Abstract
Potential participant distress is often unaccounted for in qualitative research, where topics are not pre-determined to be categorically sensitive. Additionally, researchers sometimes project their own distress with sensitive topics onto participants, inappropriately truncating or terminating data collection encounters. As a result, researchers may over- or under-react to distress, in ways that fail to attune to and meet participants’ individual needs and that are not consistent within research teams. In our proposed framework of “The Participant-Researcher Dynamics of Distress,” we advance the Dynamic of Participant Centeredness to move beyond notions of categorical sensitivity, and in alignment with principles of research ethics, we developed the Qualitative Research Distress Protocol (QRDP). The QRDP is a comprehensive tool that we co-created with an oncology social worker, for a study exploring patients’ and clinicians’ experiences of virtual supportive cancer care during COVID-19. We elaborate on the use and application of the QRDP in team science, providing examples from our experience training novice researchers and conducting interviews for the Virtual Supportive Cancer Care Research Study. This novel protocol stands apart from other available distress protocols in taking a non-categorical approach to assessing participant sensitivity and addressing distress during qualitative interviews. The QRDP can be evaluated in and adapted for use in other qualitative research studies, especially those undertaken by collaborative and diverse research teams.
Keywords
Qualitative researchers have an ethical obligation to identify, minimize, and address the risks of research-related distress. In response to human rights atrocities resulting from research endeavors, institutional and ethics boards of approval aim to ensure the ethical nature of proposed inquiry (Ahern, 2012; Snyder & Mueller, 2008). As part of their ethics board proposals, researchers often develop a number of preparatory strategies to address potential participant distress. For example, researchers may attempt to eliminate the possibility of distress by screening and excluding from participation potential research participants perceived to be at higher risk of harm (Draucker et al., 2009). To reduce the likelihood of distress for participants who are included in research studies, researchers may construct interview guides in a way that acknowledges the sensitivity of research topics and vulnerability of participants (Rogers et al., 2021). Additionally, researchers may carefully plan who will be responsible for conducting the interviews based on interviewer attributes such as clinical training (Teachman & Gibson, 2013). These preparatory strategies are helpful for minimizing the risk of potential distress in interviews. However, interviewers also need tools and resources that can guide them in adapting and responding to unforeseen participant distress that emerges during an interview. As Smith expressed, “to interview and then leave someone in emotional distress without adequate support or safeguards is morally wrong” (Smith, 1992, p. 102).
Sometimes in qualitative research, researchers find themselves unable to “deal with participants’ emotional pain” (Ahern, 2012; Burnell & O’Keefe, 2004). This experience of emotional arousal and intensity (Morse et al., 2008) often occurs when interviewers identify with the participant because of their own personal experiences (Dickson-Swift et al., 2009). By consequence, interviewers may become overly emotionally invested in the participant or overly distant, not “able to hear what is being said” (Morse et al., 2008, p. 205). As one interviewer expressed in Dickson-Swift and colleagues’ study, “I nodded to let [the participant] know that I was listening but all the while I was feeling ill, in my head I was telling her to stop, wanting her to stop talking, but I kept on smiling and nodding” (Dickson-Swift et al., 2009, p. 68). In some instances, interviewers may project onto participants their own perceptions of emotional reactions and feelings of distress (Holmes, 2014). For example, an interviewer may prematurely stop an interview, which might be “perceived as rejection of the participant and his/her feelings” (Morse et al., 2008, p. 208). Researcher countertransference can result in over-attending to some participants’ presumed distress, under-attending to others’, thereby attending to all participants’ experiences in a manner that is not participant-centered.
Distress protocols are a comprehensive and systematic means of safely and consistently assessing and responding to potential and actual participant distress rather than researcher countertransference. Published research distress protocols outline procedures to follow in the case that participants present with adverse reactions during interviews (Draucker et al., 2009), avoiding reliance on individual interviewers’ “risk-benefit perspectives” (Newman & Kaloupek, 2004, p. 383). The protocols – informed by principles of nonmaleficence, autonomy, and justice – guide interviewers to intervene if participants become distressed, providing language for offering support and stopping or continuing an interview. In addition, protocols may include lists of available resources such as crisis hotlines. Importantly, distress protocols help to protect participant wellbeing and mitigate short- and long-term risks of participation in qualitative research.
