Abstract
Background
Doctors with a history of mental health or substance use challenges face barriers accessing treatment and are difficult to recruit for research. This makes understanding their experiences challenging, yet essential. Overcoming barriers to research participation among unwell doctors is critical to advancing their overall health and wellbeing.
Methods
A qualitative interview study of 21 allegedly impaired doctors in Australia and Aotearoa New Zealand who were subject to regulatory processes relating to their health. Based in an interpretative phenomenological paradigm, this paper examines participants’ motivations for participation in this study and their reactions to participating. It highlights wider implications for sensitive research involving doctors and marginalised groups.
Results
Participants were strongly motivated to participate in our research for their own benefit (desire to be heard, catharsis, and empowerment) and to benefit others (to change the system, and to avoid other doctors experiencing what they experienced). Careful planning and communication of unique legal risks fostered a trusting researcher-participant relationship. To assist other researchers undertaking sensitive qualitative research with vulnerable participants, we describe our research protocol, challenges, and successes in detail.
Conclusions
Despite ethical concerns about research risk, researchers can and should pursue qualitative research into the experiences of under-represented groups, including allegedly impaired doctors. Without involving them in research that examines issues directly impacting on them, we may be inadvertently perpetuating the barriers and stigma that also hinder them from accessing treatment and support. This also deprives us of knowledge that can be used to improve doctors’ health. This paper has broader relevance for anyone doing research with doctors, people in vulnerable positions, people disempowered by institutional or social systems, people who are stigmatised or shamed, and people who have experienced trauma.
Keywords
Background
Doctors face unique mental health challenges, with higher rates of anxiety, depression, and suicide compared to the general population and other professional groups, according to a 2019 National Mental Health Survey of nearly 43,000 Australian doctors (Beyond Blue, 2019). Reasons for poor mental health among Australian doctors include long working hours, a stressful working environment, high work demands, regular exposure to trauma, poor work-life balance and a blame culture within the medical profession (Brooks et al., 2011). In addition, only half of Australian doctors have their own General Practitioner (Kay et al., 2004). Individual and systemic barriers can prevent doctors from engaging in treatment, especially for mental health and substance use challenges (Kay et al., 2008). These barriers include fear of professional criticism, stigma, loss of control, breaches of privacy, perceptions of weakness, or restrictions on their employment or career (Schneck, 1998). It is therefore vital to do all we can to better understand and overcome these barriers through research involving doctors with mental health and substance use challenges.
While many doctors with health challenges continue to practise medicine safely, some health conditions can impair a doctor’s capacity to safely deliver optimal patient care, by adversely impacting on cognitive functions such as attention, judgment, memory, concentration, and decision-making (Candilis et al., 2019). Therefore, poor mental health among doctors may not just be a problem for the doctors themselves, but also for their patients and the wider community (AMA Council on Mental Health, 1973). Avoiding or delaying medical treatment can lead to worsening mental health or substance use (Wijeratne et al., 2021) and greater risk to patients and the public if unwell doctors continue practising while untreated. When poor health adversely impacts a doctor’s ability to practise competently and safely, medical regulators may intervene to protect the public. Medical regulators in Australia and Aotearoa New Zealand can restrict the practice of impaired doctors by: refusing to grant registration, restricting their registration, directing them to undergo a health assessment, or referring them to a Health Panel (Health Practitioner Regulation National Law Act 2009 (Qld), ss. 55, 60, 178, 80, 181 & 156; Health Practitioners Competence Assurance Act 2003 (NZ), ss. 16, 27, 50(4), 50(3), 49 & 47).
Medical regulators exist primarily to protect the public. However, their processes can be incredibly stressful for the doctors involved and may lead to worsening burnout, depression, anxiety, insomnia, and malfunctioning (Templaar, 1997). For example, UK doctors with a current or recent complaint were approximately twice as likely as doctors without a complaint to report suicidal ideation or moderate-severe depression and anxiety (Bourne et al., 2015). Another UK study showed that sued doctors experienced higher rates of depression, anxiety, post-traumatic stress disorder, and suicidal ideation than doctors who were not (Jain & Ogden, 1999). Doctors identified the complaints process as their most stressful life experience: more so than the death of a close family member or a relationship breakdown (Charles et al., 1988). Even doctors who have not been sued identified the ‘threat of litigation’ as their most serious work-related stressor (Schattner et al., 1998) and this was associated with doctors leaving the profession (Charles et al., 1985). In the US, fear of regulatory action is a primary reason why doctors with health conditions delay or avoid seeking medical advice and treatment (Federation of State Medical Boards, 2018).
Given the gravity of the problem of poor doctor health and its association with regulatory processes, there is surprisingly little qualitative research that engages directly with doctors with mental health or substance use challenges who have been subject to regulatory inquiries and processes relating to their health (hereafter “allegedly impaired doctors”). While it is unclear why this is the case, there are a number of possible explanations. First, it may be because researchers have difficulty gaining access to potential participants due to concerns about the potential ethical, legal, or psychological ramifications of participation. Participation in sensitive research can be intense and confronting, and can involve discussion of illness, suffering, perceived injustices, or other stressful human experiences (Dickson-Swift et al., 2007). Second, the experiences of allegedly impaired doctors may be systemically undervalued by researchers, due to unjustified moral judgements associated with doctors seen to be “impaired”.
