Abstract
Background
In mechanical thrombectomy procedures, physicians are often forced to cross the clot to get more support in tortuous anatomy. Stentriever procedures always require traversing the clot. Novel delivery assist catheters with a tapered distal tip have been developed to aid aspiration catheter delivery. This study compares a novel delivery assist catheter to conventional support strategies in terms of reperfusion and distal embolization. The impact of remaining proximal versus entering the clot is also investigated, as is the impact of incorporating a Superbore 088 catheter.
Methods
Soft, medium, and stiff ovine blood clots were used to form ICA/M1 occlusions in an in-vitro thrombectomy model. Two approaches were tested: navigating the aspiration catheter with and without crossing/entering the clot with support devices. Six setups were tested: Millipede88 + Zipline88, Millipede88 + Millipede70 + Zipline70, Millipede70 + Zipline70, SOFIA Plus + 021 Microcatheter, SOFIA Plus + 3MAX, SOFIA Plus + 021 Microcatheter + Stentriever. Each setup was tested nine times totaling 72 tests. Revascularization endpoints were First-Pass-Effect (FPE), final complete revascularization, and distal embolization.
Results
Avoiding clot entry was associated with higher FPE rates, improved final revascularization, and fewer distal emboli. Using a 3MAX as a microcatheter and crossing the clot resulted in more distal emboli than a standard 021 microcatheter or stentriever procedure. Millipede88 + Zipline88 configuration achieved the highest FPE (67%) when the clot was not crossed (vs. 56% with clot entry). Both 070 aspiration catheters had the same FPE rate (33%) but using Millipede88 for flow control with Millipede70 improved the FPE rate (44%). Of the conventional techniques, SOFIA Plus + 021 Microcatheter + Stentriever was associated with the highest FPE rate (44%).
Conclusions
Remaining proximal to the clot is advantageous. Next-generation delivery catheters, like Zipline, may enhance catheter delivery without increasing embolization risk. Using larger internal catheters, such as 3MAX, leads to higher rates of emboli when crossing the clot.
Keywords
Introduction
In neuroendovascular thrombectomy procedures, aspiration and intermediate catheters have traditionally been delivered to the neurovasculature over a standard 021 microcatheter and 014ʺ guidewire. In recent years, neurovascular catheter lumen sizes have increased substantially, initially from 060ʺ to 074ʺ.1,2 More recently a class of aspiration catheter known as Superbore with an 088ʺ lumen has emerged.3–5 Studies have shown that larger aspiration catheters are associated with a higher rate of FPE, higher final recanalization, and lower rates of rescue treatment with both ADAPT and combination techniques.6,7
Successfully delivering large catheters is sometimes challenging, and physicians occasionally require greater internal support often from larger 0.027ʺ microcatheters, or the commonly used 3MAX (Penumbra), to deliver these catheters. Neurovascular wires have also increased significantly in size from the traditional 0.014ʺ to 0.018ʺ–0.024ʺ and mostly recently to the aptly named 0.035ʺ Colossus (Scientia Vascular). Despite these advances in internal support devices, failure to access the clot with the aspiration catheters occurs in up to 25% of cases. 8 In cases of failed delivery of the aspiration catheter, physicians often employ an anchoring technique, whereby they deploy a stentriever across the clot and use this to aid in delivering the aspiration catheter before performing a combined stentriever and aspiration procedure. 9
To address the issue of deliverability, and in addition to making catheters more navigable, companies have developed specialized delivery catheters designed to aid delivery of the aspiration catheter.8,10,11 These include Zipline (Perfuze), Carrier (Balt), Cheetah (Q’Apel), Tenzing (Route 92 Medical), SENDit (Penumbra), Wedge (Microvention), and AXS Offset (Stryker). The design of these catheters is summarized in Table 1. These catheters have an outer diameter (OD) that is closely matched to the internal diameter (ID) of the aspiration catheter reducing the gap, or ledge, between the devices. This design facilitates navigation through difficult anatomy such as tortuous carotid siphon and terminus and helps prevent the catheters from snagging on vessels such as the ophthalmic and posterior communicating arteries. Recent studies suggest that they improve the rates of delivery of the aspiration catheter to the target artery.8,12,13
Comparison of novel delivery assist catheters.

Note: OD: outer diameter, ID: internal diameter.
Ideally, the clot is reached without the need to disrupt it with the wire, microcatheter, or specialized delivery catheter. However, in some instances to gain additional support for an aspiration catheter, the distal tip of the support catheter may enter or even cross the clot as shown in Figure 1. In cases involving a stentriever, the clot is always crossed with the wire and microcatheter in order to deploy the stentriever. With the exception of Penumbra SENDit, the newer specialized delivery catheters have a gently tapered tip allowing the catheter to slide between the vessel wall and the clot, potentially reducing the chances for the clot to be pushed distally or fragmented.
