Abstract
Introduction
Intracranial aneurysms (IAs) affect approximately 3.2% of the global population, and their rupture leading to subarachnoid hemorrhage remains a significant cause of morbidity and mortality despite therapeutic advancements. While genetic factors have been implicated in IA pathogenesis, they account for only about 41% of heritability, suggesting that other mechanisms—particularly epigenetic modifications—may play a critical role. Epigenetic processes such as DNA methylation, histone modification, and non-coding RNA regulation have been shown to mediate gene–environment interactions, influencing endothelial function and vascular remodeling. This systematic review aims to synthesize the current literature on epigenetic mechanisms implicated in IA development, progression, and rupture.
Methods
The review was conducted in accordance with PRISMA guidelines, including both in vitro and in vivo studies available in PubMed up to November 2023. A total of 1019 studies were screened, resulting in 77 eligible full-text articles for data extraction.
Results
The most frequently studied mechanisms were microRNAs (59.7%), DNA/RNA methylation (20.8%), circular RNAs (7.8%), long non-coding RNAs (6.5%), and histone modifications (5.2%). Notably, only three overlapping epigenetic targets were identified across studies, underscoring the field's methodological heterogeneity and lack of standardization. These individual epigenetic pathways are further examined in detail within the Discussion section.
Conclusion
These findings underscore the emerging role of epigenetic research in elucidating novel pathways of intracranial aneurysm pathogenesis, while the limited reproducibility across studies highlights the need for standardized methodologies and larger, more diverse cohorts. Epigenetic regulation remains a promising target for future genetic and therapeutic investigations.
Introduction
Systematic reviews and meta-analyses suggest that the global prevalence of unruptured intracranial aneurysms (IAs) is around 3.2%. 1 Despite recent treatment advancements, spontaneous subarachnoid hemorrhage (SAH) resulting from ruptured IAs continues to have a poor prognosis, with disability and mortality rates still as high as 30–40%. 2 Therefore, further advances in understanding the pathogenesis of IAs are necessary for developing effective diagnostic and prevention strategies for aneurysms.
Genetic factors play a crucial role in the development of IA and aSAH, with the likelihood of these conditions increasing based on the number of first-degree relatives affected in a family. 3 Individuals with hereditary disorders such as Ehlers–Danlos syndrome, Marfan syndrome, and neurofibromatosis-1 have significantly higher rates of IAs (10–20%) and aneurysm rupture (8–25%) compared to the general population.4,5 Large-scale genome-wide association studies (GWAS) conducted mainly on Dutch, Finnish, and Japanese populations have identified several single nucleotide polymorphisms (SNPs) that are more common among those with IAs.6–8 Despite this, genetic variations account for only 41% of the heritability of the disease, leaving a large proportion of the risk unexplained. 9
As a result, environmental factors have been suggested as contributors to the increased familial risk, possibly through epigenetic modifications. 10 Epigenetic mechanisms, including DNA methylation and non-coding RNAs, have been shown to regulate endothelial cell proliferation, apoptosis, migration, and vascular malformation, thus being associated with formation of intracranial arterial-venous malformation. 11 Interestingly, many IA-risk loci are located in functional, noncoding regions, indicating that genetic risk may influence regulatory elements that affect gene expression rather than altering the structure of the gene product itself.5,12 Epigenetic mechanisms mediate the interaction between genes and the environment, thereby impacting susceptibility to various complex diseases. This critical role in regulating gene expression has led to a growing focus in recent years on the epigenetic regulation of IA growth and rupture as a potential target for treatment. 13
In this systematic review, we aimed to comprehensively review the current body of literature on key epigenetic mechanisms to identify prevailing trends and highlight the most promising directions for future research.
Materials and methods
Review design
Review of the literature was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The systematic search encompassed studies that examined various mechanisms of epigenetic regulation in the development, progression, or rupture of intracranial aneurysms. Both in vitro and in vivo studies were included regardless of the age of participants. Only English-language full-text papers were considered. The complete workflow is presented as a diagram in Figure 1.
