Abstract
Introduction
Venous sinus stenting (VSS) is an effective, less invasive alternative to ventriculoperitoneal shunting (VPS) for idiopathic intracranial hypertension (IIH). While efficacy is comparable, with some evidence favoring VSS for headache control, perioperative costs remain under-characterized due to reliance on reimbursement rates rather than actual expenditures.
Objective
To compare the perioperative cost of elective VSS and VPS for IIH, including outpatient workup and follow-up costs, using detailed institutional cost data.
Methods
We retrospectively analyzed IIH patients treated with VSS or VPS from 2017 to 2022 at a single center. All IIH-related costs were captured from 90 days pre-op through 90 days post-op and categorized as fixed (e.g. OR staff) or variable (e.g. supplies). Patients with fulminant IIH or unrelated elective procedures were excluded. Comparisons used Mann–Whitney U and Fisher's exact tests (p < 0.05).
Results
Forty-three patients met criteria (VSS n = 19, VPS n = 24), with no significant differences in age, body mass index, papilledema, opening pressure, or pulsatile tinnitus. Preoperative costs were significantly higher for VSS (median $14,951 [IQR 10,835–16,043] vs $4767 [1, 293, 410–11]; p = 0.008), including both variable (p = 0.008) and fixed (p = 0.015) cost components. Surgical admission costs were similar between groups (p = 0.403), as were postoperative costs (p = 0.509). Total 180-day costs remained significantly higher for VSS ($38,576 [011–43, 36, 590]) compared to VPS ($31,509 [25, 208–37, 342]; p = 0.001).
Conclusion
VSS incurs higher preoperative and total costs than VPS. Streamlining VSS workup may improve value. Further studies should assess downstream cost avoidance to determine long-term cost-effectiveness.
Introduction
Idiopathic intracranial hypertension (IIH), diagnosed according to the Friedman criteria, is characterized by elevated intracranial pressure in the absence of an identifiable cause. It most commonly affects overweight women of childbearing age and can lead to chronic headaches, visual disturbances, and, if inadequately treated, permanent vision loss.1–5 Historically, ventriculoperitoneal shunting (VPS) has served as the primary surgical intervention for patients with refractory IIH. However, venous sinus stenting (VSS) has emerged as a promising alternative, effectively reducing venous pressures and alleviating symptoms.6–9 Although prior studies have underscored the clinical efficacy of VSS—particularly with respect to headache control—its financial implications relative to VPS remain underexplored.
Accurate assessment of perioperative costs is critical for guiding treatment decisions in IIH, yet existing analyses often rely on Medicare reimbursement rates or charges rather than direct cost data. 10 This leaves an incomplete understanding of the true economic burden associated with each surgical approach, despite the significant resource utilization involved in caring for patients with IIH. In collaboration with the institution's finance department, this study addresses this gap by performing a detailed retrospective comparison of perioperative costs for VSS and VPS at a single center, including hospital-associated expenses, surgeon and anesthesia fees, and equipment costs. By clarifying these cost differences, our findings aim to help clinicians and administrators make more informed decisions on resource allocation and patient management, while also providing impetus for future prospective research that examines both long-term outcomes and broader cost-effectiveness in IIH treatment.
Methods
Study design and patient selection
We retrospectively reviewed adult patients with IIH who underwent elective VSS or VPS at a single academic center between 2017 and 2023. Patients were included if they had a diagnosis of IIH and underwent one of the two procedures as definitive treatment following multidisciplinary evaluation. Patients were excluded if they had fulminant IIH, underwent emergency procedures, lacked available cost data, or had unrelated surgeries within 90 days before or after the index procedure.
Clinical and financial data were extracted from the electronic medical record and institutional cost databases. Cost data encompassed all IIH-related inpatient and outpatient services within a 180-day window, spanning from 90 days before to 90 days after the index procedure. Costs were categorized as either fixed or variable. Fixed costs refer to expenses that remain constant regardless of the intensity or duration of patient care, such as operating room overhead, salaried non-physician personnel, and general facility or administrative fees. Variable costs fluctuate directly with the volume and complexity of care delivered and include surgical supplies and implants, surgeon and anesthesia professional fees, medications administered during the perioperative period, diagnostic imaging, laboratory testing, and other patient-specific interventions. Institutional policy precluded reporting the exact prices of individual components (e.g. specific stent or valve costs); however, all such expenses are fully captured within the aggregated fixed and variable cost categories reported here. This study was approved by the institutional review board with a waiver of informed consent (IRB # 242151) and did not receive any external funding.
Procedural details
IIH patients were primarily managed by neurologists and neuro-ophthalmologists, with surgical consultation for those whose symptoms persisted despite escalating medical therapy or who were intolerant of medications. Historically, patients were referred for VPS; however, starting in 2019, magnetic resonance venography (MRV) or computed tomography venography (CTV) became standard in IIH evaluation. If noninvasive suggested venous sinus stenosis, a cerebral angiogram was performed in the outpatient setting, under monitored anesthesia care, with venous manometry. Patients with angiographic evidence of venous sinus stenosis and a trans-stenotic pressure gradient ≥8 mmHg were offered VSS. VPS was preferred when no stenosis was present on noninvasive imaging, if no pressure gradient was seen on angiography, or in cases managed prior to VSS adoption. Seven VPS patients were treated before 2019, and one VSS patient underwent stenting prior to its routine use.
Standard techniques were used for both procedures. VSS was performed under general anesthesia after initiating dual antiplatelet therapy. Transfemoral venous access was achieved percutaneously, and a catheter system was advanced to the site of transverse or sigmoid sinus stenosis under fluoroscopic guidance. Typical equipment for VSS procedures varied between cases and practitioners based on specific anatomic considerations. However, the most common equipment are described as follows: A diagnostic arterial catheter—typically a Glidecath® Simmons catheter (Terumo Neuro)—was placed into the ipsilateral common carotid artery through a radial access sheath for control angiography during stenting. A large-bore guide catheter, most commonly a Cerebase™ (Cerenovus Stroke Solutions™, J&J MedTech) or Zoom 88 Support (Imperative Care Inc.), was advanced through a femoral venous access sheath into the ipsilateral distal internal jugular vein. This was followed by an intermediate catheter (Distal Access Catheter, Stryker Neurovascular or Zoom 45, Imperative Care Inc.) over an Aristotle® 18 or 24 (Scientia Vascular) microwire into the intracranial venous sinuses to facilitate angiography and venous manometry.
A self-expanding stent—either a Carotid Wallstent™ (Boston Scientific) or a Zilver 635® (Cook Medical)—was then deployed at the site of stenosis, followed by balloon angioplasty as needed. Intraoperative venous manometry confirmed adequate gradient reduction.
