Abstract
Background
The effectiveness of endovascular thrombectomy (EVT) for low Alberta Stroke Program Early CT score (ASPECTS) stroke patients with occlusion of the second segment of the middle cerebral artery (M2) is unclear.
Methods
This was a multicenter retrospective study. Patients with M1 or M2 occlusions and low ASPECTS (<6) who underwent successful EVT (modified treatment in cerebral ischemia score of 2b or higher) were included. Primary outcome was futile EVT reperfusion (defined as 90-day modified Rankin scale of 5 or 6). Other outcomes of interest include acceptable outcomes (modified Rankin scale of 3 or less) and intracranial hemorrhage (ICH), and all-cause 90-day mortality. Outcomes for M1 patients were compared to M2 patients with multivariable logistic regression models accounting for potential confounders.
Results
173 patients with M1 or M2 occlusions and low ASPECTS (<6) who underwent successful EVT were identified. After multivariable adjustments, M2 patients had significantly higher odds of futile reperfusion (OR 5.48 [95%CI 1.91 to 15.7], p = 0.002), lower odds of acceptable outcomes (OR 0.33 [95%CI 0.12 to 0.89], p = 0.028), and higher odds of all-cause mortality (OR 4.90 [95%CI 1.65 to 14.5], p = 0.004). These findings suggest that EVT's efficacy for low-ASPECTS stroke patients may be diminished for patients with M2 occlusions. M2 occlusion was not significantly associated with ICH.
Conclusions
Among low-ASEPCTS stroke patients who underwent successful EVT, those with M2 occlusions had significantly higher odds of poor outcome compared to those with M1 occlusions.
Introduction
Recent large randomized controlled trials have demonstrated that endovascular thrombectomy (EVT) is safe and effective compared to medical management for the treatment of acute ischemic stroke patients with low Alberta Stroke Program Early CT score (ASPECTS). 1 However, these trials overwhelmingly included patients with occlusions of the internal carotid artery or the first segment of the middle cerebral artery (M1).2–7 Whether EVT is effective for the treatment of low-ASPECTS stroke patients with occlusion of the second segment of the middle cerebral artery (M2) is unclear. 8 ASPECTS was designed to quantify the degree of early ischemic changes in the MCA territory; thus, it is possible that for M2 patients with low ASPECTS, more of the affected territory may be infarcted, and EVT may yield less benefit. Furthermore, compared to M1, EVT for M2 occlusions may be associated with higher rates of hemorrhagic complications, especially for patients who undergo EVT in which a stent-retriever was used. 9
In this study, we analyzed a large multinational registry to assess whether there were differences in the safety and efficacy of successful EVT for patients with low ASPECTS and M1 or M2 occlusion.
Methods
This was a retrospective cohort study of the Stroke Thrombectomy and Aneurysm Registry (STAR) from January 2013 to December 2023. 10 The study was approved by the institutional review board at each participating institution. The data at each institution were obtained retrospectively and collected according to a standardized protocol. Patient consent was waived due to the retrospective nature of the study.
Patient population
Adult stroke patients (18 years or older) with ASPECTS < 6 who underwent successful EVT (modified treatment in cerebral ischemia [mTICI 11 ] score ≥ 2b) for isolated M1 or M2 occlusion were included. The following patients were excluded: 1) National Institutes of Health stroke scale (NIHSS) < 6 or missing, 2) pre-stroke modified Rankin scale (mRS 12 ) > 2 or missing, 3) tandem occlusion of the extracranial carotid artery.
Primary and secondary outcomes
The primary outcome for this study was futile reperfusion, defined as a 90-day mRS of 5 or 6 despite successful EVT recanalization (mTICI 2b or better). Other outcomes of interest include 90-day mRS (ordinal analysis), all-cause 90-day mortality, acceptable outcomes (90-day mRS of 3 or better), ICH, and sICH.
Other clinical variables
Demographic data, medical comorbidities, and pre-stroke mRS were recorded for each patient. Clinical characteristics such as admission National Institutes of Health stroke scale (NIHSS), administration of intravenous thrombolysis, additional sites of vascular occlusion (posterior cerebral artery or vertebral artery), and symptom onset or last-known-well time to arteriotomy were also captured.
