Abstract
Introduction
No consensus exists on the necessity of neurocritical care unit (NCU)-level care following unruptured intracranial aneurysm (UIA) treatment. We aim to identify patients requiring NCU-level care post-treatment and determine potential cost savings utilizing a selective NCU admission protocol.
Methods
A retrospective analysis of all UIA patients who underwent endovascular treatment at a single center from 2017–2022 was conducted. Data on demographics, preprocedural variables, radiographic features, procedural techniques, intra/postoperative events, and length of stay (LOS) were collected. Multivariable analysis was performed to identify patients requiring NCU-level care post-treatment. Cost analysis using hospital cost data (not charges/reimbursement) was performed using simulated step-down and floor protocols for patients without NCU indications following a hypothetical six-hour post-anesthesia care unit observation period.
Results
Of 209 patients, 179 were discharged within 24 h and 30 had prolonged LOS. In our analysis, intra- and postoperative events independently predicted prolonged LOS. In our subanalysis, 47 patients demonstrated NCU needs: 24 with intraoperative indications, 18 with postoperative indications, and five with both. Of the 23 with postoperative indications, 20 were identified within six hours, while three were identified within six to 24 h. The median variable cost per patient for the current NCU protocol was $31,505 (IQR, $26,331-$37,053) vs. stepdown protocol $29,514 (IQR, $24,746-$35,011;p = 0.061) vs. floor protocol $26,768 (IQR, $22,214-$34,107;p < 0.001). Total variable costs were $6,211,497 for the current NCU protocol vs. $5,921,912 for the step-down protocol (4.89% savings) and $5,509,052 for the floor protocol (12.75% savings).
Conclusion
Most patients requiring NCU-level care following UIA treatment were identified within a six-hour postoperative window. Thus, selective NCU admission for this cohort following a six-hour observation period may be a logical avenue for cost reduction. Our analysis demonstrated 5% and 13% savings for uncomplicated patients using step-down and floor admission protocols, respectively.
Introduction
Unruptured intracranial aneurysms (UIA) are common, with a prevalence ranging from 2.8–7.0% in the general population.1–3 Despite the asymptomatic nature of these lesions, spontaneous rupture can result in subarachnoid hemorrhage, which is associated with a high rate of morbidity and mortality.4–6 Elective endovascular treatment, with its favorable rate of periprocedural complications, has emerged as a standard therapeutic modality for UIAs. 7 Although generally considered safe, this endovascular treatment does carry a risk of hemorrhagic and thromboembolic complications, predisposing patients to adverse outcomes.8–10
As a result, patients are typically admitted to a neurocritical care unit (NCU) after treatment for close neurological, hemodynamic, and cardiac monitoring. However, the necessity of NCU-level care following endovascular treatment for UIAs remains a subject of debate. Several studies have raised questions about the need for NCU-level care after the UIA treatment.11–13 Eisen et al. 12 suggested that selected patients could be safely placed on a regular floor unit instead of the intensive care unit (ICU) after treatment. Stetler et al. 8 explored the safety and cost-effectiveness of a step-down unit (SDU) or telemetry unit admission for immediate care following uncomplicated neurointerventional procedures. Although cost savings can reduce reimbursement in some fee-for-service contracts, most Medicare reimbursement and increasing portions of private insurance and Medicare Advantage contracts have some level of bundled payment or cost savings incentive. These incentives currently, or eventually, will penalize inefficient care and push providers to focus on costs in order to preserve financial margins.
The purpose of our study is to investigate the necessity of NCU-level care following the elective endovascular treatment of UIA. Specifically, we sought to identify indications for NCU-level care during and after the procedure and determine factors that predict prolonged length of stay (LOS) following treatment. Additionally, we assessed the potential healthcare cost savings associated with the implementation of a selective rather than a default NCU admission protocol post-UIA endovascular treatment.
Methods
A retrospective database review of patients who underwent elective endovascular treatment for UIA at a single center between 2017 and 2022 was performed. Inclusion criteria were age >18 years and undergoing elective endovascular treatment for UIA. Any patients who presented emergently without prior outpatient coordination in an elective setting were deemed non-elective and were excluded. The study was approved by our institutional review board (IRB #230342) and the requirements for informed consent were waived.
Study population
Patients with UIA were considered for prophylactic treatment based on patient factors such as age, gender, medical history, and family history as well as radiographic factors such as size, location, shape, and evidence of dynamic change. When endovascular treatment was recommended, one of the following techniques was employed: 1) primary coil embolization, 2) stent-assisted coil embolization, 3) balloon-assisted coil embolization, and 4) flow diversion (including both standard and intra-saccular flow diversion). Per current institutional protocol, all patients were admitted to NCU following UIA treatment for close hemodynamic, cardiac, and neurological monitoring.
Clinical variables
Data on demographics, past medical history, radiographic features, procedural techniques, intraoperative events (thrombus formation and/or altered intracranial hemodynamics, coil extrusion, aneurysmal rupture/perforation and/or contrast extravasation, vascular dissection, or access-related complication), postoperative events (hemodynamic instability, new or worsening neurological deficits, critical laboratory derangement, and respiratory distress), and LOS were collected. Prolonged LOS was defined as exceeding 24 h. NCU indications were defined as intra- and postoperative events requiring close neurological, hemodynamic, or cardiac monitoring, bedside interventions requiring intensive level of care, or artificial life support systems only available in an intensive care unit.