Third, allegedly impaired doctors may also be hard to reach because the fear, stigma and shame that deters them from accessing healthcare may similarly deter them from participating in sensitive research about their health (Merlo et al., 2013). In particular, allegedly impaired doctors may have privacy concerns that information obtained during research interviews may be disclosed to regulators, thus potentially worsening their legal situation. This is especially relevant in Australia and Aotearoa New Zealand, where laws mandate registered health practitioners to notify regulators if a doctor has placed the public at substantial risk of harm by practising while impaired (Health Practitioner Regulation National Law Act 2009 (Qld), ss. 140 & 141; Health Practitioners Competence Assurance Act 2003 (NZ), s.45).
A final challenge is that human research ethics committees, that seek to balance the expected benefits of a research proposal against possible harms, often classify research about distressing circumstances, drug use or mental illness as “high risk”. Schulz Moore et al. (2019) describe this phenomenon as a reluctance to ‘poke the skunk’ or inquire into matters that might disturb an otherwise placid situation. The disadvantage of this approach is that important research may be discouraged, and under-represented groups may be excluded from research (Gelinas et al., 2023). Consequently, society does not realise the benefits of the knowledge that could be gained through sensitive research directly involving allegedly impaired doctors.
The under-representation of allegedly impaired doctors in qualitative research is consistent with the under-representation of other vulnerable and marginalised groups (Friesen et al., 2022). To overcome the paucity of research in this area and to encourage other researchers to actively engage with allegedly impaired doctors, we describe our methodology in detail and report on the motivations and reactions of participants to participation in our study. The primary outcomes of this study described participants’ perspectives of the positive and negative psychological and vocational impacts of regulatory processes. These results have been published elsewhere (Bradfield et al., 2023a; 2023b), and are not reiterated here. Thus, in this paper, we focus on discussing the decisions and challenges we faced in planning, designing, and conducting our research. In so doing, we believe that publicly sharing our methodology and findings may generate broader learnings for researchers wanting to model our approach to engage with other under-represented groups through qualitative interview research.
Methods
Paradigm
We employed an interpretative phenomenological paradigm (Smith, 1996), which is exploratory in nature. This enabled us to gain information about an area in which little is known. This approach also allowed us to understand how a given person (a doctor with mental health or substance use challenges), in a given context (facing regulatory processes relating to their health), makes sense of a given phenomenon (their experiences of involvement in regulatory processes and their motivations for, and reflections of, research participation) (Fitzgerald et al., 2017). The aim is to gain knowledge of how participants make sense of their experiences; revealing rich, detailed, and personal insights, not captured by other research designs.
Participants and Setting
Participants (abbreviated to “P” in the Results) were eligible to participate if they: 1) self-reported a current or past history of mental health or substance use challenges; and 2) were the subject of one or more regulatory processes relating to their illness. Prior research has assessed the impact of regulatory processes on doctors’ health, but has not focused on the impact of those processes on the wellbeing of doctors with pre-existing mental health or substance use challenges. Hence, we wanted to recruit doctors who met both eligibility criteria, including participants who may no longer be practising as a result of suspension from practice, because this sanction can be imposed on doctors with a health impairment (Health Practitioner Regulation National Law Act 2009 (Qld), s. 178). We excluded doctors who: 1) had been the subject of regulatory processes unrelated to their health; or 2) were incapable of consenting to participate. If doctors were acutely distressed or suicidal at the time of recruitment or interview, we planned to give them the option of participating at a later stage. Participants were recruited from across Australia and Aotearoa New Zealand.
Recruitment Process
Our overall approach to recruitment was purposive (Campbell et al., 2020), and our initial strategy involved criterion sampling, because we defined the desired characteristics for inclusion of participants in this study, and deliberately selected individuals with those characteristics (Liamputtong, 2013, p.17). Before commencing this research, we recognised that doctors with mental health and substance use challenges and/or a history of regulatory sanctions would be hard to reach. Therefore, to facilitate recruitment, we formed partnership arrangements with three Australian Medical Defence Organisations (“MDOs”), the Medical Indemnity Protection Society (MIPS), Avant Insurance, and MDA National, based on the researchers’ prior professional connections with key stakeholders in the industry. We based this recruitment strategy on a prior qualitative study looking at the barriers for sick doctors returning to work, where similar partnership arrangements were formed with doctors’ organisations in a UK setting (Henderson et al., 2012).
MDOs provide medical indemnity insurance to Australian doctors, including advice and assistance to allegedly impaired doctors. They therefore represented a convenient starting point for recruiting eligible participants. Moreover, because it is compulsory for doctors to have professional indemnity insurance in Australia (Health Practitioner Regulation National Law Act 2009 (Qld), s.129), MDOs have a large membership base which we felt would increase the chances of successfully recruiting eligible participants. We provided MDOs with a copy of our approved ethics application, research protocol, proposed interview schedule, distress protocols, an advertisement, consent forms and plain language statements so that their Boards of Management could formally approve their involvement in our research. These documents described the study in detail, and outlined the eligibility criteria, and potential risks, benefits, and requirements of participation.
Once approved, MDOs then formally assisted us in finding potential participants for our study by contacting potential participants individually, or by placing our advertisement on the MDO’s website or in their print or online member communication channels. We instructed MDOs to maintain a neutral position with respect to participation and to avoid language that might encourage doctors to participate. They were instructed to pass on the researchers’ details to potential participants whom they considered might be eligible. We were not given the personal details of these potential participants. Rather, potential participants contacted us if they wanted to participate or if they wanted further information about participation. Eleven participants were recruited in this way through MDOs. We did not disclose the details of these recruited participants to MDOs.
