Abstract
Purpose
Recent advancements in flow-diverter (FD) technology have introduced various coating strategies to reduce platelet aggregation and thromboembolic complications in intracranial aneurysm treatment. This study compared postinterventional diffusion-weighted imaging (DWI) lesions, as markers of thromboembolic events, between coated and uncoated FDs.
Methods
A retrospective analysis was conducted on patients treated for intracranial aneurysms between June 2018 and December 2024. The primary outcome was the occurrence of postinterventional DWI + lesions, with periprocedural factors analyzed for their association.
Results
A total of 91 patients with 104 aneurysms were treated with 43 uncoated (n = 30 FRED, n = 13 DERIVO) and 51 coated FDs (n = 36 FREDX, n = 15 DERIVO2Heal). DWI + lesions were observed in 39.5% of uncoated and 54.9% of coated FD cases (p = 0.1). After 1:1 propensity score matching, no significant difference was found (p = 0.131). Contrast stasis (p = 0.025) within aneurysm sac after FD deployment, FD proximal/distal ratio (p = 0.007), FD size (p = 0.047), and intervention duration (p = 0.036) were significantly associated with DWI + lesions. In multivariable analysis, contrast stasis (ORa = 3.7, 95% CI 1.1–11.9, p = 0.029), FD proximal/distal ratio (ORa = 27.8, 95% CI 1.8–426.8, p = 0.017), and older age (ORa = 1.06, 95% CI 1.00–1.11, p = 0.050) increased incidence of DWI + lesions.
Conclusion
In this single-center observational study, coated FRED and DERIVO FDs did not reduce postinterventional DWI + lesions compared to their uncoated counterparts.
Introduction
With the first device receiving FDA approval in 2011 for large or wide-necked aneurysms of the internal carotid artery (ICA), flow-diverters (FDs) have become an increasingly utilized and effective treatment option, demonstrating promising long-term aneurysm occlusion rates.1,2 Constructed as braided stents with high metal coverage, FDs function by inducing flow stagnation and subsequent thrombosis in the aneurysm sac while promoting parent vessel endothelialization at the aneurysm neck. However, the need for effective dual antiplatelet therapy (DAPT) remains a significant drawback of FD treatment, as the high mesh density can otherwise induce platelet activation, potentially leading to in-stent thrombus formation and life-threatening ischemic strokes. A meta-analysis of 1656 FDs from previous generations reported an acute ischemic stroke rate of 5% and delayed ischemic events at 4.2%, highlighting thromboembolic events as a major safety concern despite the use of antiplatelet therapies. 3
To reduce the thrombogenicity of FDs without compromising their high metal coverage, FD manufacturers have, in recent years, developed various coating techniques for stent surface modification in their latest products. Preliminary ex vivo and animal studies have demonstrated enhanced hemocompatibility and reduced thrombogenicity achieved through these coating techniques.4,5 Retrospective observational studies involving small patient cohorts have further indicated the safety and efficacy of surface-modified FDs, even in patients undergoing single antiplatelet therapy (SAPT).6,7 A randomized multicenter trial aimed at comparing thromboembolic and hemorrhagic complications between coated FDs under SAPT and bare FDs under DAPT is currently ongoing. 8
To date, only limited studies have compared the postoperative short-term thrombogenicity of coated FDs with their uncoated counterparts, such as Pipeline Flex with Shield Technology versus Pipeline Flex, and have found no significant differences in the incidence of thromboembolic events. 9 Given the substantial variation in surface modification strategies among FD manufacturers, we conducted a retrospective study to compare coated and uncoated FDs, focusing on short-term thromboembolic events detected via magnetic resonance imaging (MRI). Additionally, we aimed to identify periprocedural factors associated with the occurrence of postinterventional thromboembolic events.
Methods
Study design
This retrospective study analyzed consecutive patients who underwent endovascular treatment of intracranial aneurysms with FDs at a tertiary care center in Germany between June 2018 and December 2024. Ethical approval was obtained from the institutional review board (#15-6331-BO), and the study was conducted in full compliance with the Declaration of Helsinki. Inclusion criteria were patient age over 18 years, FD treatment for intracranial aneurysms, and availability of postinterventional MRI within 3 weeks of the procedure. To facilitate a direct comparison between FDs with and without surface modification, only FDs from the same manufacturer with identical stent designs were included in the analysis.
Patient characteristics and periprocedural factors
Patient baseline characteristics, including age, sex, smoking status, and diabetes mellitus, were documented. For elective cases, patients underwent platelet function analyzer (PFA) testing and were prescribed DAPT with 100 mg aspirin and 75 mg clopidogrel daily for 1 week prior to the procedure. In cases where clopidogrel resistance was detected using vasodilator-stimulated phosphoprotein blood (VASP) testing, clopidogrel was replaced with either 90 mg ticagrelor twice daily or 10 mg prasugrel once daily. DAPT was continued for 6 months, after which aspirin was maintained for life. In acute cases of subarachnoid hemorrhage (SAH), bodyweight-adapted tirofiban was administered intravenously during the neurointervention as a bridging therapy before transitioning to DAPT.
