Abstract
The chemical reactions of titanium in air include both TiO2 and TiN. The phenomena are quite complex, and the reaction mechanisms are still unclear. However, as titanium is in widespread use all over the world, it is important that the reaction mechanisms be thoroughly investigated. Therefore, in this study, the continuous variations in the electrical contact resistance and the friction coefficient are simultaneously measured to monitor the dynamics of self-mated titanium pairs in an air, O2 or N2 gas flow. When a large amount of TiO2 formed on the surface of the plate for Ti/Ti, the results showed the tribo-physical phenomena of higher electrical contact resistance, friction coefficient almost doubled and severe wear. When a large amount of TiN formed on the surface of the plate for Ti/Ti, the results showed the tribo-physical phenomena of lower electrical contact resistance, friction coefficient almost halved and mild wear with some lubrication. Therefore, variations in the electrical contact resistance can be applied to determine whether the chemical reaction on the surface was TiO2 or Ti with TiN. The unsymmetrical frictional models and wear mechanisms of the self-mated titanium pairs in the air, O2 or N2 gas flow were proposed.
Introduction
Titanium and its chemical reactions are widely applied in industry. However, the equilibrium phase diagrams of titanium and TiO2 are quite complex. 1 Moreover, experimental results showed that the atmosphere has a strong influence on the structural evolution of titanium.2,3 Hence, it is important for engineers to clarify the dynamics of titanium sliding against titanium in different atmospheres. The study of N2 lubrication4,5 has revealed that nitrogen shows a lower friction coefficient for ceramic coatings. With reference to the above statements, the following topics should be considered:
The tribo-physical phenomena and the tribo-chemical reactions on the contact surfaces of N2 lubrication, and why the effects of lubrication can be observed in a N2 atmosphere for the experimental materials.
The effect of lubrication for other important tribological materials, such as titanium in a N2 atmosphere, and whether it is possible to show similar lubrication effects or other results of interest.
The tribo-physical phenomena and the tribo-chemical reactions on the contact surfaces of self-mated titanium pairs in a O2 atmosphere.
As mentioned above, the tribological mechanisms of titanium sliding against titanium are very complex in different atmospheres. A review of the studies of tribology6–14 shows the friction coefficient response to be the main dynamic parameter for the friction tests. However, the friction coefficient can become less distinct over time, making it necessary to introduce an improved method for monitoring the dynamics of titanium sliding against titanium. The tribo-electrification mechanisms15,16 have been experimentally studied by the authors, and the knowledge has been successfully applied to related studies. However, this method is unsuitable for direct use when measuring the contact potential difference between semiconductors, such as TiO2. Therefore, a method which used the continuous variations of the electrical contact resistance (ECR) and the friction coefficient for monitoring the dynamics of self-mated titanium pairs in different atmospheres was investigated in the previous study. 17
The interface of the pin remains in continuous contact with the surface of the plate during the unsymmetrical friction process, making it difficult to produce chemical reactions at the interface of the pin, even though its temperature is much higher than the surface of the plate. In other words, the interface of the pin remains as almost pure titanium during the unsymmetrical friction process. On the other hand, the interface of the plate comes into intermittent contact with the surface of the pin during the unsymmetrical friction process. Thus, it is always interacting with the atmosphere, producing chemical reactions on the surface of the plate, even though its temperature is lower than the surface of the pin. The chemical reaction always plays an important role in cases of mild wear and of severe wear; 18 in order to simplify the chemical reactions and wear mechanisms, it is necessary to avoid increases in the surface temperature. The main parameter influencing the rise in surface temperature is the sliding speed.19-21 Hence, the reciprocating speeds were set at lower than 600 cpm (cycles per minutes) in this study in order to investigate in depth the effects of oxygen and nitrogen gases on the dynamics between the interfaces of self-mated titanium pairs.