For VPS, a frontal burr hole was made using Medtronic AxiEM neuronavigation to access the lateral ventricle. A programmable valve (Medtronic Strata or Codman Certas®) was selected based on surgeon preference and patient anatomy. The distal catheter was tunneled subcutaneously to the peritoneal cavity and inserted laparoscopically by a general surgeon. Intraoperative confirmation of CSF flow was routinely performed.
Outcome measures
Primary outcomes included total, fixed, and variable costs, based on actual institutional expenditures obtained from internal cost databases, rather than billing charges or reimbursement estimates. Secondary outcomes encompassed clinical metrics such as operative time, estimated blood loss, reoperation rate, and length of stay (LOS).
Statistical analysis
Statistical analyses were performed using XLSTAT v2023.3.1 (Lumivero, Denver, Colorado, USA). Continuous variables were assessed for normality and compared using t-tests or Mann–Whitney U tests as appropriate. Categorical variables were analyzed with chi-square or Fisher's exact tests. Multivariable linear regression identified independent predictors of total cost. Significance for all analyses was set at p < 0.05.
Results
Baseline characteristics
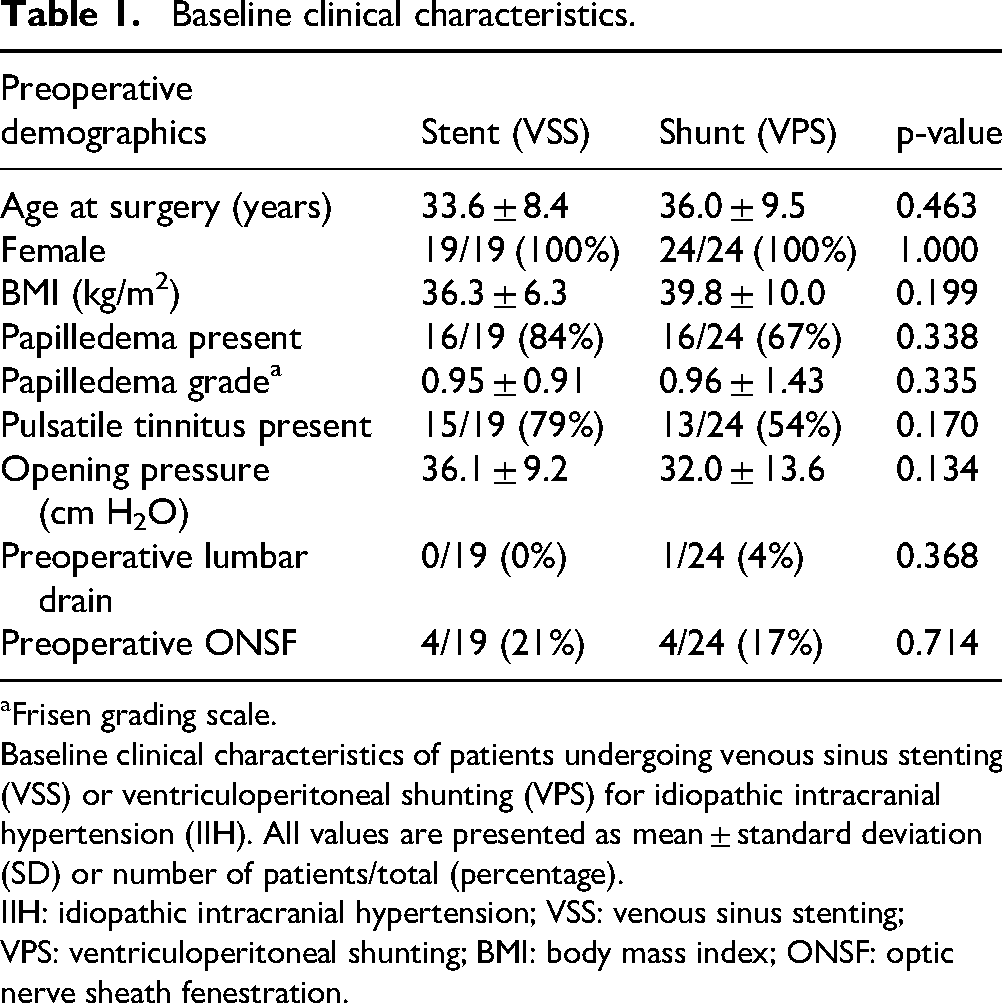
A total of 43 patients with IIH were included, of whom 19 (44.2%) underwent VSS and 24 (55.8%) underwent VPS. Baseline characteristics were statistically similar between groups (Table 1). All patients were female. The mean age at surgery was 33.6 ± 8.4 years in the VSS group and 36.0 ± 9.5 years in the VPS group (p = 0.463), and the mean body mass index (BMI) was 36.3 ± 6.3 kg/m² vs. 39.8 ± 10.0 kg/m², respectively (p = 0.199). Papilledema was present in 16 of 19 VSS patients (84.2%) and 16 of 24 VPS patients (66.7%) (p = 0.338), with comparable mean Frisén papilledema grades (0.95 ± 0.91 vs. 0.96 ± 1.43; p = 0.335). Pulsatile tinnitus was reported in 15 of 19 patients (78.9%) in the VSS group and 13 of 24 patients (54.2%) in the VPS group, without a statistically significant difference (p = 0.170). Mean preoperative lumbar puncture opening pressure was also similar (36.1 ± 9.2 cm H₂O for VSS vs. 32.0 ± 13.6 cm H₂O for VPS; p = 0.134). Preoperative lumbar drain placement was uncommon and did not differ significantly between groups (0/19 [0%] in VSS vs. 1/24 [4.2%] in VPS; p = 0.368). Preoperative optic nerve sheath fenestration (ONSF) was performed in 4 of 19 patients (21.1%) in the VSS group and 4 of 24 patients (16.7%) in the VPS group (p = 0.714).
Baseline clinical characteristics.
Frisen grading scale.
Baseline clinical characteristics of patients undergoing venous sinus stenting (VSS) or ventriculoperitoneal shunting (VPS) for idiopathic intracranial hypertension (IIH). All values are presented as mean ± standard deviation (SD) or number of patients/total (percentage).
IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting; BMI: body mass index; ONSF: optic nerve sheath fenestration.
Cost comparison
All cost data are presented below as median [interquartile range]. Both median [IQR] and mean [95% confidence interval] are reported in Tables 2, 3, 4 and 5.
Preoperative costs.
