Abstract
Base compositions of the foam glasses in this study include commercial silica, alumina, boric acid, sodium carbonate, potassium carbonate, carbon powder, and disodium hydrogen phosphate. The foam glasses with partial Na2O replaced by Li2O were prepared and investigated as a possible artificial floating island carrier of water purifying unit. The effects of Li2O dosage on the density, mechanical strengths, and microstructures were studied. Foam glass with 15 mol% Na2O replaced by Li2O displayed a good compressive strength, 3.582 MPa, while the bulk density was relatively low, about 0.398 g/cm3. The chemical stability of samples was evaluated with inductively coupled plasma atomic emission spectrometer, which showed low ion dissolution content in different pH dissolutions. The experimental results showed that the foam glass sample with 15 mol% Na2O replaced by Li2O exhibited excellent comprehensive properties, and the reason may contribute to the mixed alkali effect that proper amount of Li+ could impede the migration of large-radius ions like Na+ and K+ and thus could improve the glass network structure.
Introduction
Water eutrophication is an increasing serious social challenge since natural water source is gradually exhausted day by day and some of the human activities make the remaining water less available. 1 Finding an effective solution to purifying water is quite urgent. At present, we hope planting of hydrophytes on artificial floating island carrier will be a useful water purifying pattern. According to the history of development, artificial floating island carriers could be sorted into three types: carriers made from plant root and stem, organic polymer carriers, and inorganic material carriers. 2 However, plant root and stem carriers are easily subjected to excessive multiplication and death of plants. As a result, its range of application is limited. The widely used organic polymer carriers have an obvious advantage over mature preparation techniques, low cost, and easy reprocessing. Nevertheless, the polymers have poor aging resistance and could bring about “White Pollution” in the natural environment, whereas inorganic material floating carriers have no disadvantages in these fields. Foam glass is a lightweight material with stable mechanical and chemical stabilities. In addition, its high specific area is conducive to the adhesion, development, and biofilm growth of aquatic microbes, which would be of great help to the water purification process. All these properties make foam glass an ideal option for artificial floating island carrier.
Foam glass has already received considerable study in fields such as heat insulation and soundproofing. Chen et al., 3 Guo et al., 4 and Fernandes et al. 5 have reported preparing porous glass by fly ash, waste glass, and sheet glass cullet, respectively. However, seldom study of foam glass as artificial floating island carrier is reported in previous articles. One of the reasons may be that it is difficult to control the physical and chemical properties of different batches while using industrial wastes as raw materials. Besides, the uncertain content of heavy metal in industrial waste makes it hard to control the possible dissolution of ion types (especially heavy metal ions) after foam glass products are immersed in water bodies. Therefore, it is necessary to prepare foam glass which fits well with the application environment of outdoor water bodies.
This article concentrates on the preparation and characterization of foam glass used as environmental artificial floating island carriers in which no heavy metal is incorporated. The Li2O dosage effect on the mechanical and chemical properties of the foam glass, as well as the morphology of the samples, is observed and analyzed.
Experimental procedures
Materials
The mass percentages of compositions of foam glass in this study are given in Table 1, including commercial silica, alumina, boric acid, sodium carbonate, potassium carbonate, carbon powder, and disodium hydrogen phosphate. Carbon powder and disodium hydrogen phosphate were chosen as foaming agent and foaming stabilizer, respectively. Carbon powder was carbonized at a certain temperature, with generation of gas (CO2) which was finally surrounded by softened gas phase and formation of pores in the sintered samples. The foam glass products using carbon powder as foaming agent possessed small closed pores with homogeneous wall thickness and low bulk density. Besides, carbon powder could also act as stabilizer when carbon powder particles gather on the interface of gas–solid; then, the foams were more stable with the interface energy decreasing. Disodium hydrogen phosphate was added to improve the pore distribution of foam glass by increasing the melt viscosity at high temperatures, hindering the cell wall thinning process, and finally decreasing the ratio of foam crack or coalescence because of the fact that its pyrolysis product P2O5 could turn into [PO4] in glass network structure and act as a network former.
Material composition of the foam glass prepared in this study.

