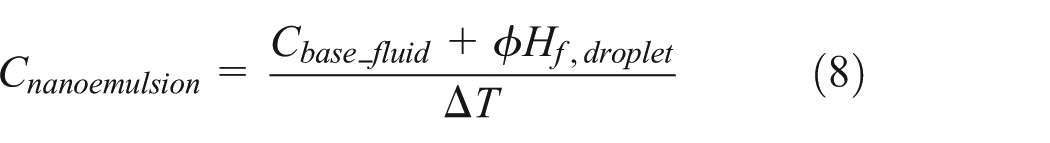Abstract
In this study, the nanostructures and thermophysical properties (thermal conductivity, viscosity, and specific heat) of one new type of nanostructured heat transfer fluid, water/polyalphaolefin nanoemulsion fluid, are investigated. The water/polyalphaolefin nanoemulsion fluids are thermodynamically stable containing dispersed water nanodroplets formed by self-assembly. It has been found that the nanostructure inside nanoemulsion fluids may affect their thermophysical properties, especially the phase change heat transfer characteristics. The small-angle neutron scattering technique has been used to help identify the nanostructure inside the water/polyalphaolefin nanoemulsion fluids. By using the 3-region Guinier–Porod model, the fitting curve shows that there is a nonlinear variation of the nanodroplets’ size and shape with water’s concentration, which also coincides with the trend of its viscosity and specific heat. On the other hand, the thermal conductivity increases linearly with higher volume fraction of water which, however, appears to be insensitive to the nanostructure change. While the water nanodroplets inside can increase the thermal conductivity of the nanoemulsion fluid by 16%, its effective specific heat can be boosted up to 90% when the water nanodroplets undergo liquid–solid phase change.
Introduction
Advanced thermal management is a complex and challenging problem widely faced in industrial and military applications.1–12 Conventional coolants, lubricants, and other heat transfer fluids used in today’s thermal management systems typically have relatively poor heat transfer properties. Hence, utilizing the phase change process to increase the heat transfer properties of conventional fluids is another promising direction.13–16 Recently, the author has proposed a new “nanoemulsion heat transfer fluid” system in which one liquid of lower boiling point is dispersed into another immiscible liquid and formed self-assembled nanodroplets which help improving the fluid thermal properties especially its phase change characteristics.7,12,17–32 The nanoemulsion heat transfer fluids belong to the family of self-assembled colloid system and are thermodynamically stable.
The customization capability of the thermophysical properties—through forming different nanodroplets inside—makes them attractive candidates for emerging heat transfer applications in various industries. For example, the added nanodroplets of higher thermal conductivity can improve the thermal conductivity of the nanoemulsion fluids and transfer heat more effectively.
In this article, the thermophysical characteristics of water/polyalphaolefin (PAO) nanoemulsion fluids—thermal conductivity, viscosity, and specific heat—are investigated experimentally, along with a structure characterization of nanostructures formed inside using small-angle neutron scattering (SANS).
Measurement methods
Structure characterization
SANS measurements were carried out for the characterization of the nanostructures. One great advantage of using SANS is that it can be applied to “concentrated” colloidal suspensions (e.g. >1% volume fraction) for the in situ determination of the size of droplets in the nanoemulsion fluids and is better suited than conventional dynamic light scattering.33–39 In the SANS experiments, samples are prepared using deuterated water (D2O) to achieve the needed contrast between the droplets and the solvent. SANS measurements were conducted on the NG-3 (30 m) beamline at the NIST Center for Neutron Research (NCNR) in Gaithersburg, MD. Samples are loaded into 2-mm quartz cells and kept at room temperature. Figure 1 shows the SANS data for water volumetric concentration covering 1.8%–3% volume fraction. The scattering intensity I versus the scattering vector
where λ is the wavelength of the incident neutrons and θ is the scattering angle. The approximation q = 2πθ/λ is used for SANS (due to the small angle θ).