Experimental equipment and procedures
Experimental equipment
A reciprocating friction tester with the measuring system, as shown in Figure 1, was utilized. A crank-slider mechanism was applied to drive the pin specimen. The length of the crank was adjusted to get a suitable stroke, which was set as 6 mm in this study. The plate specimen was placed on a stationary rest, securely connected to a load cell, in order to measure the friction coefficient. The normal load was relative to the pin through the plate and disposed along the level rule. This setup enabled the accurate measurement of the friction coefficient between the pin and plate specimens. The voltage signal from the load cell is proportional to the force which affected on it. Based on this relationship, it is therefore easy to calculate the friction force which affected on the load cell. When the pin is removed from the load cell, the polarity of the voltage signal from the load cell becomes positive. On the other hand, it shows negative. Hence, the variations of friction force appear positive and negative alternately during the reciprocating friction process. Moreover, the friction force divided by the normal load equals the friction coefficient. In addition, the pin and the plate were electrically isolated in order to measure the ECR. The voltage signals of the ECR and the friction coefficient were simultaneously recorded by the data acquisition system and then fed to a personal computer for data analysis. The specification for the ECR measuring system was 0–10 kΩ. A soft spring with an oil damper was established as part of the loading system to eliminate impact. The experimental gases were directed to the interfaces from the tanks and measured by the gas flow metre, as shown in Figure 1.

Reciprocating friction tester with the measuring system.
Experimental specimens
The specimens were made of pure titanium. The size and shape of the specimens are shown in Figure 2. To reduce the effect of the attack angle, the corner radius of the pin was machined to 0.25 mm. The plates were polished by different grades of emery papers to an average surface roughness, Ra, in the range of 0.08–0.13 µm.

Size and shape of the specimens.
Experimental procedures
The specimens were cleaned with acetone in an ultrasonic cleaner before each test and then securely locked in position. When the crank rotated clockwise at the set speed, the normal load was carefully applied to the interface of the specimens. After the friction test, the wear loss was measured through the precision balance, and the wear surface of the plates was examined by means of scanning electron microscopy (SEM). In this study, the fixed normal load was 50 N. The speed was set from 200 to 600 cpm. The O2 or N2 flow rate was fixed at 150 NL/min (NL (normal litre)). The experiments were conducted under the condition of dry friction. The room temperature remained at 25°C ± 2°C, and the relative humidity at 65% ± 5%.
Experimental results and discussion
Responses of ECR and friction coefficient
The responses of the ECR and the friction coefficient with a sliding distance produced by the self-mated titanium pairs (i.e. Ti/Ti) in an air, O2 or N2 gas flow were synchronously measured to investigate the relationships of the chemical reactions and tribological properties.
Titanium sliding against titanium in the air
The typical variations of the ECR and the friction coefficient under 200 cpm for Ti/Ti in the air are shown in Figure 3. The ECR was in the range 0–7 kΩ during the entire friction test. Moreover, the ECR was only about 0–1 kΩ at initial contact. Since TiO2 is a semiconductor, its ECR was much higher than that of either pure titanium or TiN. Hence, an ECR higher than 1 kΩ, as shown in Figure 3, was due to the fact that TiO2 was immediately produced on the surface of the plate. On the other hand, an ECR, in the range 0–1 kΩ at initial contact, was because only a small amount of TiO2 was immediately produced on the surface of the plate. Most titanium on the surface of the plate remained pure during the initial friction process.

Responses of ECR and friction coefficient under the reciprocating speed of 200 cpm for Ti/Ti in the air.
In light of this, the following two aspects need to be considered:
The insufficient time for chemical reactions to occur between the interfaces;
The temperature increase caused by residual heat 5 being too low for the chemical reactions to occur on the surface of the plate.
At a sliding distance of 0.3 m, the corresponding friction coefficient was about 0.7–1.5, which indicated severe wear under the experimental conditions. Moreover, the corresponding ECR approached 7 kΩ, which indicated that the temperature increase caused by the residual heat was quite high, and that serious chemical reactions occurred to form more TiO2.