While existing distress protocols enable a consistent, ethical response to participant distress, these protocols have been designed and used mostly in studies pertaining to research judged sensitive and populations deemed vulnerable. Ethics review boards emphasize assessing the appropriateness and safety of studies involving economically, socially, psychologically, or physically vulnerable individuals (Lockwood et al., 2018; Widom & Czaja, 2005). Researchers who develop and use distress protocols tend to study “emotionally charged topics” (Draucker et al., 2009, p. 343) such as self-harm (Lockwood et al., 2018), homelessness (Widom & Czaja, 2005), severe mental health issues (Boothroyd, 2000), sexual risk behaviors and psychosocial distress (Craig et al., 2019), and adolescent dating violence (Draucker et al., 2009). Minimizing research-related distress for participants whom researchers assume to be at elevated risk – either by virtue of their sociocultural position, the nature of the research topic, or the countertransference of researchers’ feelings – is certainly critical. However, we posit a need to break free from categorical notions of sensitive topics and vulnerable populations, as “all interviews are potentially emotional experiences for participants” (Mitchell & Irvine, 2008, p. 35). Qualitative researchers hold an ethical obligation to attend to distinct and individualized emotional reactions and needs of research participants in all studies and populations.
In this article, we look beyond sensitivity as a categorical notion and toward ethically sensitive research conduct for all human subjects participating in qualitative research. We developed a comprehensive distress protocol tailored to patient and clinician participants for a study exploring virtual supportive cancer care during COVID-19. We begin by presenting our framework “The Participant-Researcher Dynamics of Distress” upon which we built the Qualitative Research Distress Protocol (QRDP) within the purview of Beauchamp and Childress’ principles of bioethics (2019) for research purposes. We describe the process of developing the QRDP, attending to the principles of justice, nonmaleficence, autonomy, and beneficence. We elaborate on its use and application in team science and conclude with a summary of implications for replication in other studies.
Framework of Participant-Researcher Dynamics of Distress
“The Participant-Researcher Dynamics of Distress” framework elucidates two different dynamics underlying how researchers perceive and address distress. The first, the Dynamic of Categorical Sensitivity, represents a dominant approach to addressing participant distress in qualitative research (Boothroyd, 2000; Craig et al., 2019; Draucker et al., 2009; Lockwood et al., 2018; Widom & Czaja, 2005). Relying on notions of categorical sensitivity, this approach results in overlooked participant distress as well as anticipated distress misaligned with participants’ actual distress experiences. The Dynamic of Participant-Centeredness is intended as an alternative to the Dynamic of Categorical Sensitivity. The Dynamic of Participant-Centeredness leads to accurate perceptions of participant distress by moving beyond categorical sensitivity and anchoring researchers’ responses in needs of individual participants. Figure 1 illustrates the foundational and consequential differences between the two approaches. Framework of participant-researcher dynamics of distress.
The Dynamic of Categorical Sensitivity describes four possible participant archetypes based on their status of categorical sensitivity and their actual experience of distress during a qualitative interview. In this dynamic, researchers correctly align presumed and actual participant distress in only two out of the four archetypes: the first, in which researchers inaccurately address participant distress that the participant does not actually experience, and the fourth, in which researchers overlook the distress of participants who are not labeled categorically sensitive.
The Dynamic of Participant-Centeredness categorizes participant archetypes solely by their actual experience of distress during a qualitative interview. In this dynamic, researchers correctly align presumed and actual distress of all four participant archetypes thus accurately addressing distress participants experience if, and only if, they indeed experience it. Compared to the Dynamic of Categorical Sensitivity, the Dynamic of Participant-Centeredness offers a foundation for developing tools that more accurately and consistently address distress in qualitative research.
