Abstract
Purpose
To evaluate the safety, technical performance, and short-term efficacy of the ACCERO® heal stent for stent-assisted coil embolization in the treatment of intracranial aneurysms.
Methods
We retrospectively analyzed 32 consecutive patients with intracranial aneurysms treated with the ACCERO® heal stent at two German centers between September 2021 and December 2023. Data were collected on patient demographics, aneurysm characteristics, procedural technique, and complications. Aneurysm occlusion was classified using the Modified Raymond–Roy Classification (MRRC), and stent wall apposition was assessed qualitatively. Follow-up imaging was analyzed for recurrence and in-stent stenosis.
Results
A total of 34 ACCERO® heal stents were implanted in 32 patients (median age 54 years; 87.5% female). Optimal wall apposition was achieved in all cases. Immediate complete occlusion (MRRC I) was observed in 87.5%. Periprocedural complications occurred in five patients (15.6%), including one death and one non-fatal stroke in the context of ruptured aneurysms. Intraoperative in-stent thrombosis occurred in two cases (6.3%), both resolving without clinical sequelae. At follow-up, six patients (19.3%) had aneurysm recurrence or residual, and three (9.7%) required retreatment. In-stent stenosis occurred in three patients (9.7%) and resolved with conservative management.
Conclusion
The ACCERO® heal stent is a reliable and effective option for stent-assisted coiling of intracranial aneurysms. Its low-profile design and HEAL antithrombogenic coating support precise deployment and favorable early clinical outcomes. Prospective studies are needed to confirm these findings.
Introduction
Endovascular treatment of intracranial aneurysms has advanced rapidly since the initial attempts in the 1990s, with coil embolization now representing the best-established basic approach. 1 To enable treatment of wide-neck aneurysms, balloon remodeling and stent-assisted coiling were introduced.2,3 In recent decades, novel devices such as flow diverters and endosaccular flow disruptors have become available.4,5 Nevertheless, intracranial stenting remains a fundamental and versatile tool, offering individualized treatment solutions, particularly in complex cases. 3
Recent studies have reported favorable long-term angiographic and clinical outcomes using low-profile braided stents such as the Acandis ACCLINO® stent for wide-necked bifurcation aneurysms, underlining the feasibility and durability of stent-assisted coiling in these challenging cases. 6
Intracranial stents are typically deployed in the parent artery across the aneurysm neck to prevent coil prolapse into the vessel lumen. They also provide a scaffold for endothelialization, potentially increasing complete occlusion rates and reducing recurrence risk. 7 However, a key drawback of intracranial stents is the elevated risk of thromboembolic complications due to their thrombogenic potential, as well as the risk of in-stent stenosis caused by intimal hyperplasia.8,9
In recent years, device innovation has focused not only on mechanical performance but also on surface modification aimed at reducing thrombogenicity. Antithrombogenic coatings have been developed to improve hemocompatibility and endothelialization, thereby potentially reducing the need for prolonged dual antiplatelet therapy and associated hemorrhagic risk.10–12 Several coatings are now under investigation, such as phosphorylcholine-, hydrophilic polymer-, and heparin-based technologies, showing promising in vitro and preclinical results.10–14
The ACCERO® heal stent (Acandis GmbH, Pforzheim, Germany) combines a low-profile, fully visible braided stent with the HEAL coating technology. It is constructed from nitinol-composite wires with a platinum core, offering excellent traceability, precise deployment, and good wall apposition. The stent can be delivered through a 0.0165-inch inner diameter microcatheter. Figure 1 schematically shows ACCERO® heal stent depicting the fibrin–heparin coating designed to inhibit platelet activation and promote endothelialization.
