Abstract
Introduction
Flow diverting stents (FDS) are routinely used to reconstruct the arteries of the head and neck. When placed into the mobile cervical internal carotid artery (cICA) segment, the FDS runs the risk of post-procedure stent migration and proximal intimal hyperplasia reaction from physiologic movement of the neck. We report our experience using a novel proximal anchoring technique during endovascular flow reconstruction of complex petrocervical dissections to prevent this potentially deleterious result.
Methods
We reviewed a prospectively maintained IRB-approved institutional database of the senior authors to identify cases of FDS treatment in the mobile petrocervical segments which had the proximal FDS “anchored” with a nitinol stent.
Results
The proximal anchoring technique was successfully performed in the mobile cervical segment in a total of 31 cases over the study period. Each case involved a complex ICA dissection with 68% (n = 21) having an accompanying pseudoaneurysm. Fifty-two percent (n = 16) were female. Surpass Streamline and Evolve FDS were utilized in all cases. An average of 2.2 ± 0.1 FDS devices were utilized (range 2-4 FDS), with each case utilizing a laser-cut nitinol carotid stent as the proximal anchor. The average stent diameter was 5.64 ± 0.2 mm (range 4-8 mm) and length of 30.1 ± 1.5 mm (range 20-60 mm). On last follow-up angiography, there were no instances of stent migration or proximal neointimal hyperplasia.
Conclusion
Utilization of the proximal anchoring technique on FDS constructs in the mobile cICA may provide additional protection from post-procedure stent migration and intimal reaction attributed to patient neck movement resulting in augmentation of successful healing.
Introduction
Extracranial cervical dissections and pseudoaneurysms pose unique treatment challenges and are often managed first-line with antiplatelet or anticoagulant therapies. 1 For carotid dissections that are symptomatic, enlarging, have flow limitation, and pose significant risks of thromboembolism, or in patients that cannot withstand long-term anticoagulation, flow diversion treatment has been cited in recent literature as a viable treatment option. 2 While flow diverting stents (FDS) are routinely used to safely reconstruct intracranial arteries, there is limited literature investigating reconstruction in both the extracranial carotid and vertebral segments. 3
Flow-diverting stent migration and neointimal hyperplasia are known consequences of endovascular flow diversion procedure.4,5 When flow diverters are placed into the mobile cervical internal carotid artery segment, there is an enhanced possibility of stent migration and proximal intimal hyperplasia from physiologic movement of the neck post-procedure.
6
Stent migration has been well documented after intracranial implantation; the risk is higher in the cervical ICA due to its larger lumen and flow velocity compared to intracranial vessels.
6
To mitigate this potential complication, Amuluru et al. described two cases with anchoring strategies by using a carotid stent in the proximal segment of FDS and using an FDS larger in diameter than the parent vessel.
6
Proximal neointimal hyperplasia is particularly significant along with the ends of the nitinol stent where the edges of the braided FDS have been cut and the bare metal edge interfaces with the intima. Adjunctive proximal anchoring with a nitinol stent may serve to mitigate the feared complication of intimal hyperplasia. We report a novel technique of using endovascular flow diversion reconstruction for long dissecting lesions spanning into mobile cervical ICA with successful proximal anchoring with carotid stent in a 31-patient case series
Methods
Patient inclusion
Retrospective review of a prospectively maintained IRB-approved database of the senior authors revealed 31 consecutive cases of endovascular flow diversion reconstruction with proximal anchoring for ICA dissections. Cases were collected from July 2019 to September 2023. All individual identifying information, including age and sex, have been meticulously anonymized to achieve appropriate anonymity in the manuscript. As all the data gathered was de-identified and stored following IRB protocols, a waiver of informed consent was obtained. Patients identified with pseudoaneurysms or dissections, not in the mobile cervical ICA segment, defined as the petrocervical ICA, were excluded.