Developing the Distress Protocol
Guided by the Dynamic of Participant-Centeredness, we developed a comprehensive and adaptable distress protocol for the Virtual Supportive Cancer Care Research (ViSuCaRe) Study at [blinded] in the Northeastern United States. The aim of this dimensional analysis study was to examine the nature of virtual supportive cancer care during COVID-19 from the perspectives of patients, care partners, and clinicians engaged in care. From May to December 2020, members of the ViSuCaRe Study team conducted 60-minute semi-structured virtual interviews with 17 clinicians, 18 patients and three care partners.
Prior to data collection, we collaborated with the oncology social worker on the study team to co-create two distress protocols tailored to participant groups – patients/care partners and clinicians. For each protocol, we developed three sections: a triage pathway with scripted language, a tiered list of available referral resources for participant support, and directions for reporting participant distress with contact information for the study Co-Principal Investigators.
The four-step Triage Pathway offers interviewers structured language to use in cases where participants express or appear to be in distress. To script the Triage Pathway, we drew on therapeutic communication principles from our experiences and training in nursing and social work. We created a schematic prompting stepwise assessments and corresponding actions to align the pace and continuation of interviews with participants’ emotional needs (see Figure 2). Triage pathway of the qualitative research distress protocol.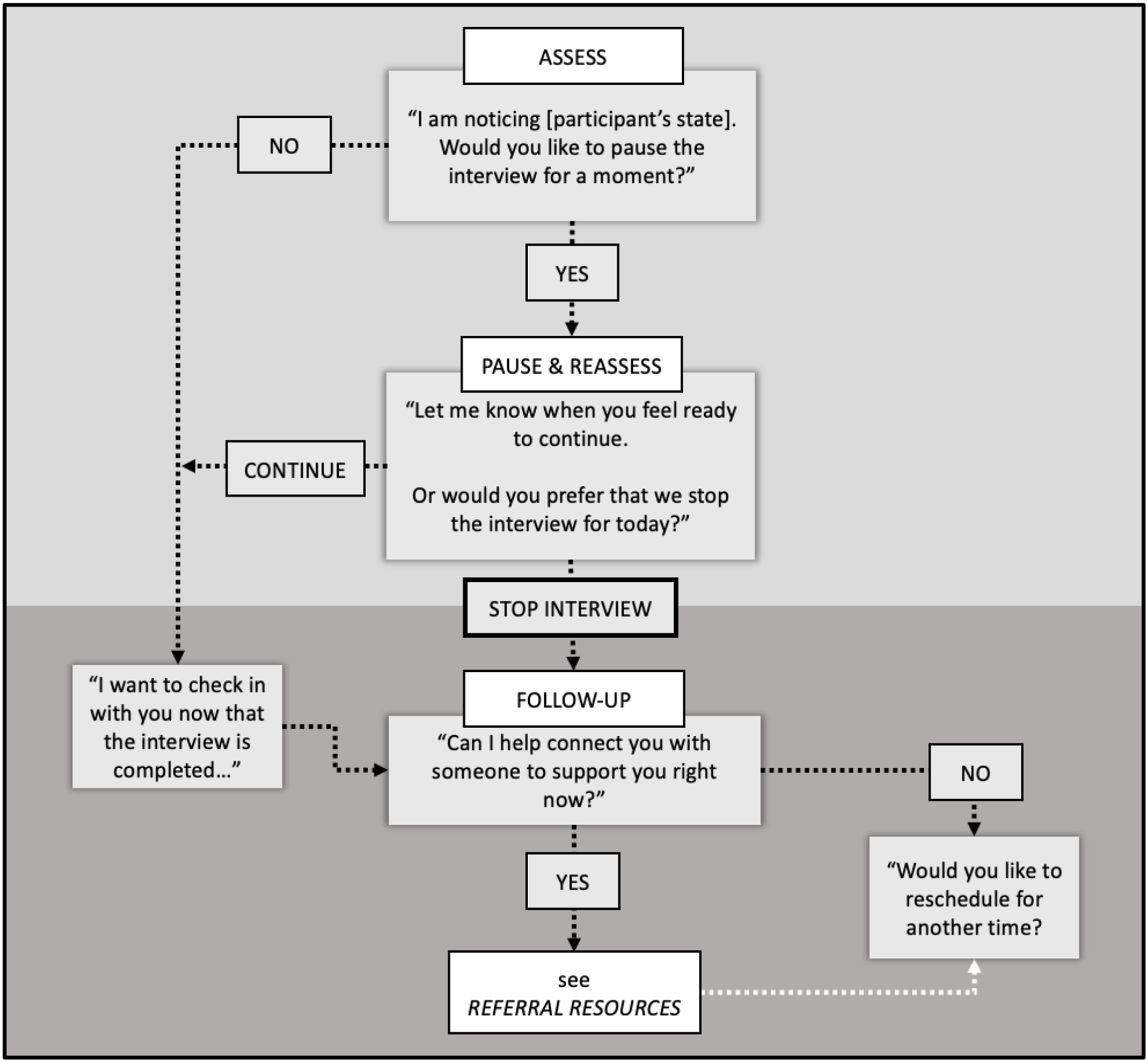
Example Referral Resources of the Qualitative Research Distress Protocol.

Reporting Directions provides the contact information for the study leaders. In the case of participant distress or referral to services, interviewers would be responsible for contacting the study leaders to notify them and determine next steps. To create Reporting Directions, Co-Principal Investigators provided current contact information where they could be reached promptly (see Figure 3). Example reporting directions of the qualitative research distress protocol.
Aligning the Qualitative Research Distress Protocol with Principles of Research Ethics
The development of the QRDP as a whole was built on the research ethics principle of justice. Justice, in the context of qualitative research, calls for the equitable distribution of burdens and benefits of research among different groups of people (Barrow et al., 2021). In research on “sensitive topics,” researchers may inadvertently exclude participants they presume to be at high risk of distress, thereby shifting a disproportionate burden of research participation onto others. Simultaneously, excluding categorically sensitive groups from