Aneurysms were categorized as either unruptured or ruptured. Additionally, a distinction was made between untreated aneurysms and those with prior treatments, such as coils and/or stents. Aneurysm dimensions were assessed using three-dimensional rotational angiography, with measurements taken for the aneurysm neck, sac height, and sac width. Dome-to-neck and ASPECT ratios were calculated accordingly. 10 Parent vessel diameters were measured at the proximal and distal landing zones of the FD as well as at the aneurysm neck. Stenosis within the stented segment was classified as less than 30%, 30% to 70%, or greater than 70%, based on a comparison of the narrowest vessel segment being stented with the vessel diameter 5 mm proximally.
Aneurysms were further categorized by morphology as saccular or fusiform and by location as sidewall, side-branch, or terminus aneurysms (e.g., carotid T or basilar apex). In cases where a single FD covered multiple aneurysms, the largest aneurysm was designated as the index aneurysm for analysis.
Flow-diverter deployment-related variables
All patients underwent general anesthesia. A bolus of heparin (50 U/kg) was administered intravenously, followed by additional boluses of 1000 units every hour. Several key parameters related to FD deployment were assessed, including complete opening of the proximal and distal stent ends, full neck coverage, and complete wall apposition along the entire device length. Aneurysm filling and blood stasis within the sac were evaluated before and after FD deployment using the O’Kelly–Marotta grading scale (OKM). 11
Procedures were classified based on whether the FD was implanted alone or in combination with concomitant coiling. Coils from two different manufacturers were mainly used: bare platinum and hydrogel-coated ones (MicroVention, Tustin, CA, USA; Stryker, Venlo, Netherlands). Intraprocedural thromboembolic events and in-stent thrombosis were systematically recorded. The duration of the intervention was defined as the time interval between the first and last digital subtraction angiography (DSA) acquisitions.
All FDs were implanted by a consistent team of three neurointerventionists: one with over 25 years of interventional experience and two with approximately 6 years each. The procedures were fairly evenly distributed among the three operators.
Postinterventional MRI evaluation
Generally, a postinterventional baseline MRI was performed for all patients before discharge. Diffusion-weighted imaging (DWI) was used to identify ischemic lesions, defined as brain tissue areas exhibiting high signal intensity on b1000 sequences with corresponding reversed signal intensity on the apparent diffusion coefficient (ADC) map. DWI + lesions were correlated with the vascular territories corresponding to the implanted FD. The total number of lesions was recorded, with the largest lesion measured in millimeters. Clinically silent DWI + lesions were defined as those without newly manifested neurological deficits. Postprocedural MRI scans were performed using either a 1.5-T (Siemens, Aera, Erlangen, Germany) or 3-T (Siemens, Skyra, Erlangen, Germany) scanner with a standardized DWI Protocoll across both systems to ensure comparability. The neuroimaging analysis was independently performed by two board-certified neuroradiologists (YL, CD), who were blinded to the type of implanted FD.
Study outcomes
The primary outcome of the study was the occurrence of postinterventional DWI + lesions. We compared the presence of DWI + lesions between patients treated with coated and uncoated FDs. Additionally, all periprocedural factors were analyzed to assess their association with the occurrence of DWI + lesions.
As a secondary outcome, the prevalence of hyperacute thromboembolic events, including in-stent thrombosis and peripheral thromboemboli, was compared between the coated and uncoated FD groups. Finally, we evaluated the clinical impact of DWI + lesions by correlating their presence with patients’ neurological status at discharge.
Statistical analysis
Categorical variables were summarized as frequencies and percentages, while numerical variables were presented as means with standard deviations. Comparisons of categorical variables were conducted using the Chi-squared test or Fisher's exact test, while numerical variables were analyzed using Student's t-test for normally distributed data and the Mann–Whitney U test for non-normally distributed data.
Individual propensity scores (PS) were calculated using a multivariable logistic regression model, with coated FD as the response variable. Based on these propensity scores, 1:1 matching was performed to create two comparable FD groups, one with and one without coating. A total of 35 periprocedural covariates were included in the regression model to assess their association with the occurrence of DWI + lesions. In an initial model, the FD proximal-to-distal diameter ratio showed a disproportionately high effect estimate, prompting further evaluation. To address potential bias, multicollinearity was assessed using the variance inflation factor (VIF), and only variables with VIF < 5 were retained in the final model. Additionally, cases with extreme values (>3 standard deviations from the mean) for continuous predictors were excluded to reduce the influence of outliers.
Additionally, Pearson correlation analysis was used to evaluate the relationship between periprocedural covariates and the number of DWI + lesions.
All statistical analyses were conducted using SPSS (version 29, IBM, Armonk, NY, USA), with a p-value < 0.05 considered statistically significant.