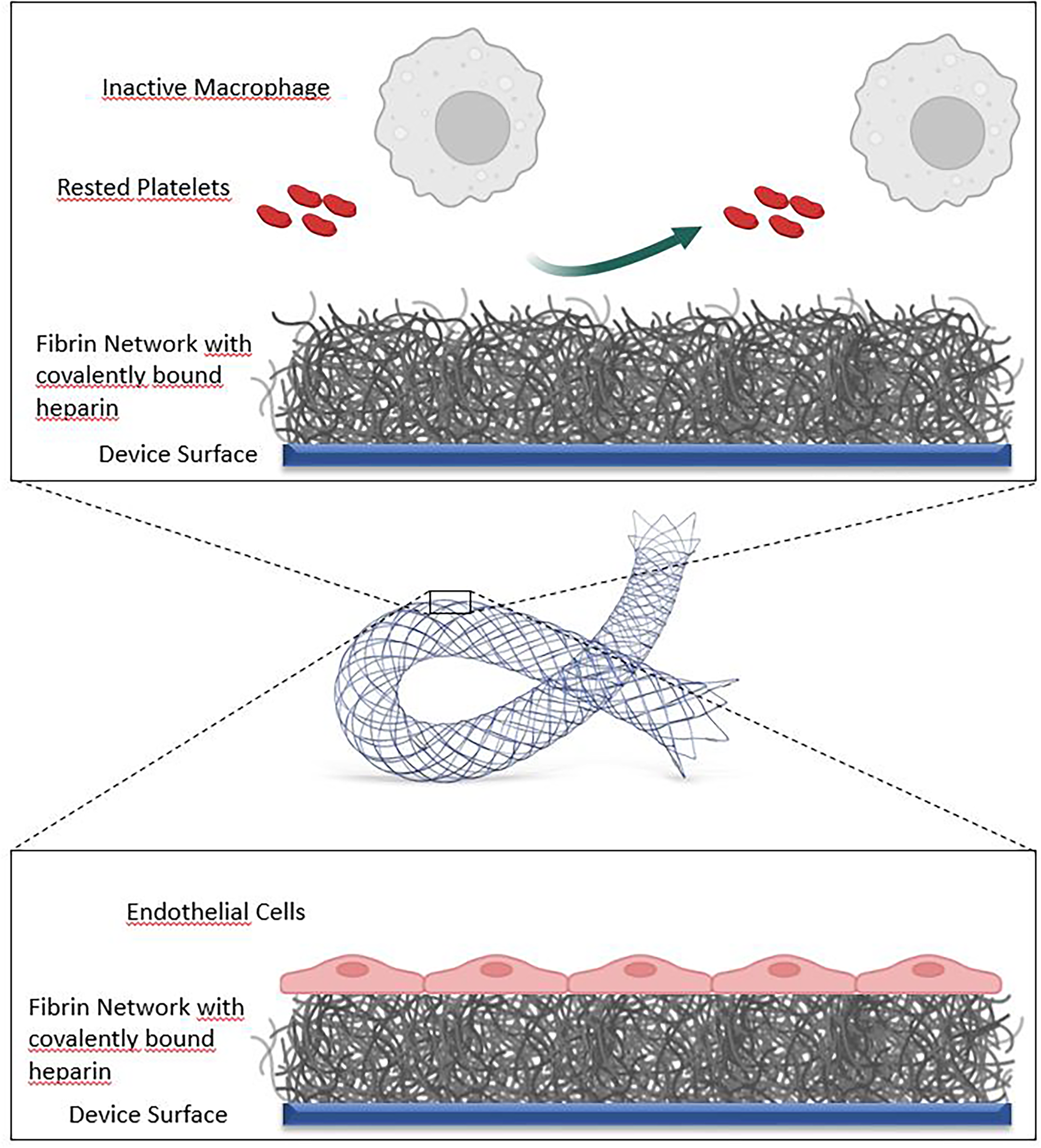
Schematic illustration of the ACCERO® heal stent depicting the fibrin–heparin coating designed to inhibit platelet activation and promote endothelialization. Image courtesy of Acandis GmbH, Pforzheim, Germany.
HEAL is a next-generation antithrombogenic coating, consisting of a thin fibrin-heparin network applied to the device surface. This configuration mimics the final step of physiological hemostasis, passivating the surface and reducing platelet activation and the coagulation cascade.13,14 In vitro studies have shown that HEAL-coating exhibits minimal immune activation—comparable to blood-only controls—and promotes endothelial cell coverage.13,14 In an animal model a significantly lower number of macrophages was detected in the vessel wall after implantation of HEAL coated devices, when compared with the same bare devices. 15 The coating is non-eluting and has no pharmacologic activity.