participating in research limits the transferability of findings in these populations. This practice of categorically excluding certain potential research participants disadvantages both those assumed to be at high risk and low risk of distress. For example, Carter and colleagues found that participating in research – even research that explicitly focused on a potentially psychologically distressing topic like suicide-related behaviors and experiences – was not associated with increased distress or harm to participants (Carter et al., 2020); adverse reactions to participation are rare (Boothroyd, 2000). A growing body of evidence even suggests that participating in ‘sensitive’ research can decrease distress and even have a beneficial therapeutic effect on participants (Carter et al., 2020; Eynan et al., 2014; Jorm et al., 2007; Legerski & Bunnell, 2010; Omerov et al., 2014). The QRDP reduces the prevalence and extent of this type of research participation inequity. By preparing researchers to assess and respond to individual participants’ distress, the QRDP ensures they do not prematurely exclude relevant groups of potential research participants in their qualitative studies.
In creating the Triage Pathway, Referral Resources, and scripted language of the QRDP, we attended to the research ethics principle of nonmaleficence. Oftentimes understood through the principle of beneficence, nonmaleficence pertains to the avoidance of preventable harm, discomfort, or exploitation of research participants (Johansen et al., 2008). In the context of qualitative research, psychological distress is a primary threat to nonmaleficence, thus as a whole, the QRDP upholds this ethical principal. The protocol specifically reduces the threat of psychological harm by preparing researchers to respond to instances of participant distress with necessary referral resources.
The research ethics principle of autonomy was foundational to the construction of the QRDP Triage Pathway. Autonomy, or respect for persons, refers to the protection of research participants’ rights to self-determination and human dignity, and it includes elements such as informed consent, voluntary participation, and freedom from coercion (Barrow et al., 2021). The QRDP protocolizes the process of assessing and reassessing distress in a way that prioritizes participant autonomy and operationalizes the practice of informed consent as continuous and ongoing rather than limited to a single transaction. While assessing participants’ distress is critical, interviewers should aim to avoid immediately stopping interviews in which participants become upset or tearful as this limits participant agency. By prompting researchers to check-in with them about if, when, and how participants would like to proceed, the QRDP protects the autonomy of potentially distressed participants.