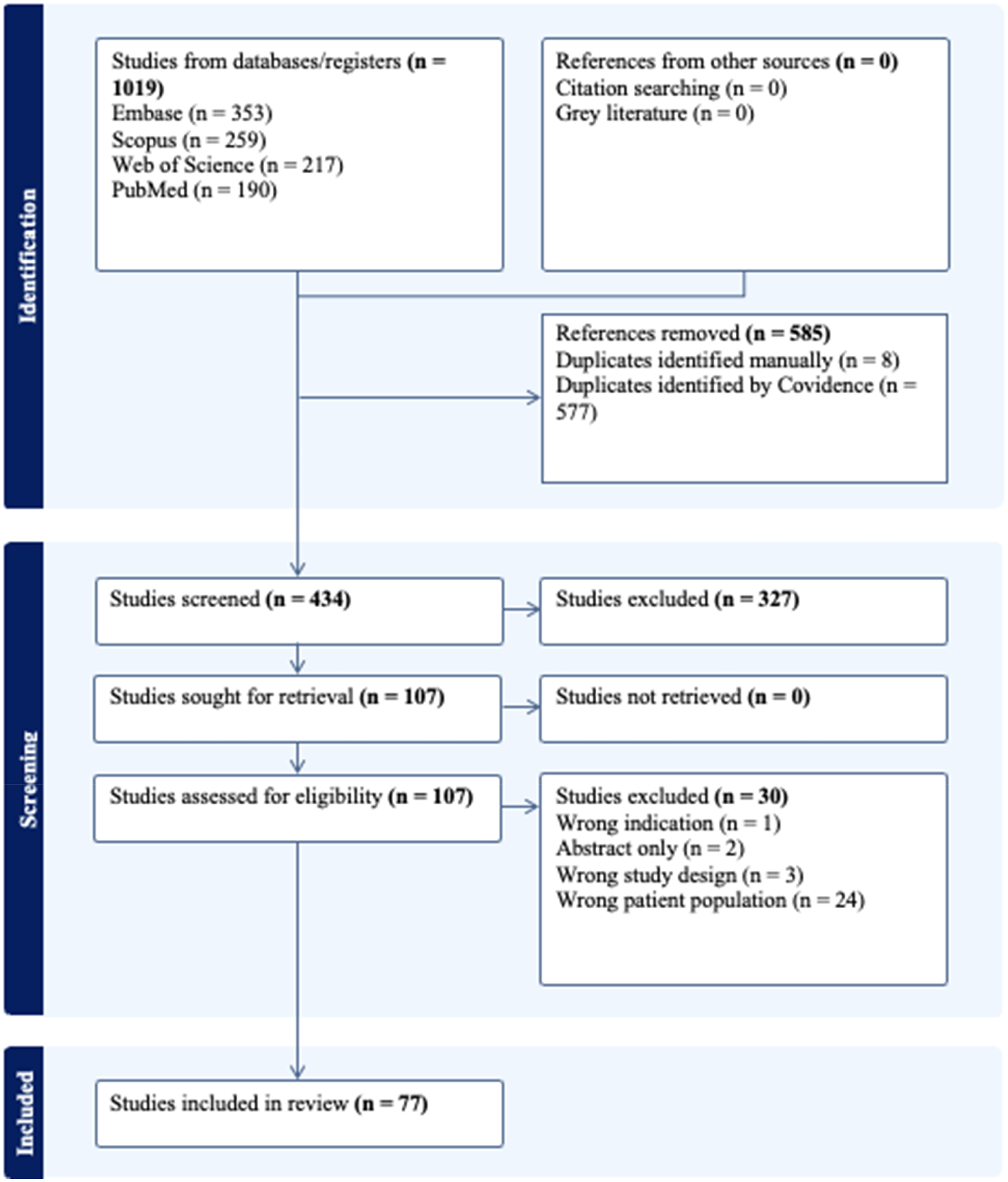
PRISMA flowchart depicting the workflow of the study selection process, in accordance with the PRISMA guidelines.
Search strategy
Keywords were identified using the MeSH (Medical Subject Headings), the NLM-controlled thesaurus used for indexing articles for PubMed. The keywords employed for the database search were “intracranial aneurysms” combined with the following epigenetic mechanisms: histone modification, histone acetylation, histone methylation, histone phosphorylation, DNA methylation, DNA acetylation, DNA phosphorylation, microRNA, circular RNA, long noncoding RNA, and small interfering RNAs.
Study selection
Studies published in PubMed by the end of 2023 were included. Additionally, a manual search was conducted concurrently to ensure no relevant documents were omitted. Titles and abstracts were thoroughly screened by two reviewers (OS and GSS), resulting in the selection of full-text articles for review, as detailed in the PRISMA flowchart. The review process utilized the Covidence app for guidance. Four reviewers (SC, MC, IMM, and GSS) screened the full articles and reached a consensus on final inclusion, followed by data extraction. Consensus on the inclusion or exclusion of studies was determined through a vote by a panel of three researchers. Studies that received a minimum of two positive votes were included for data extraction. The extracted data encompassed the authors, year of publication, country of origin, study model and number of samples, main epigenetic mechanism studied, methods, and key findings.
Results
A total of 1019 studies were screened, and all references retrieved were imported into the Covidence application. After removing duplicates, 434 journal articles were included in the study. Based on the title and abstract screening, and consensus between two blinded reviewers (MC and SC), 327 studies were excluded. Twenty-four papers were excluded due to having the wrong patient population, three due to the wrong study design, two had no full text version, and one due to the wrong indication. In the second round of review, which involved full-text assessment, 77 articles were selected for further evaluation. The breakdown by key epigenetic targets is shown in Table 1.
Breakdown of papers included by key epigenetic targets.

Other potential targets were not found in the searched studies. The distribution by country is summarized in Table 2.
Distribution of epigenetic research papers on the subject by country.

As most papers aimed at clinically applicable goals (identifying mechanisms involved in aneurysm development or risk of subarachnoid hemorrhage [SAH]), we categorized all studied epigenetic targets as associated with aneurysm formation, rupture, or prevention (“protective”). This result is shown in Figure 2.