Results
Baseline patient and aneurysm characteristics
A total of 99 patients with 114 intracranial aneurysms underwent FD treatment between June 2018 and December 2024. Of these, 91 patients with 104 aneurysms treated with 94 FDs met the inclusion criteria and were included in the final analysis. The mean patient age was 55.7 years (±10.4), with a majority being female (76%, 71/94). Smoking and diabetes mellitus were present in 57 (61%, 57/94) and 12 (13%, 12/94) patients, respectively (Table 1). The most used DAPT regimen was aspirin + clopidogrel (78.7%, 74/94), followed by aspirin + ticagrelor (14.9%, 14/94) and aspirin + prasugrel (6.4%, 6/94). Patients in the coated FD group were significantly older (58 vs. 53 years, p = 0.02); however, after 1:1 propensity score matching, this age difference was no longer statistically significant.
Baseline patient and aneurysm characteristics.
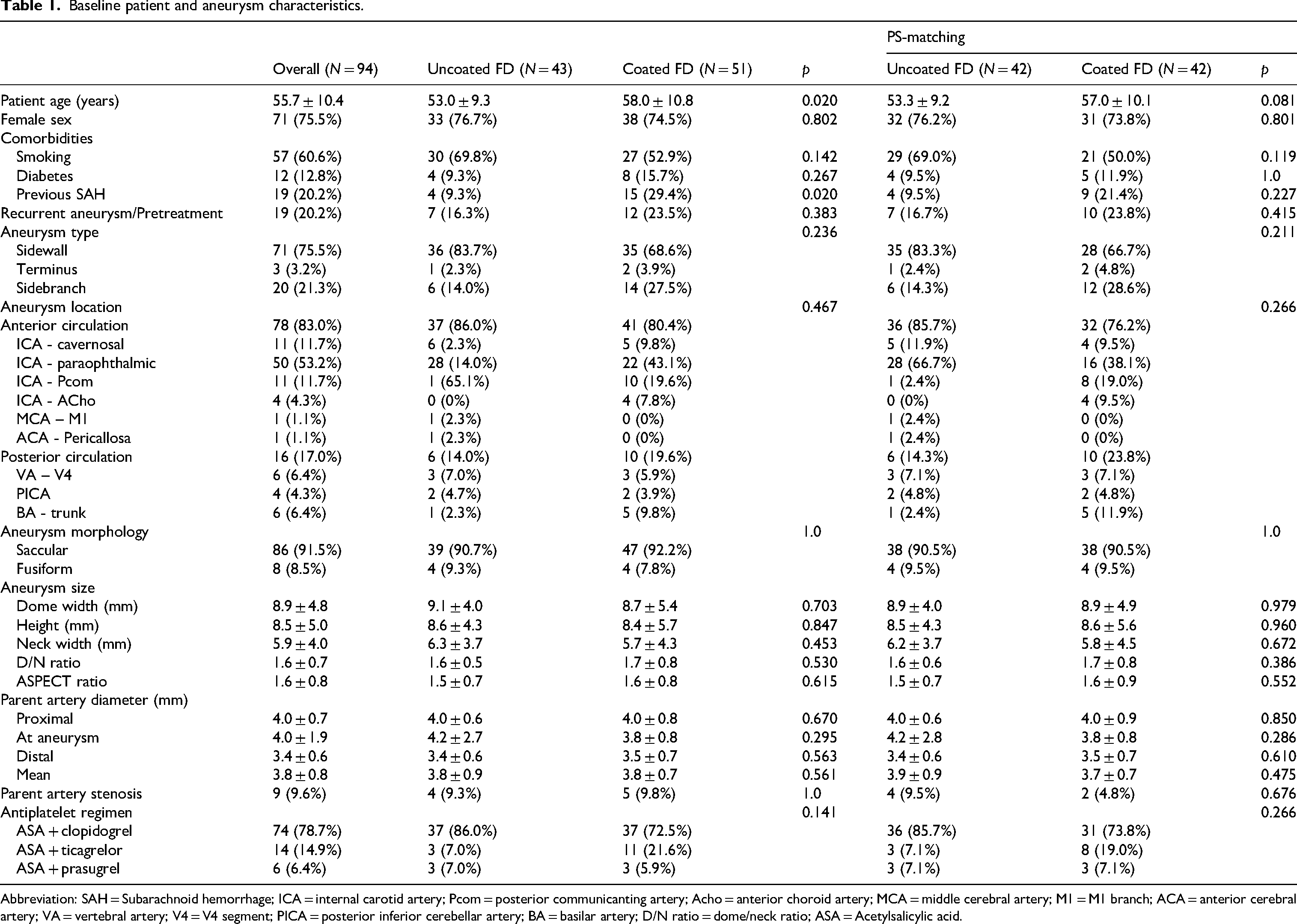
Abbreviation: SAH = Subarachnoid hemorrhage; ICA = internal carotid artery; Pcom = posterior communicanting artery; Acho = anterior choroid artery; MCA = middle cerebral artery; M1 = M1 branch; ACA = anterior cerebral artery; VA = vertebral artery; V4 = V4 segment; PICA = posterior inferior cerebellar artery; BA = basilar artery; D/N ratio = dome/neck ratio; ASA = Acetylsalicylic acid.