Titanium sliding against titanium in O2 gas flow
The typical variations in the ECR and the friction coefficient under 200 cpm for Ti/Ti in the O2 gas flow are shown in Figure 4. As compared to the air flow cases, shown in Figure 3, the ECR was in the range of 0–10 kΩ during the entire friction test. Moreover, the ECR significantly increased at a sliding distance of 0.5 m, evidence that frictional heat was carried by the O2 gas flow. Hence, the temperature rise caused by the residual heat in delayed time, and a greater sliding distance was needed to accumulate the residual heat required to form TiO2 on the surface of the plate. The homologous friction coefficient was about 0.7–1.5, indicating that the wear mechanism became severe under the experimental conditions. On the whole, the trends of the ECR and the friction coefficient for Ti/Ti in an O2 gas flow were similar to that of Ti/Ti in air. However, the ECR was slightly higher for Ti/Ti under 200 cpm, indicating that the temperature increase caused by the residual heat was relatively smaller for Ti/Ti under 200 cpm. Hence, only a small amount of TiO2 formed on the surface of the plate.

Responses of ECR and friction coefficient under the reciprocating speed of 200 cpm for Ti/Ti in the O2 flow rate of 150 NL/min.
The typical variations in the ECR and the friction coefficient under 600 cpm for Ti/Ti in an O2 gas flow are shown in Figure 5. It was observed that the ECR significantly increased after a sliding distance of 0.3 m, evidence that a great quantity of TiO2 formed on the surface of the plate. Moreover, the homologous friction coefficient substantially increased to 1.5–3.5, indicating that extremely severe wear occurred under the experimental conditions. It is well known that the temperature increases are linearly proportional to the reciprocating speeds.18,19 Hence, the temperature increase caused by the residual heat was relatively higher for Ti/Ti under 600 cpm. As a result, a severe chemical reaction occurred on the surface of the plate, and more TiO2 was formed due to both the higher temperature and the O2 gas flow.

Responses of ECR and friction coefficient under the reciprocating speed of 600 cpm for Ti/Ti in the O2 flow rate of 150 NL/min.
It was clear that the ECR increased when the reciprocating speed for Ti/Ti under an O2 gas flow increased. Moreover, since the wear resistance of TiO2 was lower than that of Ti or TiN, the severe wear occurred and the homologous friction coefficient substantially increased.
It was apparent from the above results, shown in Figures 3–5, that a large amount of TiO2 formed on the surface of the plate because TiO2 is a semiconductor, and its ECR was high. The relationships between the higher friction coefficient and severe wear will be clarified by the representative SEM micrographs of the wear track of the plate.
Titanium sliding against titanium in N2 gas flow
The typical variations in the ECR and the friction coefficient under 200 cpm for Ti/Ti in an N2 gas flow are shown in Figure 6. As compared to the cases in air shown in Figure 3, it can be seen that the ECR showed a significant decrease to 0–2 kΩ during the entire friction test. This was because the Ti surfaces were isolated by the N2 gas flow, making it difficult for TiO2 to form on the surface of the plate. The chemical reaction was mild, and the homologous friction coefficient was only 0.3–0.4, indicating that there was only mild wear and low friction under the experimental conditions.

Responses of ECR and friction coefficient under the reciprocating speed of 200 cpm for Ti/Ti in the N2 flow rate of 150 NL/min.
The typical variations in the ECR and the friction coefficient under 600 cpm for Ti/Ti in an N2 gas flow are shown in Figure 7. As compared to that for the O2 gas flow, the ECR decreased from 6–10 kΩ to 0–1.8 kΩ. Most of the titanium on the surface of the plate remained pure, or some TiN formed on the surface under the high reciprocating speed in the N2 gas flow. Therefore, the following two aspects ought to be considered:
From the physical point of view, both Ti and TiN showed a low electrical resistance, which made it difficult to discern Ti or TiN only by the ECR.
From the mechanical point of view, since the hardness of TiN is obviously larger than that of Ti, the difference of low friction and wear resistance should appear when TiN formed on the surface.