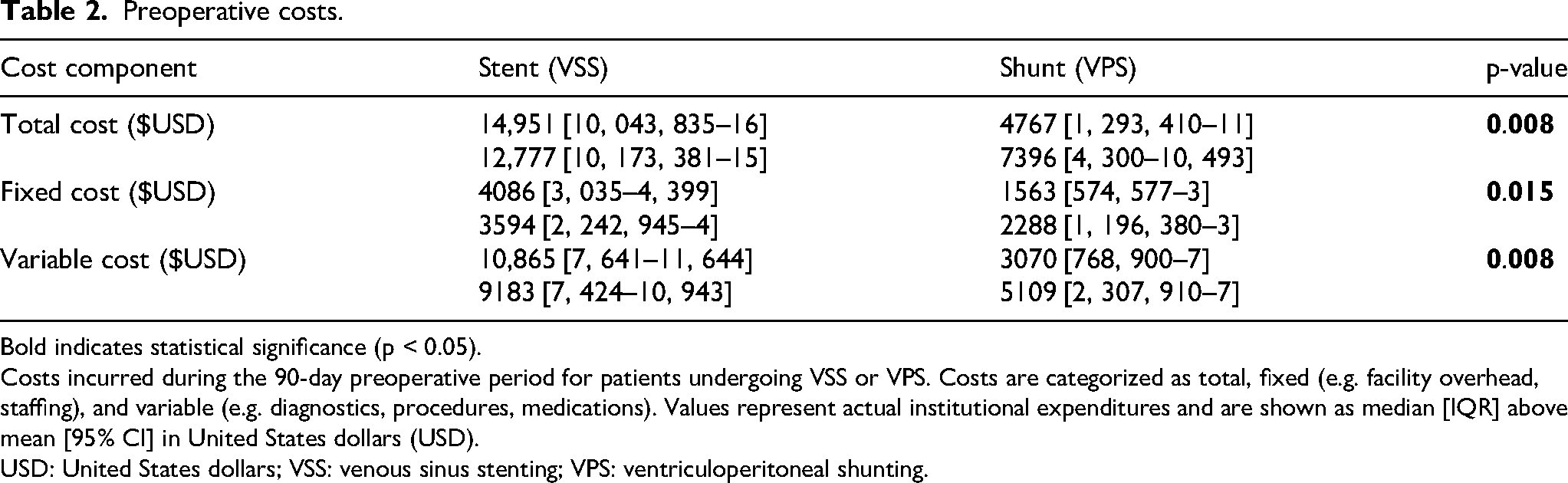
Bold indicates statistical significance (p < 0.05). Costs incurred during the 90-day preoperative period for patients undergoing VSS or VPS. Costs are categorized as total, fixed (e.g. facility overhead, staffing), and variable (e.g. diagnostics, procedures, medications). Values represent actual institutional expenditures and are shown as median [IQR] above mean [95% CI] in United States dollars (USD).
USD: United States dollars; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Surgical admission costs.
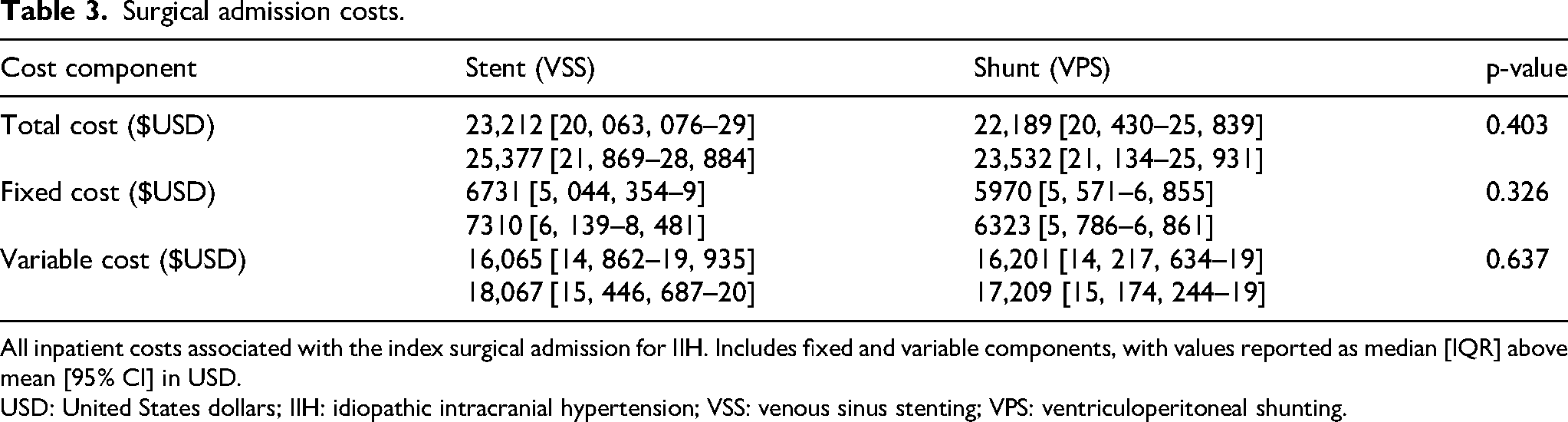
All inpatient costs associated with the index surgical admission for IIH. Includes fixed and variable components, with values reported as median [IQR] above mean [95% CI] in USD.
USD: United States dollars; IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Postoperative costs.

Costs incurred in the 90-day postoperative period following VSS or VPS. Includes outpatient visits, imaging, procedures, and readmissions. Reported as median [IQR] above mean [95% CI] in USD.
USD: United States dollars; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Total 180-day costs.