Among the 94 index aneurysms, 86 (91.5%, 86/94) were saccular, and 19 (20%, 19/94) had a history of rupture. Only 3 aneurysms (3.3%, 3/94) were treated in the acute setting of subarachnoid hemorrhage (SAH). Additionally, 19 aneurysms (20%, 19/94) had undergone previous treatment before FD implantation. The majority (83%, 78/94) were located in the anterior circulation, with most classified as sidewall aneurysms (76%, 71/94). The paraophthalmic segment of the ICA was the most frequently treated location (53.2%, 50/94).
The mean aneurysm width was 8.9 mm (±4.8), height 8.5 mm (±5.0), and neck 5.9 mm (±4.0). The mean parent artery diameters were 4.0 mm (±0.7) at the proximal FD landing zone, 4.0 mm (±1.9) at the aneurysm neck, and 3.4 mm (±0.6) at the distal FD landing zone (see Supplementary Materials).
Procedural factors and thromboembolic complications
Of the 94 implanted FDs, 30 were FRED (Flow-Redirection Endoluminal Device; MicroVention, Aliso Viejo, CA, USA) as uncoated bare FDs, and 36 were FREDX as coated devices. Additionally, 13 were DERIVO2/DERIVO (DERIVO; Acandis, Pforzheim, Germany) as uncoated FDs, while 15 were DERIVO2Heal as coated ones (Table 2). The mean FD size was 4.3 mm (±0.6), with a mean length of 19.2 mm (±7.0).
Procedural characteristics and complications.
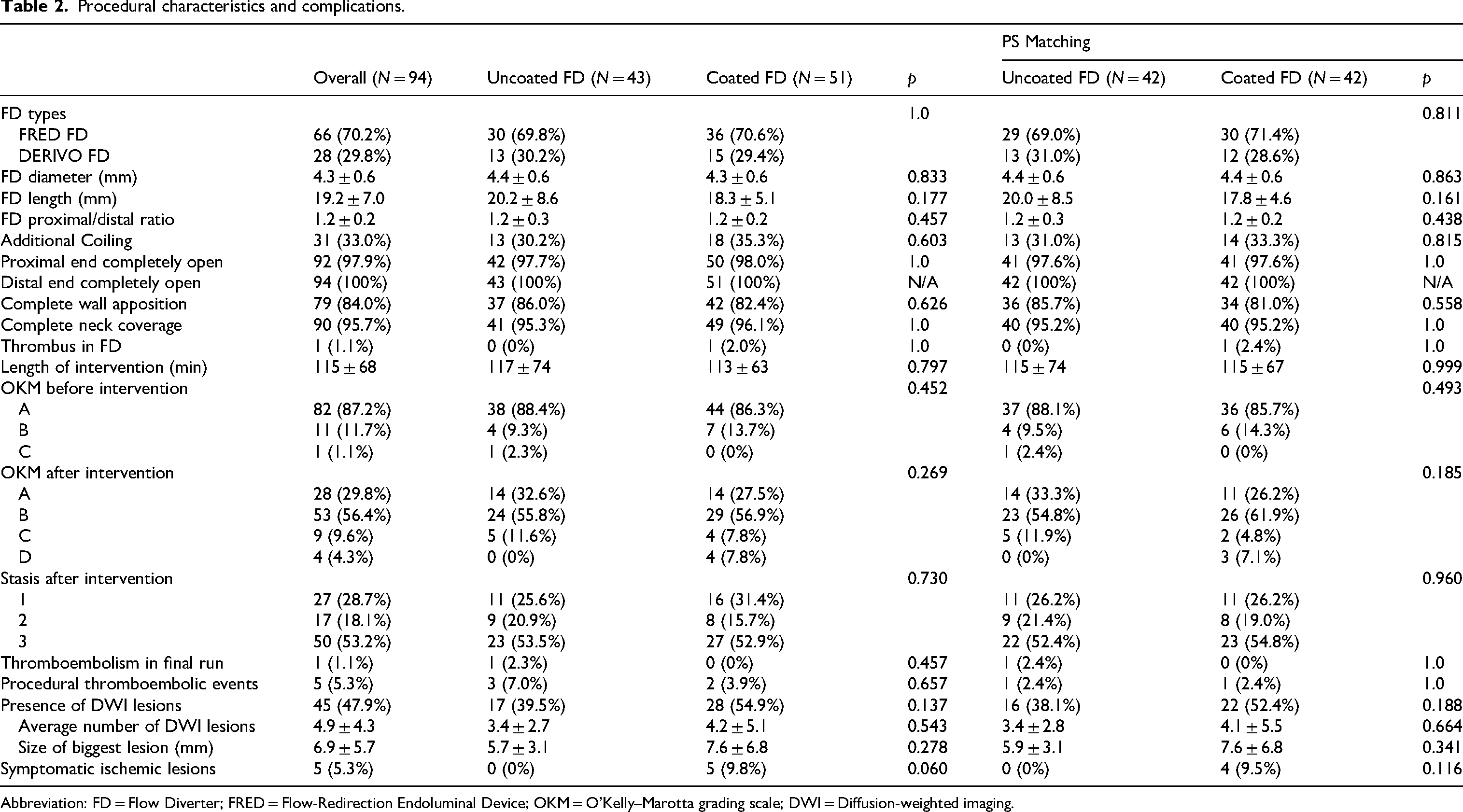
Abbreviation: FD = Flow Diverter; FRED = Flow-Redirection Endoluminal Device; OKM = O’Kelly–Marotta grading scale; DWI = Diffusion-weighted imaging.