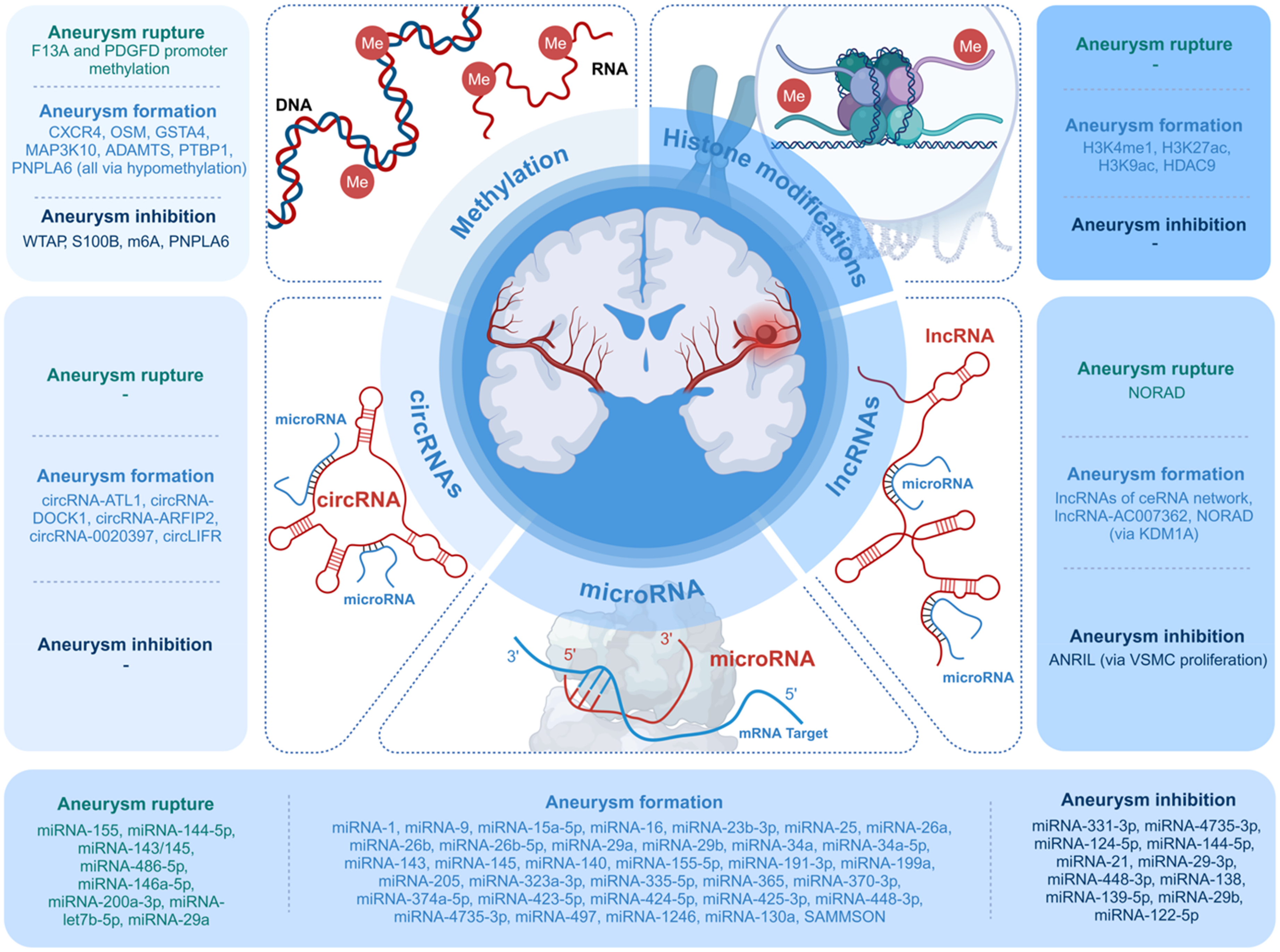
Epigenetic mechanisms associated with aneurysm formation, inhibition, and rupture.
A more detailed overview of the papers, including author, year, study model, number of samples, epigenetic target, and main findings, can be found in Tables 3–5.
Overview of studies focusing on microRNA as the primary epigenetic target.