Small-angle neutron scattering curves for water/PAO nanoemulsion fluids: water volume concentration from 1.8% to 10.3% volume fraction. Three different colors represent three different types of scattering curves. The statistical error bars are smaller than the plotting symbols.
Thermal conductivity measurement
where p is the applied electric power,
Viscosity measurement
Viscosity is defined by considering the force applied in the course of flow of a fluid, and its dissipation per unit area and velocity gradient is the viscosity
where F is the stress force, u is the velocity, y is the separation, A is the area, and
The viscosity of a nanoemulsion fluid depends largely on its microstructure, that is, the type of aggregates that are present, on their interactions, and on the concentration of the system.20,22,24,41,47–49 Therefore, the viscosity can be used to monitor the structural changes in the nanoemulsion system, and on the other hand, the structural change can also be reflected in the viscosity measured.
Specific heat measurement
A differential scanning calorimeter (DSC) was used to measure a nanoemulsion fluid’s specific heat. Usually DSC measurement can be made in two ways: by measuring the electrical power provided to heaters below the pans necessary to maintain the two pans at the same temperature (power compensation) and by measuring the heat flow (differential temperature) as a function of sample temperature (heat flux). The DSC ultimately outputs the differential heat flow (heat/time) between the measured material and the empty reference pan.
The heat capacity may be determined by taking the ratio of heat flow to heating rate. Thus
where
The specific heats of the pure PAO and water/PAO nanoemulsion fluids are measured using a heat-flux-type DSC (TA-Q100). This is accomplished by comparing the heat flux into a pan containing the sample with the heat flux into an empty pan. In this method, both the measured sample and reference sample are maintained at nearly the same temperature by adjusting heat input to them. The difference in the amount of heat supplied to the sample and the reference is recorded as a function of temperature (or time). In the curve of heat flux versus temperature or versus time, positive or negative peaks correspond to exothermic or endothermic reactions in the sample, respectively. Enthalpies of phase transitions can be calculated by integrating the peak corresponding to a given transition,
Experimental results and discussion
Structural change characterization
Figure 1 shows the SANS data for water/PAO nanoemulsion fluids with water volumetric concentration covering 1.8%–10.3% volume fraction.
It is clear from the SANS data that the neutron scattering data gradually change into three different types of curves with increasing water loading. Based on the shape of the scattering curves, they can be classified into three ranges: the 1.8%–4.5% volume fraction ones with a smooth and gradually increasing scattering intensity for low-q range (less than 0.1 Å); the 5.3%–7.8% volume fraction ones with a sharper increase of q for the high-q range (larger than 0.1 Å) and a flatter intensity curve for low-q (less than 0.03 Å); and for even higher concentrations like 8.6% and 10.3% volume fractions ones tested here, the “hump” for high q is even more obvious and the intensity for low q increases more sharply which gives very obvious three sections of scattering curves. For colloidal systems which do not experience inner structure change, the SANS data can be fitted using one model. However, most colloidal systems including nanoemulsion fluids can undergo inner structure change with change in concentration, temperature, salinity, and so on. Especially for the water/PAO nanoemulsion fluids, the simple correlation length model cannot fit all the SANS curves well for both low and high-q regions as shown in Figure 2.

Small-angle neutron scattering curves for water/PAO nanoemulsion fluids (solid symbols) and hard-sphere model curve fittings (colored lines).
The hard-sphere model fits well for low water concentrations (i.e. 1.8%–4.5% volume fraction), and nanodroplet radii are found to be 13.2, 25.6, and 96 Å for water loading 1.8%, 3.6%, and 4.5% volume fractions, respectively. For higher water concentration (i.e. 7.8%–10.3% volume fraction), the hard-sphere model does not fit well especially for scattering variable q less than 0.1 Å region, which suggests that those nanodroplets are not simply spherical.
To describe the structure variation inside the water/PAO nanoemulsion fluids more accurately, another fitting model is used to describe the structure change. The 3-region Guinier–Porod empirical model is used to determine the nanodroplet geometry by fitting the SANS data especially for scattering q less than 0.1 Å region, and the fitting curves are shown in Figure 3.35,36,38,50

