Abstract
This study aims to provide a clear relationship of how the storage status and duration of the urea affect the efficiency and performance of the SCR system. Focusing on the marine environment, the research compares NOx emissions using new urea solution versus urea solution stored for 5 years on ships. The results indicate that using new urea solution reduces NOx emissions by an average of 30%. Additionally, the study confirms that the impact of the SCR system on the combustion process is negligible. These findings the importance of regular urea solution replacement to optimize the performance of SCR systems installed to meet environmental regulations. Currently, there are no detailed procedures or regulatory standards for urea management and replacement on ships. Considering the strict NOx emission regulations and harsh storage conditions in ship, the study proposes the establishment of effective urea replacement cycles and management procedures.
Introduction
Cargo transportation by ships accounts for about 80% of the world’s freight traffic, which means that ships account for a larger proportion of the world’s freight than any other mode of transport. 1 In addition, both the global shipping capacity and the cargo volume are showing a continuous upward trend. Shipping capacity, which was 1537.5 million DWT in 2012, increased by about 43% to 2194.2 million DWT in 2022,2,3 and cargo volume increased by about 20% from 9948 million tons in 2012 to 11,920 million tons in 2022. These statistical facts show that ships play an important role in the global transportation market. However, these ships consume a large amount of fuel and emit a large amount of harmful gases during the combustion process of the main propulsion and generator diesel engines.4,5 These emissions are attracting global attention due to their negative impact, and regulations regarding them are gradually being tightened. 6
The International Maritime Organization (IMO) approved a convention to prevent air pollution from ship emissions at the 40th session of the Marine Environment Protection Committee (MEPC), and it was established as a new annex, Annex VI (Regulations for the Prevention of Air Pollution from Ships), to the International Convention for the Prevention of Pollution from Ships (MARPOL 73/78). 7 Additionally, in terms of nitrogen oxide emissions, regulations have been applied since 2016 through the implementation of Tier III, which reduces emissions by approximately 80% compared to Tier I standards. 8
To address the tightening exhaust gas regulations, post-treatment technologies such as scrubbers, exhaust gas recirculation systems (EGR), and selective catalytic reduction (SCR) have been commercialized and are known to be effective in reducing atmospheric pollutants like sulfur oxides (SOx) and nitrogen oxides (NOx).9,10 Especially, the SCR system is a device that can reduce more than 90% of the nitrogen oxides contained in the exhaust gas. By adding ammonia (NH3) to the exhaust gas and passing it through the SCR device, it initiates a catalytic reaction that reduces it to environmentally benign nitrogen and water. Compared to the EGR method, it has the advantages of higher combustion pressure and compression ratio, increases engine thermal efficiency, and improves fuel efficiency, making it an effective solution.11,12 Against this backdrop, existing research has generally focused on topics related to the performance of SCR systems. 13
Xia et al. 14 focused on the issue of urea crystallization in SCR systems of both two-stroke and four-stroke engines, publishing results that demonstrate how the composition of urea deposits is significantly influenced by the composition of exhaust gases. Especially, it was found that the chemical composition and concentration of urea deposits within the SCR system vary depending on their location, and in the SCR system of marine two-stroke engines, urea was identified as the main component of the deposits at the bottom of the urea nozzle. Furthermore, Liu et al. 15 explored the impact of using hydrocarbon-based biodiesel blends on marine SCR systems and investigated strategies for optimizing urea dosing accordingly. When hydrocarbon-based biodiesel is blended, to solve the issue of failing to meet NOx regulations with the urea dosing used in pure diesel mode, a three-dimensional model of marine engines and SCR systems was constructed. Optimal blending ratios and engine injection timing were determined. Through this method, a strategy for optimizing the urea dosing of SCR systems when blending hydrocarbon-based biodiesel was presented. Kang et al. 16 investigated the physical and chemical properties of urea solutions as a function of urea concentration and analyzed the characteristics of exhaust gases. Through this, it was confirmed that as the concentration of urea increased, the content of biuret, aldehydes, and phosphates in the urea solution increased, but the changes in the emissions of carbon monoxide (CO), hydrocarbons (HC), and particulate matter (PM) in the exhaust gas were minimal. However, it was found that the emission of nitrogen oxides (NOx) decreased as the urea concentration increased, and in urea solutions of more than 30.0%, the reduction efficiency of nitrogen oxides was confirmed to be over 80%. As such, there is a lack of research on the urea solution as a catalyst for SCR, especially quantitative research on the performance changes according to the storage period and environment of the urea.
Table 1 shows the shelf life of urea solution according to the storage temperature provided in ISO 18611. When exposed to high temperatures, urea in the urea solution can decompose into ammonia gas and carbon dioxide, which can degrade the performance of the SCR system. Unlike urea solution for land vehicle use (Urea content 32.5%), the urea solution for marine use (Urea content 40%) is made with a 7.5% higher concentration of urea, making the impact of environmental conditions more significant. However, research on urea solution, which is the catalyst for SCR, is lacking, and in particular, quantitative studies on performance changes according to the storage period of urea solution are insufficient.17–19
Shelf life as a function of temperature (ISO 18611).