This technology has also been applied to other Acandis devices, including DERIVO®, CREDO®, ACCLINO®, and CARESTO®. 16
Here, we present a double-center clinical experience evaluating the safety and efficacy of stent-assisted coil embolization using the ACCERO® heal stent.
Materials and methods
We retrospectively analyzed 32 consecutive patients with intracranial aneurysms treated using the ACCERO® heal stent between September 2021 and December 2023 at our two centers (see affiliations). All data had been previously entered into our institutional anonymized database and were extracted from clinical records, procedural reports, and imaging studies. The requirement for formal ethics approval was waived by the responsible local ethics committee due to the non-interventional, retrospective nature of the study and the use of anonymized patient data.
Inclusion criteria required the implantation of at least one ACCERO® heal stent. Both ruptured and unruptured aneurysms were included. Treatment indication was determined by interdisciplinary consensus between neuroradiologists and neurosurgeons. All procedures were performed under general anesthesia by a neuroradiologist with a minimum of seven years’ experience in neurovascular interventions. Imaging was retrospectively reviewed by consensus between two independent experienced neuroradiologists who were not involved in the procedures.
We collected demographic data, aneurysm characteristics, procedural outcomes, and perioperative morbidity. Stent wall apposition was evaluated using a four-point Likert scale: “not sufficient,” “sufficient,” “suboptimal,” and “optimal”. Aneurysm occlusion was assessed using the Modified Raymond–Roy Classification (MRRC). Available follow-up data were analyzed for recurrence and in-stent stenosis.
Follow-up protocols varied between centers. One center performed angiographic follow-up at 90 days, and optionally at 180 days in case of recurrence or stenosis. The other center conducted MRI at 180 days and DSA at 12 months.
Results
Patient and aneurysm characteristics
Thirty-two patients (28 female; median age 54 years) were included in the study. Eleven of these were also enrolled in the post-market Clinical follow-up study “Evaluation of Safety and Efficacy of the ACCERO® (heal) Stent for Intracranial Aneurysm Treatment”. Table 1 summarizes patient and aneurysm characteristics. Each patient had one aneurysm treated. In two cases, two ACCERO® heal stents were implanted, totaling 34 stents.
Summary of patient demographics, aneurysm characteristics, and parent artery diameters.

Acom = anterior communicating artery; ICA = internal carotid artery; MCA = middle cerebral artery; BA = basilar artery; Pcom = posterior communicating artery; ACA = anterior cerebral artery.
Aneurysm locations were distributed as follows: 11 in the middle cerebral artery (MCA), 8 in the anterior communicating artery (ACom), 7 in the basilar artery (BA), 2 in the internal carotid artery (ICA), and 4 in other locations including the posterior communicating artery (PCom) or anterior cerebral artery (ACA). The average aneurysm size was 6.9 ± 3.6 mm (range: 3.2–23.0 mm).
Clinical presentation and antiplatelet management
Most patients (28/32, 87.5%) received elective treatment either for unruptured or previously ruptured aneurysms. Three of these were treated electively for recurrent initially ruptured aneurysms. One patient presented with a 4-weeks-old thunderclap headache from a basilar aneurysm rupture and was treated urgently after loading with aspirin and ticagrelor. Three additional patients with ruptured aneurysms underwent emergent treatment. Modified Rankin Scores (mRS) preoperatively were 0 or 1 in all elective cases. Two emergency patients had mRS 5 and one had mRS 1.
All elective patients were premedicated with 500 mg aspirin and 300 mg clopidogrel at least five days before the procedure, followed by 100 mg aspirin and 75 mg clopidogrel daily. Clopidogrel responsiveness was confirmed using Multiplate or VerifyNow testing. Three patients with clopidogrel resistance received 180 mg ticagrelor one day prior to treatment and were again tested for responsiveness on the following day.