Concomitant coiling was performed in one-third of all cases. Significant stasis (OKM 3) of the contrast agent at final angiographic control was observed in 53% of cases. The median intervention time was 114 minutes (±6.8). There was one case of intraprocedural hyperacute peripheral thromboembolism with FRED and one case of in-stent thrombosis with FREDX.
Postinterventional DWI + lesions
The median time interval between neurointervention and MRI was 46 hours (±67). DWI + lesions were observed in 37% of FRED, 58% of FREDX, 46% of DERIVO/DERIVO2, and 47% of DERIVO2Heal cases (Figure 1).
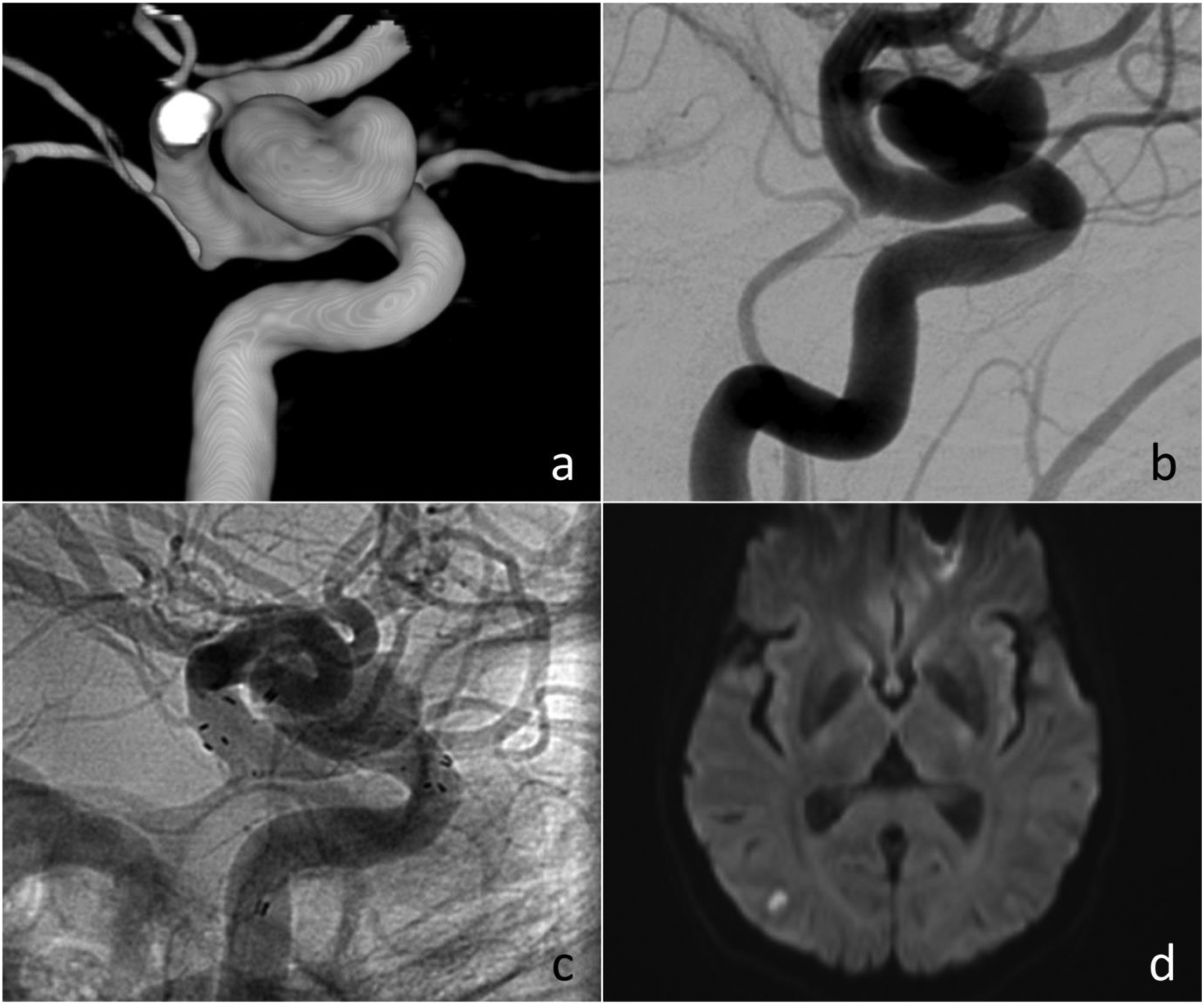
DWI + lesion in a 45-year-old patient following treatment with flow-diverter. (a, b) Saccular supraophthalmic aneurysm of the right ICA shown in 3D rotational angiography and DSA. (c) Flow-diverter (FRED) implantation. (d) DWI + lesion in the right temporal lobe detected 21 hours postimplantation.