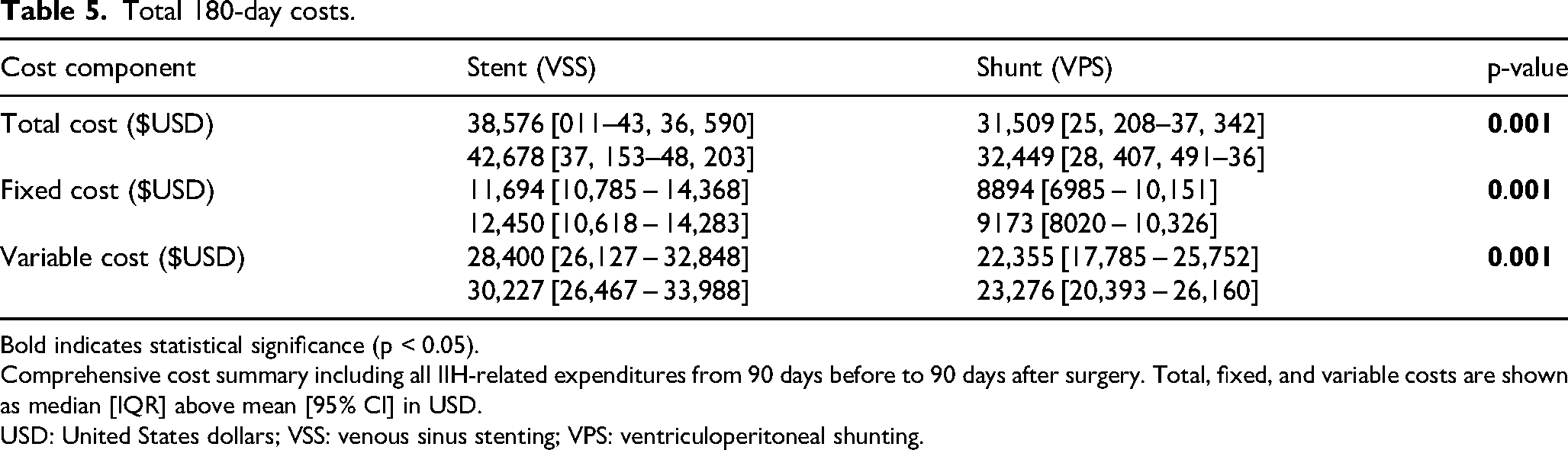
Bold indicates statistical significance (p < 0.05). Comprehensive cost summary including all IIH-related expenditures from 90 days before to 90 days after surgery. Total, fixed, and variable costs are shown as median [IQR] above mean [95% CI] in USD.
USD: United States dollars; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Preoperative costs were significantly higher for VSS ($14,951 [10, 043, 835–16]) compared to VPS ($4767 [1, 293, 410–11]; p = 0.008) (Table 2, Figure 1). This was driven by both higher fixed costs ($4086 [3, 035–4, 399] for VSS vs. $1563 [574, 577–3] for VPS; p = 0.015) and higher variable costs ($10,865 [7, 641–11, 644] vs. $3070 [768, 900–7]; p = 0.008).
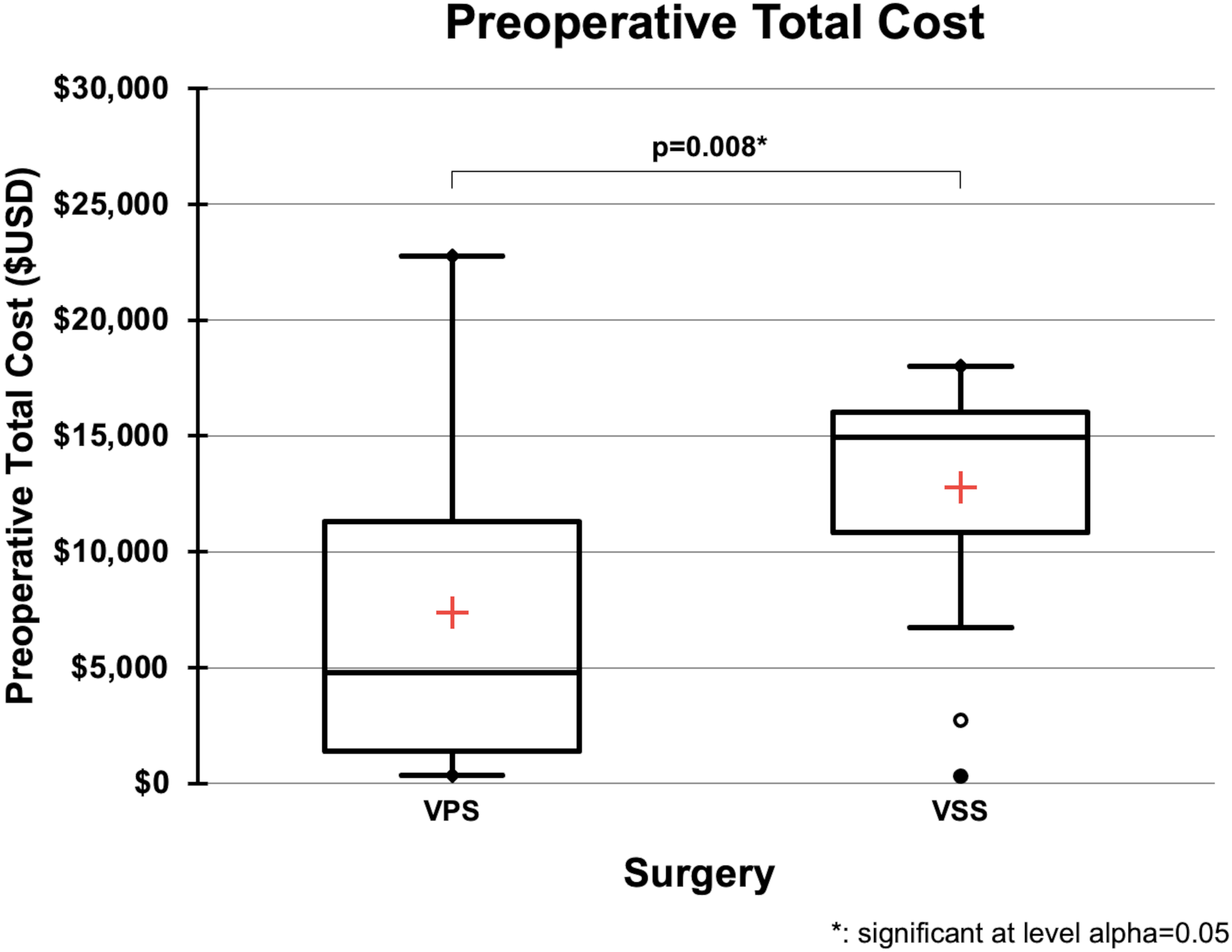
Box-and-whisker plot showing costs incurred during the 90-day preoperative window for patients with idiopathic intracranial hypertension (IIH) undergoing either ventriculoperitoneal shunting (VPS) or venous sinus stenting (VSS). The red cross indicates the mean; boxes represent the interquartile range (IQR); whiskers extend to 1.5×IQR; and circles/dots denote outliers. VSS was associated with significantly higher preoperative costs). USD: United States dollars; IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting; * indicates statistical significance at α = 0.05.