For emergent cases and in case of intraoperative thrombosis, a weight-adapted bolus of eptifibatide was administered intraoperatively, followed by continuous infusion. All patients received 5000 IU of heparin intraoperatively, and catheters were flushed with heparin and nimodipine solutions.
Procedural technique
All procedures were performed via femoral access using either a 7 or 8 French sheath and Envoy DA (Cerenovus), Cerebase DA (Cerenovus), or Neuronmax (Penumbra) as guiding catheter. Intermediate catheters were not required. In eight cases, balloon-assisted coiling was performed first, followed by stent implantation. A Scepter XC (MicroVention) balloon catheter was used.
The “jailing” technique was used in 20/32 cases. In two cases, the microcatheter was navigated through the stent mesh. One case required Y-stenting using the ACCERO® heal and Neuroform Atlas (Stryker) stents. One patient with three A2 segments had a stent placed in one segment while another was protected with a balloon. In another case, rescue stenting from the left PCA to the basilar artery stabilized a displaced WEB device.
The NeuroSlider 17 DLC (Acandis) was used in 26 stent deployments. Other microcatheters included the Echelon (3 cases), Headway 17 (1 case), and Scepter XC balloon catheter (4 cases). Puncture site closure was achieved using Angio-Seal.
Immediate angiographic outcomes and complications
Wall apposition of the stent was rated as “optimal” in all cases (100%). At the end of the procedure, complete aneurysm occlusion (MRRC Class I) was achieved in 28 patients (87.5%). One case resulted in MRRC Class II (3.1%), and another in Class IIIa. One ruptured aneurysm and one case with intraprocedural thrombosis led to MRRC Class IIIb. An example of good occlusion result and optimal wall apposition of the stent is shown in Figure 2.
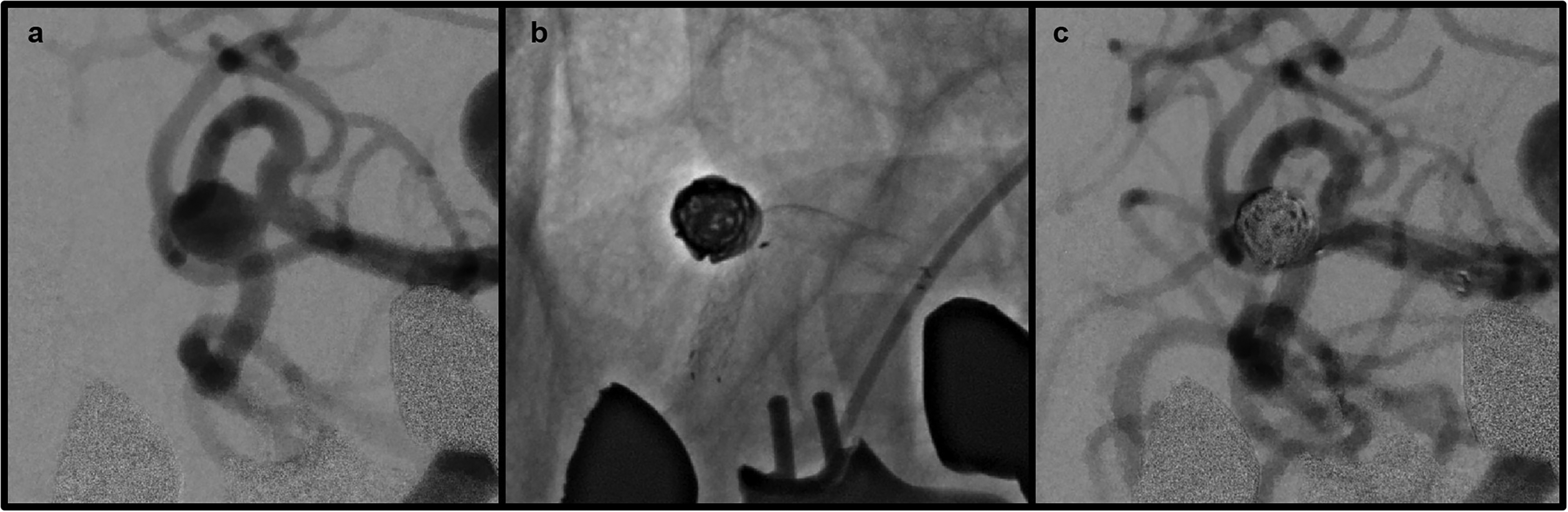
Aneurysm of the right middle cerebral artery before stent-assisted coiling (a). Unsubtracted image without contrast after placement of an ACCERO® heal stent and coiling of the aneurysm (b) and subtracted image showing good occlusion result (c).