Procedural details
The choice to employ endovascular reconstruction with proximal anchoring as the treatment for ICA dissections with or without an accompanying pseudoaneurysm was determined by the surgeon's preference and the established practices of the institution. Embolizations were performed under general anesthesia with the institution of at least 2000U of systemic heparin. The procedure utilized 6-, 7-, or 8-French femoral access. In all cases, a triaxial platform was used. The guide sheaths utilized for access are as follows: Walrus (Q’Apel Medical), Ballast (Balt USA), Infinity (Stryker Neurovascular), Armadillo (Q’Apel Medical), TracStar (Imperative Care), and RIST (Medtronic Neurovascular). The distal access catheters (DACs) utilized include Cat 5 (Stryker Neurovascular), AXS Offset (Stryker Neurovascular), Vecta 071 (Stryker Neurovascular), and Navien (Medtronic Neurovascular). The microcatheters used include XT-27 (Stryker Neurovascular), SL-10 (Stryker Neurovascular), and Prowler (Cerenovus). The following stents were used: Surpass Evolve (Stryker Neurovascular), Surpass Streamline (Stryker Neurovascular), Neuroform Atlas (Stryker Neurovascular), Resolute Onyx (Medtronic Neurovascular), Precise (Cordis), Protégé (Medtronic Neurovascular), and Enterprise (Cerenovus).
Endovascular procedure
All patients were initiated on preoperative dual antiplatelet therapy consisting of aspirin 325 or 81 mg daily, ticagrelor twice daily, or clopidogrel daily. 7 VerifyNow was used for platelet function testing on the day of the procedure per routine. An 8F guide catheter was advanced over a Select catheter in the aortic arch and used to select the proximal ICA. Catheter angiography was performed, which revealed and characterized the petrocervical dissection. A coaxial system consisting of a DAC, such as Vectra 071″ and an Offset catheter, were used to gain access across the petrocervical dissection into the intracranial petrous segment. The Offset was removed, and the first FDS was deployed from the horizontal petrous back into the mid-cervical and access was reestablished. A second FDS was typically telescoped within that to create a construct reaching back to the level of the carotid bulb. Access was reestablished with a DAC, and the flow-diverting system was removed. The distal end of the construct was tacked down with a stent, such as an Atlas 4.5 × 21. Attention was then turned to the proximal anchoring technique. Typically, a laser-cut nitinol carotid stent was used to tack the proximal end of the flow-diverting construct back into the common carotid artery. Final angiography was then performed. Technical success for the procedure was determined by the successful implementation of flow diversion implantation construct with a proximal anchor stent as described above.
Data collection
Demographic and clinical data, encompassing age, sex, comorbidities, and utilization of antiplatelet, anticoagulant, or aspirin medications, were collected from the medical records. The pseudoaneurysm location was also collected. Procedural details, including the type, size, and quantity of guide catheter, distal intracranial catheter, microcatheter, FDS, and stent were collected. The Plavix VerifyNow Platelet Reactivity Unit (PRU) test results (P2Y12 reaction units), total fluoroscopy time (min), total procedural contrast (cc/mL), and radiation dose (mGy) were also recorded. Procedural outcomes, including complications, pseudoaneurysm grading as defined by the Modified Raymond-Roy Classification (MRRC) system, 8 and the incidence of stent migration or proximal in-stent intimal reaction, were assessed at the most-recent follow-up angiography. The amount of proximal in-stent stenosis was categorized into 1 of 5 categories: none, 1% to 25%, 25% to 50%, 50% to 75%, and 75% to 100%.
Results
Over the study period from December 2019 to September 2023, 31 endovascular flow diversion with proximal anchoring cases were identified with an average age of 58.1 years ± 3.43 (range 22-93 years) (Table 1). Sxiteen patients (52%) were female. Case presentations were trauma (12, 39%), headache (10, 32%), weakness (6, 19%), and other (3, 10%). Among these patients, 29 (93.5%) had pseudoaneurysms with a mean size of 9.38 ± 0.71 mm. All (100%) pseudoaneurysms were in the petrocervical region of the ICA. Average PRU was 81.3 ± 13.9.
Case Summary.