Small-angle neutron scattering curves for water in PAO nanoemulsion fluids (solid symbols) and 3-region Guinier–Porod model curve fittings (colored lines).
Generally, the scattering intensity is given by the two contributions in the Guinier–Porod model
Here
Based on the fitting curves using the 3-region Guinier–Porod model, there are two dimensionality parameters
Thermal conductivity characterization
Figure 4 shows the thermal conductivity enhancement in water/PAO nanoemulsion fluids as a function of the loading of water from 0.47% to 8.6% volume fraction at room temperature using the

Thermal conductivity of water/PAO nanoemulsion fluids versus water volume fraction. The prediction from the Maxwell equation is shown for comparison.
It can be seen that the thermal conductivity increases linearly with water volume fraction. A maximum increase of 16% is observed at a water volume concentration equals 8.6% volume fraction. The observed enhancement in thermal conductivity agrees well with that predicted by the classical Maxwell equation with the assumption of spherical droplets. The Maxwell equation for suspensions of well-dispersed, non-interacting spherical particles51,52 reads
where
Viscosity characterization
The dynamic viscosity of water/PAO nanoemulsion fluids was measured at room temperature and the viscosity dependence upon the water volumetric concentration is shown in Figure 5. The calibration is carried out using the pure PAO and its dynamic viscosity is found to be 7.3 cP which compares very well with the literature value. The test samples exhibit a shear-independent characteristic of Newtonian fluids for the spindle rotational speed from 10 to 30 r/min.

Dynamic viscosity of water/PAO nanoemulsion fluids versus water volume fraction.
One interesting thing can be seen in Figure 5 that the viscosity first increases with water concentration, reaches a maximum at 5.3% volume fraction, and then decreases. This trend is different from the thermal conductivity shown in Figure 4, and the maximum viscosity can be attributed to the attraction force between droplets within the nanoemulsion fluids. As the amount of water is increased, the surfactant molecules become hydrated while releasing their counterions into water. The exchange of surfactants molecules and counterions between the droplets could make themselves charged oppositely. The interdroplet attraction increases with water concentration until the hydration process is complete. This leads to a maximum viscosity in water/PAO nanoemulsion fluids. It also coincides with the nonlinear inner structure change with increasing water concentration as seen in Figure 3.
Heat capacity
Meanwhile, another important liquid–solid phase change property, the heat capacity of water/PAO nanoemulsion fluids, has also been investigated and shown in Figure 6. Theoretically, the heat capacity can be enhanced through two different mechanisms: one is due to the high specific heat of the dispersed phase; the other is due to the latent heat of the dispersed phase-changeable nanodroplets.24,41,53–55 The latter one, that is, use of phase-changeable nanodroplets, is much more efficient for the heat capacity enhancement. In water/PAO nanoemulsion fluids, the fluid’s heat capacity can be increased by the high specific heat of water (i.e. Cwater = 4.2 J/g C, CPAO = 1.88 J/g C) or the latent heat of water (