Surgical admission costs (defined as all costs incurred during the inpatient hospitalization for the surgical procedure) were similar between groups (Table 3, Figure 2). Median surgical admission costs were $23,212 [20, 063, 076–29] for VSS and $22,189 [20, 430–25, 839] for VPS (p = 0.403). Fixed costs were $6731 [5, 044, 354–9] for VSS and $5970 [5, 571–6, 855] for VPS (p = 0.326), while variable costs were $16,065 [14, 862–19, 935] vs. $16,201 [14, 217, 634–19] (p = 0.637).
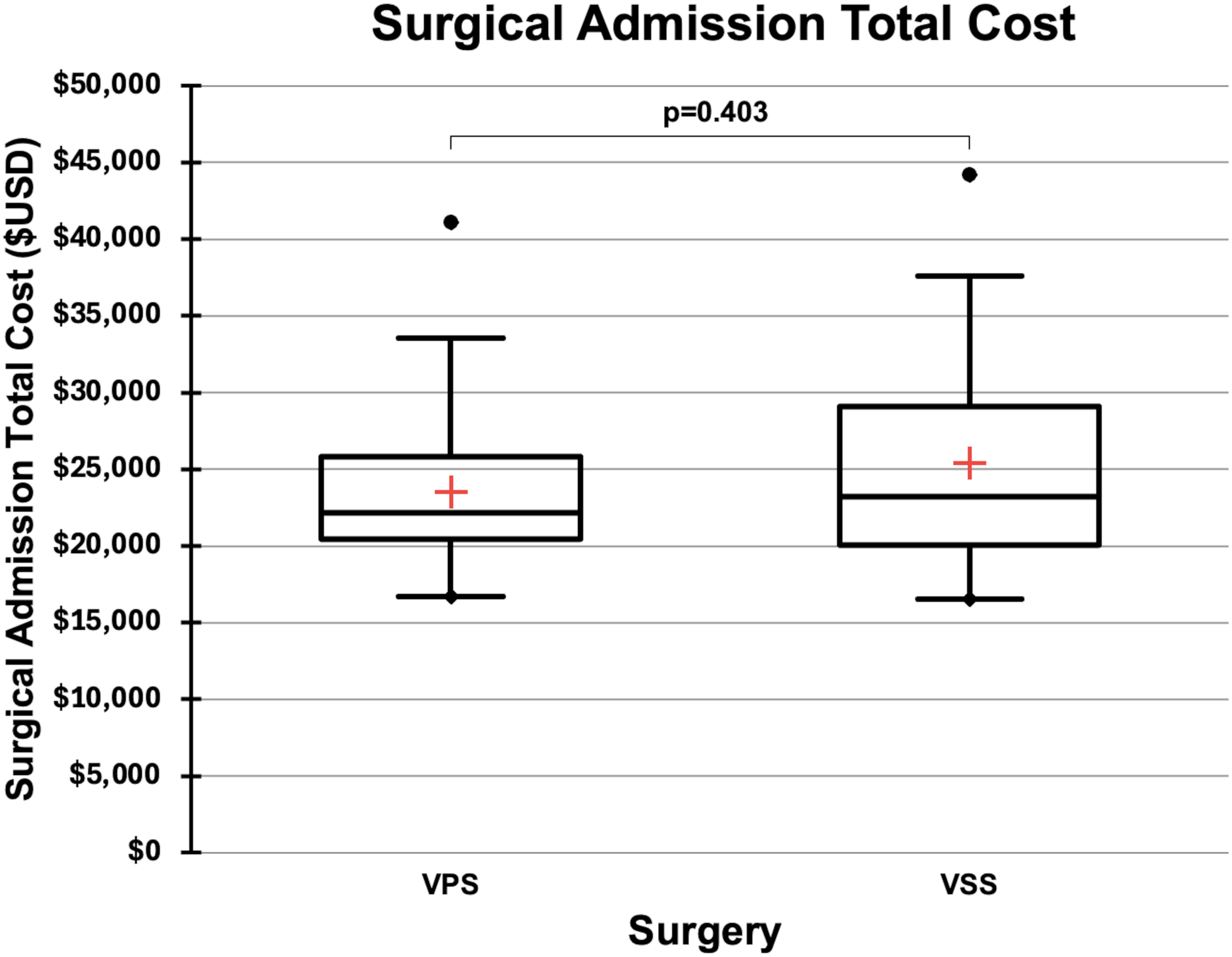
Box-and-whisker plot comparing total inpatient surgical admission costs for IIH patients undergoing VSS or VPS. Costs include all expenditures related to the index procedure. Red crosses represent means; boxes indicate IQR; whiskers show 1.5×IQR; and dots reflect outliers. No statistically significant difference was observed. USD: United States dollars; IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Postoperative costs were also comparable (Table 4, Figure 3). Total postoperative costs were $764 [304–1, 989] for VSS and $581 [121, 180–3] for VPS (p = 0.509). Fixed costs were $180 [77–783] for VSS and $224 [75–1, 147] for VPS (p = 0.966), while variable costs were $554 [242–1, 305] vs. $377 [117–1, 971] (p = 0.407). In the VSS group, three high-cost postoperative events—one emergency department presentation for retroperitoneal hematoma, one repeat stenting procedure, and one follow-up angiogram for recurrent headache—accounted for variability. Sensitivity analysis excluding these outliers did not change the overall findings, with no statistically significant difference in postoperative costs between groups (p = 0.861).