Therefore, this study analyzed the effect of the storage period of marine urea solution stored in the actual ship environments on the SCR efficiency and performance. This study aims to provide a clear relationship of how the storage status and duration of the urea solution affect the efficiency and performance of the SCR system and provide basic data for the efficient operation of the SCR system in practice.
Materials and methods
Principles of SCR
The SCR System has a De-NOx efficiency close to 90%, making it an effective post-treatment method for controlling NOx emissions from diesel engines.4,20 The method reduces NOx emissions from exhaust gases using a catalyst and a reducing agent, primarily using urea solution instead of the difficult to handle ammonia. It includes High Pressure SCR (HP SCR), which is installed before the engine’s turbocharger and operates under relatively high pressure and temperature for the catalytic reaction, and Low pressure SCR (LP SCR), which is installed after the turbocharger. Urea solution, used as a reducing agent, decomposes into ammonia and carbon dioxide through the chemical reaction of equation (1) in the presence of high temperature gas vapors.

The decomposed urea solution is converted into nitrogen molecules and water, which have no impact on air pollution, through a chemical reaction process as shown in equation (2) on the catalytic surface within the SCR chamber, and then released into the atmosphere. The temperature at which the catalyst is most reactive ranges from 290°C to 450°C; if the temperature is too high, there is a risk of ignition,

and if it is too low, it does not react, causing corrosion by ammonium sulfate and contamination by ammonium nitrate. 21

Experimental configuration and equipment specifications
The LP SCR system used in this experiment consists of a four-stroke diesel generator, SCR chamber, Urea solution storage tank, Soot blowing unit, Urea dosing unit, and Gas analyzer, as illustrated in Figure 1. Urea solution is transferred from the Tank to the Urea Dosing Unit, where the flow rate is adjusted according to the engine load. The urea solution supplied through the Injection Pipe mixes with the high temperature exhaust gases emitted from the diesel engine, cause the chemical reaction described in equation (1). Inside the SCR Chamber, catalyst modules are positioned to facilitate the chemical reaction shown in equation (2), reducing NOx emissions. To prevent soot deposition on the catalyst surface from the exhaust gases, a Soot Blowing Unit is installed.

Configuration of LP SCR.
As shown in Figure 2, the urea solution is stored in a designated location inside the engine room, which is a urea solution storage tank. Urea solution stored in the storage tank is exposed to conditions in which the minimum temperature in the engine room in winter is 15°C, the minimum temperature in seawater is 13.6°C, the maximum temperature in the engine room in summer is 39.6°C, and the maximum temperature in the engine room is 33.5°C. In addition, since this storage tank is installed on a floor higher than sea level, it is exposed not only to temperature changes but also to the vibration and movement of the hull. When these factors are considered comprehensively, the external environment is expected to have a significant impact on the composition of the urea solution.