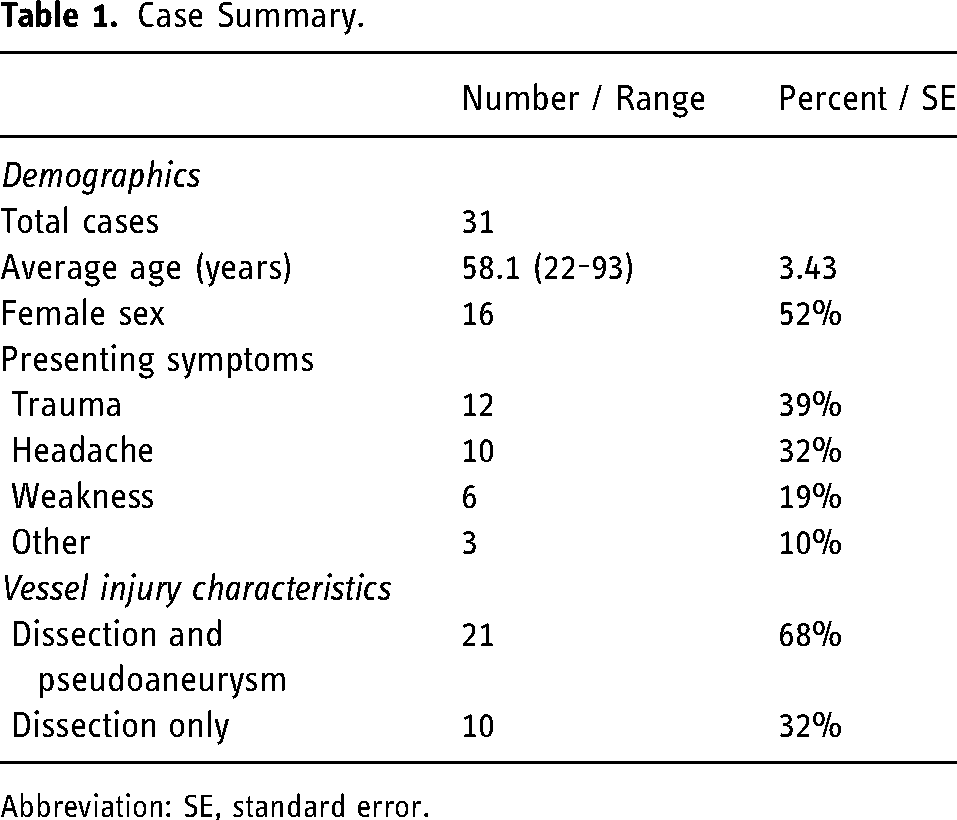
Abbreviation: SE, standard error.
The average fluoroscopy time was 41.4 ± 4.6 min, contrast was 41.6 ± 5.4 mL, and radiation dose was 915 ± 124 mGy (Table 2). Patients were placed on dual antiplatelet therapy with a combination of aspirin 81 mg or 325 mg (29, 93.5%) and ticagrelor 60 mg or 90 mg (11, 35.5%), or clopidogrel 75 mg (6, 19%). 7 On average, 2.28 flow diverters per patient were implanted over the 31 cases. Implanted Surpass Evolve and Streamline flow diverters had an average width of 4.8 ± 0.18 and 4.75 ± 0.06, respectively. Average width of implanted nitinol stents was as follows: Protégé (8.2 ± 0.44, n = 5), Precise (7.14 ± 0.18, n = 23), Onyx (4.77 ± 0.10, n = 11), Atlas (4.5, n = 22), Enterprise (4.07 ± 0.07, n = 9), and Synergy (4, n = 1).
Procedural Details.
