Abstract
In this work, a voltage-heating actuated flexible solvatochromic film is developed using the composites of Nafion film-nickel complexes with spray-coated silver nanowires (AgNWs). The nickel complexes, inside Nafion film through ion exchange, serve as colorable components due to the coordination number change of water molecules in solvatochromism. The color-changing process of the composites was characterized experimentally by adjusting the water content in films, which exhibit distinct coloration properties from red to green with an absorbency shift (0.25–0.60 at 420–550 nm). To achieve an electrical actuation, the electrothermal joule effect of AgNWs is studied to accelerate the hydrolysis of the nickel complexes. A mask patterned spraying process is developed to increase the adhesion between AgNWs and Nafion film, which can be beneficial in a low (2–5 V) voltage-regulated coloration. This study provides a systematic investigation of flexible electrothermally responsive color-changing film of complex metal ions, the composite film prepared from flexible smart materials and well-adhered electrodes have potential applications in the fields of adaptive camouflage, wearable display devices and e-skins.
Rapid and active coloration in functional material is an importance feature that can be implemented in vivid display, camouflage, visible device and temperature-controlled equipment.1–8 Among these techniques, different physical stimulus-responsive chromic have been developed, such as electrochromism, 9 thermochromism, 10 photochromism, 11 and solvatochromism. 12 Solvatochromism has a unique character by the absorption or emission spectrum of a substance in the vapor state, exhibiting a solvent-dependent coloration.13–15 This mechanism can be further implemented into engineering applications in sensor or actuator if an effective regulation methodology is developed.16,17 Electrical actuation in chromic system by a voltage or a current is a useful strategy that incorporating colorfully device into mechatronic systems.18,19 Examples include a colorful artificial muscle, 20 camouflage robot, 21 flexible visual indicator. 22 The application of thermal management technology determines the active solvochromic performance during the preparation and demonstration of related composite devices.23–26 Electrothermal actuation through flexible electrodes and smart materials enables the application of thermal management technology in color-changing displays.27,28
In this paper, we propose a methodology that integrating the solvatochromic response in nickel complex with electro-thermal actuation by fabricating aNafion film composite. Nafion is a perfluorinated ion-exchange polymer, Figure 1(a), with a short side chain terminated by a sulfonic acid group. Long polymer backbones qualify its mechanical strength, and short side chains provide ionic groups that react with some cation and form channels. 29 In addition, a mass of nanopores distribute in the polymer matrix allow the formation of nanoscale ionic channels (Figure 1(b)). The ion channels provide passes for the ion exchange of cation in Nafion with other solvatochromic metal-ligand, which incorporating solvatochromism in Nafion. We then sprayed sliver nanowires (AgNWs) onto the surfaces of Nafion, to achieve voltage-heating regulation by thermal evaporation of solvent inside the Nafion. The porous property of AgNWs offers a transparent appearance and do not affect coloration by the coordination change of nickel complexes with molecules.