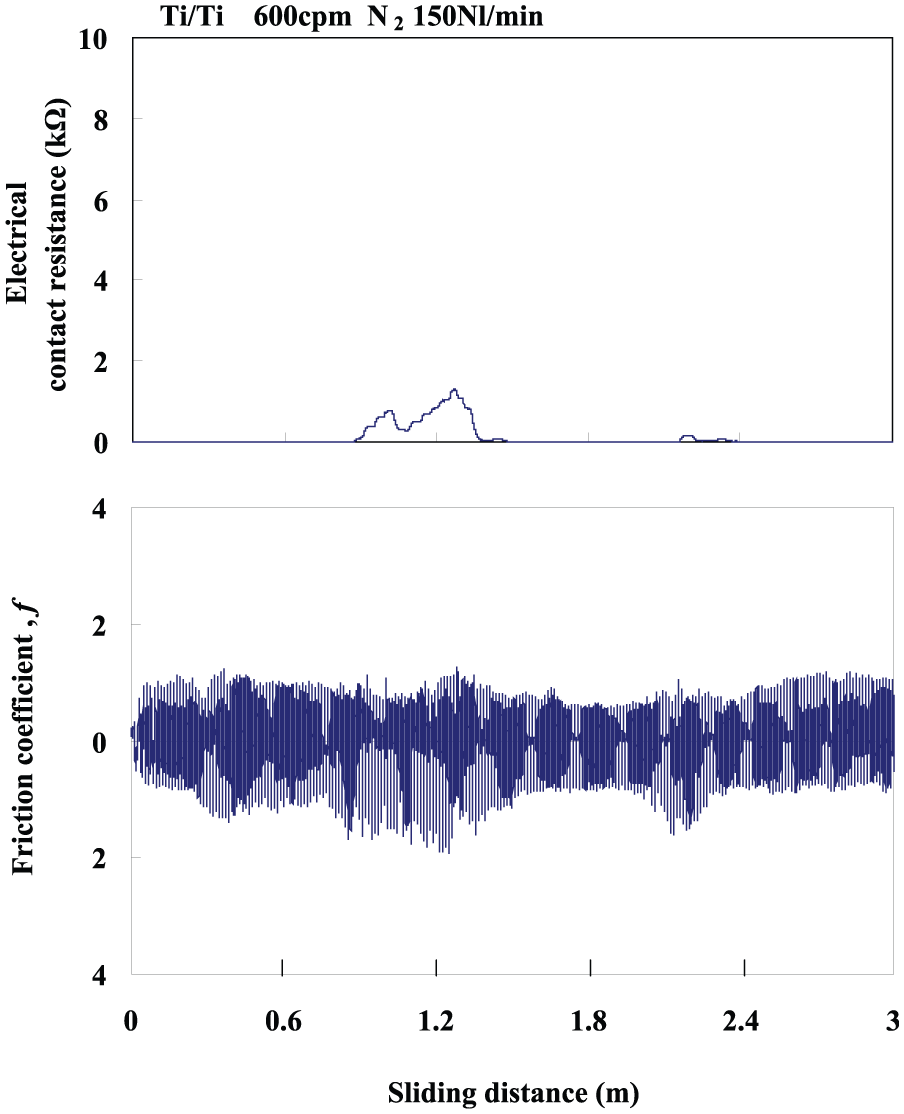
Responses of ECR and friction coefficient under the reciprocating speed of 600 cpm for Ti/Ti in the N2 flow rate of 150 NL/min.
The corresponding friction coefficient, shown in Figure 7, significantly decreased to 0.5–1.2. It was much smaller than for Ti/Ti in an O2 gas flow. The low friction level was in agreement with the results of the references.4,5 On the whole, both the ECR and the friction coefficient under 600 cpm for Ti/Ti in an N2 gas flow were lower than for the cases of Ti/Ti in an air or O2 gas flow. This indicated that the temperature increase caused by the residual heat was higher under the high reciprocating speed and more TiN was formed. Moreover, TiN on the surface significantly influenced the tribological properties, resulting in both a low ECR and a low friction coefficient.
Wear rate
Figure 8 shows the wear rates, defined as the wear loss per friction distance of 1 m, for the test specimens under 50 N and different sliding speeds. The positive values are defined as the weight loss at the worn surfaces, while the negative values indicate weight gain.

Effects of nitrogen and oxygen gases as well as the speeds on the wear rate of self-mated titanium pairs.
For the two cases (Ti/Ti in an air or O2 gas flow), the wear rates of the pin showed a negative value, while those of the plate showed a larger positive value. This indicated that the wear particles were caused primarily at the plate and were somewhat transferred to the pin. Moreover, the wear rate of the plate was higher for the case of Ti/Ti in the O2 gas flow. The following two reasons should be considered:
Mechanical analysis: this might be explained by the effect of the attack angle.