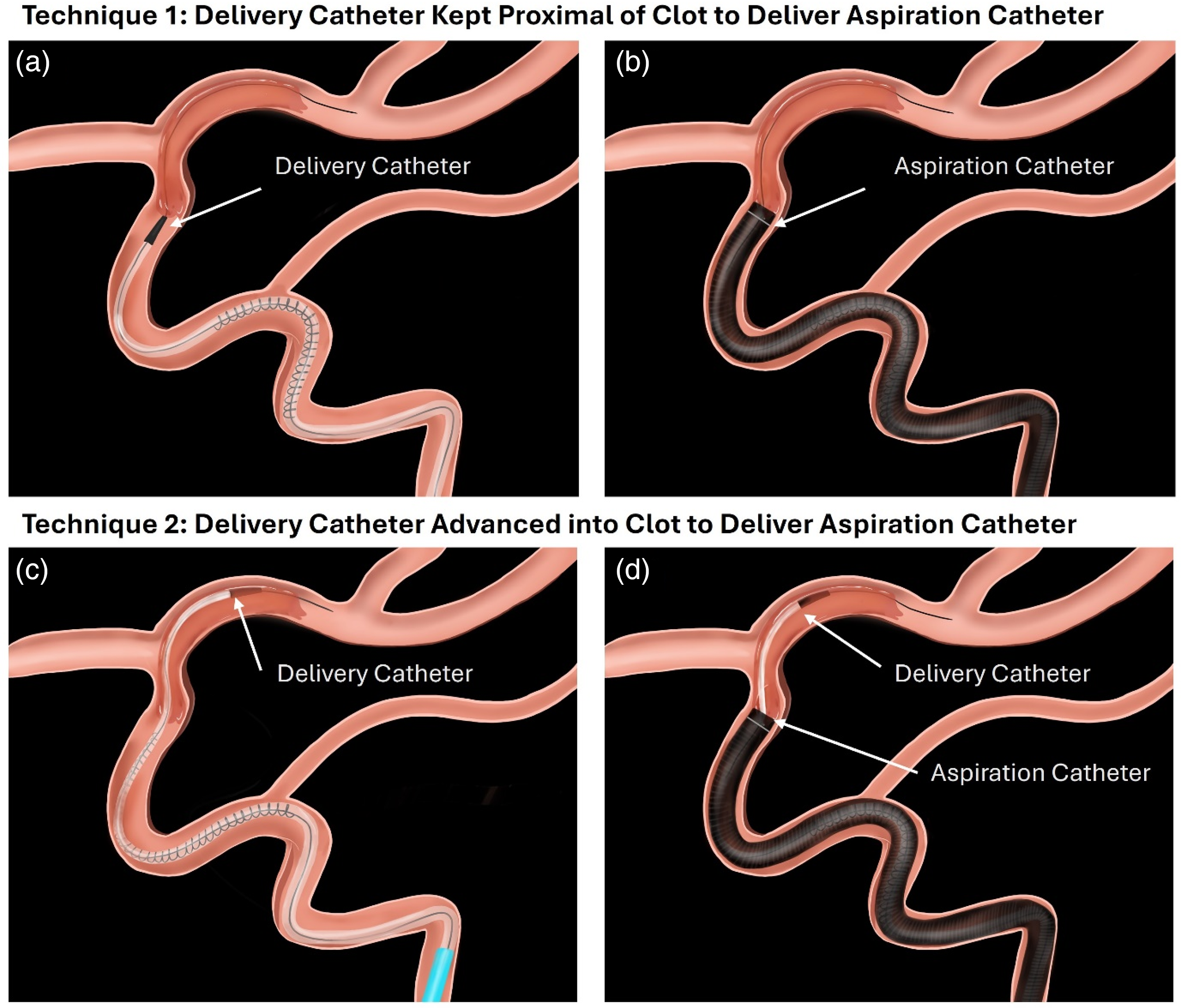
Schematic of techniques evaluated in the study. (a) Delivery catheter kept proximal of the clot face while delivering the aspiration catheter (b). (c) Delivery catheter entering the clot to facilitate delivery of the aspiration catheter/stentriever (d).
Whilst the delivery performance of these novel devices has been proven,8,10,11 the effect of clot interaction with such large devices has not been studied. The aim of this in-vitro study is to compare novel strategies for the delivery of aspiration catheters to traditional techniques in terms of rates of revascularization and distal embolization. Several different clot types are used to represent the diverse compositions encountered clinically. 14
Materials and methods
The data that support the findings of this study are available from the corresponding author upon reasonable request. Ethical approval was not required as the study did not involve human or animal subjects or tissue. Ovine blood for testing was obtained under license from Ellax Medical Ltd (Galway, Ireland).