Structure of Nafion: (a) a ball-and-stick model of polymer groups of Nafion molecular structure and (b) nanopores and ionomeric channels of Nafion.
We prepared nickel complexes as follows. Nickel nitrate hexahydrate (J&K Chemical Inc., Beijing, China) was treated by diketone and diamine ligands, which were provided by acetone, acetyl acetone, and tetramethylenediamine. The treated solution was ion exchanged with sodium tetraphenylboron (NaBPh4) and the final product was obtained after repeated purification, as shown in Figure 2(a). The complex has strong ability of coordination with methanol molecules, which can show color changes from red to blue-green. Then, the solvatochromic Nafion membrane were obtained by immersing in the methanol solution of these salts for 48 h.
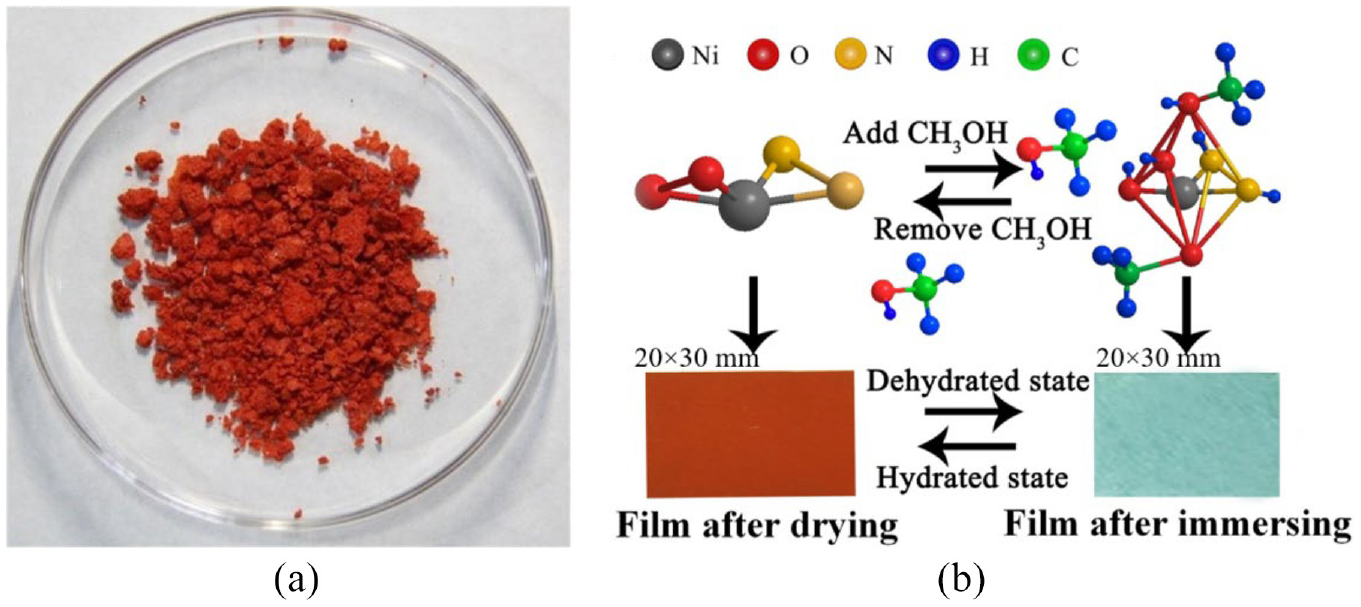
Synthesis of nickel complex and coloration: (a) nickel complexes in solid and (b) scheme diagram of coordination process of nickel complex in methanol solution.
As shown in Figure 2(b), the nickel complex with characteristic chemical structure contains diamine and diketone ligands, which turns the complex into different colors when immersed in different solutions. Reversibility of color changes can be observed during the process, which is based on the long-term stability of the sample film. Table 1 records the discoloration effect diagram of the solvent discoloration film after being placed for a long time, and it can be seen that the sample has good maintenance of discoloration performance. Here, we only use this film as a carrier, a red color changing film obtained after vacuum drying can coordinate with water molecules (vacuum drying, constant temperature 90°C, 48 h). The nickel complexes in color-changing film can be reconstructed by coordination with water molecules and produce rich color transformation between red and green.
Fabrication of AgNWs/Nafion composite film.