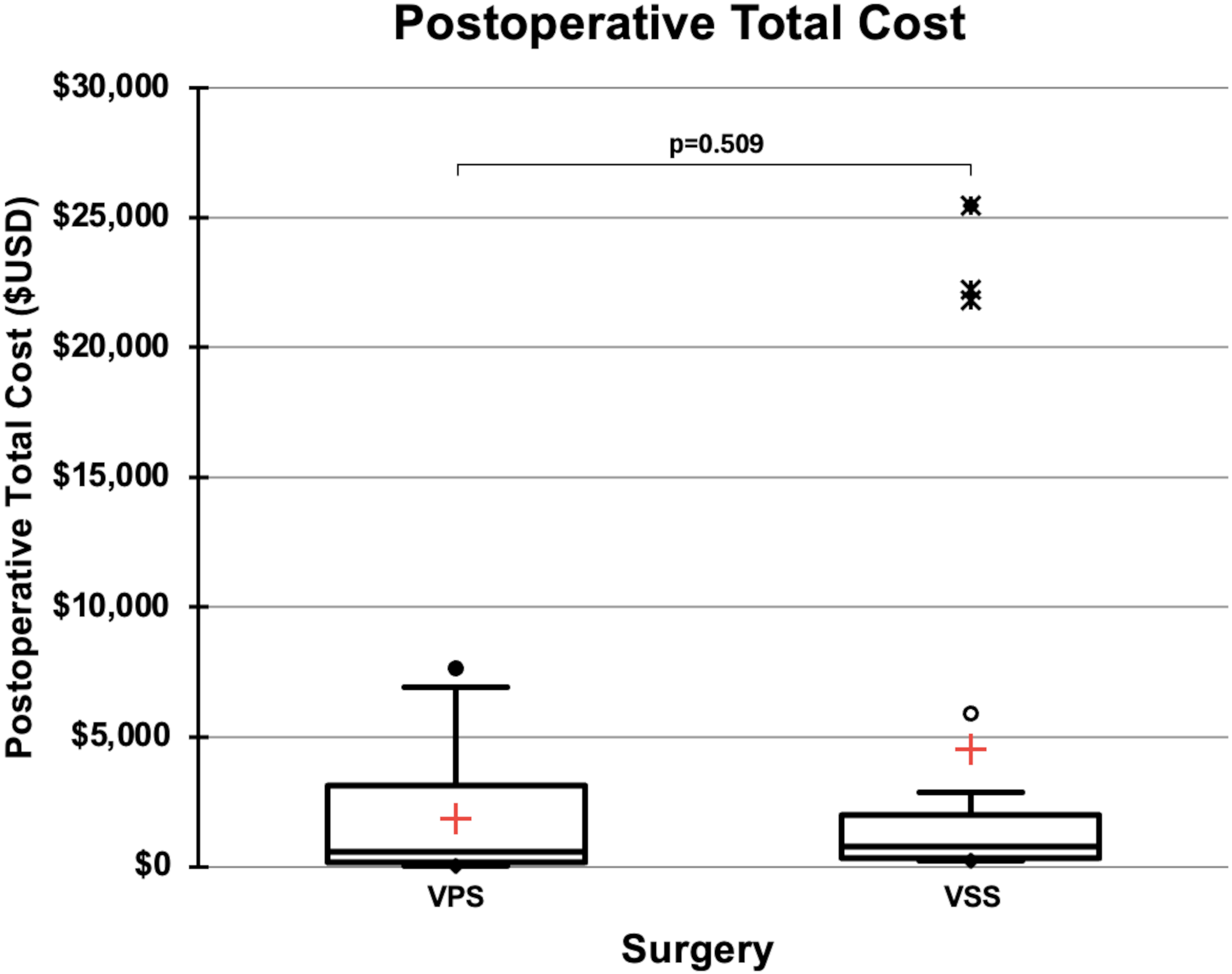
Box-and-whisker plot displaying costs incurred in the 90-day postoperative period for IIH patients treated with VSS or VPS. Red crosses mark group means; boxes represent IQR; whiskers extend to 1.5×IQR; and symbols indicate outliers. No significant cost difference was found between groups. USD: United States dollars; IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Total 180-day costs were significantly higher in the VSS group ($38,576 [011–43, 36, 590]) compared to the VPS group ($31,509 [25, 208–37, 342]; p = 0.001) (Table 5, Figure 4). This included significantly higher fixed costs ($11,694 [10, 368, 785–14] for VSS vs. $8894 [6, 151, 985–10] for VPS; p = 0.001) and variable costs ($28,400 [26, 127–32, 848] vs. $22,355 [17, 752, 785–25]; p = 0.001).
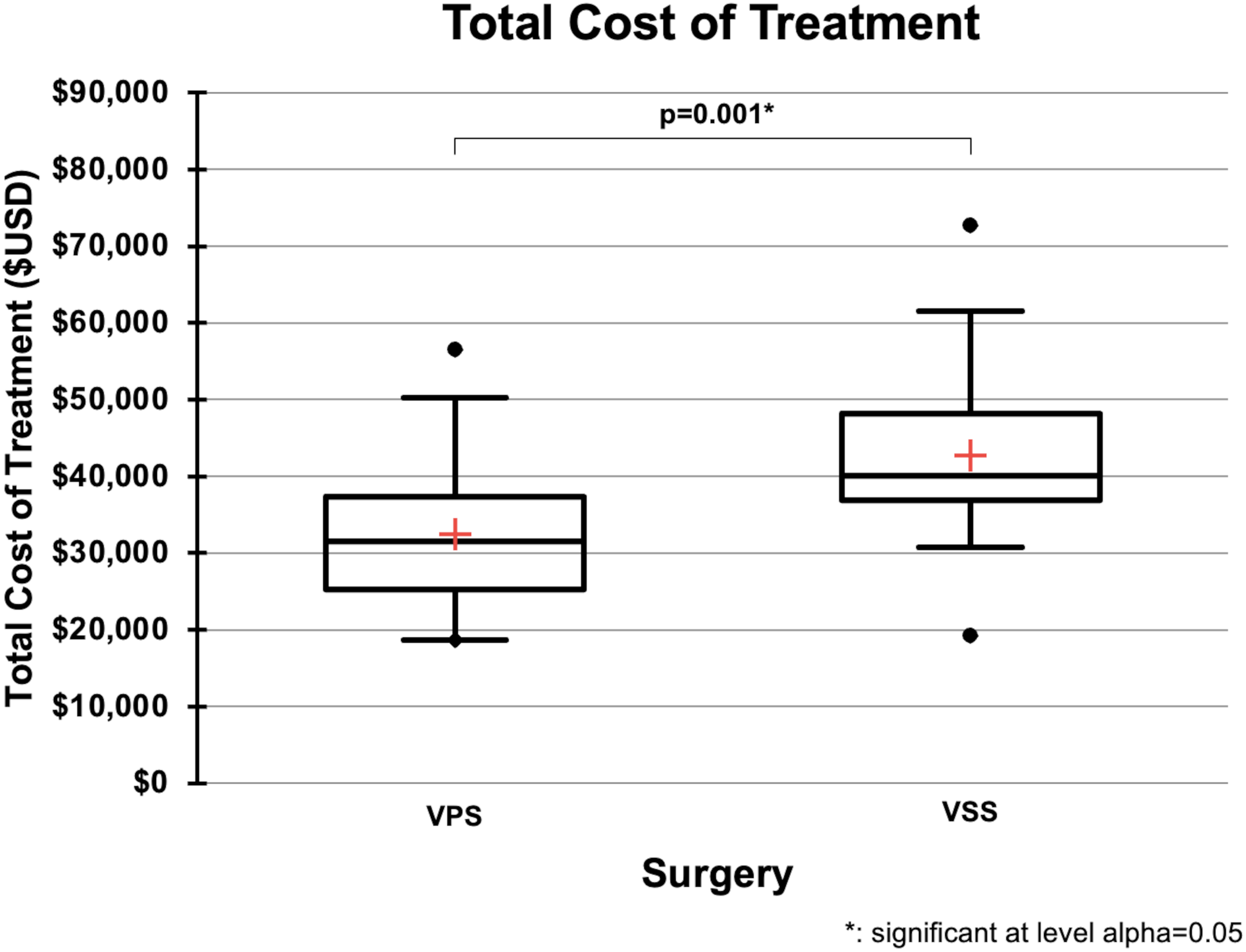
Box-and-whisker plot comparing the total 180-day cost of treatment for VSS versus VPS in patients with IIH. Total cost reflects all IIH-related expenditures from 90 days before to 90 days after surgery. Red crosses indicate the mean; boxes represent IQR; whiskers extend to 1.5×IQR; and dots identify outliers. VSS was significantly more costly overall. USD: United States dollars; IIH: idiopathic intracranial hypertension; VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting; * indicates statistical significance at α = 0.05.