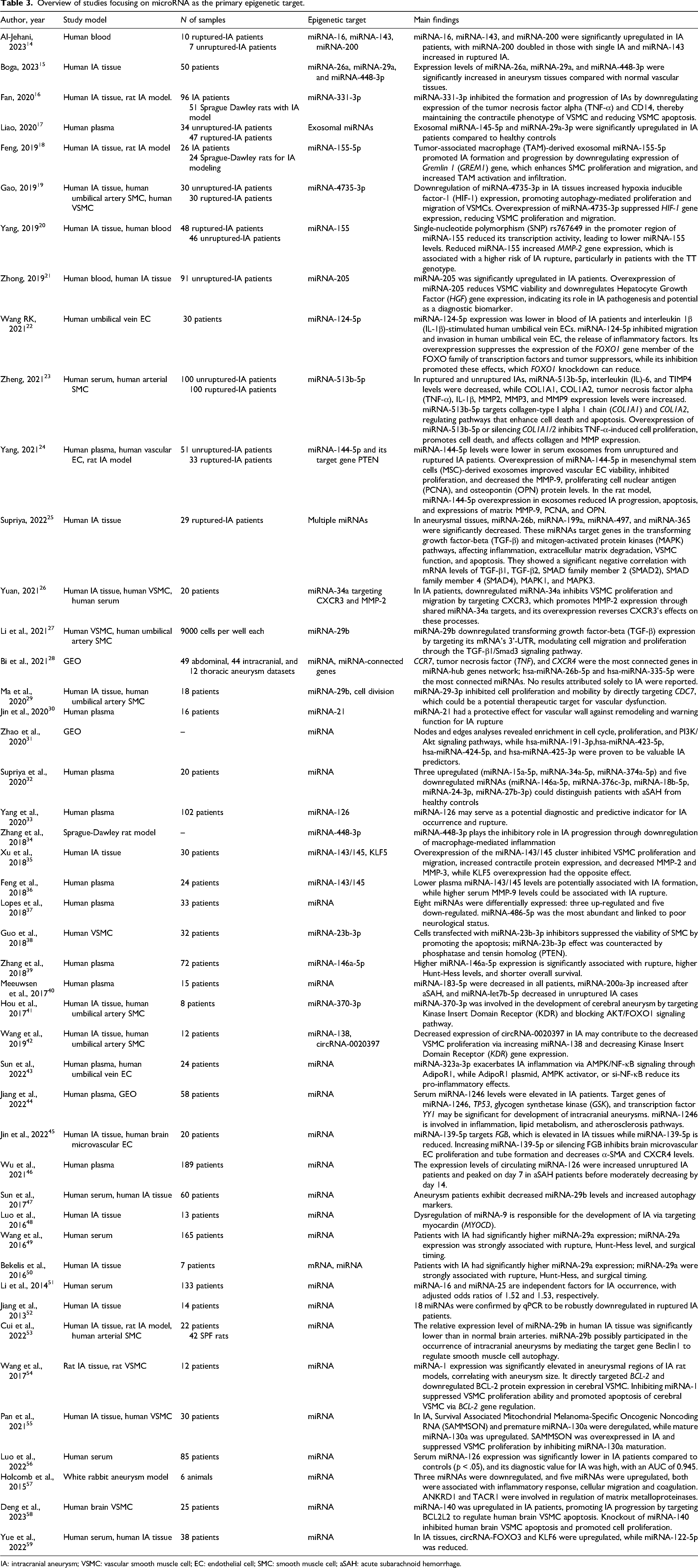
IA: intracranial aneurysm; VSMC: vascular smooth muscle cell; EC: endothelial cell; SMC: smooth muscle cell; aSAH: acute subarachnoid hemorrhage.
Overview of studies focusing on DNA/RNA methylation, circRNA, lncRNAs, and histone modifications.
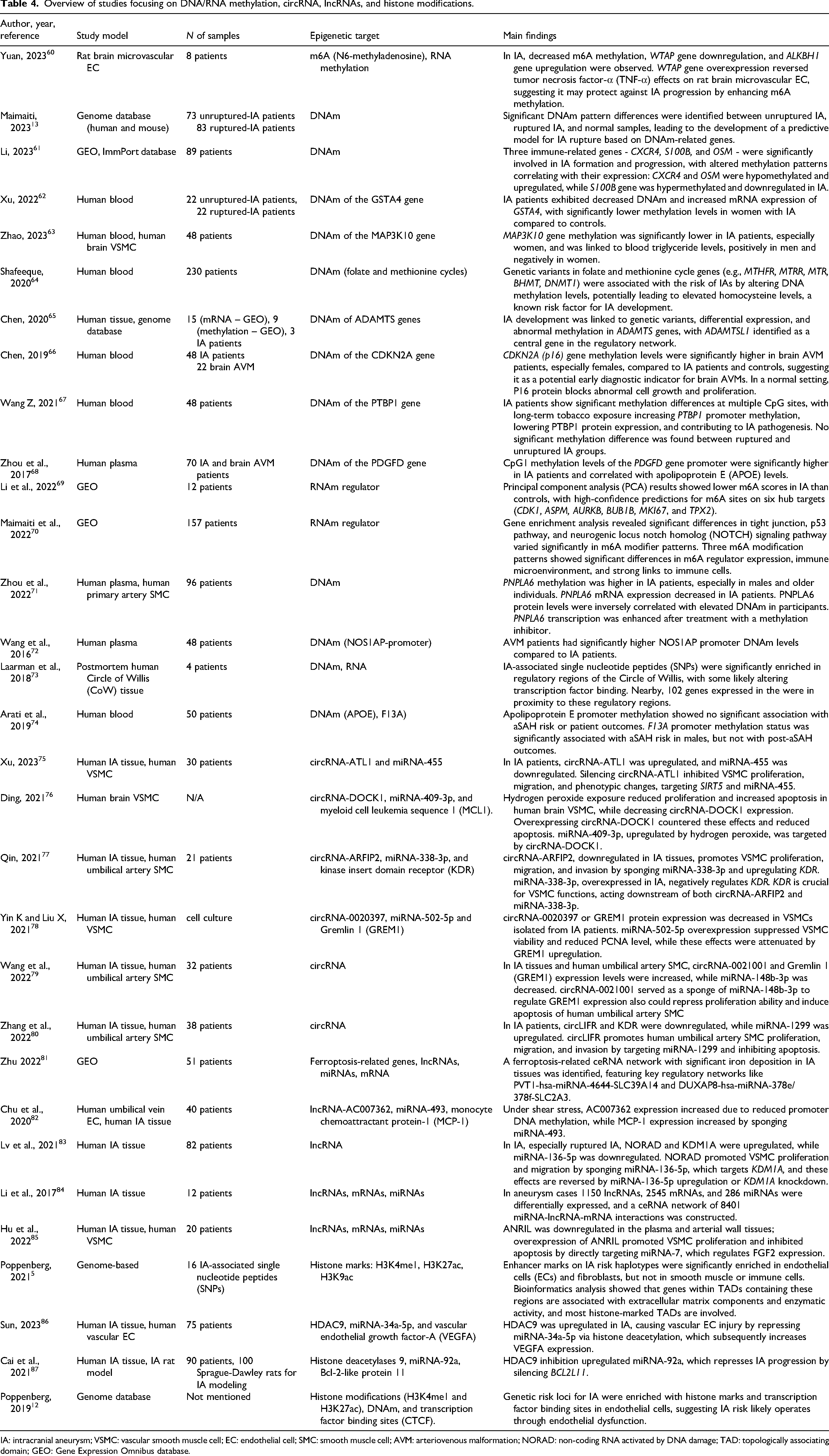
IA: intracranial aneurysm; VSMC: vascular smooth muscle cell; EC: endothelial