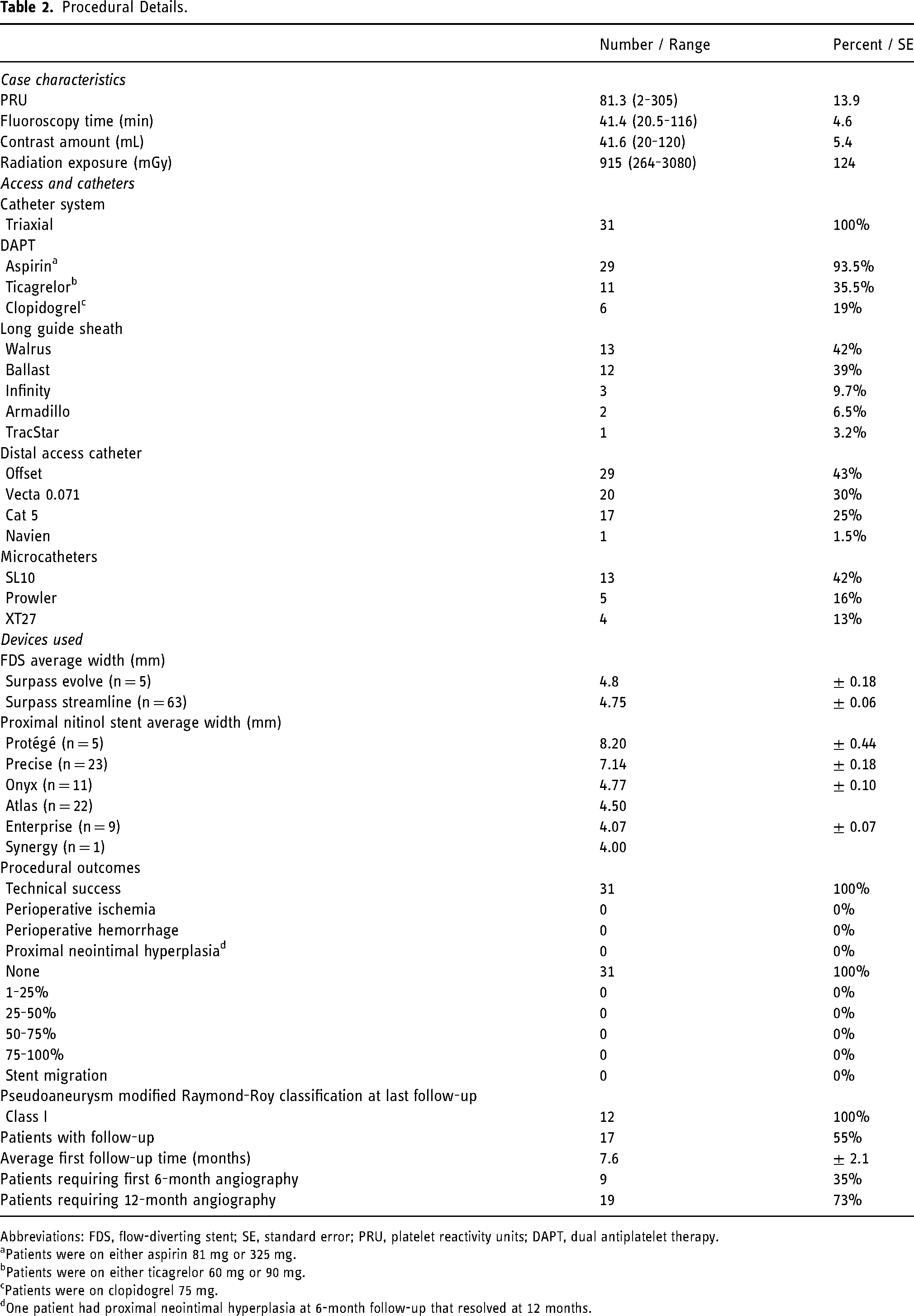
Abbreviations: FDS, flow-diverting stent; SE, standard error; PRU, platelet reactivity units; DAPT, dual antiplatelet therapy.
Patients were on either aspirin 81 mg or 325 mg.
Patients were on either ticagrelor 60 mg or 90 mg.
Patients were on clopidogrel 75 mg.
One patient had proximal neointimal hyperplasia at 6-month follow-up that resolved at 12 months.
Technical success was achieved in all (31 cases, 100%) patients. There were no reported cases of perioperative ischemia or hemorrhage. Follow-up imaging was available in 20 (67%) patients at a mean of 12.3 ± 1.52 months post-procedure. Of these 20 patients, 19 (95%) achieved Raymond grade 1 occlusion while 1 patient had decreased aneurysm size to Raymond grade 2 occlusion. No patients (0, 0%) had stent migration or proximal neointimal hyperplasia on their most recent angiography. One patient (1, 5%) had proximal neointimal hyperplasia at 6 month follow up, which resolved at the 12 month follow up.