Sample preparation
At first, the compounds were wet-milled in a ball mill for 2–3 h (balls:deionized water:powders = 2:1.5:1). Then, the uniform mixture was dried overnight for further milling together for about 2–3 h. Finally, the powders were sieved through 150-mesh sieve. The green samples were filled in a graphite crucible, outside of which a corundum crucible was used to avoid its oxidation, and sintered at a heating rate of 5 °C/min, holding at 750 °C for 30 min, and then annealed at 600 °C for 20–30 min. Finally, the samples were naturally cooled down to room temperature.
Characterization techniques
The foaming temperature and soften temperature were determined by the NETZSCH STA449C differential thermal analyzer (Germany) using a heating rate of 5 °C/min.
The bulk density of foam glass samples with regular shape was calculated with mass and volume. The mean compressive strength of samples was measured with a universal tester with a span of 20 mm at a cross-head speed of 10 mm/min. Each result was given as the mean value of five measurements. The pore morphology of the samples was studied with a field emission scanning electron microscope (FESEM; JEOL JSM-6700F, Japan).
The chemical stability test was performed by recording the weight change of samples (10 mm × 10 mm × 10 mm) which were immersed in water bodies with different pH values (pH = 3, pH = 7) at regular intervals and detecting the dissolution of ions in solutions through the inductively coupled plasma atomic emission spectrometer (ICP-AES; VISTA-MPX EL02115765, Japan). The solution volume was 1000 mL, and each result was given as the mean value of five measurements.
Results and discussion
Bulk density, compressive strength and microstructure
Figure 1 illustrates the samples’ density and compressive changes with different Li2O contents. It is seen that with the Li2O content increasing, the bulk density increases at first, from 0.341 to 0.398 g/cm3, and then decreases, to 0.380 g/cm3. However, with the Li2O content continuously increasing, large amounts of Li+ existing in the structure add to the gathering effect, leading to phase separation and finally an inhomogeneous glass structure. Besides, Li2O content changes the melt viscosity, as well as the fluidity, affecting the high-temperature reacting process.6,7 Consequently, the sintered foam glass density and the porous structure are influenced.

Effect of Li2O content on the bulk density and compressive strength of foam glass sintered at 750 °C.
The compressive strength values of samples demonstrate similar features. This may be due to the function relationship between compressive strength and relative density. The classical model for the compressive strength of cellular structural materials provided by Gibson and Ashby 8 predicts the dependence of compressive strength (σcr) on the porosity (p), that is, the ratio between the bulk density and the true density in a combination of exponential and linear terms, as follows

where C is a dimensionless calibration constant (0.2). The bending strength σfs of the reference borosilicate glass (Pyrex 7740) is 68 MPa, a typical value for the Pyrex glass employed as commercial material. The quantity (1 − Φ) expresses the fraction of solid positioned in cell faces; if the foam is open-celled, the pores are fully interconnected with material only on the cell edges so that Φ = 1 (1 − Φ = 0). On the contrary, for closed cell foam, Φ is lower with the solid phase constituting mostly cell walls and thus enhancing the linear term. As a result, foam glass compressive strength value mostly depends on its cellular microstructure.
Figure 2 is the scanning electron microscope (SEM) microstructure graphs of samples with different Li2O contents. We can find that a lot of pores with different diameters (0.1–1 mm) exist in all the samples. Figure 2(a) and (b) exhibits pore distributions with many pores on the cell wall. And sample no. 2 possesses thinner cell walls and a more even pore size distribution than sample no. 1. Thus, the compressive strength of sample no. 2 (2.422 MPa) is slightly higher than that of no. 1 (2.258 MPa). Sample no. 3 features with nonuniform cell size distribution and through holes. The average pore size decreases, although some large pores still exist in Figure 2(c). And in Figure 2(d), the pore size is much more balanced and the wall thickness is the largest of the five, leading to the highest density (0.398 g/cm3) and compressive strength (3.582 MPa) despite some tiny pores on the cell wall.