Chemical composition: since the interfaces of the pin remained in continuous contact with the surface of the plate during the unsymmetrical friction tests, it was difficult for the gas to enter. Hence, the surface of the pin remained almost pure titanium. However, the interface of the plate came into intermittent contact with the surface of the pin during the unsymmetrical friction process, and the ongoing interaction with the atmosphere produced the chemical reactions. As a result, a large amount of TiO2 formed on the surface of the plate for Ti/Ti in an air or O2 gas flow. Thus, the wear resistance of the plate decreased significantly.
For the case of Ti/Ti in the N2 flow, the wear rates of the test specimens under the lower reciprocating speed were similar to those of Ti/Ti in an air or O2 gas flow. Notably, the wear rates of the test specimens were higher than those of Ti/Ti in an air or O2 gas flow, with a large amount of material being transferred from the plate to the pin. This was because it was difficult for TiO2 to form on the surface of the plate for Ti/Ti in an N2 gas flow. Moreover, the temperature increase caused by the residual heat was lower under the lower reciprocating speeds; hence, it was also difficult for TiN to form on the surface. Since the material on the surface of the plate remained pure Ti, a large amount of mass transferred from the plate to the pin.
However, the results were different under the higher reciprocating speed. The wear rate of the pin showed a positive value, while that of the plate showed a larger negative value. This demonstrated that the wear particles came primarily from the pin and to some extent were transferred to the plate. This was because the temperature increase caused by the residual heat was higher under the higher reciprocating speed, making it easy for TiN to form on the surface of the plate. Moreover, the wear resistance of TiN on the plate was quite high. As a result, the wear particles came mainly from the pin and were partly transferred to the plate. The SEM micrographs were used to verify these results.
SEM images of the wear tracks
In order to investigate the effects of air, O2 and N2 on the tribological properties of Ti/Ti, the wear surfaces of the plates were observed by SEM, as shown in Figure 9.

(a) Representative SEM micrographs on the wear track for titanium sliding against titanium at a constant load of 50 N in the air. (b) Representative SEM micrographs on the wear track for titanium sliding against titanium at a constant load of 50 N in the O2 flow rate of 150 NL/min. (c) Representative SEM micrographs on the wear track for titanium sliding against titanium at a constant load of 50 N in the N2 flow rate of 150 NL/min.
As shown in Figure 9(a), the breadth of the wear track for Ti/Ti in air clearly increased as the reciprocating speed increased, at 500 µm under 200 cpm, several mm order under 600 cpm. The dynamic actual contact areas increased with the increase in the reciprocating speed for Ti/Ti in air. Moreover, there was no fractional projection, known as junction growth, evident with the wear tracks under 600 cpm. Therefore, the material transfer from strong adhesion remained insignificant for Ti/Ti in air under a higher reciprocating speed and higher temperature increase.
Figure 9(b) shows that the breadth of the wear track for Ti/Ti in the O2 gas flow was about 400 µm under 200 cpm. Moreover, the wear surface appeared smoother with few wear particles or junction growth, as only a minor amount of material transfer occurred with a lower temperature increase and a lower reciprocating speed. When the reciprocating speed increased to 600 cpm, the breadth of the wear track showed an obvious increase. Moreover, the wear surface appeared to be rougher, with large wear particles or junction growth, evidence of severe wear. The results of Figure 9(b) agreed with that of Figures 4 and 5.
As Figure 9(c) shows, the breadth of the wear track for Ti/Ti in the N2 gas flow was about 400 µm under 200 cpm. The deep wear track showed evidence of some material transfer from the plate to the pin. However, the wear surfaces appeared smoother with few wear particles or junction growth under 600 cpm. This indicated that the wear resistance increased with the increased speed. The results of Figure 9(c) agreed with that of Figures 6 and 7.