Discussion
In this report, we describe the largest known series of petrocervical dissection endovascular flow diversion treatment and bring forth a novel proximal anchoring technique. Sun et al. reported the use of telescoping flow diverters for a giant petrous and cavernous ICA aneurysm. 9 Moreover, Kühn et al. describe in their report on ICA reconstruction one patient who received telescoping flow diverters and 3 patients who received a combination of FDS and carotid stent. 10 However, there were no cases of endovascular flow diversion with a proximal anchoring carotid stent. Our case series is the first and largest that describes patients who received flow diversion with a proximal anchoring carotid stent technique. All 31 cases involved a complex ICA dissection with 68% having an accompanying pseudoaneurysm. An average of 2.2 FDS was deployed per case with one proximal anchoring stent in each case ranging in 4 to 8 mm in diameter and 20 to 60 mm in length. Of the 17 patients with angiographic follow-up of 6 months or more, there were no instances of stent migration or significant or flow-limiting proximal neointimal hyperplasia. Complete occlusion of the pseudoaneurysm was achieved in 100% of cases as defined by MRRC Class I (Figure 1). 8 This innovative technique exhibited a favorable safety and efficacy profile across the cohort.
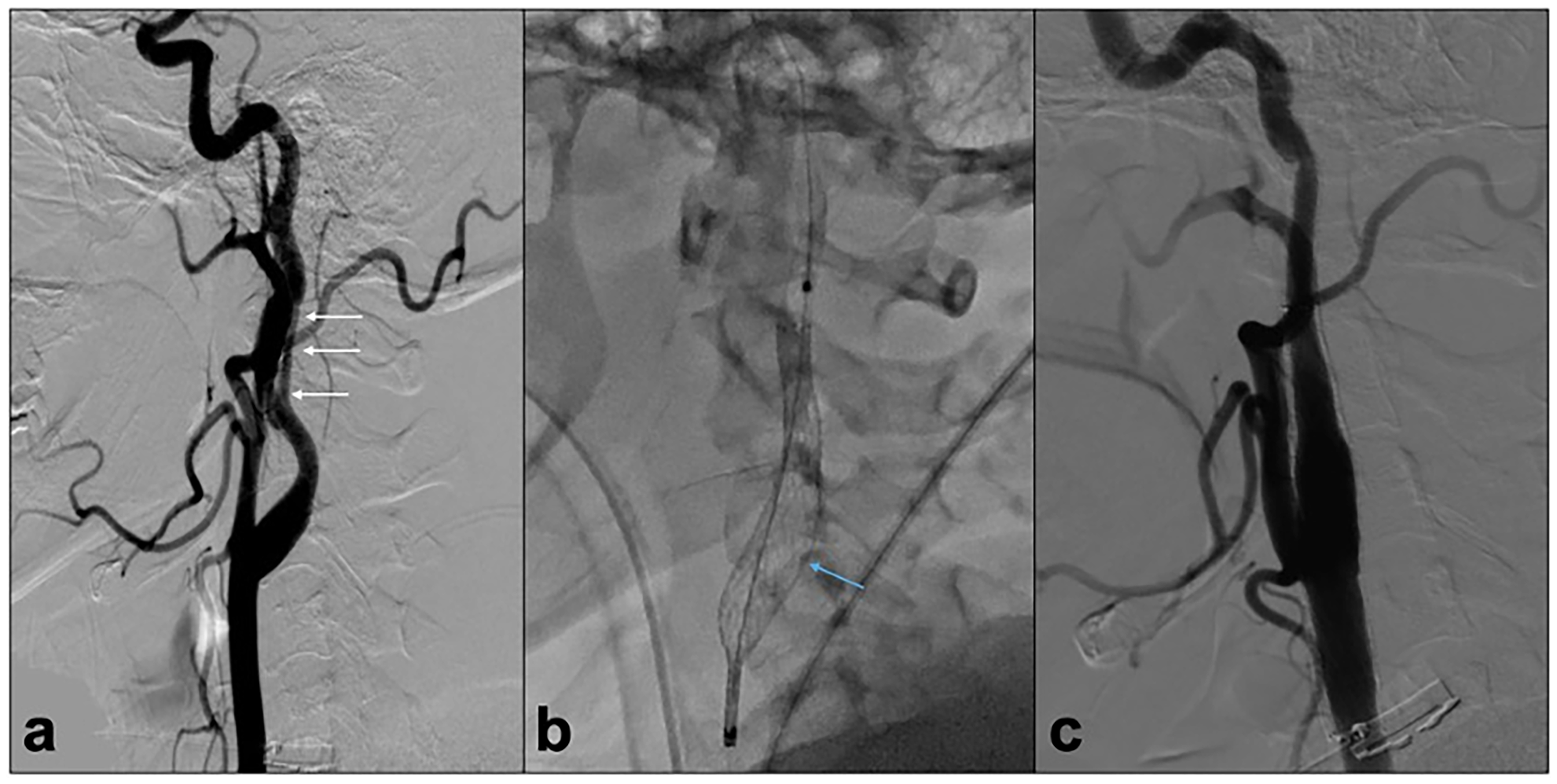
(a) Digital subtraction angiogram of lateral common carotid artery injection demonstrating a complex double-barrel dissection with accompanying pseudoaneurysm (arrows) in a patient in their 30s. (b, unsubtracted lateral view) Use of proximal anchoring technique with Protégé 8 × 60 mm carotid stent (arrow) at the proximal end of the long Surpass Streamline construct. (c) Twelve-month control angiogram demonstrates definitive reconstruction without migration or proximal in-stent intimal reaction.