AgNWs were selected as electrodes due to low stiffness, high transparency, and conductivity, which had little effect on the color of film itself. This process mainly utilizes the porous properties of Nafion membrane materials and the overlapping properties of silver nanowire materials. Adhesion can be formed between the two materials to complete the attachment of surface electrodes. A facile spraying process is developed as shown in Figure 3.
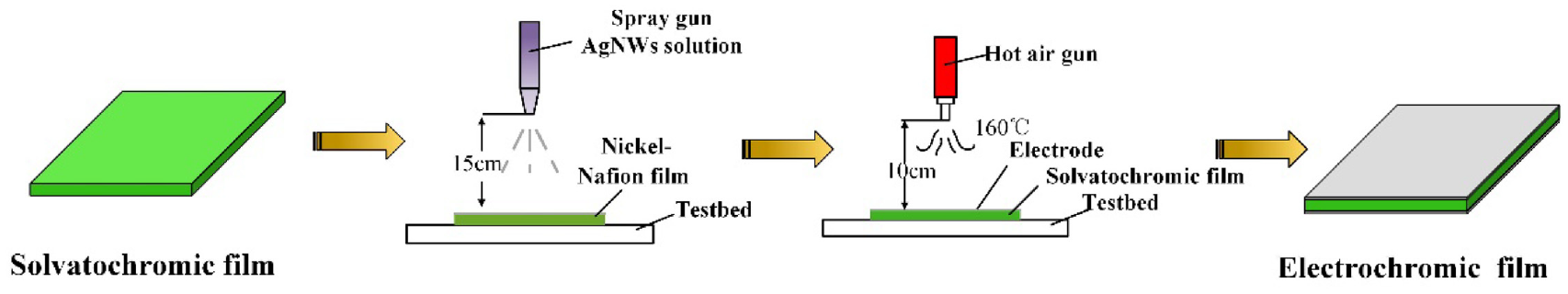
Fabrication of AgNWs/Nafion composite film.
The SEM image of the AgNWs/Nafion composite film is displayed in Figure 4. The electrode layer of AgNWs was deposited uniformly with networked distribution. The overlapping morphology of AgNWs increases the current collecting area of the electrode, which reduce the electrode resistance to about 30 Ω/□. The diameter range of AgNWs we used is about 30–100 nm, and the length of some AgNWs can reach 4.27 μm. The above properties enable the AgNWs to as electrode for regulating solvatochromic film.

SEM image of a silver nanowire electrode.
In order to evaluate the coloration, we firstly investigated the water coordinate capacity of the film samples and the change of its water content vs. time due to dehydrating or hydrating in air by the weighing method, (the environmental humidity was about 63% RH in the lab). The solvatochromic film and the flexible composite film were soaked in DI water for 8 h to absorb water to a saturated state. After that, the samples were removed from DI water and cleaned the surface moisture to be weighted. The transient masses mt at a time interval were recorded until the sample reached an equilibrium state. The values at the initial moment are set to the completely hydrated mass. Then the films were vacuum dried at 80 degrees centigrade in a drying oven for 8 h to sufficiently remove the internal water. And we measured the transient mass mt in the same way. Under this condition, the initial masses are set as the dry mass md of solvatochromic film and flexible composite film. The water content w can be calculated as,

Based on equation (1), we got the water content of the films from the initial saturated state or from the dry state to equilibrium as shown in Figure 5. Twenty-two points (S1–S11 and E1–E11) were selected respectively as the conditions of the color change test.

The moisture content curves of (a) solvatochromic film and (b) flexible composite film.
The temperature and humidity measured at the initial point have a slight deviation, In Figure 5(a), the absorption curve of solvatochromic film fast grows and gradually reaches to equilibrium from 0 to 5400 s. The water contents increased from 0% to 1.85% due to the absorption of water. After 5400 s, the water content of the solvatochromic film slowly increases to 2.1%. In fact, there are three types of water involved in the coordination state with the nickel complexes inside the samples, namely free water, loosely bound water and freezing bound water, which depends on the bonding level between the water molecule and the complexes. During absorption process, the absorbed water molecules coordinate with the nickel complexes, which result in color change of the sample. For the dehydration process, the solvatochromic film presents a tendency from sharply dropping to slow decrease and finally reach an equilibrium state. The water content decreased from 12.75% to 4.09% (0 –5400 s), and then decreased to 4.01% (5400–10800 s). The sample experienced from losing free water to the loss of bound water lost. From Figure 5(b), the flexible composite film presents a similar result as solvatochromic film. During the absorption process, the curve displays a liner increase from 0 to 10,800 s and then gradually reaches to the equilibrium. During dehydration process, the decline slope of the flexible composite film is smaller than that of the solvatochromic film, which exhibits a moderate decline. This is probably attributed to the barrier property of porous AgNWs layer on surfaces of the sample.
Through the relationship between the marked moments and water content in Figure 5, we conducted tests at a specified moment to observe the color change of the sample in the air. The sample is placed on the platform of the LED background light for better observation, and the color change process is recorded with the camera backlit.
Figure 6(a) shows the optical images of the color evolution of solvatochromic film during process of absorption and dehydration, respectively. Each color corresponds to the specific water content (S1–S11 and E1–E11). During absorption process with the water content from 0% to 2.01% (S1–S6), the color of the film changed from red, orange, yellow to light green. The solvatochromic film in fully dry state optically appears red because of the planar quadrilateral crystal configuration of the nickel complexes. When undergoing a coordination reaction with water molecules, the crystal of nickel complexes will become a spatial octahedral configuration, which appears optically red to green. During the dehydration process with water content from 12.7% to 4.09% (S7-S11), the color presents a slightly change. Excess water molecules do not change the crystal structure of the nickel complex, or the crystal of the nickel complex is unable to absorb extra water molecules, which are stored in the Nafion matrix. In fact, from S1 to S6, the solvatochromic film mainly absorbs freezing bound water with loosely bound water combined with nickel complexes, which resulting in a change in crystal configuration. From S7 to S11, the film mainly these waters, which has a relatively small effect on the color change.