In vitro thrombectomy model for clot retrieval testing
The fresh blood was centrifuged at 1,200 RPM for 20 min to separate it into its constituents. Plasma was removed by pipetting and the remaining Red Blood Cells and Buffy Coat were mixed together by inverting. Three clot phenotypes were created for clot retrieval testing, Red Blood Cell-Rich (Soft), Mixed (Medium), and Fibrin/Platelet-Rich (Stiff), as per Supplementary Table 1. Thrombin (1 NIH/mL, Roche Diagnostic GmbH, Germany, #T6884) and 5% Calcium chloride (CaCl2) solution (Sigma Aldrich, Ireland, #C1016) were added to stimulate coagulation. The tube containing the clot analogue mixture was quickly mixed by inversion and drawn into 3 mL syringes. The syringes were spun overnight at room temperature on a blood tube rotator (Stuart SB3 Blood Rotator, Cole Palmer, UK) at 40 RPM to mimic the dynamic flow conditions of the human vasculature.
The in-vitro anatomical model has been described previously.15,16 A peristaltic pump (Economy EV3000, Verderflex, UK) is connected to a patient representative 3D printed iliofemoral, aortic arch, and Circle of Willis (COW) model made of REFLEX silicone (Mentice Inc, Illinois, USA), and with individual physiologically representative outflows. The model contains a relatively straight cervical ICA, a 90° petrous bend radius, and a U-shaped carotid Siphon. The pulse rate (72 BPM), intracranial flow rate (750 mL/min), and temperature (37°C) were controlled to replicate in-vivo conditions.
Clots 10 to 15 mm in length were inserted into the cervical ICA under dynamic flow and subsequently lodging at the ICA-T and extending into the M1 segment. Test sieves (>1000, 500 and 200 µm) were used to capture procedure-related distal emboli which occurred during the retrieval attempts.
Devices and techniques
In this study the Perfuze Zipline was compared to a range of more traditional support strategies for aspiration catheter delivery; Penumbra 3MAX and 021 microcatheter. Zipline has a similar design to some of the approved predicates such as Balt Carrier, Q’Apel Cheetah, and Route 92 Medical Tenzing, with a tapered and radiopaque distal tip. One difference between Zipline and the predicates is the presence of a radiopaque coil providing visualization of the catheter at the region of greatest outer diameter and support under fluoroscopy. There are currently two sizes of Zipline available: Zipline70 and Zipline88 with maximum outer diameters of 0.061″ and 0.080″, respectively. These can be used to aid the delivery of Millipede70 large bore and Millipede88 Superbore catheters, respectively. Millipede88 has been approved for use in the USA (FDA Product Code QJP) and Europe. Millipede70 has been approved for use in the USA (FDA Product Code NRY).
SOFIA Plus (Microvention) was chosen as a representative of commonly used 6F aspiration catheters. 3MAX (Penumbra) is an aspiration catheter that is commonly used for providing additional internal support in interventional procedures. A solitaire 4 × 40 mm stentriever device was chosen as a representative of a commonly used stentriever diameter and length. A range of 021 microcatheters were used during testing. For procedures involving Millipede88, an 8F Destination long sheath (Terumo, USA) was placed in the cervical ICA for proximal support. For all other procedures, a Neuron Max, (Penumbra) was placed in the cervical ICA. In one group Millipede88 was used as a guiding catheter for Millipede70, thus providing flow control and distal access.
An aspiration pump (Dominant Flex, Medela AG, Switzerland) was used to supply a static vacuum (≈700 mmHg). An 0.014ʺ micro guidewire was used in all tests. Two principal approaches were tested: crossing/entering the clot with the internal support devices, and without crossing/entering the clot. Nine replicates of each experiment were performed totaling 72 tests. A summary of all test setups can be found in Supplementary Table 2.
Tests without entering the clot
The delivery catheter tip was navigated over a micro guidewire to the face of the occlusion. The aspiration catheter was advanced over the delivery assist catheter to the face of the clot. The delivery catheter was removed, and aspiration tubing was connected to the hub of the aspiration catheter. Aspiration was applied until free flow was observed, or if free flow was not observed, for 2 min after which the aspiration catheter was withdrawn under continuous aspiration.
Tests crossing or entering the clot
In all tests, an 0.014ʺ micro guidewire was used to cross the clot, such that it exited the distal end of the clot. For the tests involving the microcatheter or 3MAX, it also fully crossed the clot. In tests involving the Zipline, it was advanced over the microwire and into the clot such that the distal end of the catheter was in the clot but did not exit the distal end of the clot as shown in Figure 1.
The aspiration catheter was advanced to the face of the clot. The support catheter was removed, and the aspiration tubing was connected to the hub of the aspiration catheter. Aspiration was applied through the aspiration catheter until free flow was observed, or if free flow was not observed, aspiration was applied for 2 min after which the aspiration catheter was withdrawn under continuous aspiration.