Following initial interviews, it became clear that many participants were members of close and mutually-supportive informal networks comprising other allegedly impaired doctors. This is consistent with the existing literature (Wile & Jenkins, 2013). Many participants who completed their interviews knew of other allegedly impaired doctors whom they thought might meet the eligibility criteria for inclusion into our study and shared information about our study within those networks. This seemed to generate discussion about our research among these doctors: [Previous participant] tells me it was very cathartic talking to you (P4). In our support group, a few of us have been talking about this project (P6).
Seven participants were recruited through this snowball sampling strategy (Seale, 2012). We believe that referral from peers assisted in overcoming possible fears or hesitancy that allegedly impaired doctors might have faced in participating in this type of sensitive research, especially since we know that privacy concerns and stigma are common reasons why doctors with mental health and substance use challenges avoid seeking help.
We also posted information about our study to the Facebook groups Creative Careers in Medicine and Women in Medicine. Three doctors who responded to these posts participated.
We then determined the eligibility and suitability of potential participants against our inclusion criteria. Two potential participants were excluded because their experiences of regulatory processes were unrelated to their health. One was excluded because they had never qualified or practiced as a doctor. All participants were informed that the research was being undertaken to better understand the experiences of allegedly impaired doctors who were subject to regulatory processes relating to their health.
Consent
We assumed that all participants were capable of consenting and then tested that assumption by engaging in a discussion with participants about the research goals and processes prior to interviews. Based on the first author’s assessment of these interactions, all participants were deemed to be capable of consenting and provided specific written consent. Participants were informed that participation was voluntary and that they could terminate or withdraw from interviews at any time, and could ask that interview transcripts be destroyed within four weeks of interview. No doctors withdrew or changed their mind about participation.
Data Collection
Data were collected using one-on-one in-depth semi-structured interviews. We considered that the use of individual interviews (as opposed to focus groups) was the most appropriate method of data collection, given the need to thoroughly understand each participant’s unique experience and the highly sensitive nature of the ideas and issues to be raised during interviews. Interviews were conducted using a pre-prepared interview guide that was drafted by the first and last authors, both of whom are dual-qualified doctors and lawyers with clinical and legal experience supporting allegedly impaired doctors. Consistent with our interpretative phenomenological approach, the interview schedule was flexible and non-directive to facilitate participants telling their story. The interview guide was not formally piloted but was subject to minor modifications as interviews progressed, to improve the clarity of the questions. All interviews were conducted by the first author, who reconfirmed eligibility and consent prior to interviews and answered any further participant questions about the nature and purpose of the study. Given the first author’s experience supporting unwell doctors, he did not require formal training prior to conducting interviews.
Given the onset of the COVID-19 pandemic before this research commenced, a decision was made that all interviews would occur online. With the exception of one interview that was conducted by telephone at the participant's request, all interviews were conducted using Zoom™ between September 2020 and February 2022. All interviews were conducted in a confidential setting and all interviewees attended alone. The length, breadth and depth of interviews was determined by the interviewee. The use of online interviews also facilitated recruitment across a larger geographical area.
Given that discussion of mental illness, substance use, and regulatory processes could be triggering and traumatic for some participants, a trauma-informed approach to interviews was adopted (Isobel, 2021). Non-threatening, non-judgmental, empathic, and appreciative language and attitudes were employed (Collins & Cooper, 2014). We allowed participants to decide what and how much information to divulge. Concern for the welfare and interests of participants drove the nature and direction of questioning. Interviewees were asked to describe: their motivation for participation; their personal and medical history; their ability to access treatment and support; the nature of the regulatory process they faced and its outcomes; their experience of, and reaction, to the regulatory process; the most stressful aspects of the process; and availability of supports. They were also asked to discuss how they felt about the interview and the research after the conclusion of the interview.
Interviews were audio-recorded, transcribed verbatim, and uploaded into NVivo™ (Version 1.6.1, QSR International, 2020). We then carefully considered whether to offer participants the opportunity to review interview transcripts. On the one hand, we felt that this would empower participants (DeCino & Waalkes, 2018), improve the accuracy and quality of the data (Birt et al., 2016), and contribute to the credibility and trustworthiness of the process (Goldblatt et al., 2011). On the other hand, interview transcript review can be potentially re-traumatising for participants who are “drawn into dialogue with their written speech” (Forbat et al., 2005). Having weighed these competing considerations, we decided to offer all participants the opportunity to review transcripts, particularly given the sensitive nature of our research and the unique legal risks outlined below. Only two participants wanted to review the transcripts. Upon reviewing the transcripts, one had no concerns, while the other (described below) made minor changes to details that they considered could re-identify them. The other 19 participants declined to review transcripts, citing their trust in the research process.