On multivariable regression analysis, procedure type (VSS vs. VPS) was the only variable independently associated with total cost (β = –12,304; p = 0.011). LOS (p = 0.155), operative time (p = 0.793), estimated blood loss (p = 0.678), age (p = 0.798), BMI (p = 0.122), opening pressure (p = 0.249), and prior procedures such as lumbar drain placement (p = 0.436) and ONSF (p = 0.640) were not significantly associated with cost (Table 6). Assessment of multicollinearity demonstrated low variance inflation factors (all <2.0), indicating no significant collinearity among model variables.
Multivariable linear regression for variables associated with total cost.
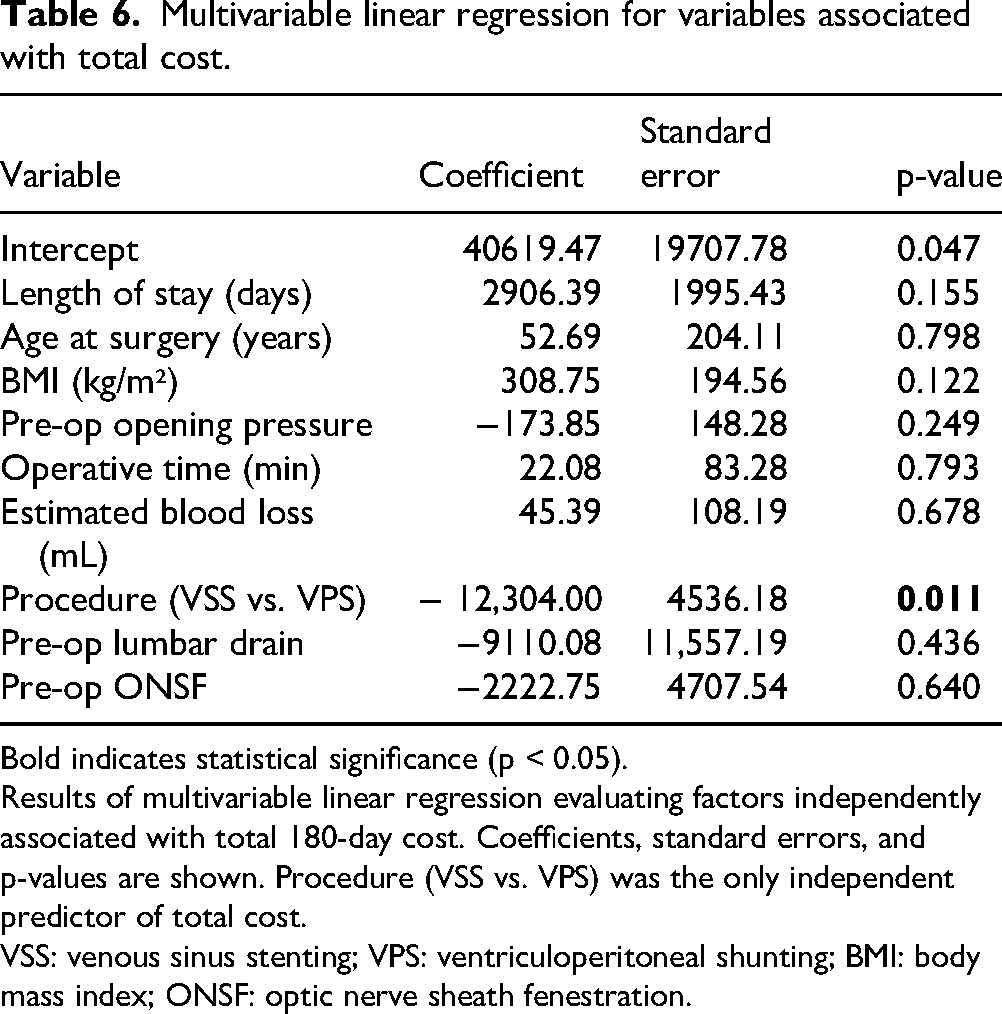
Bold indicates statistical significance (p < 0.05). Results of multivariable linear regression evaluating factors independently associated with total 180-day cost. Coefficients, standard errors, and p-values are shown. Procedure (VSS vs. VPS) was the only independent predictor of total cost.
VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting; BMI: body mass index; ONSF: optic nerve sheath fenestration.
Clinical outcomes
Operative time was significantly longer for VSS compared to VPS (96.0 ± 26.4 min vs. 68.3 ± 18.2 min; p = 0.001), while estimated blood loss was significantly lower in the VSS group (9.2 ± 7.2 mL vs. 26.5 ± 20.5 mL; p < 0.0001). Length of hospital stay was comparable between groups (1.26 ± 1.15 days for VSS vs. 1.17 ± 0.48 days for VPS; p = 0.795). Total follow-up duration was also similar, with mean time to last neurosurgical follow-up of 404.4 ± 367.0 days for VSS and 594.2 ± 574.7 days for VPS (p = 0.434). Reoperation rates through last documented follow-up did not differ significantly, occurring in 3 of 19 VSS patients (16%) and 4 of 24 VPS patients (17%) (p = 0.735); all reoperations were of the same type as the index procedure (Table 7).
Key clinical and surgical outcomes.

Bold indicates statistical significance (p < 0.05). Key clinical and surgical outcomes for patients treated with VSS or VPS. Data are reported as mean ± SD or number (percentage).
VSS: venous sinus stenting; VPS: ventriculoperitoneal shunting.