Endpoints
Complete recanalization was defined as the complete removal of the clot from the cerebral vasculature in the absence of any large distal emboli (>1000 µm). Up to three passes were attempted to achieve recanalization. In this study First Pass Effect is defined as the complete retrieval of the clot in the first pass in the absence of any large distal emboli >1000 µm considered analogous to TICI 2c/3, Final Complete Revascularization mTICI 2c/3 (complete retrieval of the clot in the absence of any large distal emboli >1000 µm within three passes), and procedure-related distal emboli (>1000 µm, 500–1000 µm, and 200–500 µm).
Statistical analysis
All statistical analyses were performed using Microsoft Excel. One-way ANOVA was used to compare success and emboli rates across the groups. Tukey HSD post-hoc tests were used to assess pairwise comparisons. T-tests were used to assess statistical differences between treatment groups.
Results
Overall, avoiding clot entry or crossing was associated with higher FPE rates, improved final revascularization, and fewer distal emboli.
Revascularization performance—proximal versus entering clot
Catheter delivery without entering the clot resulted in better FPE rates than entering the clot for the Millipede88 + Zipline88 combination (67% vs. 56%), Figure 2. Remaining proximal also resulted in a better complete revascularization (100% vs. 78%). For the Millipede70 + Zipline70 combination the same FPE rate (33%) was observed whether the clot was entered or not. However, delivery without entering the clot resulted in better final complete revascularization rates than when the clot was entered with the Zipline70 (67% vs. 56%).
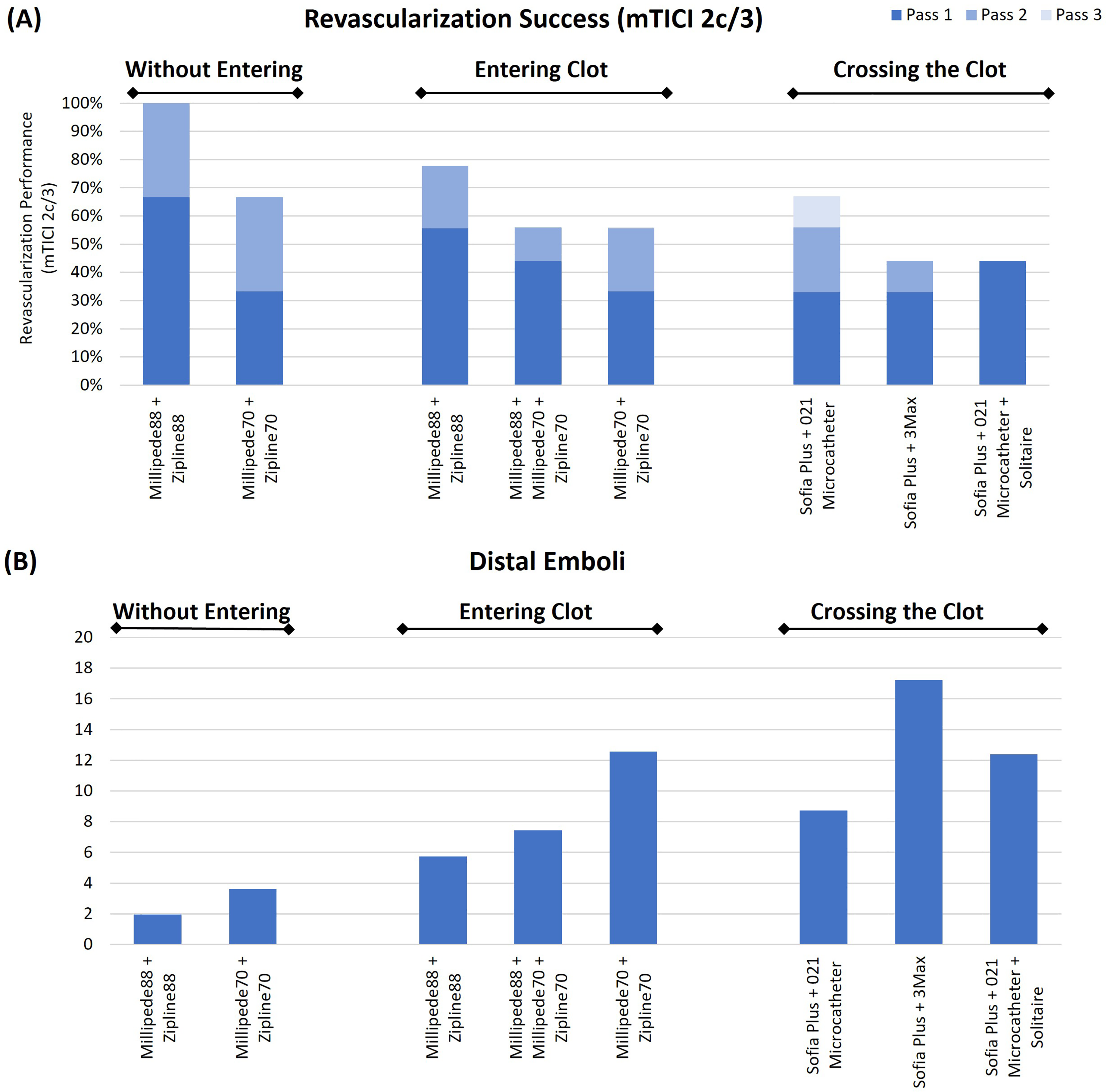
(a) Revascularization success (mTICI 2c/3) and (b) distal embolization rates for the various thrombectomy setups evaluated. The weighting applied to the distal emboli was as follows; ((5 × 1000 µm) + (2.5 × 500 µm) + (1 × 200 µm)).
