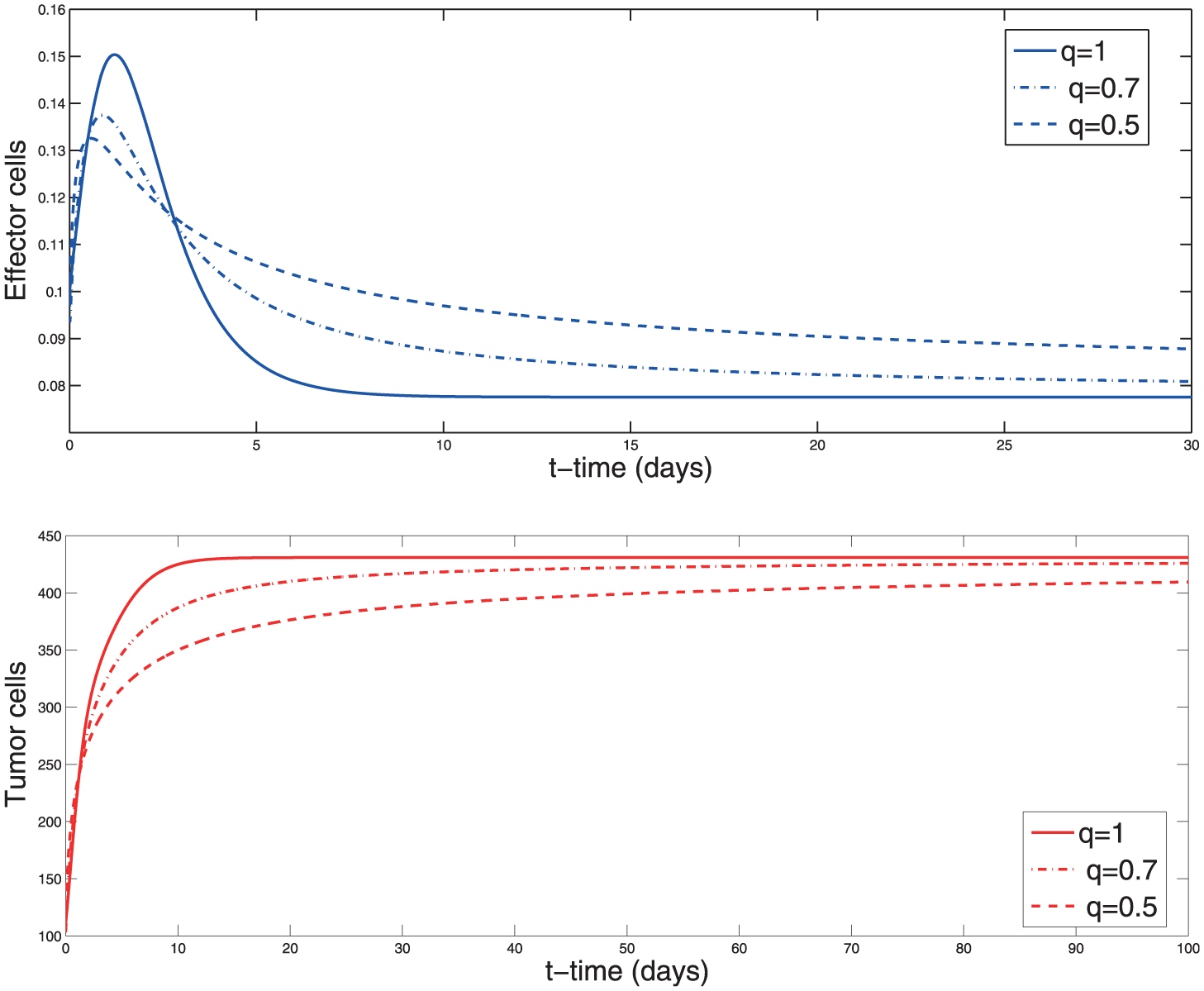
Solution of the system (2) with
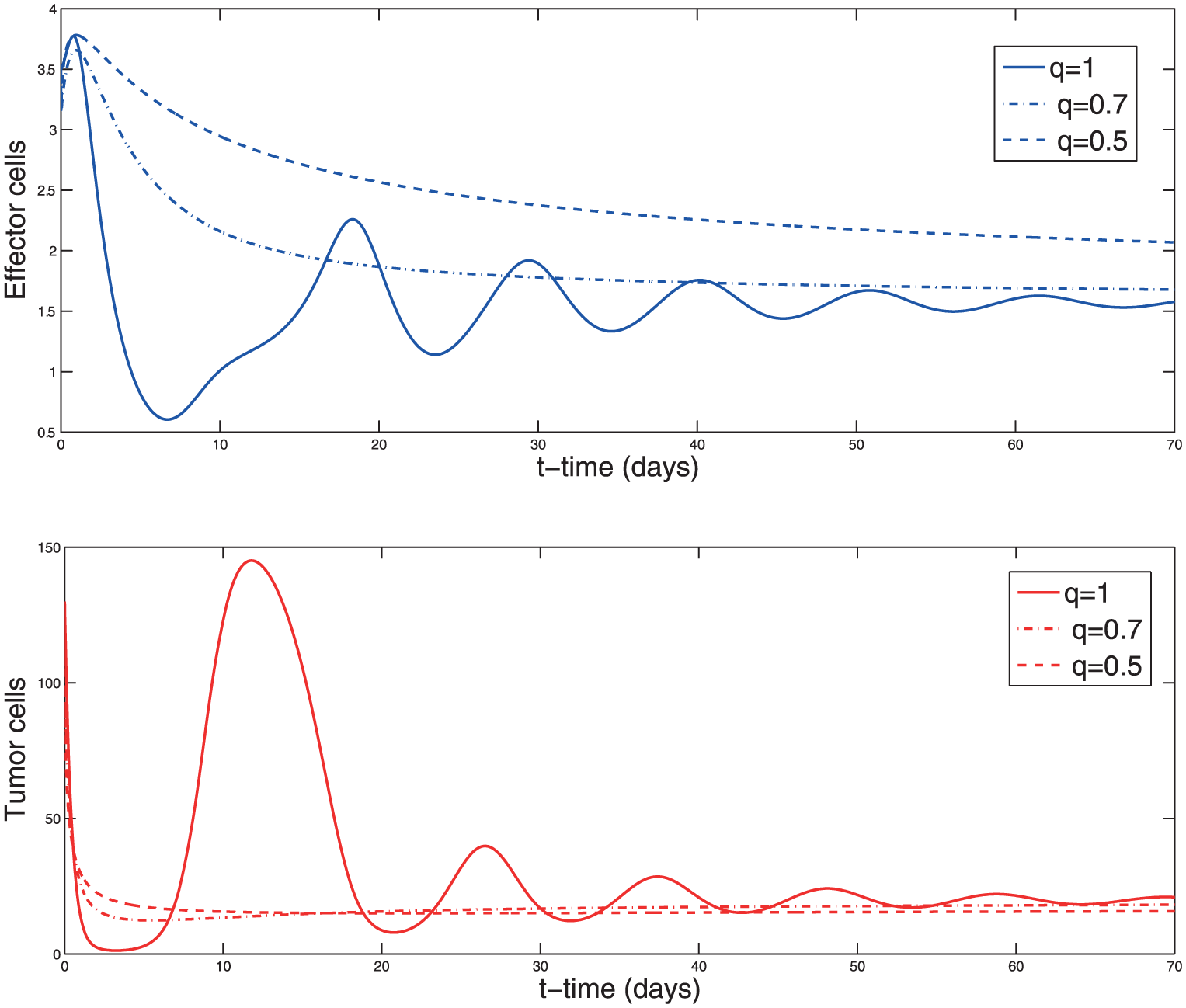
Solution of the system (2) with

Solution of the system (2) with
Table 5 shows that when
Discussion and conclusion
In this article, we have studied an immunogenic tumor model of fractional order. Dynamics of the model depends on the stability of the equilibrium points:
Tumor-free equilibrium:
Tumor-infection equilibrium:
Medically, if the trajectories of the fractional order model will converge to tumor-free equilibrium, then tumor will eliminate in the long run (hence the patient is ultimately cured) and if trajectories will converge to the tumor-infection equilibrium point, then tumor will persist in the body. Mathematically, in order to find cancer treatment, we have to find the analytical conditions under which trajectories of the system will converge to tumor-free equilibrium. This analytical condition is given in Theorem 3.2, that is, the tumor-free equilibrium point of the system is locally asymptotically stable if

This relation depends on three parameters:
Precisely, variation of these three parameters plays a key role in cancer treatment and doctors have to adjust these parameters according to above relation in order to eliminate tumor. The parameters
Numerical simulations are presented for various values of fractional order q indicating that antitumor reactivity might be due to cytotoxic effector cells. Fractional calculus may improve the understanding of biological processes because the fractional differential equations can capture past evolution of the function. The memory kernel in the solution of fractional differential equations decays as a power law whereas in ordinary differential equations the memory kernel turns into the Dirac Delta function. Mathematical models can help the physicians to choose optimal dosage. Cancer patients have different tendencies during tumor growth which is difficult to capture by integer order derivative. Fractional derivative can be varied to best fit the real data according to progression of different tumors. Thus, more reliable model can be obtained by choosing the relevant fractional index according to real data which can help clinician to suggest treatment to each individual patient.
Footnotes
Acknowledgements
The research is supported by a grant from the “Research Center of the Center for Female Scientific and Medical Colleges”, Deanship of Scientific Research, King Saud University. The authors are also thankful to visiting professor program at King Saud University for support.
Academic Editor: José Tenreiro Machado
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.