Statistical analysis
Descriptive statistics were presented as median (interquartile range) for continuous variables or percentage for categorical variables. The time from symptom onset or last known normal to arteriotomy variable was dichotomized into early treatment window (< 6 h) or extended treatment window (≥ 6 h or unknown). 90-day mRS values of 0 and 1 were binned into one category. For unadjusted comparisons of baseline patient characteristics, continuous and ordinal variables were compared between groups using Mann-Whitney U test, and categorical variables were compared using chi-squared analyses. For unadjusted comparisons of study outcomes, binary or ordinal regression analyses were used to calculate odds ratios (ORs) and common odds ratios (cORs), respectively. For adjusted comparisons, binary or ordinal regression analyses were conducted with additional adjustments such as for age, NIHSS, treatment time window, pre-stroke mRS, intravenous thrombolysis, ICH type (none, asymptomatic, or sICH), frontline EVT technique (aspiration, stent-retriever, or combined), and ASPECTS, based on previous reports of significant predictors for post-EVT outcomes.13–18 P-values less than 0.05 was deemed statistically significant. Statistical analyses were conducted using SPSS v29.0.
Results
Patient characteristics
A total of 173 patients met inclusion criteria: 138 with M1 occlusion, and 35 with M2 occlusion. Patient characteristics are outlined in Table 1. Overall, M2 patients were more likely to be male (62.9% vs. 43.5%, p = 0.040), more likely to have received intravenous thrombolysis (54.3% vs. 29.7%, p = 0.006), and more likely to have undergone EVT with contact aspiration (80.6% vs. 57.1%, p = 0.015).
Patient characteristics.
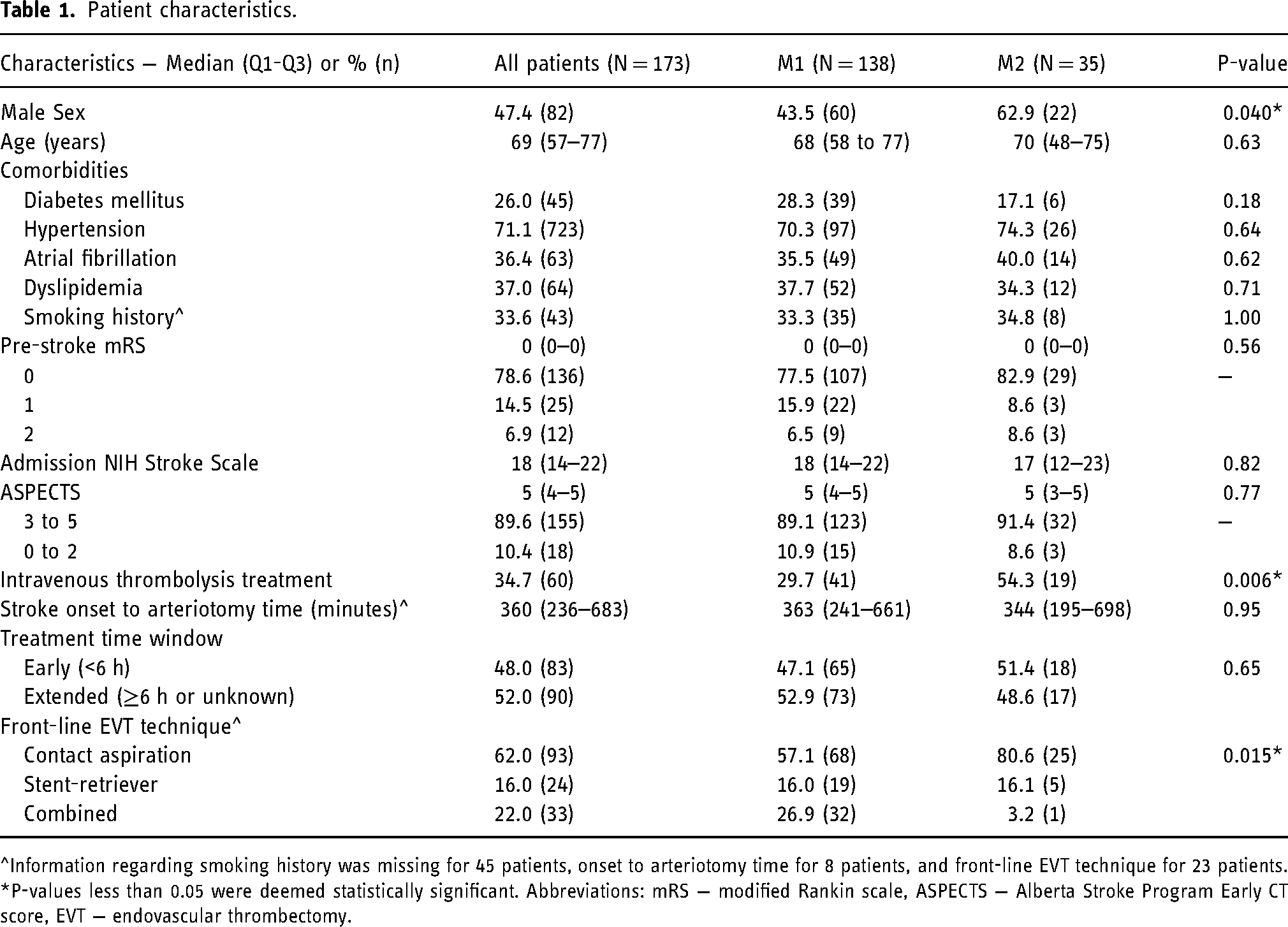
^Information regarding smoking history was missing for 45 patients, onset to arteriotomy time for 8 patients, and front-line EVT technique for 23 patients. *P-values less than 0.05 were deemed statistically significant. Abbreviations: mRS − modified Rankin scale, ASPECTS − Alberta Stroke Program Early CT score, EVT − endovascular thrombectomy.
Study outcomes
Distribution of 90-day mRS outcomes of M1 and M2 patients is presented in Figure 1. In unadjusted comparisons, M2 patients had significantly higher odds of futile reperfusion (50.0% vs. 29.9%, OR 2.34 [95%CI 1.09 to 5.03], p = 0.029; Table 2) and all-cause 