Revascularization performance—zipline (entering) versus conventional techniques (crossing)—6Fr aspiration
Looking specifically at procedures involving 6Fr aspiration catheters, use of the novel Zipline delivery device resulted in similar outcomes to conventional delivery techniques. The FPE of the Millipede70 + Zipline70, SOFIA + 3MAX, and SOFIA Plus + 021 microcatheter were all 33%. SOFIA Plus + 021 Microcatheter + Stentriever had the highest FPE rate (44%). However, SOFIA + 021 microcatheter had a better final complete revascularization outcome (mTICI 2c/3) (67%) than the Millipede70 + Zipline70 (Entering), SOFIA + 3MAX, and SOFIA Plus + 021 Microcatheter + Stentriever (56%, 44%, and 44% respectively), Figure 2. Entering the clot with the Zipline70 resulted in less distal emboli than crossing with a 3MAX, similar distal emboli to the stentriever procedure, and more distal emboli than crossing with the 021 microcatheter (Figure 2).
Revascularization performance—impact
of 088 catheter
Superbore Millipede88 and Zipline88 combination resulted in the highest rates of FPE and final recanalization, regardless of whether the Zipline88 entered the clot or not (Figure 2). Use of Millipede88 for flow control in combination with Millipede70 + Zipline70 resulted in improved FPE rates when compared with Millipede70 + Zipline70 alone (44% vs. 33%), Figure 2.
Distal emboli rates
There is a statistically significant effect of delivery catheter position on weighted distal emboli across all clot types combined (p = .040*). Not entering the clot resulted in lower rates of distal emboli than entering the clot for both the Millipede88 + Zipline88 and Millipede70 + Zipline70 combinations, Figure 2.