cell; SMC: smooth muscle cell; AVM: arteriovenous malformation; NORAD: non-coding RNA activated by DNA damage; TAD: topologically associating domain; GEO: Gene Expression Omnibus database.
Epigenetic mechanisms associated with aneurysm formation, inhibition, and rupture.
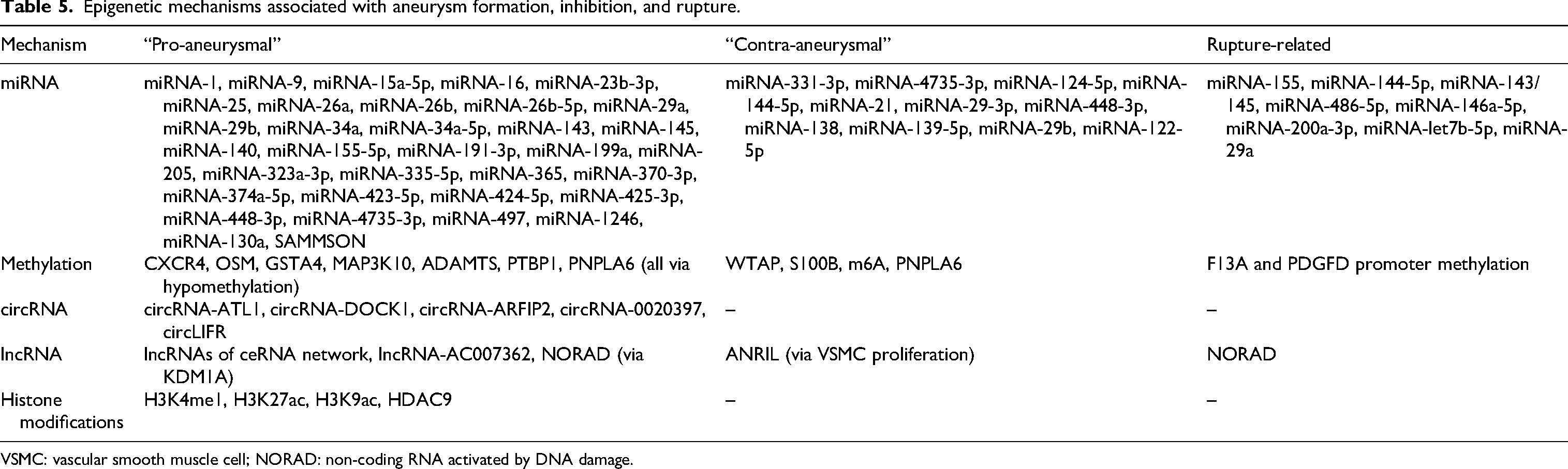
VSMC: vascular smooth muscle cell; NORAD: non-coding RNA activated by DNA damage.