Discussion
This study presents a comprehensive comparison of VSS and VPS for the treatment of IIH, focusing on perioperative cost and clinical outcomes using real-world institutional cost data. While VSS was associated with significantly higher total perioperative costs, this difference was almost entirely attributable to the preoperative phase. Surgical admission costs, postoperative expenses, LOS, and reoperation rates were statistically comparable between groups, suggesting that the procedure itself and its immediate recovery are not responsible for the financial disparity. Rather, the diagnostic workup required to determine candidacy for VSS appears to be the primary driver of its higher short-term costs.
The preoperative evaluation for VSS typically includes MRV or CTV, diagnostic cerebral venography, trans-stenotic venous manometry, and initiation of dual antiplatelet therapy. These components, while essential for ensuring appropriate patient selection, incur significant fixed and variable costs. In contrast, patients undergoing VPS generally proceed to surgery based on standard imaging and clinical criteria, without the need for invasive hemodynamic confirmation. This distinction highlights a key opportunity for cost containment: optimization of the VSS diagnostic pathway. Venous manometry, in particular, is resource-intensive and often performed as a separate procedure, which adds further cost and logistical burden.
Emerging strategies may help streamline this process. Combining diagnostic manometry and stenting into a single session for patients with a clear pressure gradient may be an option for reducing procedural inefficiency. 11 Recent studies support performing venous manometry and stenting under conscious sedation, which provides treatment and physiologic pressure measurements while avoiding the cost and complexity of general anesthesia.11,12 Experimental approaches, such as computational fluid dynamics modeling based on noninvasive imaging, may eventually allow clinicians to predict which patients have significant pressure gradients without invasive catheter-based measurements.13,14 These innovations warrant further investigation, as they may preserve diagnostic accuracy while significantly reducing the cost and complexity of the VSS preoperative workup.
An important consideration in interpreting these findings is that this study included only elective cases of IIH. Fulminant presentations—characterized by rapid vision loss or severe symptoms requiring urgent intervention—were excluded. These emergent cases often bypass the full preoperative diagnostic algorithm and proceed directly to VPS, which may shorten time to treatment but also introduce more variability in perioperative resource utilization. By focusing exclusively on elective cases, this study likely captured a more controlled comparison between VSS and VPS in patients who were stable enough to undergo a complete diagnostic workup. While this improves internal validity, it also limits generalizability to the broader IIH population, especially those requiring urgent intervention. Future analyses should separately examine the cost and outcome profiles of emergent IIH cases, where the balance between speed, access, and procedural choice may differ significantly.
While VSS appears more expensive in the short term, it may offer important long-term advantages that are not captured within the perioperative cost window. Chief among these is the avoidance of permanent intracranial hardware. VPS is associated with substantial long-term morbidity due to shunt failure, obstruction, migration, and infection. Revision rates for VPS in IIH are high, with studies citing cumulative failure and revision rates of 30–50%.6,15,16 Shunt infections, though less frequent, are clinically serious and financially burdensome, often requiring complex, multi-stage surgical revisions. 17 Agarwal et al. found that each shunt revision can lead to supply costs of $1500–$3500. 18 Moreover, shunt dependency imposes a chronic healthcare burden, including frequent imaging, emergency department visits, and repeat interventions, all of which add to the long-term economic and psychosocial toll on patients.
In contrast, VSS avoids these hardware-related risks. Long-term follow-up studies have consistently shown low reintervention rates after successful stent placement, with up to 87% of patients remaining intervention-free over extended periods. 19 Severe complications from VSS are rare, and stenting appears to provide durable symptom relief for well-selected patients. 6 Thus, while VSS carries higher up-front costs, it may reduce downstream utilization and improve long-term quality of life—a hypothesis that must be tested in future longitudinal studies.
Several limitations should be acknowledged. This study is retrospective and based at a single academic institution, which may limit generalizability and introduce selection bias. The sample size, while sufficient to detect significant cost differences, may underpower analyses of less common clinical outcomes or subgroup effects. Because treatment was not randomized, selection bias may have influenced both procedural choice and associated costs. Patients undergoing VSS may have differed systematically in terms of referral patterns, disease severity, or access to subspecialty evaluation, which could influence preoperative utilization. Although we used institutional cost data rather than reimbursements or charges, we were unable to itemize surgical costs into more granular components (e.g. operating room personnel, implants, imaging) due to institutional disclosure restrictions. There may be underlying patient- or procedure-specific factors—such as stent type or valve programmability—that influence costs but were not captured in this analysis.
Finally, while we captured costs over a 180-day perioperative window, this time frame does not reflect the long-term costs and complications that are relevant for procedures like VPS, where hardware failure and revisions often emerge well beyond the early postoperative period.
Future research should address these limitations through prospective, multi-center studies with long-term follow-up. Comparative effectiveness and cost-utility analyses over multi-year timeframes will be critical to understanding the full economic and clinical trajectories of VSS and VPS. These studies should incorporate patient-reported outcomes, visual function, pulsatile tinnitus, health-related quality of life, and total healthcare utilization to provide a more comprehensive assessment of value. Particular attention should be paid to emergent IIH presentations, which may follow different diagnostic and therapeutic pathways. Efforts should also focus on risk stratification tools to guide patient selection, the ability to avoid permanent hardware through utilization of VSS, and strategies to streamline the VSS diagnostic workflow, including integration of manometry into the therapeutic session, sedation optimization, and development of noninvasive alternatives.
In summary, this study demonstrates that while VSS incurs higher short-term costs compared to VPS, this difference is not due to the surgical procedure or its complications, but rather to the diagnostic intensity required for appropriate patient selection. By targeting the preoperative workup—particularly venous manometry—for optimization, it may be possible to enhance the cost-effectiveness of VSS without compromising clinical rigor. At the same time, the long-term complications and cumulative costs associated with VPS highlight the potential value of durable, hardware-free interventions such as VSS. A more complete understanding of these trade-offs will require prospective evaluation over extended follow-up, but these findings lay a foundation for such future work.
Conclusion
In this retrospective analysis of IIH, VSS was associated with higher total perioperative costs compared to VPS, driven primarily by more extensive preoperative diagnostics. Surgical admission and postoperative costs were comparable, and outcomes did not differ, indicating that the procedure itself is not the main contributor to the cost difference.
These findings suggest that the preoperative workup for VSS is a key target for cost optimization. Techniques such as combining diagnostics and treatment in a single session, using conscious sedation, or exploring noninvasive pre-stent evaluation may improve value.
While VPS carries lower initial costs, it is associated with significant long-term risks, including hardware failure and revision surgeries. VSS may offer greater long-term durability by avoiding shunt-related complications. Future multi-center, prospective studies with extended follow-up are needed to clarify the long-term cost-effectiveness and clinical impact of each approach.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