Data Analysis
The six steps of reflexive thematic analysis, as described by Braun et al. (2018), were employed to identify, analyse, and report themes within the data. This provided flexibility and a rich, detailed, and complex evaluation. The first author immersed himself in the data to familiarise himself with the depth and breadth of the narratives provided by participants, by actively re-reading transcripts to search for tacit meaning and patterns, while being reflexive, honest, and vigilant about his own perspectives and beliefs as a doctor. The first and last authors systematically reviewed and analysed transcripts and identified and labelled important sections of text. They identified components or fragments of ideas or experiences arising from the data and then organised these into codes using NVivo™ (QSR International, 2020). Codes were iteratively reviewed for interconnectedness to see where themes should be merged or split. The researchers compared their codes and resolved any discrepancies in the final coding framework by consensus. A copy of our final thematic map is available on request.
The first and last authors regularly reflected and debriefed with each other about their professional positions as doctors and lawyers. Without seeking objectivity, they discussed and recognised that their interpretation of the data could be shaped by their past professional experiences assisting allegedly impaired doctors, their shared ideological commitment to patient safety, and their motivation to improve the health of doctors.
Sample Size
We employed the paradigm of theoretical saturation to determine our sample size (Carpenter & Suto, 2008). We determined saturation by consensus when additional information obtained from subsequent interviews no longer generated new understanding but, instead, continued to fit into existing categories and codes that had already been developed in the data analysis process (Braun & Clarke, 2019). As a result, we stopped recruiting new participants into the study after completing 21 interviews.
Data Security and Privacy
Given the sensitive nature of our research, protecting the privacy of participants was of paramount importance. All documents (including audio-recorded interview files) were stored electronically on encrypted drives on password-protected personal computers. No paper records were generated. Participants signed and returned consent forms to researchers electronically. Only signed consent forms contained the name and signature of participants. No other documents (except our crossover file) contained identifying information. We did not collect date of birth, residential address, or names of employing health services, as this was not required for our study and might unreasonably re-identify participants. We did not use participant names during recorded interviews to preserve their anonymity when interviews were transcribed. Participants were assigned a unique participation code. This was used to identify interview recordings and transcripts. A crossover file (Excel spreadsheet) was used to record the name and participation code of participants, which was stored separately to other data to reduce the risk of re-identification. We used a professional transcription service that signed a confidentiality agreement with the researchers confirming that its employers, contractors and/or agents would not share data with third parties, would securely store and share all data with us, and would delete all audio and typed transcription files within 30 days of transcription. As described above, there was no exchange of personal information about potential or actual participants between researchers and MDOs. As a result, they were unaware who participated, and we were unaware who was approached and declined to participate.
Management of Legal Risks of Participation
In Australia, sections 140 and 141 of the Health Practitioner Regulation National Law Act 2009 (Qld) require an Australian registered health practitioner to notify the Australian Health Practitioner Regulation Agency (Ahpra) as soon as possible if, in the course of practising their profession, they form a reasonable belief that another registered Australian health practitioner has practised the profession while intoxicated, or has placed the public at risk of substantial harm by practising the profession while impaired. This requirement was relevant to our research for three reasons. First, it was foreseeable that participants might disclose notifiable conduct during the course of interviews. Second, medical research is included in the Medical Board of Australia’s broad definition of “medical practice” (Medical Board of Australia, 2012). Therefore, by conducting research interviews, the first author was deemed to be engaging in medical practice. Third, the first author is a registered doctor in Australia and was bound by the obligation to report notifiable conduct that became known to him while practising medicine. Although Aotearoa New Zealand has similar mandatory reporting obligations (Health Practitioners Competence Assurance Act 2003 (NZ), s. 45), the first author was not registered to practise medicine in Aotearoa New Zealand. Therefore, this risk pertained to Australian participants only.
The requirement to report impairment or intoxication was a novel but serious risk inherent in the conduct of our research. Not only could it undermine the trust that participants place in us in conducting this research respectfully and confidentially, but also making a mandatory notification could be particularly devastating and triggering for participants with negative prior experiences of regulatory processes, including mandatory notification. To manage this risk, all consent forms and plain language statements emphasised this risk, which was also specifically discussed at the beginning of all interviews. Participants were strongly encouraged only to discuss matters that had previously been reported to the regulator and participants were informed that the interviewer would interrupt the participant if he suspected that a participant was about to raise such matters during an interview. No new notifiable matters were raised during any of the interviews and no mandatory disclosures needed to be made by the researchers.
Management of Psychological Safety Risks of Participation
Prior to interviews:
Identifying participant distress prior to interview.


Responding to participant distress prior to interview.
During interviews:
Assessing participant distress during an interview.