The QRDP as a whole maximizes the potential for participants to experience benefits of research participation. Beneficence, in the context of research ethics, refers to the ways in which participating in research can benefit participants, while protecting and promoting their welfare and safety (Beauchamp, 1990). By upholding the principle of justice, the QRDP helps to assure inclusion of all interested participants, thereby preventing premature exclusion of participants who could benefit from participation. Standardized directives promote the principle of nonmaleficence and by consequence increase researchers’ confidence to pursue data collection procedures in a fashion that protects all research participants. The Triage Pathway’s participant-centered approach champions autonomy, reducing researcher-driven truncation or termination of interviews. Taken together, the integration of justice, nonmaleficence, and autonomy into the Triage Pathway, Referral Resources, and Reporting Directions ultimately maximizes the opportunity for participants to benefit from the experience of participating in research. While research qua research cannot claim to guarantee therapeutic outcomes, a growing body of evidence suggests that participating in qualitative research frequently has positive therapeutic effects on participants (Carter et al., 2020; Lockwood et al., 2018). Grounding and growing the QRDP in ethical principles optimizes the opportunity for beneficence to the extent possible in the qualitative research setting.
Using the Distress Protocol for Team Science
Rigorous qualitative inquiry often calls for the concerted effort of multiple critical and collaborative scholars. The QRDP supports this team science approach by offering a standardized tool that can be used by multiple researchers. We used the QDRP in three key phases of the ViSuCaRe study where the team science model can prove challenging: training research assistants with varying levels of experience, standardizing interviewer responses to participants’ emotional reactions, and debriefing difficult situations.
Conducting team science with researchers who have varying levels of experience requires comprehensive training that is adaptative and provides opportunities for formative and summative feedback. As study Co-Principal Investigators and nurse scientists, we trained members of the data engagement team – which included undergraduate and doctoral nursing students, a clinical nurse administrator, and a premedical postgraduate student – to use the QRDP. We designed role-playing exercises to give data engagement team members an opportunity to practice employing the QRDP, drawing on simulation education techniques (Fey & Kardong-Edgren, 2017; Waznonis, 2015). This interactive training supplemented institutionally required instruction on informed consent and methodologically necessary preparation in qualitative interviewing. The straightforward and accessible nature of the QRDP facilitated training researchers – those new to practice and those with long-developed habits or preferences – in a way that was efficient and consistent.
While evidence-based and effective training exercises are key to preparing a whole team to conduct interviews, even the most realistic simulation cannot fully prepare interviewers for the range of situations that may be encountered during actual data collection. In the second key phase of the ViSuCaRe study, multiple members of the data engagement team conducted interviews with patients, care partners, and clinicians. Interviewers included the QRDP in the group of materials they had available during interviews, alongside the informed consent document, interview protocol, field note log, and post-interview demographic questionnaire. Having a tangible tool accessible and immediately ready to deploy if needed reinforced and provided real-time support for applying learnings from initial QRDP training.
Processes for collating reflections during data collection enable qualitative research teams to benefit from emerging procedural insights. The QRDP supports post-interview debriefing by directing interviewers to connect with study leadership after interviews with instances of participant distress. For example, in the ViSuCaRe Study, when one clinician participant expressed discomfort at the beginning of the interview about her participation in the study, the interviewer paused and assessed her distress. The interviewer effectively attended to the participant’s requests for additional measures to protect her confidentiality before resuming the interview. Afterwards, the interviewer spoke with the Co-Principal Investigators in an effort to both debrief and determine a plan to protect the participants’ confidentiality. Based on this debriefing session, we limited access to her audio-recording and complete transcript to only the interviewer and study Co-Principal Investigators as requested by the participant. The QRDP supported interviewers in the iterative and adaptive learning that occurs during team-based data collection.