Color change versus the water content of films: (a) color change of solvatochromic film and (b) color change of flexible composite film.
Figure 6(b) shows the color change process of the flexible composite film with different water content. It can be clearly observed that the flexible composite film presents excellent color change effect similar to the solvatochromic film due to the high transparency of the AgNWs layer. In order to obtain good electrothermal effect and high transparency, the surface resistance of the AgNWs layer was set to about 20–40 Ω/□. Meanwhile, from the figure that the coverage of the silver nanowires obscured the discoloration process of the film to a certain extent, which is also the place where the follow-up research needs a breakthrough.
The absorbance tests were carried on using the self-made test platform as shown in Figure 7. After picked up from the DI water and cleaned the surface water, the sample was placed on glass plate. And the distance was set to 20 mm from the test plate to the LED side light source. The sample were tested by Maya2000Pro MAYP111232 and white incident light source. Meanwhile, their absorption spectrum data were collected and recorded by OceanView 1.6.5 and plotted in Figure 8.

Absorbance spectrum experiment.

Absorbance spectra of solvatochromic film and flexible composite film: (a) absorption of solvatochromic film, (b) dehydration of solvatochromic film, (c) absorption of flexible composite film, and (d) dehydration of flexible composite film.
The abscissa in the spectrum is the wavelength λ, and the ordinate is the absorbance A. The water content of the sample was regulated, and the absorbance is calculated by equation (2).

A is the absorbance, l0 and l1 are the intensity of the incident light passing through the sample surface and the intensity of the transmitted light passing through the sample surface, respectively. The absorbance is affected by factors such as temperature, humidity, and the degree of coordination of the nickel complexes. From the theory of chemistry, organic compounds can absorb ultraviolet and visible radiation, and its absorption spectrum is produced by the transition of valence electrons in the molecule. Figure 8 shows the absorption spectra of the solvatochromic film and electrochromic film. The extracted and recorded spectrum belongs to band spectrum rather than sharp line spectrum, which is mainly due to the transition of electrons in the molecule during the energy absorption process. The dry state (0% of solvatochromic film and electrochromic film) and saturated water state (12.7% of solvatochromic film and 13.3% of electrochromic film) of the films were selected to analyze color change process, and four strong absorption peaks can be observed at λ1 = 474, λ2 = 468, λ3 = 468, and λ4 = 469 nm, respectively. Under certain experimental conditions, with the increase of water content, the absorbance of the solvochromic film and the flexible composite film at the maximum peaks decreased from 0.92, 1.23 to 0.57, 0.59, respectively. Usually, the absorption of light by a substance is proportional to the concentration of the substance. During the discoloration process, the concentration of nickel ion complexes decreases due to the water coordination reaction, resulting in a weakening of the substance’s absorption of light. It can be seen that the as-prepared nickel ion complex groups play a major role in absorbing light during the spectral measurement. When an absorption peak appears in the ultraviolet-visible region (200–700 nm) of the absorption spectrum, it indicates that the n–p* valence electron transition exists in the molecule and the molecule contains heteroatomic unsaturated groups. The coordination reaction of nickel complexes with water molecules leads to the generation of wave crests. In addition, the absorption peak moves to the left along the abscissa as shown in Figure 8 shows, which is called a blue shift. According to the optical image of Figure 6, the blue shift corresponds to a change process of the sample from red to light green. In Figure 8(b) and (d), there is a very small blue shift, which can correspond to the color of the solvatochromic film and flexible composite film did not change significantly during the process.
Figure 9(a) shows a patterned electrochromic results of a flexible composite film under different voltage. Both ends of the sample are clamped by Kelvin clamps, and the DC power supply is used to adjust the voltage. A relatively stable humidity environment (about 60%) was selected for the color change display experiment, and optical recording photos, electrical parameter changes and thermal imager images during the color change of the sample were respectively given. Original AgNWs/Nafion flexible composite film was light green in an indoor environment. The film gradually transferred to red with increasing voltage, the color turned red obviously as the voltage changed from 2 to 4 V. We can see the most prominent color variance at 5 V, and there is no obvious damage on the AgNWs surface electrode. Higher voltages would cause thermal damage to the electrodes ending up in irreversible oxidation. The surface current changed from 0 to 238 mA to 210 mA also confirmed the thermal damage of the AgNWs. Temperature of electrode and composite film increased from 23.1°C to 79.7°C during discoloration process, which is used to demonstrate the occurrence and completion of structural coordination reaction.