90-day mortality (38.2% vs. 19.7%, 2.53 [95%CI 1.12 to 5.67], p = 0.025; Table 2). After multivariable adjustments, M2 patients had significantly higher odds of futile reperfusion (aOR 5.48 [95%CI 1.91 to 15.7], p = 0.002; Table 2), higher odds of all-cause mortality (aOR 4.90 [95%CI 1.65 to 14.5], p = 0.004), lower odds of acceptable outcomes (aOR 0.33 [95%CI 0.12 to 0.89], p = 0.028; Table 2), and overall worse 90-day mRS outcomes (cOR 3.05 [95%CI 1.33 to 7.01], p = 0.009; Table 2). M2 occlusion was not significantly associated with ICH in unadjusted and multivariable analyses (Table 2).
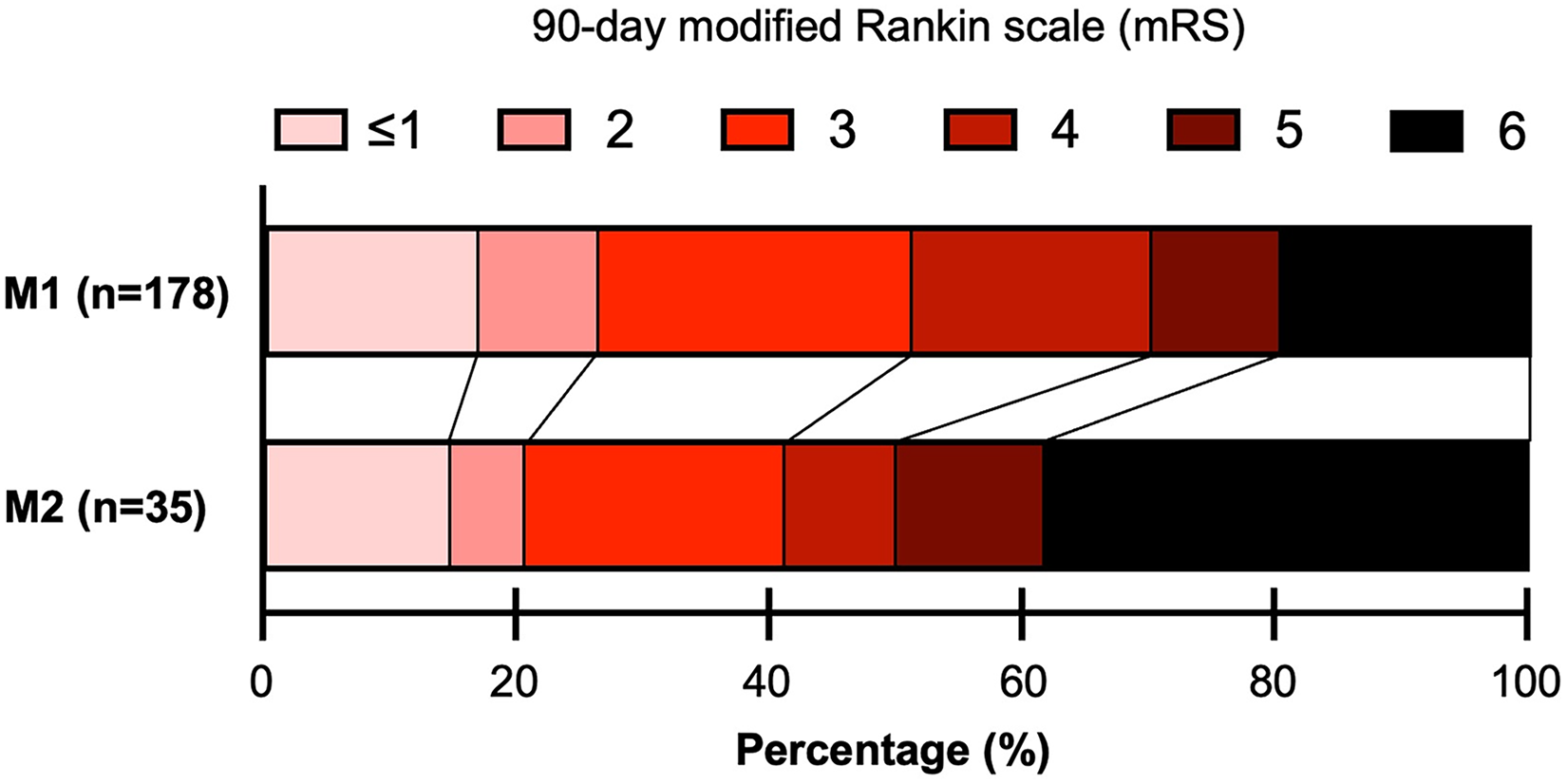
90-day outcomes of low-ASPECTS stroke patients who underwent successful endovascular revascularization of M1 vs. M2 occlusions.
Treatment outcomes following successful M1 vs. M2 thrombectomy for low ASPECTS patients.
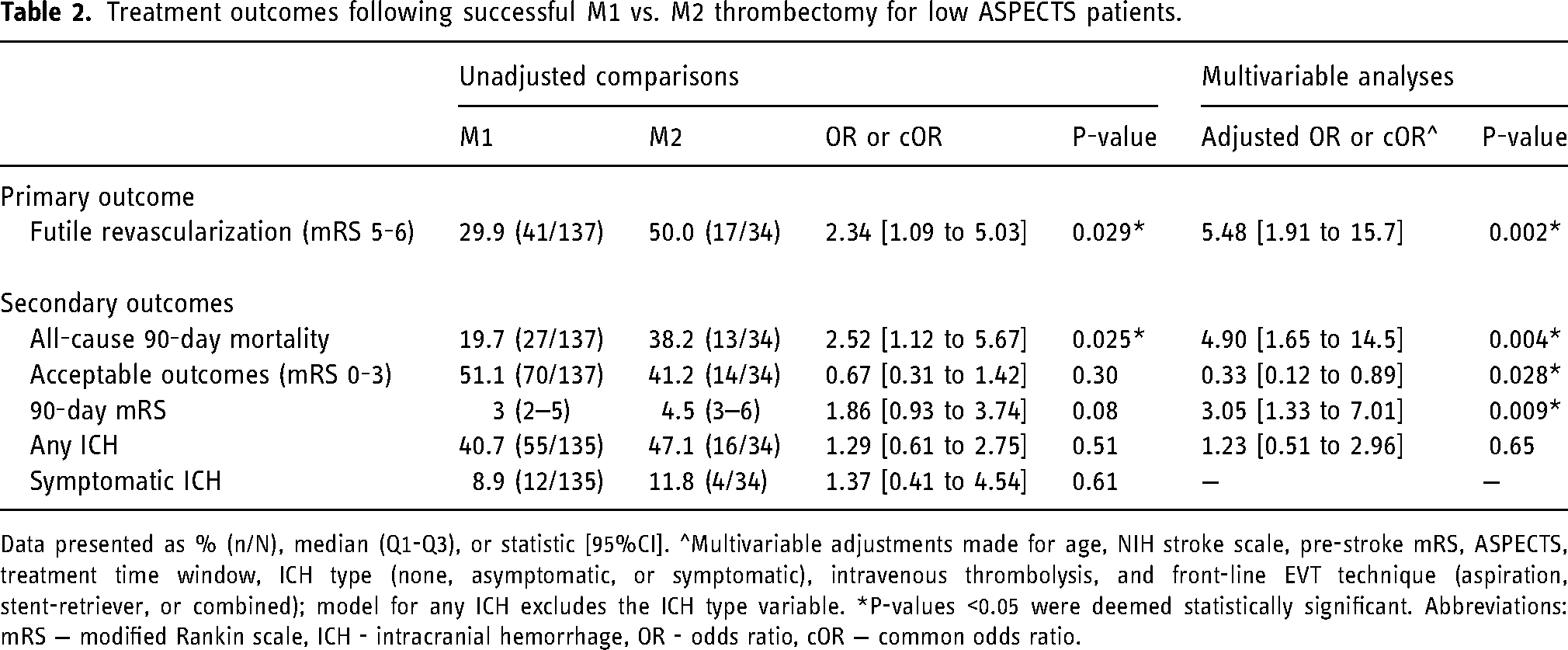
Data presented as % (n/N), median (Q1-Q3), or statistic [95%CI]. ^Multivariable adjustments made for age, NIH stroke scale, pre-stroke mRS, ASPECTS, treatment time window, ICH type (none, asymptomatic, or symptomatic), intravenous thrombolysis, and front-line EVT technique (aspiration, stent-retriever, or combined); model for any ICH excludes the ICH type variable. *P-values <0.05 were deemed statistically significant. Abbreviations: mRS − modified Rankin scale, ICH - intracranial hemorrhage, OR - odds ratio, cOR − common odds ratio.
Given the large discrepancy in intravenous thrombolysis treatment between M1 and M2 patients, we performed additional subgroup and interaction analyses. Here, among those treated with intravenous thrombolysis, 9 of 16 (56.3%) M2 patients experienced futile reperfusion versus 29 of 97 (29.9%) M1 patients (OR 3.02 [95%CI 1.02 to 9.29], p = 0.049). For those not treated with intravenous thrombolysis, 8 of 19 (42.1%) M2 patients experienced futile reperfusion versus 12 of 41 (29.3%) of M1 patients (OR 1.76 [95%CI 0.56 to 5.49], p = 0.33). These effect sizes were not statistically significantly different (interaction p = 0.50). Based on these results, there was no heterogeneity in effect sizes between IVT and non-IVT treated patients.
Discussion
In this international multicenter retrospective cohort study of low-ASPECTS patients who underwent successful EVT, patients with M2 occlusions had higher odds of worse outcomes compared to patients with M1 occlusions.