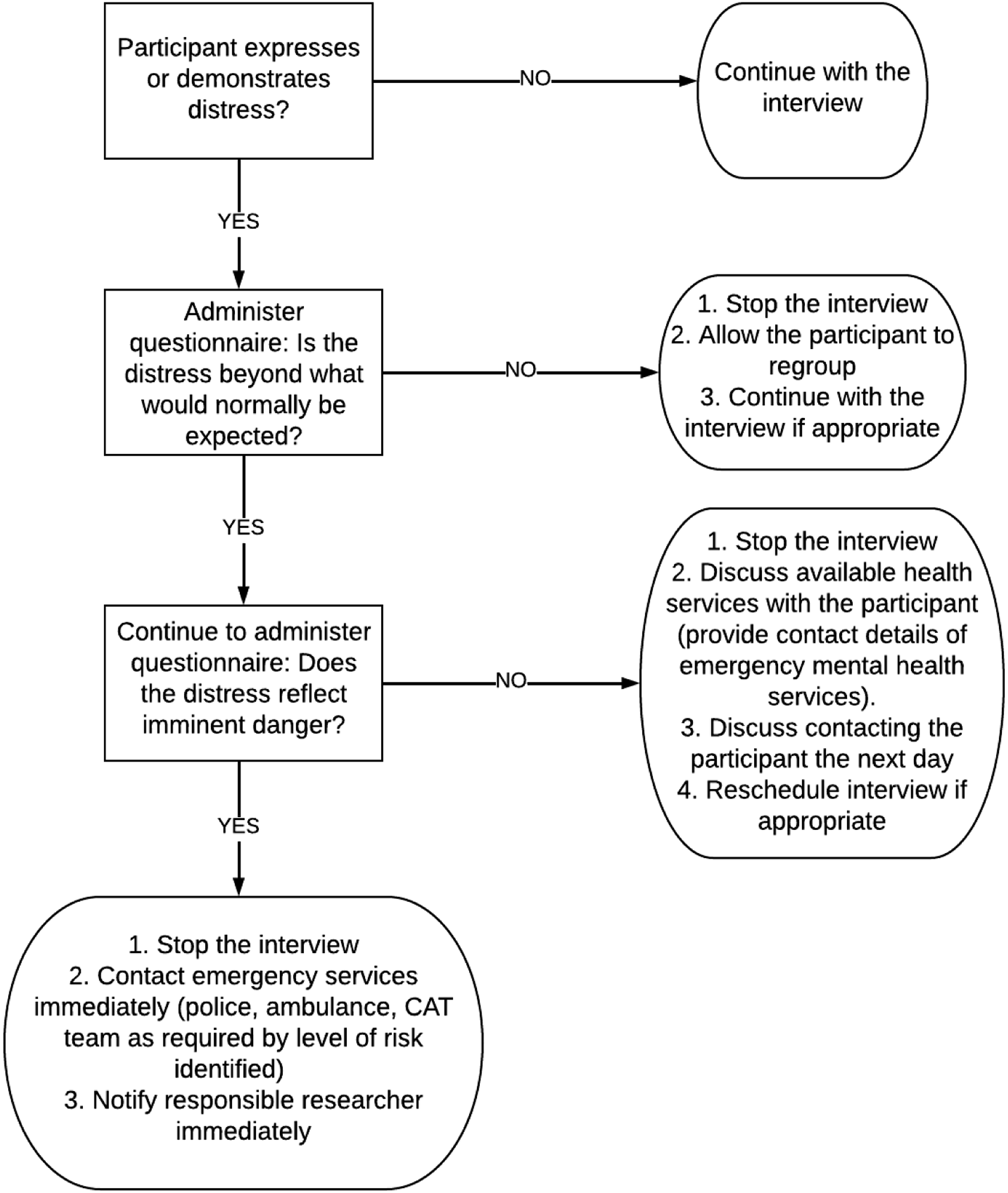
Responding to participant distress during an interview.
After interviews:
Participants were also provided with an opportunity to reflect and debrief following interviews, to identify the possible impact of the research on their wellbeing. If the interview raised any issues for participants, they were provided with the details of support and referral services and were encouraged to contact their treating health practitioner. In line with existing literature, we felt that it was important to spend time with participants after interviews had concluded to demonstrate that we were interested in their wellbeing, rather than just their narratives (Warren, 2012). All participants were contacted by telephone or email 48 hours after interviews to check in with them and ensure that they were not experiencing any distress or ongoing upset. No participants expressed distress or ongoing upset when contacted.
Participant Reimbursement
Financial reimbursement of participants in sensitive research acknowledges the valuable contribution that participants make in advancing scientific knowledge, and recognises the time, inconvenience, expenses, and risks of their participation (Winter et al., 2022). However, in certain situations, reimbursement may undermine voluntary consent by inducing vulnerable participants to participate without full regard to the benefits and risks of participation (National Health and Medical Research Council, 2019). We decided not to compensate participants because: 1) we were concerned that creating a financial incentive to participate might influence their decision to participate and that this might skew their response to questions assessing their motivation for participation; and 2) as interviews were conducted online, we considered it unlikely that participants would incur any (significant) expenses associated with participation.
Dissemination of Results
We undertook to make the results of this work available to participants. Following publication of the primary results of this work elsewhere (Bradfield et al., 2023a; 2023b), there was notable interest from the local Australian medical press and medical community (Attwooll, 2023; RACGP, 2023; Sparke, 2023; Swannell, 2023). Copies of the final published manuscripts, related news articles and media releases were sent to participants for their information. We felt it was important to demonstrate respect to participants by showing how the risks they assumed by participating translated into tangible findings and policy reform recommendations (Doernberg & Wendler, 2016). Moreover, as participants were doctors, we felt they would be especially interested in reading about our scientific methodology, results, and conclusions (Gulland, 2015).
Five participants responded to the first author following dissemination of this material. Four participants responded positively and expressed satisfaction with the project outcomes. They felt that their thoughts and views had been accurately incorporated into the manuscripts and were pleased to see the medical press reacting favourably to their experiences. However, one participant expressed dissatisfaction with the conclusions drawn. They felt that their specific circumstances had not been adequately captured and reflected in the manuscript. Support and an opportunity to explain the limits of thematic analysis were offered to this participant.