The Millipede88 and Zipline88 combination resulted in the lowest rates of distal embolization compared to all other techniques. Use of Millipede88 for flow control in combination with the Millipede70 + Zipline70 resulted in lower distal emboli rates when compared with the Millipede70 + Zipline70 without Millipede88, Figure 2. The SOFIA + 021 microcatheter had lower rates of distal emboli than the SOFIA + 3MAX and SOFIA Plus + 021 Microcatheter + Stentriever (Figure 2 and Table 2).
Impact of thrombectomy strategy and clot composition on revascularization outcome and distal emboli.
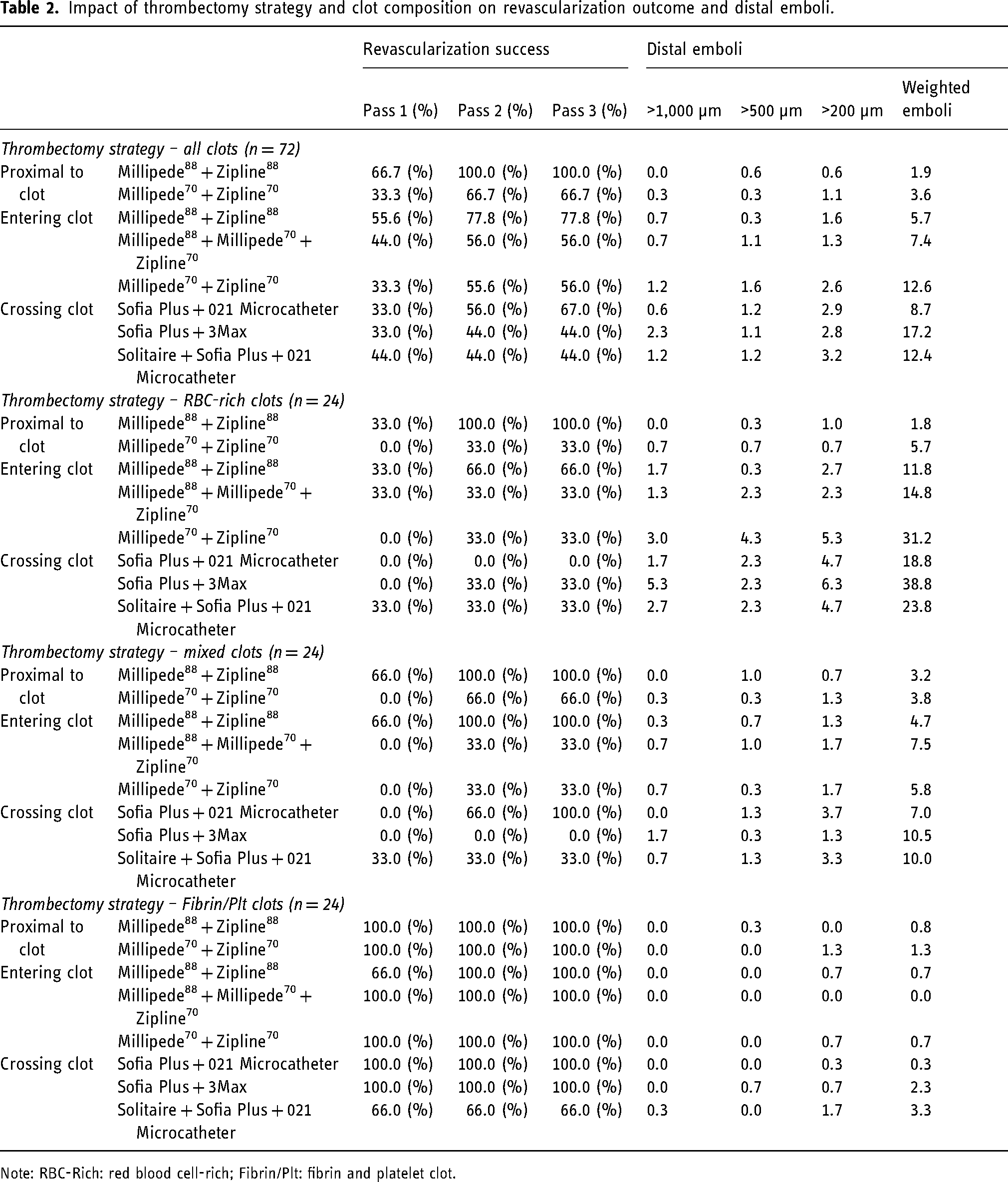
Note: RBC-Rich: red blood cell-rich; Fibrin/Plt: fibrin and platelet clot.
Impact of clot composition
The effect of crossing/entering a clot is statistically significant only within the soft RBC-Rich clots, crossing/entering soft RBC-Rich clots leads to a significantly larger number of emboli than staying proximal (p = .021*), as shown Table 3. Crossing/entering soft RBC-Rich clots also resulted in a concomitant decrease in final complete revascularization (mTICI 2c/3) outcome (22% vs. 67%), Figure 2. Crossing/entering Mixed clots lead to a non-significant increase (p = .082) in the number of emboli while also reducing the final revascularization (mTICI 2c/3) outcome relative to remaining proximal (83% vs. 44%), Figure 2. Crossing/entering stiff, Fibrin/Platelet-rich clots did not lead to an increase in emboli (p = .861) and was associated with a marginally smaller final revascularization (mTICI 2c/3) outcome (94% vs. 100%), Figure 2.
Impact of clot composition on revascularization outcome and distal emboli.
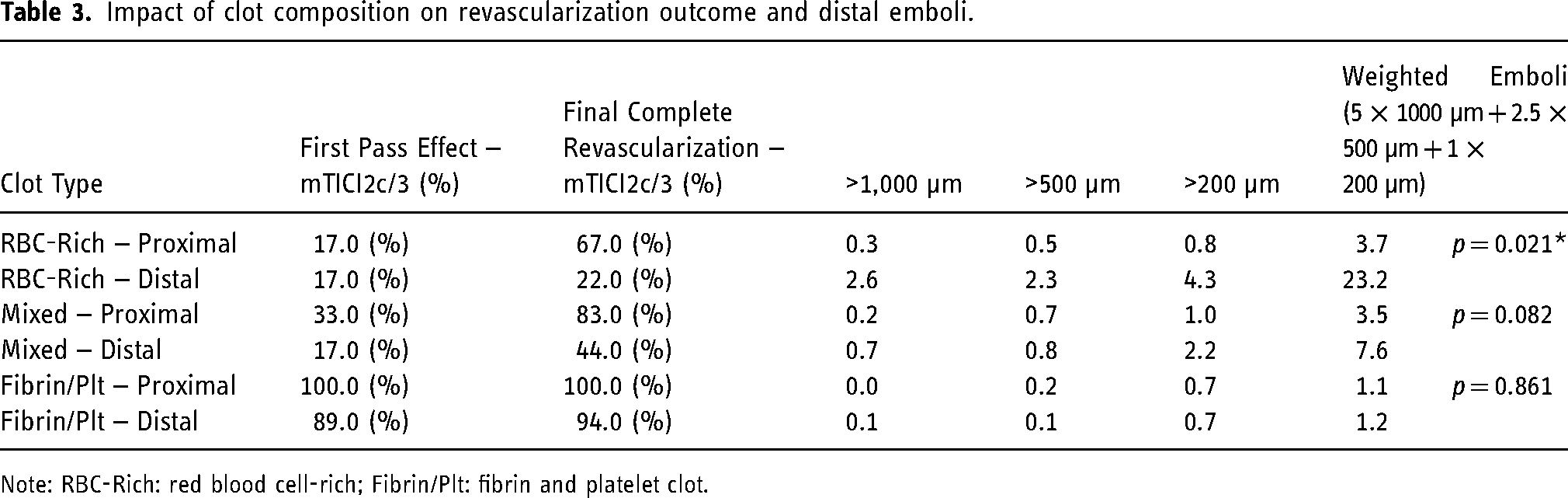
Note: RBC-Rich: red blood cell-rich; Fibrin/Plt: fibrin and platelet clot.