The prevalence of DWI + lesions was slightly higher in the coated FD group compared to the uncoated group (55% vs. 40%, p = 0.137 for the unmatched comparison; 52% vs. 38%, p = 0.188 after propensity score matching) (Table 2).
Among the 35 periprocedural factors tested in the univariate analysis, FD size, proximal/distal landing zone ratio, post-deployment contrast stasis within the aneurysm sac (OKM > 1), and intervention duration were significantly different between the coated and uncoated groups (see Supplementary Materials). In the multivariable logistic regression analysis, after adjusting for confounders (Table 3), increased patient age (ORa = 1.06, 95% CI 1.00–1.11, p = 0.050), a higher proximal/distal landing zone ratio (ORa = 27.8, 95% CI 1.8–426.8, p = 0.017), and post-deployment contrast stasis (ORa = 3.7, 95% CI 1.1–11.9, p = 0.029) were significantly associated with an increased likelihood of DWI + lesions. No association was found between the occurrence of DWI + lesions and FD coating status.
Multivariate logistic regression analysis on the presence of DWI lesions.
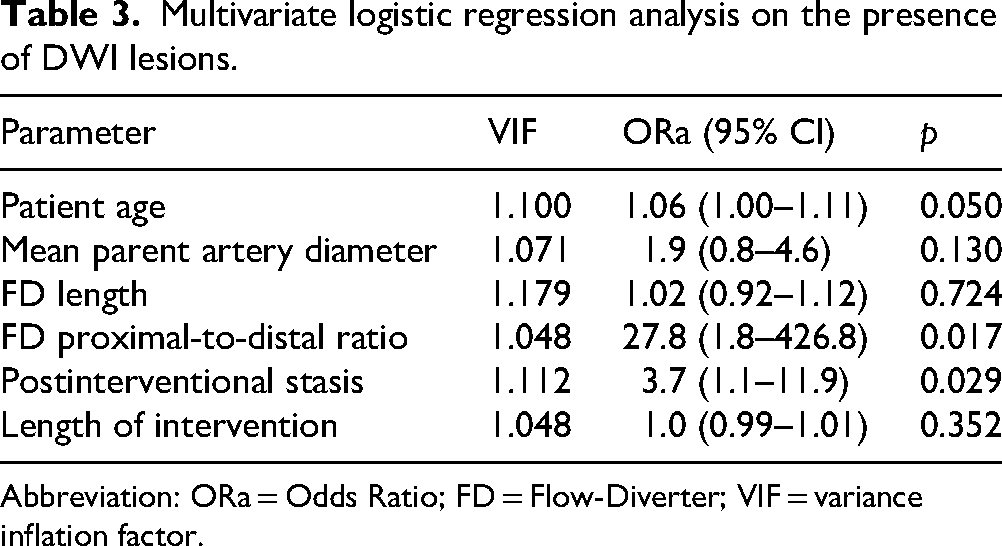
Abbreviation: ORa = Odds Ratio; FD = Flow-Diverter; VIF = variance inflation factor.
Pearson correlation analysis showed that patient age (ρ = 0.289, p = 0.005), aneurysm dome height (ρ = 0.212, p = 0.04), and FD proximal/distal ratio (ρ = 0.297, p = 0.004) were significantly correlated with the number of DWI + lesions (see Supplementary Materials).
Postprocedural neurological outcomes of patients
Of the 94 FD cases, newly developed neurological deficits were observed in 5 patients at discharge (5%, 5/94). Among these, 4 cases exhibited acute onset within 24 hours, while 1 patient developed delayed symptoms after 8 days. Corresponding DWI + lesions were present in all symptomatic cases (11%, 5/45), whereas the remaining lesions were clinically silent (89%, 40/45).
Patients without DWI + lesions did not exhibit any new neurological symptoms at discharge. However, two patients (4%, 2/49) experienced delayed hemorrhagic strokes with parenchymal hematoma occurring 3 and 5 days postprocedure, respectively (Figure 2).