SEM micrographs of sintered samples with different Li2O contents: (a) No. 1, (b) No. 2, (c) No. 3, (d) No. 4, and (e) No. 5.
The effect of enhancing melting point by Li2O is the reason. After bodies begin to soften under the gas expansion pressure, the foaming process occurs, which brings large amounts of pores in samples. The viscous flow sintering reduces as a result of the generation of a large amount of liquid phase with increasing viscosity, which prevents small pores from completely developing into large pores or disappearing.6,9,10 In the end, a dense cellular structure is formed. This may also explain why sample no. 4 owns the highest bulk density and compressive strength. When the Li2O content keeps increasing, an excess of Li+ in network structure enlarges the gathering effect, which gives rise to phase separation in foam glass. Finally, the microstructure homogeneity of foam glass is affected. As is shown in Figure 2(e), the pore shape and size distribution is almost disorderly. The number of pores is less than that of sample no. 4, and some pores are not regularly spherical, which may cause an uneven stress distribution under compressive load (the fracture surface after cracking is rugged). All these lead to a decrease in compressive strength value.
Chemical stability discussion
Generally speaking, the chemical stabilities of glass materials mainly consist of the properties of water resistance and acid resistance. Table 2 shows the mass changes of foam glass sample nos 1–5 in this study, which were totally immersed in water solution with different pH values: sulfuric acid solution (pH = 3) and deionized water (pH = 7), respectively.
Effect of Li2O on some properties of foam glass samples.
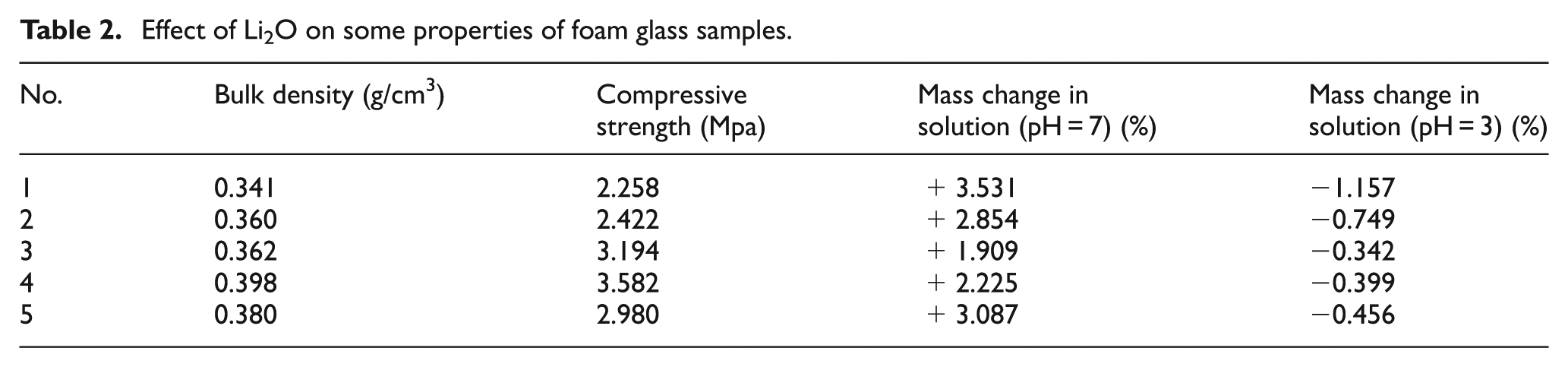
It can be seen from Table 2 that samples in these two solutions have mass variations after a certain period of time, and such mass changes vary with the different Li2O contents. The more Li2O is added, the steadier is the sample mass variation. The mixed alkali effect could be applied to explain this phenomenon since the chemical stabilities of glass mainly depend on the migration of alkali ions. Proper amount of Li+ fills in the blanks of glass network structure and impedes the migration of large-radius ions such as Na+ and K+, thus lessening the glass erosion by water solution at a large extent and ultimately contributing to the enhancement of chemical stabilities. However, excess Li+ existing in the structure adds to the gathering effect, causing the decline in chemical stabilities. 11
For samples immersed in deionized water (pH = 7), their masses rise as time goes on, which follows a parabolic trend. The peak appears after 30 days of immersion in water. And finally, a flat trend is obtained. Sample nos 3 and 4 own the best water resistance because of the mixed alkali effect.
The glass erosion in water consists of two parts: (1) the exchange of H+ in water with Na+ in glass and (2) direct reaction of H2O with Si-O skeleton. The reaction product Si(OH)4 is a kind of polar molecule which could polarize and absorb the water molecule nearby. Finally, it turns into silicic acid gel, Si(OH)4·nH2O, which is slightly dissolved in water and mostly adhered to the surface of glass, forming a thin film with advantages in water resistance and acid resistance. Moreover, this kind of thin film has a strong adsorption of positive ions like Na+ and K+, shaping cationic colloidal particles. 12 This could explain why the glass mass increases at first and finally is steady.
As is shown in Table 2, the mass change is negative at first, then positive, and finally stays at a low change rate (about 0.10%). Sample nos 3 and 4 own the best acid resistance.
Glass erosion by acid is realized through media—water. Therefore, the erosion by concentrated acid is less than that of diluted acid. One product of the erosion reaction is the hydroxide of metal which is neutralized by acid. The neutralization here has two opposite functions: 12 (1) accelerates the ion exchange reaction between glass and water solution, adding to the weight loss of glass; (2) lowers the pH of solution and decreases the solubility of Si(OH)4, reducing weight loss. For high alkali metal content glass, process (1) is primary; while for high SiO2 content glass, process (2) is the dominating one. 13 Therefore, the mass of samples immersed in sulfuric acid solution first decreases due to the active ion exchange reaction on the glass surface and the generated Si(OH)4 mostly dissolves in solution; then the mass increases as more Si(OH)4 is accumulated and the ion exchange reaction on the glass surface is weakened. In the end, their mass changes level off, meaning that a dynamic balance is acquired between weight loss and gain.
ICP-AES
In order to understand the ion dissolution content quantificationally, sample no. 4 is chosen as an example and an ICP-AES is used to detect the ion concentration of the following six elements: Si, Al, B, Li, Na, K. The results are shown in Figure 3, and the ion dissolution content is confined in a low level. All of them are less than 3 mg/L. The ion dissolution content in deionized water (most of it is below 1 mg/L, except for Na) is much lower than that of sulfuric acid solution. In view of the outdoor application of foam glass carrier, the ion dissolution concentration in those large area waters would be much lower. As a result, the aim to protect environment and purify water is expected to be achieved.

The ion concentration of sample no. 4 after 90 days of immersion in solutions with pH = 3 and pH = 7.
Conclusion
The conclusions can be drawn from the results discussed above. Li+-introduced foam glasses are good to act as the artificial floating island carrier, which have suitable bulk density (0.341–0.398 g/cm3), proper compressive strength (2.258–3.582 MPa), and good chemical stabilities (ion dissolution content is confined to a small range, less than 3 mg/L). Foam glasses with different Li2O contents are prepared below 750 °C using carbon powder as foaming agent. And samples with 15 mol% Na2O replaced by Li2O have better properties than other samples.
Footnotes
Academic Editor: Guoqiang Li
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by Tianjin Municipal Science and Technology Commission (No. 11ZCKFSF01200).