There are several possible explanations for these study findings. First, ASPECTS is a quantitative system that assesses the dispersion of early ischemic changes throughout the MCA territory. 1 While patients with a proximal M1 occlusion could eventually present with an ASPECTS of zero, the worst ASPECTS is limited for patients with M2 occlusions as only part of the total MCA vascular territory is affected. Thus, it is possible that M2 patients with low ASPECTS may have had further disease progression compared to M1 patients with the same ASPECTS, which could explain the higher odds of EVT futility among M2 patients. Given that M2 occlusions may have a higher “minimum” ASPECTS, it is also possible that a low ASPECTS in this population may be associated with poorer collateral circulation compared to similar ASPECTS in patients with M1 occlusions, influencing outcomes. 19 M2 patients with low ASPECTS may also have a higher burden of cortical infarcts compared to M1 patients with the same numerical ASPECTS, which could be associated with deficits in language and cognition 20 that have an outsized impact on 90-day mRS. It is also possible that low-ASPECTS patients with M2 occlusions may have initially had more proximal occlusions that lysed spontaneously or with intravenous thrombolysis 21 ; in this scenario, early ischemic changes despite recanalization of proximal occlusion may indicate advanced infarction, thus rendering further recanalization with EVT less effective and potentially futile. Finally, recent medium vessel occlusion EVT trials (ESCAPE-MeVO 22 and DISTAL 23 ) have suggested that EVT may have limited clinical benefit over medical management; trial data also showed that EVT may be associated with higher rates of symptomatic hemorrhage, which have also been corroborated by prior retrospective studies.24–28 Thus, it is possible that for low-ASPECTS patients who may have more limited penumbra and salvageable tissue, the procedural risks associated with EVT for M2 occlusion strokes may have a larger marginal clinical impact, culminating in overall worse clinical outcomes compared to EVT for low-ASPECTS M1 strokes.
Importantly in our study, the lower bound of the 95% CI of the M2 group's negative association with worse 90-day outcomes (adjusted cOR 1.33) was numerically similar to the pooled effect size of EVT's benefit over medical management in previous randomized trials of EVT in low-ASPECTS patients (generalized OR 1.54). 2 Thus, our study suggests that providers should be cautious when extrapolating such positive trial results of EVT for low ASPECTS to patients with M2 occlusions.
Our study has several limitations. First, as a retrospective study, our results are vulnerable to selection bias and hidden confounders. Second, ASPECTS was reported by each institution, and was not adjudicated at a core lab. Third, we did not have a medical management arm and thus we cannot draw conclusions regarding the relative effectiveness of EVT versus medical management for low-ASPECTS M2 patients. Fourth, other factors, such as collateral status and infarct volume derived from perfusion imaging, 29 were not accounted for in our study and could have confounded our results. Finally, our sample size was modest; larger prospective studies are needed to confirm our study findings.
Conclusion
Among low-ASEPCTS stroke patients who underwent successful EVT, those with M2 occlusions had significantly higher odds of poor outcome compared to those with M1 occlusions. These results suggest that EVT may have a more restricted therapeutic benefit for M2 occlusions compared to M1 occlusions among low-ASPECTS.
Footnotes
Acknowledgments
None.
Author contributions
HChen, MC, PK conceived the study idea. MC, HM, CC, IM, PJ, JK, SQW, AR, RMS, MP, AS, NG, SY, H.Cuellar, BH, A.Alawieh, A.Alaraj, ME, DGR, OT, JM, IF, AP, FS, JO, RC, RG, CM, MSP, MRL, WB, MM, ED, RW, PN, SC, DJA, AMS, and PK collected the data. H.Chen and HM analyzed the data. HChen, MC, and PK wrote the manuscript. CM, RG, A.Alawieh, MRL, and AMS revised the manuscript.