Ethics Approval
All procedures contributing to this work comply with the National Health and Medical Research Council’s National Statement on Ethical Conduct in Human Research (2018) and with the Helsinki Declaration of 1975 (World Medical Association, 2013). All procedures involving human participants were approved by the University of Melbourne Human Research Ethics Committee (approval number 2056342). We did not experience any challenges obtaining ethical approval to conduct this research, with no major amendments requested. We believe that this was due to: 1) our focus on ensuring that participants would be informed of the key risks of participation; 2) our detailed adverse event protocol that outlined how we intended to mitigate those risks; and 3) the steps proposed to protect the security of data collected. In recent years, there has also been a shift in the general attitude and risk appetite of ethics committees to the conduct of sensitive research. Ethics committees are now more willing to balance risks of participation against the potential benefits of participation and participants’ free choice to accept some risks (Bredal et al., 2022).
Results
Demographics
Characteristics of participants.

aSome doctors faced regulatory processes in more than one jurisdiction.
bSome doctors had more than one illness type.
Participants’ Attitude and Approach to Our Research
We began our interviews by asking participants to tell us what motivated them to participate in our research. We felt that this allowed participants to begin their story on their terms. Despite the potential personal and professional risks of participating in our research, we were struck by how open and candid participants were during the interviews and how driven they were to participate. Not only did many speak enthusiastically about our research and the benefits that they hoped it would achieve, but some spoke for more than the 90 minutes anticipated. The mean duration of interviews was 91 minutes, and the median was 98 minutes. Interviews ranged from 44 minutes to 128 minutes. Two participants spoke for over two hours in one sitting, despite being offered the opportunity to split the interview into multiple sittings. In all, we felt that this bespoke a trusting researcher-participant relationship. Well done for what you’re doing. I think it’s fabulous (P1)
Some felt that they had been unable to tell anyone else their entire story. They did not want to burden family members, or did not feel that they could tell their legal representatives how they really felt. I haven’t talked to anyone about this before. I know you’re not my counsellor, you’re not my GP, you can’t offer me any sort of help in this, but I just needed to unload it, get some of this off my shoulders…When I’m talking to my lawyer, I can’t cry. I can’t do any of that sort of emotional stuff. It’s all business and it’s all trying to save my backside (P3).
Related to this was a strong desire for participants to be heard. I have a story that needs to be told, and I want to get it out there (P4)
Many recounted distressing experiences dealing with regulatory processes and wanted others to hear about this because they felt silenced by regulators and regulatory processes. I’m grateful to you for undertaking this project and for me to have had an avenue to air things. It’s a good opportunity to get things heard. It can be difficult when dealing with the regulator to get anyone to hear you (P2). Addicts know what triggers them and what causes their downfall and relapse, but we are just never listened to (P6). I made formal complaints both to Ahpra and the Ombudsman [about my regulatory experiences], that were both whitewashed (P11).
Some participants felt that disclosing their experiences to others made them susceptible to skepticism and marginalisation by peers, colleagues, and employers on account of their illness or the stigma of having received a regulatory notification. No one listens if you complain about the [regulator] because people assume that you must have done something wrong (P7). People are a bit more open about depression. But it’s still not accepted that doctors might be alcoholics or heroin addicts (P6).
Many participants reported that the fear of regulatory processes and notifications resulted in them avoiding or delaying treatment, resulting in worse health outcomes. Some participants spoke of their desire to participate in this research as a means of breaking down these perceived barriers, destigmatising mental illness and overcoming the fear. They wanted mental illness among doctors to be more visible and acceptable. One of my roles as a doctor with a depressive illness is that I should be open about it to destigmatise it (P21). The main reason why I wanted to participate in this study is because I think the regulatory process deters doctors from getting help when they have a mental health condition, for fear of punitive action and disciplinary action. I don’t want somebody else to seek help too late because they were afraid, because that’s exactly how I felt (P17).
Others talked about regulatory processes being extremely distressing and that they knew of some doctors who had suicided as a result of their experiences. Therefore, another commonly cited reason for participation was a desire to help current and future doctors facing regulatory processes relating to their health. By participating in this research, they wanted us to contribute to shared knowledge that could support recommendations to improve the lives of other doctors with mental health or substance use challenges. I don’t want this to happen to others…people die because of [regulatory processes] and people’s families suffer (P4). A systematic voice…will be most useful in preventing future doctors from going through the kind of traumatic [regulatory] experiences that I’ve been through (P7). I’m really glad that you’re doing this research because I’m sure other doctors have been deprived of basic human rights through these processes (P10).
Another key theme was that many participants complained about the slowness and perceived inhumanity of the regulatory process. They wanted to effect system reform by participating in this research and forming a united collective voice. I hope discussing my experience might add to the impetus for change (P7). I think it’s an important area, and a scholarly research project such as this may assist [the regulator] get it right (P12). We can’t have doctors dying because of [regulatory processes]…there’s got to be better ways to do it (P13). There’s got to be another solution out there… I can’t just forget about all the horrifying things that have just transpired in the last five years. If that’s the solution, then there’s no accountability and no-one’s going to learn anything (P15).
Finally, one doctor wanted to participate because they enjoyed positive experiences when dealing with the regulator in their jurisdiction. They were concerned that many participants would want to discuss negative experiences and felt that it was important for the researchers to receive balanced viewpoints and to recognise that negative experiences were not universal. What happened to me was short, brief, very easy, and sensible… straightforward and boring. I think it’s important you have the positive angle as well. Simple things can get dealt with well (P20).