Electrochromic results of a 5 mm × 20 mm AgNWs/Nafion composite film. (a) Discoloration of different voltages applied. (b) The transmittance of electrochromic film after several cycles. (c) Discoloration after bonding the electrochromic film to a model.
Figure 9(b) shows the transmittance of electrochromic film after multiple electrochromic cycles. The transmittance tested by 721 spectrophotometer increases slowly from 21.3% to 22.4%. Good transmittance can ensure that the electrochromic film has an excellent color display effect. The slow curve rising rate confirms stable sample performance and long material life. Repeated use of the sample would cause degradation and debonding of surface electrodes. We selectively deposited silver nanowires on the surface of the film to form a three-letter pattern as shown in Figure 9(c). Each character in turn became red in a few seconds when 4 V voltage was applied. The patterning of electrodes could potentially make voltage induced discoloration technology more applicable and provide some supports for research in the field of flexible electronics based on the satisfying performance exhibited in the experiments (Figure 10).

Discoloration response and recovery time of flexible composite films.
In summary, we developed a fabrication of a flexible solvatochromism based on Nafion film and nickel complexes. A nickel complex was prepared, which could coordinate with water molecules. When a nickel complex molecule is coordinated with two water molecules, its crystal configuration changes from red to light green, then Nafion film was immersed in a methanol solution containing nickel complexes to prepare the solvatochromic film. The AgNWs/Nafion composite flexible composite film was prepared by spraying AgNWs on the surface of the solvochromic film and regulating the surface resistance of the electrode to be about 30Ω/□. In order to better study the mechanism of color change, we studied water content effect by absorbance spectrum analysis. We revealed the effect between free water, loosely bound water, freezing bound water, and discoloration. Through comprehensive analysis of the dehydration process and water absorption process, the color of the solvatochromic film and electrochromic film changed from red to green and gradually deepened with the increase of water content from 0% to about 13%. The color change was characterized by absorbance spectroscopy, and the blue-shift phenomenon of the wave peak with a width of about 5 nm verified the coordination change process of the metal complexes. demonstration was illustrated to validate the flexible electrochromic display of the AgNWs/Nafion composite film. The flexible composite film exhibits fast color-changing response and patternable solvatochromism under voltage gradient changes of 2–5 V. In view of the aforementioned performances, the electrothermal response as well as active solvatochromics of ionomer substrate containing nickel complexes under two electrodes entail the entire composite device to be an candidate solution with appropriateness and superior efficiency for adaptive camouflage, wearable display devices and e-skins.
Material and method
Footnotes
Handling Editor: Chenhui Liang
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by the National Natural Science Foundation of China (51975184 and 52075411), the Fundamental Research Funds for the Central Universities (B210202124 and B200203148).