microRNAs
MicroRNAs are the most studied targets due to their crucial role in gene regulation, remarkable stability in biological fluids, high evolutionary conservation across species, and consequent therapeutic potential. In this review, 46 papers with 38 unique microRNA targets were identified; microRNA-143, microRNA-29a, and microRNA-155 were the most frequently studied. These studies have investigated aneurysm association, formation, inhibition, and/or rupture. Key findings are summarized in Table 3, which includes the targets, study models, and primary results.14–59
Over half of the identified microRNA targets are associated with formation or progression of intracranial aneurysms. For example, microRNA-16, microRNA-143, microRNA-200, microRNA-26a and microRNA-29a, and microRNA-448-3p were significantly upregulated in IA patients compared to the controls, with microRNA-200 and microRNA-143 showing particularly high levels in patients with multiple or ruptured aneurysms.14,15 On the other hand, several microRNAs have been identified as “protective” against the formation and progression of intracranial aneurysms, including miRNA-331-3p, miRNA-4735-3p, miRNA-124-5p, miRNA-144-5p, miRNA-21, miRNA-29-3p, miRNA-448-3p, miRNA-138, miRNA-139-5p, miRNA-29b, and miRNA-122-5p. For example, microRNA-331-3p inhibited IA formation and progression by downregulating TNF-α and CD14, thus maintaining the contractile phenotype of VSMCs and reducing VSMC apoptosis. 16
Finally, some microRNAs have been found to be associated with both promoting and inhibiting aneurysm formation, depending on the context. MicroRNA-513b-5p downregulation in ruptured aneurysms (RA) and unruptured aneurysms (UA) targeted COL1A1 and COL1A2 genes, regulating RIP1-RIP3-MLKL and MMP pathways to enhance cell death and apoptosis. 23
Circular RNAs
CircRNA is a type of non-coding RNA molecule that forms a closed loop structure through a covalent bond between its 3′ and 5′ ends, providing high stability. CircRNAs have been shown to modulate microRNA activity, transcription, and protein interactions, and exhibit tissue-specific expression. These characteristics make circRNAs potential biomarkers with high utility. Six studies explored the association and/or role of circular RNAs, including circRNA-ATL1, circRNA-DOCK1, circRNA-ARFIP2, circRNA-0020397, and circLIFR, in IA formation42,75–80
These studies utilized various experimental models, including human tissue samples from IA patients and controls and in vitro models using human vascular smooth muscle cells (VSMCs). These studies consistently demonstrated that circRNAs play crucial roles in regulating VSMC function, which is central to IA pathogenesis. Specifically, most identified circRNAs (circ-ATL1, circ_DOCK1, circ-ARFIP2, circ_0020397, and circLIFR) were shown to modulate VSMC proliferation, migration, and/or invasion.42,75–78,80 Additionally, circ_DOCK1 and circLIFR were found to attenuate apoptosis, while circ_0021001 silencing induced apoptosis in VSMCs. Several studies identified important downstream targets of circRNA-miRNA axes, including SIRT5, MCL1, KDR, and GREM1, which are involved in various cellular processes relevant to IA formation.42,75–78,80
Long non-coding RNAs
Five studies investigated the role of lncRNAs in IAs.81–85 The studies revealed several mechanisms and pathways involving lncRNAs: competing endogenous RNA (ceRNA) networks and miRNA–lncRNA–mRNA interactions,81,84 the ANRIL/miR-7/FGF2 pathway, 85 the NORAD/miR-136-5p/KDM1A axis, 83 and the AC007362/miR-493/MCP-1 pathway. 82 The identified lncRNAs were associated with various functional changes in VSMCs and endothelial cells.
Li et al. 84 found that mRNAs were involved in muscle contraction and vascular smooth muscle contraction, providing insights into mechanisms of IA formation. Similarly, Zhu et al. 81 conducted bioinformatics analysis on public datasets to investigate ferroptosis-related genes and non-coding RNAs in IAs. They identified key regulatory pathways and validated their findings by showing significant iron deposition in IA tissues, suggesting the role of ferroptosis in IA pathogenesis.
Chu et al. 82 studied lncRNA AC007362 in human IA samples and endothelial cells, discovering that shear stress increased AC007362 expression due to reduced promoter DNA methylation. It was found that AC007362 sponged miR-493, increasing MCP-1 expression, suggesting that altered hemodynamics might contribute to IA formation via epigenetic regulation. Similarly, Hu et al. 85 investigated the role of lncRNA ANRIL in IAs, finding lncRNA ANRIL downregulation in IA patients. They demonstrated in vitro that ANRIL promotes vascular smooth muscle cell proliferation and inhibits apoptosis via the ANRIL/miR-7/FGF2 pathway, suggesting therapeutic potential for IA. Studies have shown that the presence of certain lncRNAs is associated with aneurysm rupture. Lv et al. 83 used RNA-seq to study lncRNAs in IA, finding upregulated lncRNAs NORAD and KDM1A, and downregulated miR-136-5p expression in IA. These results are more pronounced in ruptured IA versus unruptured IA. It was proposed that NORAD acts as a competing endogenous RNA for miR-136-5p, upregulating KDM1A and affecting VSMC phenotype regulation in IA formation and rupture.