DSC cyclic curves of water/PAO nanoemulsion fluids for different water loadings. Exothermal peaks are observed at −20 °C, corresponding to the freezing of water nanodroplets, while endothermal peaks are observed at 0 °C, corresponding to the melting of water nanodroplets.
When water nanodroplets do not undergo solid–liquid phase change, the specific heat of the water/PAO nanoemulsion fluids follows the simple mixing rule
where
where
DSC cyclic curves of water/PAO nanoemulsion fluids under different water loadings are shown in Figure 6. During the heating and cooling cycles, water nanodroplets undergo a melting–freezing transition in the nanoemulsion fluids. The observed melting–freezing hysteresis (i.e. supercooling) varies from 20 °C to 45 °C, depending on the size of the droplets and the structure of the microemulsion, while most of the peaks occur at about −20 °C). The hysteresis in the melting–freezing cycle may be due to the interfacial energy change inside nanoemulsion fluids. On the other hand, the presence of a single freezing peak in Figure 6 indicates a correspondence of the structural change with increasing water concentration (or water-to-surfactant molar ratio) as observed in our previous SANS measurement result. There is no obvious melting–freezing peak for water concentrations less than 4.5%, while the exothermic crystallization peak starts at around −20 °C when water concentration is higher or equal to 4.5%. When the water concentration is increased further to above 8.6%, the freezing peak shifts to effect lower supercooling and peak values.
To quantify the specific heat of each sample, the result is also calculated and summarized here in Table 1. It can be seen here that the specific heat sharply increases from 9.8 to 26.72 J/g when the water concentration is increased from 4.5% to 5.3%. It gradually increases with higher water concentration and decreases again when the water concentration is higher than 8.6%, while the freezing peak temperature is decreased. All these happen to coincide with the structure transition from spherical to cylinder shape with increasing water concentrations as observed from SANS measurement.
Specific heat of water/PAO nanoemulsion fluids with its supercooling temperature.

PAO: polyalphaolefin.
Assuming ΔT = 20 °C, the effective volumetric specific heat can be increased by up to 80% for the nanoemulsion fluid containing 8.6% water nanodroplets when the water nanodroplets undergo phase transition. The use of phase-changeable nanodroplets is expected to provide a way to simultaneously increase the effective specific heat and thermal conductivity of conventional heat transfer fluids. Meanwhile, the heat of fusion Hf of pure water is 334 J/g, an outstanding value among phase-changing materials. The calculated Hf values of water/PAO nanoemulsions for different water loading from 5.3 to 8.6 volumetric percent are 17.7 and 28.724 J/g, respectively, in agreement with the measured results shown in Table 1, which are 26.72 and 34.17 J/g, respectively. The volumetric heat capacity of water is about 4.18 J/mL K, and is over two times the heat capacity of PAO (1.74 J/mL K). 26 For a temperature increase from −20 °C to 0 °C, 1-mL PAO absorbs 37.6-J heat. For the nanoemulsion containing 8.6% water nanodroplets, the melting of ice nanoparticle absorbs 34-J heat, which means that upon melting the ice nanoparticles in the nanoemulsion, the heat capacity of PAO has increased up by about 76%. Together with the enhancement in heat capacity caused only by the addition of water without phase change, totally a maximum heat capacity increase of 88% is obtained in the 8.6% water/PAO nanoemulsion fluids.
Conclusion
In summary, thermophysical properties such as the thermal conductivity, the viscosity, and the specific heat have been investigated along with the inner structure of water/PAO nanoemulsion fluids. The thermal conductivity increase is rather moderate in these fluids, for example, a 16% increase for 8.6% volume fraction sample, which agrees well with the classical colloidal theory. The dynamic viscosity exhibits a maximum value of 10.7 cP at a water volume concentration around 5.3% which coincides with the variation of the nanosrtructure inside at different water concentrations. The effective specific heat can be greatly enhanced when the water nanodroplets undergo phase change: it can be increased up to 90%. Future research will aim at correlating the phase change process with inner structure change, and/or nucleation mechanism will be particularly beneficial, as such phenomena are typically not well understood in complex colloidal systems. Additionally, to enhance the thermophysical properties further, future investigations could focus on a systematical study and comparison of different nanoemulsion systems.
Footnotes
Appendix 1
Academic Editor: Moran Wang
Authors note
The identification of commercial products does not imply endorsement by the National Institute of Standards and Technology nor does it imply that these are the best for the purpose.
Declaration of conflicting interests
The authors declare that there is no conflict of interests regarding the publication of this article.
Funding
This study is financially supported by National Science Foundation (CBET-0730963). The SANS measurements performed at the NIST-CNR are supported in part by the National Science Foundation under agreement no. DMR-0944772.