The primary objective in treating petrocervical ICA dissections is to prevent embolic events and neurological deficits and to restore the natural lumen of the ICA with nonflow limiting anterograde flow. As compared to antiplatelet medications alone, neuroendovascular intervention is an option for treating such lesions where this can be achieved. This is performed by using a combination of FDS and/or coils to exclude the vascular tear and resulting pseudoaneurysm from arterial flow and facilitate endoluminal reconstruction. There remain no formal guidelines for the endovascular management of ICA dissections/pseudoaneurysms, in part due to the literature on this matter being limited to small case series and case reports. In a case series of 14 patients, Phogat and colleagues demonstrated the safety of stenting, stent-assisted coiling, coiling with glue, and flow diversion for the management of pseudoaneurysms of the ICA with only one reported complication in the coiling group. 11 Other studies describing the successful application of FDS for pseudoaneurysm treatment report a similarly low complication rate across small cohorts.12,13 These results are not surprising as the proximal location of these devices in the petrocervical segments, as well as the lack of perforators, branch coverings, and microscopic vessel size that come from intracerebral flow diversion, make management with FDS nearly assured.
The use of FDS in petrocervical dissections is not without its limitations: use of DAPT, lack of immediate aneurysmal thrombosis, and the availability of FDS and laser-cut nitinol stents specifically designed for this application are just a few. 7 Devices such as Surpass Streamline and Pipeline Embolization Device (Medtronic Neurovascular) are indicated for deployment in and distal to the petrous ICA and are thus only available in diameters up to 5 mm. The Surpass Streamline implant is known to be able to go up to sizes of 6 mm and greater due to its construction on a mandrel that approximates these sizes. There are reports of the use of larger than 5 mm FDS, but these devices are not currently commercially available in the USA. 14 A case series by Baz et al. demonstrated that the cervical and petrous segments of the ICA measure on average 5.49 mm (±0.64) and 4.53 mm (±0.43) in diameter, respectively. 15 Thus, even the largest commonly used FDS (Pipeline Embolization Device) may alone not achieve complete apposition to the wall of the cervical ICA. Furthermore, physiologic neck turning and respiration result in the carotid artery undergoing longitudinal wall motion, which can potentially cause movement of an FDS within the cervical ICA. 16 Such displacement of the FDS could lead to neointimal hyperplasia with subsequent in-stent stenosis, flow limitation, and even ischemic stroke.
To cover and flow divert long-segment ICA dissections, we used endovascular flow diversion with a proximal anchoring technique to prevent flow diverter migration. This technique leverages the rigidity of carotid artery stents (CAS), which have long been used for the management of carotid artery stenosis. 17 Carotid artery stents, such as the Protégé or Enterprise, are crafted with strong micromesh that resists compression, migration, and enhances wall apposition. In contrast, flow diverters are more compliant, made with smaller diameter wires (28-31 μm), and are intended for use in the nonmobile intracranial vasculature where the risk of stent migration is known to be low. 18 Proximal pinning of the flow diverter via nitinol stents serves to prevent comparatively softer FDS from migration as well as preserve vessel wall apposition and reduce intimal reaction. Anchoring corrects for small FDS that are placed in wide carotid artery segments such as the bulb. This technique allows for the remodeling of complex dissections and pseudoaneurysms without the disadvantages that FDS placement in the mobile cervical segment may carry.