Urea solution storage tank in the engine room: (a) side of the urea solution storage tank, (b) front of the urea solution storage tank.
The specifications of this SCR system are shown in Table 2, which indicate that the system meets IMO Tier III standards with a nitrogen oxide emission rate of 2.31 g/kWh and an allowable exhaust gas temperature of up to 400°C.
Specification of LP SCR.

Table 3 presents the main specifications of the four-stroke diesel engine system developed by Hyundai Heavy Industries, used in this study. The engine is a four-stroke Trunk Piston Type equipped with a Turbocharger and Intercooler, designed as a generator drive engine to operate at a constant rated speed of 900 rpm across the entire load range to produce electricity at a consistent frequency.
Specification of diesel generator.

The gas analyzer is a portable combustion gas analysis system that can analyze the exhaust gases emitted from diesel engines. This measure device is composed of a control unit, analyzer box, condensate trap, dust filter, external air inlet filter, and gas dilution filter, and the measurement range and accuracy is shown in Table 4. It adopts the testing methods required by the IMO NOx Technical Code 2008 and MARPOL Annex VI for NOx emission rate certification on ships. It has been certified from credible classifications, like DNV(Certificate No. TAA00001K0 Rev. 4) and NK(certificate no. TA23536M).
Specification of gas analyzer.

In this study, the chemical composition of urea solution stored for 5 years in harsh marine environments was compared to that of new urea solution to verify performance degradation. And the NOx reduction rates at specific engine loads, like 25%, 50%, and 75%, as specified by the official measurement method (D2 cycle) for ship engine exhaust emissions, were analyzed using a diesel engine. This experiment allowed us to examine the impact of urea solution condition on NOx reduction in diesel engines.
Results
Engine performance
Table 5 presents the engine operating results and the urea solution feed rate for each load on the four-stroke diesel engine used in this experiment. It was found that the urea solution, which initiates chemical reactions, was injected at a consistent flow rate across the same loads. The ambient temperature conducive to vigorous decomposition reactions of urea solution was deemed appropriate between 300°C and 450°C, and it was verified that the exhaust gas temperature during this experiment was within this range. The scavenging pressure indicating the air flow supplied for combustion was also found to be similar, showing no significant difference. This confirms that other factors such as engine operating conditions and the amount of urea injection were excluded from affecting the performance of the urea solution in this experiment.
Operation results and urea solution feed rate according to engine load.