Effects of oxygen and nitrogen gases on tribological properties of self-mated titanium pairs
From the above results, the experiments demonstrated that the method of using continuous variations of ECR and friction coefficient has great potential for dynamic monitoring the tribo-chemical reactions and the tribo-physical phenomena of self-mated titanium pairs in different atmosphere. Moreover, the following clarifications could be made as to the effects of the chemical reactions on the friction and wear for Ti/Ti:
Electrical properties: since TiO2 is a semiconductor material, its electrical resistivity is quite high, whereas Ti and TiN are conductor materials, so their electric resistivity is low. Therefore, the region of the greatest ECR assumed to be the chemical reaction at the interface was TiO2. However, it remained unclear whether the chemical reaction at the interface was Ti or TiN due to the smaller ECR. Because of this, it was necessary to further consider the results of the corresponding friction coefficient and wear rate.
Mechanical properties: since TiN showed a lower friction and wear resistance than Ti, its friction coefficient would also be low. Hence, for the regions with a lower ECR and lower friction coefficient, it could be assumed that the chemical reaction at the interface resulted in TiN. Moreover, the results of the wear rate, in Figure 8, also showed that the wear resistance increased for the cases when TiN formed on the surface.
Therefore, the responses of ECR made it reasonable to assume that TiO2, or Ti with TiN, were produced between the interfaces. The corresponding friction coefficient could then be used to determine whether Ti or TiN existed at the interfaces, and the effects of TiO2, TiN or Ti on the friction and the wear resistance investigated. From the above results and analyses, the unsymmetrical frictional models and wear mechanisms for titanium sliding against titanium were proposed, as shown in Figure 10.

Unsymmetrical frictional models and wear mechanisms for titanium sliding against titanium: (a) in the air, (b) in the O2 flow and (c) in the N2 flow.
As shown in Figure 10(a), the surface of the plate came into intermittent contact with the surface of the pin during the unsymmetrical friction process. Hence, some TiO2 formed and the rest stayed as almost pure titanium. This mixture had poorer tribological properties than the pure Ti of the pin. The result was high friction (see Figure 3) and severe wear (see Figure 9(a)) for Ti/Ti in air.
As Figure 10(b) shows that there were two effects for Ti/Ti in the O2 gas flow: (1) tribo-chemical reactions – a large amount of TiO2 formed on the surface of the plate and (2) tribo-physical phenomena – TiO2 on the plate showed significant wear and tear from the pin sliding against it. The high friction level is evident in Figures 4 and 5. Severe wear can also be easily observed in the SEM micrographs shown in Figure 9(b).
Figure 10(c) shows the two effects found for Ti/Ti in the N2 gas flow: (1) tribo-chemical reactions – TiN film formed on the surface of the plate and (2) tribo-physical phenomena – the high hardness of TiN makes it significantly wear resistant. The very low friction level is evident in Figures 6 and 7. The mild wear can also be observed in the SEM micrographs shown in Figure 9(c). These results were in agreement with the study results of N2 lubrication.4,17
Conclusion
Continuous variations in the ECR and the friction coefficient were used to monitor the dynamics of the self-mated titanium pairs in an oxygen or nitrogen gas flow under different reciprocating speeds. From the experimental results, the following conclusions were drawn:
Variations in the ECR were successfully applied to determine whether the chemical reaction on the surface was TiO2 or Ti with TiN.
A large amount of TiO2 formed on the surface of the plate for Ti/Ti in the O2 gas flow due to the tribo-chemical effects in comparison with that in the air. The results showed the tribo-physical phenomena of higher ECR, friction coefficient almost doubled and severe wear.
A large amount of TiN formed on the surface of the plate for Ti/Ti in the N2 gas flow under the higher reciprocating speed due to the tribo-chemical effects in comparison with that in the air. The results showed the tribo-physical phenomena of lower ECR, friction coefficient almost halved and mild wear with some lubrication.
The unsymmetrical frictional models and wear mechanisms of the self-mated titanium pairs in the air, O2 or N2 gas flow were proposed. The tribo-chemical reactions and the tribo-physical phenomena at the interface for Ti/Ti in different atmospheres can be appeared from these models.
Footnotes
Academic Editor: Stephen D Prior
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to express their appreciation to the Ministry of Science and Technology in Taiwan, R. O. C. for their financial support under grants MOST 105-2622-E-150-001-CC2 and MOST 104-2221-E-168-019.