One patient with an acute rupture and an mRS of 5 died three days post-procedure due to brain herniation caused by the initial intracranial bleeding and brain swelling. Another patient with an mRS of 5 developed an embolic PICA occlusion during the procedure, likely related to difficult vessel access. Due to cerebellar swelling following the PICA infarction, he underwent decompressive craniectomy and was eventually discharged with an mRS of 4.
In two cases, intraoperative in-stent thrombosis—classified as “low grade” and “severe grade,” respectively—resolved after treatment with eptifibatide without clinical sequelae. In both cases, stent apposition and opening were considered optimal, and no further stent manipulation was necessary.
One aneurysm perforation occurred after microcatheter advancement through the stent meshes. This resulted in subarachnoid hemorrhage and prolonged hospitalization but resolved completely.
In one case, a WEB device implanted in a basilar aneurysm dislocated into the patent lumen immediately after detachment. The device was successfully repositioned into the aneurysm sac and stabilized with the ACCERO® heal stent as a rescue strategy.
One elective procedure was complicated by unintended coil detachment during an angiographic system malfunction. The coil partially protruded into the patent artery between the stent meshes and vessel wall, but this resulted nevertheless in complete occlusion without sequelae.
Follow-up and recurrence
Six patients (19.3%) had aneurysm recurrence or residual (MRRC II or III) at first follow up with DSA, which took place at 90 days in five of these cases and at 120 days in the other case. Three (9.7%) underwent reintervention, achieving complete occlusion. In-stent stenosis was observed in three patients (9.7%) and was assessed as “low grade” in two cases and as “moderate” in the other one. All in-stent stenosis resolved completely with prolonged dual antiplatelet therapy.
Discussion
This retrospective analysis indicates that the ACCERO® heal stent is an effective tool for stent-assisted coil embolization in the treatment of intracranial aneurysms with an acceptable safety profile. Immediate complete occlusion was achieved in 87.5% of cases, consistent with outcomes reported for other modern intracranial stents such as the Enterprise, Neuroform or LVIS EVO.17,18 This high rate of occlusion reflects the mechanical stability and robust coil support provided by the stent.
Importantly, the one Class IIIb occlusion occurred in an emergency case involving a ruptured aneurysm, which may partially explain the suboptimal occlusion outcome due to the urgent clinical context and higher procedural complexity. Furthermore, the single Class IIIa occlusion observed immediately post-procedure demonstrated spontaneous improvement, achieving Class I occlusion at follow-up without the need for retreatment.
Device performance and technical considerations
The ACCERO® heal stent offers favorable handling characteristics due to its full radiopacity and compatibility with low-profile microcatheters. These features facilitated successful deployment even in complex scenarios, including Y-stenting, balloon-assisted coiling, and rescue procedures. These findings are in line with early clinical experience reported by Nania et al., who described the stent's enhanced visibility and deliverability as key advantages in treating wide-neck and bifurcation aneurysms. 19
Antithrombogenic coating and safety profile
The HEAL coating, a fibrin-heparin-based antithrombogenic surface, may reduce thrombotic risk and promote endothelialization. While our study did not include a direct comparison with uncoated stents, we observed a low rate (6.3%) of intraoperative thrombosis, all of which were successfully managed with eptifibatide. The only severe in-stent thrombosis occurred in case of Y stenting of the MCA bifurcation with Neuroform Atlas, which is an uncoated stent. Higher rates of in-stent thrombosis have also been described for Y stenting technique. 20 It is therefore not possible to know how exactly the interaction of the two stents influenced the thrombogenicity of the whole system in this case. Preclinical data suggest that the HEAL coating reduces platelet activation and enhances endothelialization compared to bare-metal surfaces.13,14 This may contribute to the low incidence of thromboembolic events and in-stent stenosis observed in our cohort.