Ingredients analysis of urea solution
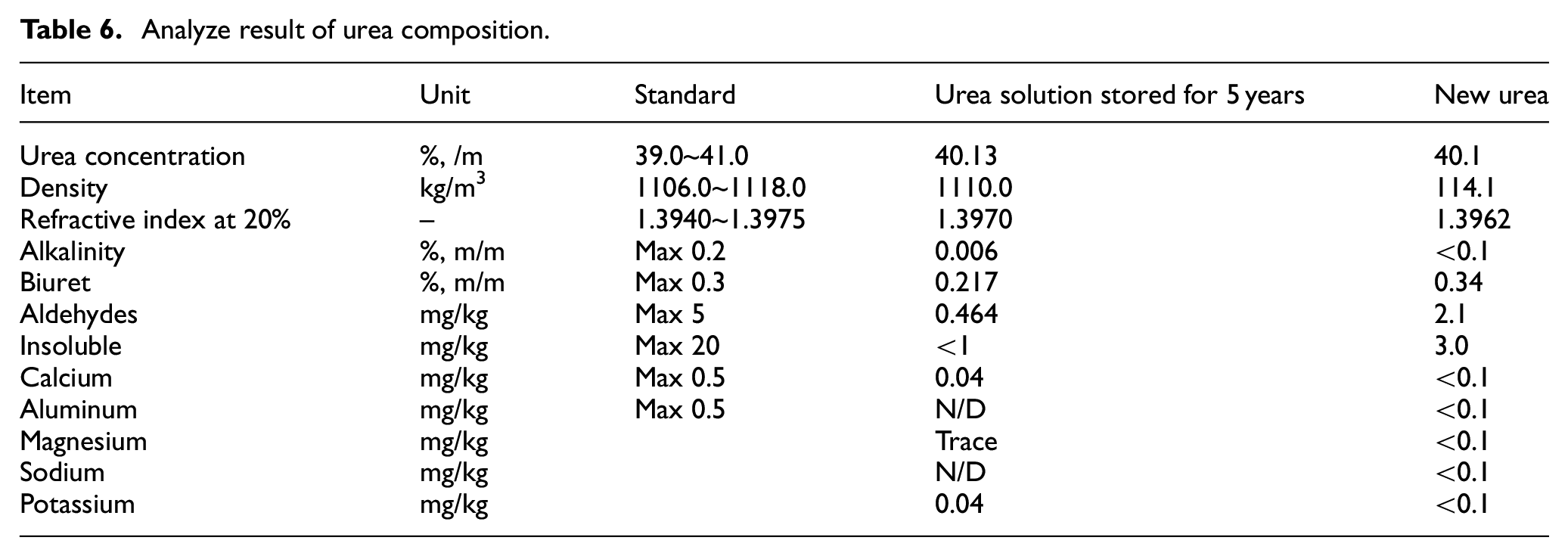
The analysis results for two types of urea used in this experiment are as shown in Table 6. The urea solution stored for 5 years was stored in a marine environment for 5 years, while the Urea solution stored for 5 years was a new product received from the manufacturing plant. The test method was carried out according to KS R ISO 22241-2 and ISO 3675 (Density) standards through a credible testing organization. Since there is no global standard for marine urea, the criteria, excluding urea content, were based on automotive urea standards. The urea content was evaluated with a benchmark of ±1%. Although the urea content slightly decreased over the storage period, it was found to meet the specified standards. Considering the change in density, the urea content is deemed to be within the test’s error range, and it is concluded that the urea content has substantively decreased over the storage period. Biuret is a typical impurity formed after the chemical reaction of urea, as illustrated in equation 1. This indicates that decomposition has occurred through chemical reactions during long term storage before the urea is injected into the SCR system.22,23
Analyze result of urea composition.
Emission analysis
NOx
Thermal NOx is known to be generated through chemical reactions with air during engine combustion. To mitigate the environmental pollution caused by this, a post-treatment equipment, the SCR system, is installed. 24 Urea, the catalyst in SCR, is essential for converting NOx into harmless nitrogen and water. Figure 3 shows the changes in NOx emissions when using new urea solution (▲) and Urea solution stored for 5 years (▼) under various load conditions in a four-stroke diesel engine equipped with SCR. As seen in the graph, under all engine load conditions, the new urea measured on average 28% less NOx emissions compared to when the urea solution stored for 5 years was used. NO decreased by up to 33% at 25% load, and NO2 decreased by up to 36% at 50% load.
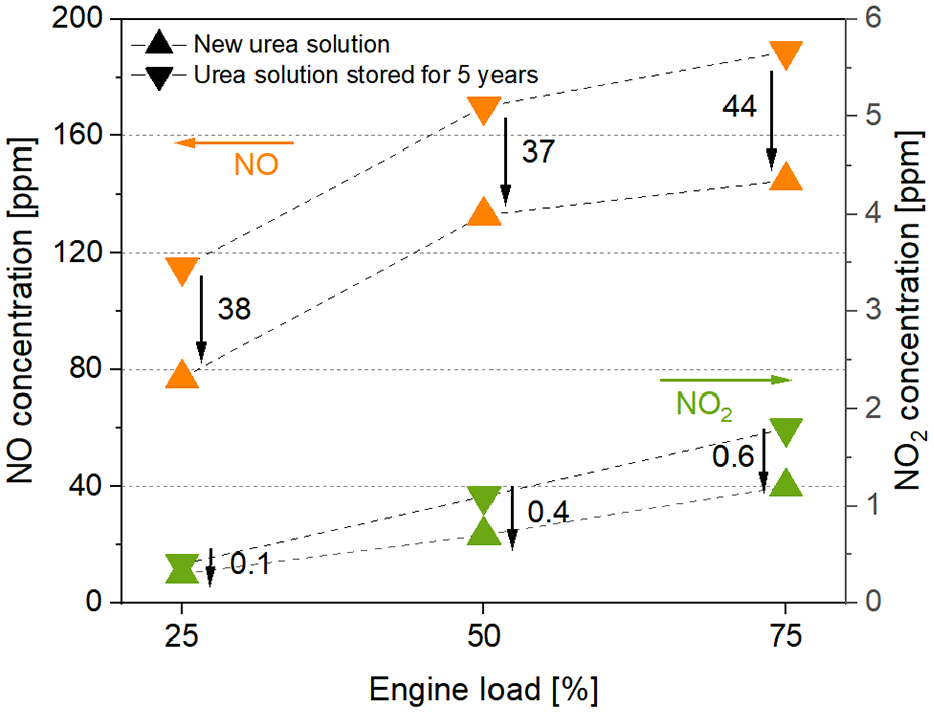
Changes in NOx emissions according to urea condition.
Figure 4 illustrates the reduction rates of NO and NO2 per load due to the change of urea. At 25%, the reduction rate of NO is shown to be higher, while at 50% and 75%, NO2 shows a higher reduction rate. This indicates that the high exhaust gas temperatures at high engine loads enhance the chemical reaction of urea, significantly improving the NOx conversion efficiency. It is known that the NO in the exhaust gas reacts with O2 to form NO2, especially at high temperatures, this reaction occurs rapidly and is further promoted if the reaction time is sufficiently long.25,26