Discussion
The data presented in this study suggest that entering or crossing the clot with internal support devices is associated with poorer revascularization performance and higher rates of distal emboli than remaining proximal to the clot. The use of next-generation delivery assist catheters, such as Zipline, to facilitate navigation does not necessarily result in worse reperfusion or higher rates of distal embolization in aspiration thrombectomy. Revascularization performance and distal emboli are impacted by clot composition, with soft RBC-Rich clots being significantly more prone to embolization and poorer revascularization rates when entered/crossed.
In patients with challenging anatomy, the physician may be forced to cross the clot with their delivery catheters and guidewires in order to get the support required to deliver the aspiration catheter to the clot. However, physicians sometimes choose to cross the clot, even if they do not need to deliver a stentriever or require additional support. This study suggests that entering or crossing the clot with internal support devices is associated with poorer revascularization performance and higher rates of distal emboli than remaining proximal to the clot. Where possible, it is better to remain proximal to the clot in order to reduce the risk of fragmenting the occlusive clot. Fragmentation of the clot can lead to distal emboli and consequentially reduced rates of complete revascularization and poorer neurological outcomes. 17
If one does require the additional support achieved by crossing the clot, these data suggest that it is best to cross with a smaller 021 microcatheter than a larger internal support device such as a 3MAX, as larger internal support catheters lead to higher rates of distal emboli. 18 Entering the clot with the novel Zipline70 delivery catheter resulted in less distal emboli than crossing with a 3MAX, similar distal emboli to the stentriever procedure, and more distal emboli than crossing with the 021 microcatheter. This is likely due to the tapered tip design which has the potential to slide between the clot and vessel wall and appears to result in less fragmentation than the blunt edge effect of the 3MAX tip which is often used as a ‘large micro catheter’. The novel delivery catheters described in Table 1, tend to have a tapered design. Zipline (Perfuze), Carrier (Balt), Tenzing (Route92 Medical), Cheetah (Q’Apel), and Offset (Stryker) have a consistent proximal outer diameter with a gently tapering distal segment. The Wedge (Microvention) has a slightly distinctive design with a bulb and distal microcatheter segment. SENDit (Penumbra) would likely impact the clot more due to its blunter distal face.
It is noteworthy that despite having the largest profile of all the internal support devices, Zipline88 did not result in poorer reperfusion or higher rates of distal embolization when advanced into the clot. The effect of advancing the delivery catheter into the clot may be mitigated somewhat by using a super bore aspiration catheter for flow control and achieving flow reversal to prevent distal emboli, as shown previously. 16 Despite entering the clot with the larger Zipline88 delivery catheter, the Millipede88 achieved a higher FPE rate of 57% than all other thrombectomy setups evaluated. Interestingly, when the Millipede88 was used in combination with the Millipede70 and Zipline70, the FPE rate increased from 33% to 44% and the rate of distal emboli reduced relative to the Millipede70 and Zipline70 alone. The improvements in revascularization performance and flow control benefits of Superbore aspiration catheters have been established previously in vitro.16,19,20 Recent clinical studies support the in-vitro data, showing improved revascularization outcomes regardless of when the 088ʺ catheter is used for aspiration or flow control.4,19,21
Histological and mechanical characterization of clots from ischemic stroke procedures has shown that a diverse range of clot compositions exist,22–24 and that the mechanical and fracture properties of clots are influenced significantly by their cellular composition.25,26 Red-blood cell-rich clots, which are known to be friable and mechanically soft, are more prone to fragmentation when crossed, as shown in this study. Fibrin/Platelet-rich clots are mechanically stiffer, and despite typically being more difficult to cross, they do not tend to fragment as easily. Mixed clots, as their name suggests, are in the middle ground with a mixture of behaviors. 19 Physicians can gain an understanding of the clot composition using diagnostic imaging. 27 RBC-Rich clots typically have a Hyperdense Artery Sign (HAS) on non-contrast CT, 28 or a Susceptibility Vessel Sign (SVS+) on MRI while Fibrin/platelet-rich clots are iso-dense on CT and SVS- on MRI.29,30 This study suggests that when an RBC-Rich clot is identified, it should only be crossed with the internal catheters when necessary. The effect of entering or crossing stiff Fibrin/platelet-rich clots on distal emboli and revascularization performance is minimal.