Participants’ Reactions to Participating
We also asked participants to tell us how they felt after disclosing their story. Feedback was overwhelmingly positive. Participants expressed gratitude at being able to tell us their stories and found the experience to be therapeutic. It hasn’t brought up any traumas or anything like that. I feel good that I’ve been able to talk about it. I feel invigorated. You’ve inspired me to do some advocacy work in this area (P2). It was good to get it out and talk to someone who doesn’t really know me (P3). I feel good because I had the opportunity to tell my story (P4). Disclosing my story to you has helped me (P13).
Many participants told us that they had never previously told anyone else their story or their emotional reaction to it. Some experienced mild upset as a result of the interview, but did not experience extreme distress or retraumatisation. I probably haven’t told anyone else the whole story from beginning to end before, so it’s a bit emotionally raw. I’m still upset at what happened to me, but I will be okay (P16).
The remainder of participants experienced neutral responses to the interview. For doctors involved in support groups, they were experienced at narrating their stories and reflecting on their thoughts and emotions. I’m not going to fall apart. I’ve been through worse than this (P4). You get used to sharing your story at the support group meetings (P6). You haven’t traumatised me (P7). I feel comfortable talking about my story because I attend a lot of NA meetings (P17).
One participant expressed concern that, given the uniqueness of their story, they might be re- identifiable. How could I trust anyone?… I’m really scared that my story is so unlike anybody else’s. I am starting to question how on earth you could possibly deidentify this (P15).
This participant was reassured that interview transcripts and publications would be fully deidentified. They were provided with a copy of the interview transcript once it was prepared to assist them verify whether they thought that any of the information contained in it might be re-identifiable. They made some minor amendments and felt reassured by this and reconfirmed their consent to the information in the transcript being analysed in this study.
Discussion
Summary of Findings and Comparison to Prior Research
Allegedly impaired doctors have few opportunities to voice their opinions about regulatory processes that directly impact on them. Our research sought to overcome this. We describe in detail our approach to conducting novel qualitative research with allegedly impaired doctors, a group known to be hard to reach. We also report the thoughts and ideas of participants about our research before and after their involvement. We found that a desire to be heard, to help other allegedly impaired doctors, and to change perceived unfair systems were the most common reasons for participation. This is consistent with prior qualitative research examining the experiences of research participants with mental illness (McDonald et al., 2013), and of unwell doctors specifically, which shows they want to actively participate in research to improve their quality of life and that trust is a key ingredient in ensuring that this can occur successfully (Taylor et al., 2021).
Reflecting on their involvement after being interviewed, many participants felt positive about their participation, and expressed a sense of gratitude, relief, inspiration, and catharsis. Many found the experience therapeutic and were grateful for the opportunity to tell their entire story for the first time. This is consistent with a prior systematic review of the experiences of participants in psychiatric research, which found that reactions were overwhelmingly positive and that most participants perceived benefits from their involvement (Jorm et al., 2007). A small minority of participants in other psychiatric research studies indicated some unexpected upset, but of those, very few regretted their participation or rated the overall experience as negative (Newman & Kaloupek, 2004). In our study, one participant was dissatisfied without our final conclusions and recommendations upon reading our other published manuscripts (Bradfield et al., 2023a; 2023b).
Unlike prior research involving unwell doctors, this research involved talking to allegedly impaired doctors about their experiences of regulatory processes relating to their health. For many participants, they disclosed a sense of shame, anger, and disillusionment with regulatory processes, including mandatory reporting (Bradfield et al., 2023a; 2023b). Therefore, our a priori hypothesis was that this would be a potentially significant barrier to participation in our research because there was a foreseeable risk that participants might disclose information during interviews that was notifiable by the researchers to regulators. On the contrary, participants were highly motivated to participate, even when warned of this risk, and still spoke frankly about their experiences. Many also recommended participating in our research to their colleagues with health challenges. We attribute the positive reactions of participants to several factors.
First, as part of our application for ethical review, we carefully considered the potential risks for participants and the ways in which we could minimise and manage those risks. This involved the preparation of detailed supporting information to assist participants make an informed decision about participation early on. This information included a recruitment letter, advertisement, plain language statement, consent form, interview schedule, data management plan, adverse event protocol, and an organisational letter of support. Through careful planning, preparation, and communication with potential participants about possible research risks, we sought to facilitate an environment of mutual trust that respected potential participants and allowed them to feel safe disclosing personal and sensitive information and recounting distressing situations, even though it involved some personal, emotional, and legal risk to themselves.
Second, we offered all participants the opportunity to review transcripts. Consistent with prior studies (Rowlands, 2021), this may also have allowed participants to more freely engage in our research, by giving them greater control over the future use of sensitive data generated from their interviews, thus reassuring them about the trustworthiness of the process. Finally, two of the researchers (including the first author who conducted all interviews) were dual-qualified doctors and lawyers with clinical and legal experience working with unwell doctors. We believe that their insights and skills helped them build trust and rapport with participants before, during and after interviews, and contributed to a sense of participants feeling “safe” throughout the research process on account of a sense of shared identity between researchers and participants (Walton et al., 2022).