Histone modifications
Histone modifications have emerged as critical regulators in the pathophysiology of intracranial aneurysms (IAs), with a growing body of evidence highlighting their role in aneurysm growth and rupture. Four studies5,12,86,87 (see Table 3) have explored the association of specific histone modifications, including H3K4me1 (methylation of histone H3), H3K27ac, H3K9ac (acetylations of histone H3), and the epigenetic factor HDAC9, with IAs.
Poppenberg et al. 12 identified genetic risk loci enriched with histone marks and transcription factor binding sites in endothelial cells, linking these modifications to endothelial dysfunction, a key driver of aneurysm formation. In 2021, Poppenberg's team 5 further analyzed IA-associated SNPs and histone marks, including H3K4me1, H3K27ac, and H3K9ac, uncovering significant enrichment of enhancer histone marks in endothelial cells and fibroblasts. These findings emphasize the epigenetic regulation of genes within topologically associating domains (TADs), many of which are differentially expressed in IA tissues. HDAC9 has been identified as a pivotal factor in IA development. Cai et al. 87 demonstrated that inhibiting HDAC9 in a rat IA model and human IA samples upregulates miR-92a, which represses IA progression by silencing BCL2L11. Conversely, Sun et al. 86 found that HDAC9 upregulation in IA tissues promotes vascular endothelial cell injury by repressing miR-34a-5p. This repression leads to increased VEGFA expression, resulting in endothelial cell proliferation, enhanced migration, suppressed apoptosis, and increased vascular permeability, all processes critical to aneurysm growth and rupture.
DNA and RNA methylation
In this review, the roles of DNA and RNA methylation in the pathogenesis of IAs were studied in 16 papers.13,60–74 Several studies have shown that the methylation status of specific genes is associated with aneurysm rupture. For example, methylation status of F13A gene 74 and the PDGFD gene promoter 68 were found to be associated with the aneurysm rupture. DNA methylation patterns show significant differences between unruptured IA, ruptured IA, and normal samples, leading to the development of predictive models for IA rupture risk. 13
Hypomethylation of several genes, including CXCR4, OSM, GSTA4, MAP3K10, ADAMTS, PTBP1, and PNPLA6,61–63,65,67,71 has been identified as being associated with the aneurysm formation. In contrast, aneurysm inhibition was found to be associated with the methylation changes in WTAP, S100B, m6A, and PNPLA6 genes.60,61,71 Several studies have highlighted that decreased N6-methyladenosine (m6A) methylation levels, including the downregulation of WTAP and upregulation of ALKBH1 gene expression, may play a protective role against IA progression.60,69 Gender-specific differences in methylation patterns have been observed, including lower MAP3K10 methylation in women 63 and higher CDKN2A methylation in female patients with brain arteriovenous malformations compared to IA patients and controls. 66 The observed gender-specific differences and the impact of genetic variants further emphasize the need for personalized approaches in understanding and treating IA, with methylation emerging as a potential biomarker and therapeutic target for future research.
Discussion
Epigenetics definition
Epigenetic changes are regulatory processes that modulate gene expression without altering the primary DNA sequence. We have chosen five of the most extensively studied epigenetic mechanisms (DNA methylation, histone acetylation, lncRNAs, miRNAs, and circRNAs) while newer mechanisms such as histone ubiquitination, SUMOylation, and ADP-Ribosylation and others have only recently emerged and represent promising avenues for future research. Among the five mechanisms studied, histone acetylation (loosens chromatin to promote transcription), lncRNAs (serve as scaffolds or guides to enhance gene regulation), and circRNAs (act as miRNA sponges to protect gene expression) primarily support transcription. In contrast, DNA methylation (silences gene promoters) and miRNAs (bind target mRNAs to block translation or promote degradation) are generally associated with transcriptional suppression.
Research rationale
Recent advancements in sequencing technologies, reduced costs, and growing insights from genetic research have enabled neurosurgeons to approach their most challenging diseases (such as intracranial aneurysms [IAs]) with new precision. Epigenetic mechanisms have emerged as a key research focus in the context of intracranial aneurysms (IAs) due to their involvement in essential biological processes (such as inflammation, vascular smooth muscle cell proliferation, and extracellular matrix remodeling) that are fundamental to aneurysm development and rupture.5,44,63,75,80
If successfully validated, epigenetic targets may serve as biomarkers or therapeutic targets for IA diagnosis, disease screening, and personalized treatment solutions. Established modifiable risk factors such as smoking, hypertension, and age are well known to be strongly associated with aneurysm formation, growth, and rupture.88–91 While emerging work has identified DNA methylation, histone modifications, and non-coding RNAs as potential epigenetic contributors,13,92,93 direct comparisons between these mechanisms and traditional clinical risk factors are lacking. If epigenetic pathways are more clearly defined, a potential advantage would be the ability to pharmacologically mitigate smoking- or hypertension-related vascular injury without relying solely on behavioral modification.