Data availability statement
Data available on request due to privacy/ethical restrictions
Declaration of conflicting interests
The STAR registry receives research support from Penumbra, Microvention, Medtronic, Stryker, RapidAI, Brain Aneurysm Foundation. HChen: None. MC: None. HM: None. CC: None. AA: None. DA: Consultant for MicroVention, Stryker, Q'apel, Synchron, and Cerenovus, Investor Von Vascular, Research Support The Bee Foundation. WB: Holds equity in Nested Knowledge, Superior Medical Editors, Piraeus Medical, Sonoris Medical, and MIVI Neurovascular. He receives royalties from Medtronic and Balloon Guide Catheter Technology. He receives consulting fees from Medtronic, Stryker, Imperative Care, Microvention, MIVI Neurovascular, Cerenovus, Asahi, and Balt. He serves in a leadership or fiduciary role for MIVI Neurovascular, Marblehead Medical LLC, Interventional Neuroradiology (Editor in Chief), Piraeus Medical, and WFITN. SC: Consultant and proctor for Medtronic and Microvention. RC: none. HCuellar: Consultant for Medtronic and Microvention. ED: None. RDL: PI for Imperative Trial; Research grants from Siemens Healthineers and Kaneka Medical. Consultant for Cerenovus, Stryker Neurovascular and Sim & Cure. Minor equity interest Vastrax, Borvo medical, Synchron, Endostream, Von Vascular, Radical catheters and Precision Recovery Inc. ME: Consultant for Viz.ai and Imperative Care. Investments in Galaxy Therapeutics. IF: None. NG: None. RG: Consultant for Balt Neurovascular, Cerenovus, Medtronic Neurovascular, Rapid Medical, and Stryker Neurovascular. BH: None. PJ: None. PK: Grants from the NIH (1U18EB029353-01) and unrestricted educational grants from Medtronic and Siemens. Consultant for Imperative Care and Stryker Neurovascular. Stock ownership in Vena Medical. JTK: None. MRL: Unrestricted educational grants from Medtronic and Stryker; consulting agreement with Medtronic, Aeaean Advisers and Metis Innovative; equity interest in Proprio, Stroke Diagnostics, Apertur, Stereotaxis, Fluid Biomed, and Hyperion Surgical; editorial board of Journal of NeuroInterventional Surgery; Data safety monitoring board of Arsenal Medical. IM: speakers honoraria from Pfizer and Bristol-Myers Squibb. JM: None. CM: Consultant for Stryker, Medtronic, Microvention, Penumbra, and Silk Road Medical. Speaker for Penumbra and Silk Road Medical. Contact PI for NIH Grant R21NS128641. MM: None. PN: Consultant for Penumbra, Medtronic, Stryker, Cerenovus and Balt. JO: None. MSP: Consultant for Medtronic. AP: None: MNP: Grants from the Swiss National Science Foundation (SNF) for the DISTAL trial (33IC30_198783) and TECNO trial (32003B_204977), Grant from Bangerter-Rhyner Stiftung for the DISTAL trial. Unrestricted Grants for the DISTAL trial from Stryker Neurovascular Inc., Phenox GmbH, Penumbra Inc. and Rapid Medical Inc., Sponsor-PI SPINNERS trial (Funded by a Siemens Healthineers AG Grant), Research agreement with Siemens Healthineers AG, Local PI for the ASSIST, EXCELLENT, TENSION, COATING, SURF and ESCAPE-NEXT trials. Speaker fees: Stryker Neurovascular Inc., Medtronic Inc., Penumbra Inc., Acandis GmbH, Phenox GmbH, Siemens Healthineers AG. AR: None. DR: Consultant for Penumbra, Balt, Microvention, Phenox. AShaban: None. ASpiotta: Consultant for Penumbra, Terumo, RapidAI, Cerenovus. RS: RMS research is supported by the NREF, Joe Niekro Foundation, Brain Aneurysm Foundation, Bee Foundation, Department of Health Biomedical Research Grant (21K02AWD-007000) and by National Institute of Health (R01NS111119-01A1) and (UL1TR002736, KL2TR002737) through the Miami Clinical and Translational Science Institute, from the National Center for Advancing Translational Sciences and the National Institute on Minority Health and Health Disparities. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. RMS has an unrestricted research grant from Medtronic and Balt and has consulting and teaching agreements with Penumbra, Abbott, Medtronic, Balt, InNeuroCo, Cerenovus, Naglreiter, Tonbridge, Von Medical, and Optimize Vascular. OT: Consulting Agreements: Viz.AI, Inc., Penumbra, Inc, Balt, Inc, Stryker Inc, Imperative Inc. Proctor: Microvention Inc, Medtronic Inc. Educational/Research Grants: Q’apel Inc, Steinberg Foundation. RW: Consultant for Medtronic, Stryker, and Synaptive Medical. SQW: None. SY: lecture fee from Stryker, Medtronic, Johnson & Johnson, Kaneka Medics. AE: None. AA: Consultant for Cerenovus
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stryker, Medtronic, RapidAI, Microvention, Brain Aneurysm Foundation, Penumbra.