Reduction rate of NOx emissions according to urea condition.
These results confirm that the storage period of urea has a significant impact on the performance of the SCR system. Therefore, it can be stated that regular replacement and management of urea are very important for maintaining consistent environmental performance of the engine.
Engine emissions
Figure 5 shows the concentrations of CO, CO2, and O2 in the exhaust emissions by engine load. CO is produced by incomplete combustion and is associated with O2, 27 while CO2 is known to be emitted during the engine combustion process as a representative greenhouse gas substance. 28 During SCR operation, CO2 is generated through the decomposition reaction of urea and the chemical reaction with nitrogen oxides, while O2 is consumed.

Engine emissions concentration according to urea solution condition.
Regardless of the load, the urea solution stored for 5 years showed higher concentrations of CO2 compared to the new urea solution, and O2 was found at lower concentrations. This result can be mistaken for the chemical reaction of the urea solution stored for 5 years being more active.
This can be explained by the experimental results related to the urea solution stored for 5 years in Table 5, where the scavenge pressure was 0.1 bar lower than that in the experiments with the new urea solution, resulting in less oxygen supplied.29,30 Furthermore, the concentration of CO was inversely proportional to the production of CO2, which can be attributed to improved combustion efficiency due to adequate mixing of fuel and oxygen after 50% load, understanding in conjunction with the decrease in O2 concentration. 31 These results indicate that the total impact of the chemical reactants during SCR operation is negligible compared to the chemical reactants produced during the combustion process, confirming that the direct effect of the SCR post-combustion treatment equipment on the engine combustion process is minimal.
Conclusions
In this study, the effect of urea condition on nitrogen oxides emissions was quantitatively evaluated in a stroke diesel engine equipped with a SCR system. Experiments conducted under various engine load conditions revealed that using new urea solution resulted in an average reduction of NOx emissions by about 30% compared to when using urea stored on a ship for 5 years. These results indicate that regular replacement of urea is essential for optimizing the performance of the SCR system during diesel engine operation.
While basic conditions for the replacement cycle of the SCR catalyst module and the storage of urea based on operational time are established, detailed procedures and regulatory conditions have not been set. To faithfully achieve the objectives of the stringent NOx regulations implemented since 2016, it is crucial not only to install and initially verify equipment like the SCR but also to establish procedures for the management and verification of urea, considering the harsh environmental conditions on ships. The results of this study are expected to significantly advance the maritime industry’s approach to NOx reduction, ultimately contributing to a cleaner and more sustainable future.
Footnotes
Handling Editor: Silambarasan Rajendran
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