Several pharmacological classes have been investigated for their potential to modulate epigenetic mechanisms in intracranial aneurysms. The most robust aneurysm-specific evidence comes from SIRT1 activators, where resveratrol reduced rupture rates in a mouse model by upregulating Sirt1 and downregulating NF-κB-mediated inflammation. 94 Other categories remain more speculative. DNMT inhibitors such as 5-aza-2′-deoxycytidine have been proposed on the basis of DNA methylation changes identified in aneurysm tissue, 62 although direct therapeutic data are lacking. HDAC inhibitors, including valproic acid and trichostatin A, have shown anti-inflammatory and neuroprotective effects in central nervous system models; 95 however, findings from thoracic aortic aneurysm models indicate that broad HDAC inhibition may increase aneurysm susceptibility, 96 cautioning against direct extrapolation. Finally, miRNA-based strategies remain of interest, as several dysregulated miRNAs (notably miR-126) have been linked to intracranial aneurysm pathophysiology and rupture risk, highlighting potential diagnostic and therapeutic applications. 97 Moreover, a recent bioinformatics study by Lai et al. 98 highlighted the potential involvement of estrogen receptor signaling (ESR1), suggesting that hormonal regulation may also contribute to IA pathophysiology and represent an additional therapeutic target.
Epigenetic changes, along with genetic alterations, can be detected in liquid biopsy samples (such as blood, saliva, or urine) enabling the development of non-invasive diagnostic and monitoring tools based on distinct molecular signatures.99,100 Epigenetic mechanisms represent an exceptionally prominent focus in intracranial aneurysm research, as evidenced by the fact that 80.5% of relevant publications have been produced in the last five years (2018–2023), with Chinese groups contributing greatly, comprising 66 (83.6%) papers in this review.
Current landscape and outlook
The study by Boga 15 reported significant upregulation of miRNA-448-3p in aneurysm tissues compared to normal vascular tissues, while Zhang et al. 34 showed that miRNA-448-3p is associated with reduced IA progression through modulation of macrophage-driven inflammation. Rather than representing conflicting evidence, these findings may reflect the multifaceted role of miRNAs as components of complex epigenetic networks. It is possible that the observed upregulation of miRNA-448-3p represents a secondary or compensatory response to ongoing inflammation within aneurysmal tissue, rather than a direct driver of aneurysm formation. Therefore, such outcomes should be interpreted with caution in the context of underlying pathophysiological mechanisms.
Of the 79 papers analyzed, there were only three instances of overlapping targets identified: circRNA-0020397,42,78 miRNA-143/145,14,35,36,50 and miRNA-29b.27,29,47,53 The limited overlap in identified targets across studies, as observed in our analysis, reflects a broader challenge in genetic and epigenetic research, one driven by variability in sample types, sequencing strategies, data processing pipelines, and cohort characteristics. Similar inconsistencies have been reported in other fields of genomics and are often attributed to insufficient standardization, underpowered study designs, and population heterogeneity. To address this, researchers have proposed several solutions, including harmonized protocols, meta-analyses, and cross-validation using multi-cohort datasets.101,102
More recently, AI-based integrative approaches and machine learning algorithms have shown promise in identifying robust patterns across heterogeneous datasets, helping to uncover signature genes despite methodological diversity, particularly in contexts such as abdominal aortic aneurysm growth 103 or intracranial aneurysm (IA) rupture. 104
Limitations
This review provides a thorough and current summary of the existing evidence on the role of epigenetics in the pathophysiology of IA. However, it is not without its limitations. The studies included in this review show significant variability in both the types of samples and the methodologies employed to investigate the epigenetic mechanisms underlying intracranial aneurysms (IAs). Researchers utilized a wide range of human tissue samples, including those from IA patients, normal arterial tissue, and various vascular cell types such as vascular smooth muscle cells (VSMCs) and endothelial cells or serum. Furthermore, several studies incorporated animal models, particularly rats, to simulate IA conditions, while others relied on bioinformatics analyses using publicly available datasets to supplement experimental data. In addition, many of the manuscripts show associations between the targets and outcomes (aneurysm formation, inhibition, and rupture), rather than causality, highlighting the need for further experimental studies to demonstrate causal relationships.
Conclusion
Epigenetic mechanisms will likely continue to be a central focus in intracranial aneurysm (IA) research due to their potential to unravel the intricate and multifactorial nature of this condition. Unlike genetic mutations, which are relatively stable, epigenetic changes are dynamic and can be modulated by environmental factors, lifestyle choices, and various disease states. This dynamic nature makes epigenetic mechanisms a critical link between genetic predisposition and environmental influences in the pathogenesis and progression of IAs. Moreover, the reversible nature of epigenetic modifications presents a promising avenue for the development of novel therapeutic interventions. By specifically targeting these epigenetic alterations, future research may pave the way for strategies aimed at preventing aneurysm formation, stabilizing existing aneurysms, or reducing the risk of rupture.
Footnotes
Abbreviations
Authors’ contributions
Conceptualization: VMS, SC; methodology: OS, GSS; formal analysis: IMM, OS, MC, SC, IK; resources: OS, GSS, IMM, NM, SK; project administration: JKB, VMS; supervision: VMS, JKB; writing–original draft: OS, GSS, IMM, NM, SC, IK, GP; writing–review and editing: JKB, VMS, OS, GSS, GP, SK.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