This study has limitations. First, as mentioned several tapered tip delivery catheters of varying sizes are currently available (Table 1), yet only Zipline (Perfuze) was evaluated. Devices that have a similar purpose but different distal tip geometry such as Microvention Wedge and Penumbra SENDit were not evaluated. Secondly, only one anatomical model was used and therefore did not allow an evaluation of the impact of varying degrees of tortuosity and associated manipulation of the catheters and wires on clot disruption and fragmentation. In this work, the clots resided across the ICA-T and proximal M1. This clot burden represents a challenging scenario, resulting in lower reperfusion rates than previously reported for this test model. Other, less challenging scenarios such as standard M1 occlusions were not investigated. Thirdly, the impact of guidewire size (0.018ʺ vs. 0.014ʺ) was not investigated in this study, but the impact of the wire profile is likely to be minimal compared to the type of support catheter used. One could assume that larger neurovascular wires, such as the Aristotle 024ʺ or Colossus 035ʺ, will cause more micro emboli than an 0.014ʺ wire, particularly in Red-Blood Cell-Rich clots. Finally, the thrombectomy model is anatomically representative but not overtly challenging and all catheters were successfully able to make it to the target location in all tests. However, the aim of the study was to assess the impact of the delivery catheter position, not to assess the navigation and delivery success of delivery catheters.
Conclusions
To optimize reperfusion success in aspiration thrombectomy, operators should aim to position the internal support catheter proximal to the clot whenever possible. Next-generation delivery assist catheters with a tapered design, such as Zipline, offer improved delivery advantages without compromising reperfusion success or increasing the rates of distal emboli. Using larger internal support devices, such as 3MAX, leads to higher rates of emboli when crossing the clot. The composition of the occlusive clot significantly influences its response to being entered/crossed with internal support devices.
Supplemental Material
sj-docx-1-ine-10.1177_15910199251349672 - Supplemental material for Impact of next-generation delivery assist catheters and crossing strategy on reperfusion success in aspiration thrombectomy
Supplemental material, sj-docx-1-ine-10.1177_15910199251349672 for Impact of next-generation delivery assist catheters and crossing strategy on reperfusion success in aspiration thrombectomy by Raul G Nogueira, Seán Fitzgerald, Miquel Murillo Artigues, Akorede Rei Adedeji, Liam Mullins and John Thornton in Interventional Neuroradiology
Footnotes
Author contribution
RGN and JT contributed to the study design, analysis, and manuscript review. MMA and ARA performed the testing, data analysis and manuscript review. SF and LM were involved in the study design, data analysis and drafting the manuscript.
Data availability statement
Data are available from the authors upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Raul Gomes Nogueira reports consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Philips, Hybernia, Hyperfine, Imperative Care, Medtronic, Phenox, Philips, Prolong Pharmaceuticals, Stryker Neurovascular, Shanghai Wallaby, Synchron, and stock options for advisory roles with Astrocyte, Brainomix, Cerebrotech, Ceretrieve, Corindus Vascular Robotics, CrestecBio Inc., Euphrates Vascular, Inc., Vesalio, Viz-AI, RapidPulse and Perfuze. RGN is one of the Principal Investigators of the “Endovascular Therapy for Low NIHSS Ischemic Strokes (ENDOLOW)” trial. Funding for this project is provided by Cerenovus to Emory University. RGN is the Principal Investigator of the “Combined Thrombectomy for Distal MediUm Vessel Occlusion StroKe (DUSK)” trial. Funding for this project is provided by Stryker Neurovascular to University of Pittsburgh. RGN is an investor in Viz-AI, Perfuze, Cerebrotech, Reist/Q’Apel Medical, Truvic, Tulavi Therapeutics, Vastrax, Piraeus Medical, Brain4Care, Quantanosis AI, and Viseon. Seán Fitzgerald declares the following competing interest; Perfuze (stock options). Miquel Murillo Artigues declares the following competing interest; Perfuze (stock options). Akorede Rei Adedeji declares no competing interests. Liam Mullins declares the following competing interest; Perfuze (stock options). John Thornton declares the following competing interests; Perfuze (Physician Advisory Board, shareholder, stock options); Consultancy fees: Microvention.
Ethical approval and informed consent statements
Ethical approval was not required as the study did not involve human or animal subjects or tissue. Ovine blood for testing was obtained under license from Ellax Medical Ltd (Galway, Ireland).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