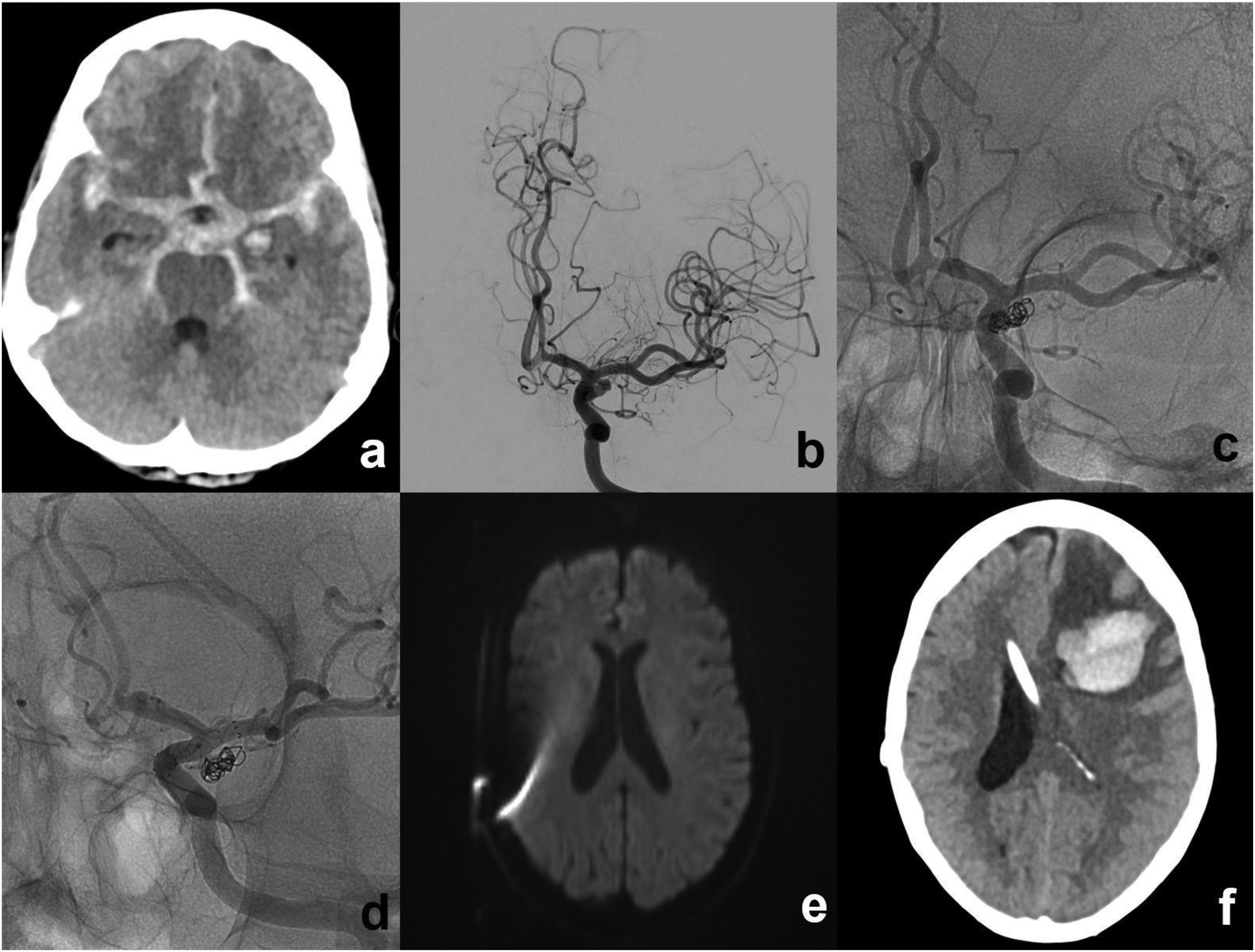
Delayed hemorrhagic stroke in a 63-years-old patient. (a) Presence of acute SAH on CT. (b) DSA showing aneurysm of left anterior choroid artery. (c) Aneurysm treatment with loose coil package for preservation of anterior choroid artery. (d) Staged aneurysm treatment with flow-diverter after 34 days. (e) Absence of any DWI lesions at patient's discharge. (f) Acute onset of right-sided arm-dominant hemiparesis due to acute parenchymal hematoma 3 days after discharge.
Discussion
The main finding of this single-center observational study with a limited patient number was that coated FRED and DERIVO FDs under DAPT were not associated with a reduction in postinterventional DWI + lesions compared to their bare metal counterparts. Instead, advanced patient age, contrast-agent stasis within the aneurysm sac after FD deployment, and a high FD proximal/distal ratio were significantly correlated with the occurrence of DWI + lesions.
Although most DWI + lesions remained clinically silent, efforts to minimize them remain crucial, as 11% of cases resulted in neurological impairment. As Goyal et al. highlighted, the brain warrants protection from even minor injury, especially given our limited understanding of periprocedural DWI lesions. 12 In our study, symptomatic DWI + lesions were defined as newly occurring neurological deficits, although this does not capture the full spectrum of brain functions, such as cognition.
A key question in FD treatment is the origin of these lesions. The incidence of DWI + lesions following FD implantation is markedly higher than that observed in diagnostic cerebral angiography, 13 and unlike diagnostic angiography, some of these lesions were symptomatic. Our results suggest that coating technology did not reduce DWI + lesion incidence, consistent with findings from Cortez et al., who reported similar rates between coated and uncoated FDs (62% in Pipeline Shield vs. 61% in Pipeline Flex). 9 Another study investigating the coated p48-MW-HPC device found an even higher DWI + rate (70%). 14 These findings suggest that, regardless of coating strategy, additional surface modifications do not necessarily reduce DWI + lesions under DAPT. A study of significant clinical relevance, comparing coated FDs under SAPT with uncoated counterparts under DAPT, is still ongoing. 8
The prevalence of DWI + lesions in both the coated and uncoated groups (52% vs. 38% after propensity score matching) in our study was lower than previously reported in the literature. This may be partly explained by structural differences between different FDs and their specific coating techniques (see Supplementary Materials), which warrants further investigation. The time interval between intervention and MRI likely also influences detection rates. Additionally, MR technical factors such as differences in MR vendors, field strength, slice thickness, and applied b-values of DWI among studies may have contributed to the observed variability.