The success of the technique largely depends on meticulously following specific considerations that ensure the effectiveness and safety of the proximal anchoring technique when dealing with complex vascular lesions. A critical factor is ensuring an adequate overlap between the FDS construct and the proximal anchoring stent, which typically spans 5 to 10 mm, to secure a firm engagement. Additionally, employing a standard length of 40 mm for the carotid stent ensures proper position within the common carotid artery. It is also crucial to avoid oversizing the proximal anchoring stents, as they can significantly expand FDS when deployed within them, raising concerns of damaging the FDS. Finally, the appropriate selection of the laser-cut, nitinol stent is paramount. We predominantly utilize carotid stents, with a preference for the Precise, due to its compatibility with a 0.070-inch DAC. In our practice, we often configure the device assembly to include two interlocking Surpass Streamline devices of 50 mm in length, anchored with an Atlas 4.5 mm × 21 mm distally and an 8 mm × 40 mm Precise delivered on the proximal end. This technique has been found proficient, allowing for the deployment of all necessary devices through a Vecta 071 115 cm DAC. This setup obviates the need for an exchange technique by facilitating the use of the same DAC for the entire procedure, thereby enhancing procedural efficiency and safety.
While endovascular flow diversion is not the sole technique available for the treatment of petrocervical dissections, it may become the preferred method.2,3,9–13 When compared to alternative historical methods, such as the off-label use of laser-cut nitinol stents, we believe FDS are more effective as their finer mesh helps the damaged (nonatherosclerotic) artery to heal and undergo controlled reconstruction and remodeling. Endovascular flow diversion devices with their finer cobalt chromium mesh and slimmer profile strut design are designed to be minimally traumatic to endothelium as they are primarily purposed to be implanted in intracranial vasculature.19–21 Finally, the delivery systems of modern flow diversion devices are low profile (as compared to carotid or coronary stents) and can readily be placed in tortuous cervical ICA segments where these complex dissections are prone to occur.
Limitations
The current study was a nonrandomized, single-arm, and retrospective study that only allows for correlations, and not causation, to be made. Furthermore, additional investigation is needed with extended follow-up to determine if this technique would be effective over the decades of life the patients in this study will likely have. Surgeons should be cautious when applying this technique to other intracranial locations as its safety and efficacy were demonstrated in a limited cohort of patients restricted to petrocervical dissections.
Conclusion
A petrocervical flow diverter reconstruction with a proximal laser-cut nitinol stent anchoring technique is a safe and effective treatment for carotid dissections and pseudoaneurysms. We recommend this treatment option to achieve complete aneurysm occlusion and mitigate stent migration and neointimal hyperplasia with physiologic neck movement. Comparative and prospective studies are required to delineate the optimal treatment regimen for petrocervical carotid dissections and pseudoaneurysms.
This study did not receive any financial support for the research presented here. This research was accepted as an abstract at the 2022 Annual Society of Neurointerventional Surgery Meeting in Toronto, Canada.
Footnotes
Authors’ note
IRB: MetroWest Medical Center IRB: 2020-039.
Authors’ contribution
JKC and BMM contributed equally to this work. JKC, BMM, DAZ, and FJL assisted with the conception and design of the manuscript. JKC, BMM, and FJL drafted and revised the manuscript for important intellectual content. JKC, BMM, MWK, JCCdB, and GA assisted with the data acquisition and analysis. NBB, MTB, SS, GPC, and ALC reviewed the important intellectual content presented in the manuscript. ALC, MWK, and FJL performed treatment procedures and critically revised the important intellectual content. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alexander L. Coon, MD, is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Stryker Neurovascular, Cerenovus, Rapid Medical, Avail MedSystems, Imperative Care, Deinde, InNeuroCo, Q'apel; a proctor for MicroVention-Termo, Stryker Neurovascular, and Medtronic Neurovascular. Geoffrey P. Colby, MD, PhD, is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Rapid Medical, Cerenovus, and Stryker Neurovascular. Shuichi Suzuki, MD, PhD is a consultant for Medtronic Neurovascular. Matthew Bender, MD, is a proctor Stryker Neurovascular. Narlin B. Beaty, MD, is a proctor for Medtronic Neurovascular, Stryker Neurovascular, and CMO of NeuroMedica.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