Complications
In this cohort, procedure-related complications occurred in 18.7% of cases, with permanent morbidity in 3.1%. Periprocedural mortality was 3.1%, and both the patient death and the only major stoke (PICA occlusion) occurred in the context of ruptured aneurysms, which are known to have significantly higher rates of adverse outcomes compared to unruptured cases.21,22 Other complications included one aneurysm perforation. As previously described, the high mesh density of the ACCERO® stent may make it more challenging to navigate microcatheters through its strut, possibly increasing the risk of aneurysm perforation during this attempt. 23 On the other hand, this also provides a better support for the implanted coils as well as an endothelialisation scaffold. For this reason, a different approach with “jailing” of the microcatheter inside the aneurysm before placing the stent might be preferred. The other procedure related complications, such as the angiographic system malfunction with consequent unintended coil detachment and the WEB device dislocation, are not related to ACCERO® heal stent.
Importantly, no cases of permanent disability were observed in patients undergoing elective treatment.
Follow-up outcomes
At follow-up, aneurysm recurrence or residual was noted in six patients (19.3%). Of these, three represented true recurrences, all classified as MRRC Class II. The two cases classified as Class III, as well as the remaining MRRC Class II case, represented residual aneurysms rather than new recanalization. Only three patients (9.7%) required retreatment. These findings are within the expected range for stent-assisted coiling, particularly when considering challenging morphologies such as wide-neck or ruptured aneurysms, which are known to be associated with higher recurrence and residual rates.24,25 This underscores the importance of structured follow-up imaging to monitor treatment durability.
In-stent stenosis was observed in 9.7% of patients, all of which resolved without the need for reintervention. This suggests a favorable response to the HEAL coating compared to bare-metal stents, which may lead to longer-term vessel compromise. 13
Limitations
This study has several limitations. First, the sample size of 32 patients limits the statistical power and generalizability of the findings. While this reflects a typical early clinical experience, larger multicenter cohorts are necessary to confirm reproducibility. Second, the retrospective nature of the study introduces potential selection and information bias, as treatment strategies were not standardized prospectively. Third, the inclusion of both ruptured and unruptured aneurysms reflects real-world practice but introduces clinical heterogeneity, with different procedural risks and antiplatelet regimens that may have influenced complication rates. Fourth, angiographic follow-up duration was relatively short, precluding evaluation of long-term aneurysm stability and incidence of in-stent stenosis. Finally, while this study focused on early safety and feasibility, it did not include direct comparison with uncoated stents, which would be valuable for assessing the true impact of the HEAL coating on thromboembolic and inflammatory outcomes.
These limitations underline the need for larger, prospective, and ideally randomized studies with standardized imaging and clinical follow-up to better determine the long-term safety and efficacy profile of the ACCERO® heal stent.
Conclusion
The ACCERO® heal stent appears to be a reliable and versatile device for endovascular treatment of complex intracranial aneurysms. Its novel coating may offer antithrombogenic and endothelialization-enhancing benefits, contributing to favorable procedural outcomes. Larger, prospective comparative studies are warranted to further define its role in the neuroendovascular field.
Footnotes
Acknowledgments
Ethics statement
This retrospective study was conducted in accordance with the ethical standards of the Declaration of Helsinki. The need for formal ethical approval was reviewed by the Ethics Committee of the Medical Association of Westfalen-Lippe (Ärztekammer Westfalen-Lippe), which granted an exemption due to the non-interventional, retrospective nature of the study and the use of anonymized patient data.
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contributions statement
FC and CL took care of conceptualization and methodology. EC and CL supervised the study. FC, HW and AG curated and analyzed data. FC wrote the main manuscript text and prepared figures and tables. All authors reviewed the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Acandis GmbH (Pforzheim, Germany),
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Not applicable