Discussion
Distress protocols are integral to carrying out ethical research endeavors aligned with a Dynamic of Participant-Centeredness, guiding researchers in more accurately and consistently addressing participant distress. The QRDP uniquely aids researchers in bringing attention to potential distress all participants may experience regardless of their presumed categorical sensitivity. Using this tool can help researchers align their study procedures with the principles of justice, nonmaleficence, autonomy, and beneficence and standardize how interviewers respond when distressing situations arise during data collection.
We intend the distress protocol to be adaptable and transferable tool to use in other qualitative studies (See Appendix 1). Researchers can tailor the Triage Pathway to their distinct participant groups within and across studies. Specifically, researchers can adapt the phrasing to meet the developmental, cultural, or language needs of their participants. In modifying the Referral Resources, researchers can consider resources in their institutional settings (e.g., participants’ employee health services, participants’ health or social care providers), local settings (e.g., community agencies, support groups), and national contexts (e.g., crisis hotlines, public health departments). Reporting Directions can be updated with the study leaders’ contact information and institution-specific processes for ethics board notification. As a whole, researchers can also redevelop this entire tool with other ethical frameworks rooted in traditions outside of a Western context of bioethics and human subjects research.
In the 36 interviews conducted for the ViSuCaRe study, the QRDP was only deployed in one interview. While consistent with typical low rates of participant distress in qualitative studies – with positive evaluations of research participation are more common than negative ones (Carter et al., 2020; Lockwood et al., 2018) – this limited our ability to fully assess its fidelity, acceptability, and appropriateness. Therefore, evaluating the outcomes of the QRDP when it is deployed can help to inform its implementation across research settings and populations. Other evaluation metrics, such as researcher preparedness and comfort, should similarly be evaluated in future inquiry. Indeed, one ViSuCaRe Study team member expressed: Having the distress protocol gave me confidence to explore the difficult or emotional themes of supportive care within the COVID-19 pandemic with participants. I knew that if someone became upset, I would know how to handle the situation in a dignified and respectful way with the protocol’s guidance.
– PhD student, Data Engagement Team member
Future inquiry can also examine the variety of ways that debriefing after instances of deploying the QRDP can reduce preventable instances of participant distress in future data collection such as omitting, rephrasing, or reordering questions that recurrently evoke participant distress.
Finally, evaluative research of the QRDP should assess both participant and interviewer experiences engaging with the tool either during training or when employing the protocol. For example, studies could focus on the experiences of practitioner-researchers, such as clinicians, educators, and social service providers using the QRDP in their research endeavors. These individuals, who have professional training in practical fields other than research, may feel compelled to attend to peoples’ distress in a manner informed by their training rather than the appropriate scope of their researcher roles, as “moving toward a pseudotherapist role can be ethically inappropriate” (Mitchell & Irvine, 2008, p. 35). Thus, assessing if and how the QRDP can aid practitioner-researchers in appropriately responding to distress is a critical next step in understanding the utility of the QRDP.
Conclusion
The QRDP is a comprehensive, adaptable, and participant-centered tool grounded in ethical principles and the Dynamic of Participant-Centeredness. The QRDP can help research teams to consistently, appropriately, and sensitively assess and respond to participant distress, decreasing negative consequences of researcher countertransference. It is our hope that other qualitative researchers will find this protocol adaptable and transferable to their own work. Future evaluation of the QRDP will support expansion of its use and potential across research settings, participant communities, and researchers.
Footnotes
Acknowledgments
The authors would like to thank Lucy Andersen, Rachel Benenson, Radha Vyas, Rohan Krishnan, and Sarah Kagan as well as the entire ViSuCaRe study team for their contributions and Penn Medicine Abramson Cancer Center for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: While working on this paper, Jane Evered was supported by the University of Wisconsin-Madison Department of Family Medicine and Community Health Primary Care Research Fellowship, funded by grant T32HP10010 from the Health Resources and Services Administration.
Appendix