Strengths and Limitations
This is one of a handful of qualitative interview studies that recruited allegedly impaired doctors. It is the first to examine their experiences of regulatory processes. It revealed novel findings of relevance to allegedly impaired doctors, their treating practitioners, MDOs, medical regulators, and policy makers. The semi-structured and in-depth nature of our interviews facilitated unanticipated findings to emerge. There were no adverse outcomes reported and no participants were excluded because of distress or inability to consent. The sample studied was also relatively heterogeneous, with almost equal gender representation, and representation from a range of ages, specialties, and geographical locations. Although we have no information about any potentially eligible participants who declined to participate, we believe that the combination of purposive and snowball sampling techniques reduced potential coercion and best protected confidentiality. We also believe that it may have assisted in overcoming privacy or mandatory reporting fears that might have otherwise dissuaded some people from participating.
One possible limitation is that only three participants were from Aotearoa New Zealand. Although Aotearoa New Zealand shares many similarities to Australia in terms of its regulatory framework for impaired doctors (Bradfield et al., 2022; 2023c), this may have implications for our claims of saturation beyond the themes identified and may require further research.
Implications for Research and Policy
While many doctors consider research to be as important as seeing patients (Gulland, 2015), there is little research examining the attitudes of doctors to their own involvement in research as participants. In this study, we found that allegedly impaired doctors valued participating in our research and expressed an interest in learning about our research outcomes, because they perceived personal and professional benefits. Consistent with their sense of social conscience, they wanted us to use their experiences and perspectives to improve the quality of life for other doctors with mental health and substance use challenges. Our findings suggest that we should be promoting the participation of doctors generally, not just allegedly impaired doctors, in research that directly impacts on them. To do this, we need to impart the benefits, not just the risks, of research participation. Indeed, the attitude of human research ethics committees can be pivotal in determining whether research into hard-to-reach populations is viable. Historically, ethical review processes have attracted criticism by adopting a risk-averse approach that prioritised the protection of individuals from research risks over the need for greater inclusion (Rennie et al., 2019), and the right of an individual to voluntarily assume risks associated with research participation (Brandenburg et al., 2021). However, in recent decades there has been growing recognition in the US (McDonald et al., 2013), Australia (Ries et al., 2020) and New Zealand (Douglass & Ballantyne, 2019) of the individual and social benefits of facilitating those with lived experience of traumatic events to participate in sensitive and potentially distressing research that is carefully planned and monitored. Indeed, in the case of our research, we believe that participants and the communities they serve will benefit from the knowledge gained by better understanding the potentially adverse aspects of regulatory processes that are otherwise designed to protect the public.
Conclusions
The Lancet recently described doctors’ health as a “global health-care predicament” demanding a system-wide response (Horton, 2019). We know that fear of regulatory processes is one of the most common reasons why doctors avoid or delay seeking help for mental health or substance use challenges (Beyond Blue, 2019). Therefore, addressing these problems necessitates research that engages directly with allegedly impaired doctors to better understand their experiences of regulatory processes. However, allegedly impaired doctors are difficult to reach. Our study demonstrates that inroads can be made into this predicament through carefully constructed research that limits the potential for coercion and privacy breaches and harnesses the expertise of researchers with experience in talking to doctors about their health and wellbeing. However, doctors are also a highly motivated cohort, many of whom are instinctively driven to help others and address perceived social injustices that they see as barriers to achieving health. Researchers and ethics committees need to focus not just on the risks, but also the benefits, that may flow from this type of research. Allegedly impaired doctors face barriers in accessing early interventions and this contributes to poor health outcomes for doctors and their patients (Bradfield et al., 2023c). It is critically important that researchers seek to defeat these barriers through the active and inclusive participation of allegedly impaired doctors and by learning from their perspectives and experiences. Only then can we advance the health and wellbeing of doctors.
Footnotes
Acknowledgments
We thank the Medical Indemnity Protection Society (MIPS), Avant Insurance, and MDA National for their assistance in recruiting doctors into this study. We also sincerely and gratefully acknowledge the participants in this study, who generously gave up their time to candidly discuss challenging and potentially upsetting experiences. Without their input and openness, this project would not have been possible. Finally, we acknowledge the traditional custodians of the Aboriginal and Māori lands on which the researchers and participants engaged in this research.
Author’s Contributions
All authors contributed to the study conceptualisation and design of the research. OB contributed to literature review, application for ethics approval, data collection, data analysis and writing and editing of the manuscript. MB was involved in data analysis, literature searching to inform the manuscript and contributed to writing and editing the manuscript. All authors reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OB was previously employed by Avant Insurance and is currently employed by MIPS, but was not employed by either organisation when this research was conducted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Owen Bradfield was funded by the Australian Government on a 2020 Melbourne Research Scholarship, by the Australian-American Fulbright Commission and the Kinghorn Foundation on a 2020 Fulbright Future Scholarship, and by the Avant Foundation on a 2021 Grant. Matthew Spittal was funded by the Australian Government on an Australian Research Council Future Fellowship (FT180100075). Marie Bismark was funded by the Australian Government on a National Health and Medical Research Council Investigator Grant (APP1195984).
Ethical Approval
This study was approved by the Medicine and Dentistry Human Ethics Sub-Committee of the University of Melbourne Human Research Ethics Committee (approval number 2056342).
Informed Consent
Participants gave consent to use de-identified data in scholarly publications and presentations.
Data Availability
The data generated and analysed for this study are not available due to participant confidentiality reasons. Reasonable requests for further information may be directed to the corresponding author.