Our results indicate that postinterventional DWI + lesions cannot be solely attributed to the intervention or the FD itself. Instead, we hypothesize that a portion of these lesions may originate from thromboemboli within the aneurysm sac. Post-deployment contrast-agent stasis (OKM 2 or higher) was a strong predictor of DWI + lesions (ORa = 3.7, 95% CI 1.1–11.9, p = 0.029). Stasis reflects reduced flow velocity, decelerated inflow rates, and lower shear stress, all of which promote intra-aneurysmal thrombus formation.15,16 While such pro-thrombotic hemodynamic changes are desirable for aneurysm occlusion, they may also facilitate thrombus escape before endothelialization is complete, leading to DWI + lesions. Supporting this hypothesis, aneurysm dome height was significantly correlated with the number of DWI + lesions (ρ = 0.212, p = 0.04), likely reflecting the proportional relationship between aneurysm dome size, intra-aneurysmal thrombus formation, and distal thromboembolism.
Another key finding was that a higher FD proximal/distal ratio was strongly associated with both the likelihood (ORa = 27.8, 95% CI 1.8–426.8, p = 0.017) and number of DWI + lesions (ρ = 0.297, p = 0.004).
Proper FD sizing is crucial for optimal flow diversion at the aneurysm neck. Oversizing can increase stent porosity and cell size, potentially diminishing the flow-diverting effect and increasing the risk of in-stent stenosis and braid deformation.17,18,19 A higher proximal/distal ratio, often a consequence of FD oversizing, may lead to increased porosity at the aneurysm neck, facilitating microthrombus escape from the sac. 20 Additionally, greater diameter discrepancies between the FD and distal landing zone may enlarge stent cell openings at transition zones, resulting in reduced metal coverage and an increased risk of thromboembolism. 21 In the multivariable analysis, the proximal-to-distal diameter ratio of the FD remained a significant predictor of DWI + lesion occurrence (ORa = 27.8; 95% CI: 1.8–426.8; p = 0.017). Despite the large effect size, the low variance inflation factor (VIF = 1.048) and the robustness of the association after excluding outliers argue against substantial multicollinearity or undue influence from extreme values. However, the wide confidence interval highlights statistical imprecision related to the limited sample size and number of events. Therefore, the observed association should be interpreted with caution and warrants validation in larger studies.
Furthermore, increased patient age was significantly correlated with DWI + lesion occurrence, a finding consistent with multiple previous studies.13,22–24 Since patient age is an immutable factor and post-deployment contrast stasis is often desirable for aneurysm occlusion, 15 optimizing FD sizing remains the most critical modifiable factor for reducing short-term thromboembolic complications.
However, one should keep in mind that an unremarkable postprocedural MRI does not guarantee a complication-free course, as two patients in our study experienced intracerebral hemorrhages after hospital discharge.
The main limitations of our study include its retrospective, single-center design and the inclusion of two different FD models. Additionally, uncoated FDs were primarily used before 2022, while coated devices were predominantly implanted thereafter. As operator experience increased over time, implantation quality may have improved with coated devices, introducing a potential selection bias. However, since neither the specific FD model nor the coating status was associated with the occurrence of DWI + lesions, these factors are unlikely to compromise the validity of our findings. Nevertheless, additional data, particularly from prospective studies, are needed to directly compare uncoated FDs under DAPT with coated FDs under SAPT. Furthermore, the lack of randomization in assigning patients to treatment groups introduces the potential for intergroup comparisons to be influenced by unmeasured confounders not captured in the dataset. To mitigate this, we utilized patient matching and multivariable regression models to reduce the impact of the key confounders identified within our analysis.
Conclusion
In this single-center observational study, coated FRED and DERIVO FDs did not reduce postinterventional DWI + lesions compared to their uncoated counterparts. Contrast stasis within aneurysm sac after FD deployment, higher FD proximal/distal ratio of landing zones, and older age of patient increased incidence of postinterventional DWI + lesions.
Supplemental Material
sj-docx-1-ine-10.1177_15910199251367504 - Supplemental material for Impact of surface coating on postinterventional DWI lesions: A comparative analysis of coated and uncoated FRED and DERIVO flow-diverters in the treatment of intracranial aneurysms
Supplemental material, sj-docx-1-ine-10.1177_15910199251367504 for Impact of surface coating on postinterventional DWI lesions: A comparative analysis of coated and uncoated FRED and DERIVO flow-diverters in the treatment of intracranial aneurysms by Christoph Ziegenfuß, Natalie van Landeghem, Adrian Engel, Ramazan Jabbarli, Philipp Dammann, Ulrich Sure, Michael Forsting, Isabel Wanke, Lukas Goertz, Christoph Kabbasch, Cornelius Deuschl and Yan Li in Interventional Neuroradiology
Footnotes
Ethics approval
This study was approved by Ethics Committee of the University of Duisburg-Essen (#15-6331-BO) and conducted in accordance with the Declaration of Helsinki. Informed consent was waived by the Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