Characteristics of UIAs
Among the 209 UIA cases, the mean volume and diameter of the dome and the mean diameter of the neck were 37.51 mm3, 8.28 mm, and 4.48 mm, respectively. Of included patients’ aneurysms, the following locations were represented: 51/209 ophthalmic/superior hypophyseal artery, 49/209 posterior circulation (basilar bifurcation and trunk, vertebral, and PICA), 40/209 ACA/ACOM, 28/209 PCOM/anterior choroidal artery, 26/209 ICA terminus, 13/209 MCA, and 4/209 PCA. The number of aneurysms with irregular conformation was 154/209, regular conformation 43/209, and fusiform or dissecting 10/209.
Care capabilities of floor, SDU, and NCU
At our institution, the floor unit maintains a 4:1 patient-to-nurse ratio with a limited number of telemetry-equipped beds. Nursing staff are not authorized to administer continuous infusions of IV vasoactive medications, although PRN medications for blood pressure management can be given. Neurological checks are performed every four (Q4) hours.
The SDU operates with a 3:1 patient-to-nurse ratio, allowing for closer monitoring of patients, including neurological checks every two (Q2) hours. All SDU beds are equipped with telemetry, and nursing staff are authorized to administer IV vasoactive medications via peripheral lines. The SDU's proximity to the ICU facilitates prompt consultations with the critical care team.
In the ICU, patient care is primarily overseen by highly trained critical care providers with a patient-to-nurse ratio of 2:1. All ICU beds are telemetry-equipped, and neurological checks are performed every hour (Q1). Nursing staff in the ICU are authorized to administer continuous infusions of IV vasoactive medications through either peripheral or central lines, and to monitor blood pressure minute-by-minute via arterial lines. Additionally, EVD placement and mechanical ventilation can only be performed in the ICU.
Statistical analysis
Data are presented as mean and standard deviation for continuous variables and as frequency for categorical variables. Pearson test or Fischer's exact test were used to compare dichotomous variables between groups, while an independent sample two-tailed t-test was used to compare continuous variables, as appropriate. One-way analysis of variance or the Kruskal-Wallis test was used for comparisons of multiple groups. Univariate analysis was performed to identify potential covariates that are predictive of prolonged LOS. Multivariable logistic regression analysis was then performed using the stepwise backward elimination process to determine independent predictors of prolonged LOS. Descriptive statistics were performed to analyze the timeframe of postoperative complications.
Cost analysis
UIA patients under the current NCU protocol had all their surgical and hospital costs (not charges or reimbursements) recorded. A cost analysis was performed substituting base overnight NCU costs with base step-down (SDU) and floor admission costs to simulate alternative protocols. All patients also had their post-anesthesia care unit (PACU) costs adjusted to a hypothetical six-hour observation time window, which was calculated using internal PACU cost data. This cost was applied based on the patient's PACU stay: 1) for patients admitted directly to NCU post-treatment, the six-hour PACU cost was added, 2) for patients with a PACU stay shorter than six hours, the cost was increased to match the six-hour PACU cost, and 3) for patients with a PACU stay cost exceeding the six-hour cost, the higher cost was retained to remain conservative. A cost analysis was then performed using the simulated SDU and floor admission protocols for patients without NCU indications identified by the end of six-hour PACU observation period. Statistical significance in variable costs among the NCU, SDU, and floor protocols was assessed using the Mann-Whitney U test. Thirty patients were excluded from the analysis due to incomplete cost data from a combination of ongoing negotiation or administrative processes and the common practice of lumping same-service elective operations together if patients had multiple embolization or diagnostic angiograms over a short period, which made it difficult to accurately divide costs.
Results
Description of samples
A total of 209 patients who underwent elective endovascular treatments for UIAs were identified. Of these, 30 patients (30/209) had prolonged LOS, while 179 patients (179/209) were discharged from the hospital within 24 h. Mean age for LOS ≤ 24 h group was 58.98 (12.95, SD) and for LOS > 24 h was 53.73 (14.70, SD) (p = 0.0455). Prolonged LOS was associated with intraoperative events; thrombus formation and/or altered intracranial hemodynamic (23.33% vs. 3.91%, p < 0.001), rupture/perforation and/or contrast extravasation (6.67% vs. 1.12%, p = 0.040), coil extrusion (10.00% vs. 1.68%, p = 0.012), and vascular dissection (6.67% vs. 0%, p < 0.001), as well as postoperative events; retroperitoneal bleeding (3.33% vs. 0%, p = 0.014), hemodynamic instability for other causes (30.00% vs. 2.23%, p < 0.001), new or worsening neurological deficit (16.67% vs. 0.56%, p < 0.001), critical laboratory derangements (20.00% vs. 1.68%, p < 0.001), and respiratory distress (6.67% vs. 0.56%, p = 0.009). Aneurysms of the posterior cerebral artery (PCA) were also associated with prolonged LOS (6.67% vs. 1.12%, p = 0.040) (Table 1).
Description of study samples.

Predictors for prolonged LOS
Of 209 patients, 179 (85.65%) were discharged within 24 h post-treatment and 30 (14.35%) had prolonged LOS. On the univariate model, prolonged LOS was associated with intraoperative events such as thrombus formation or altered intracranial hemodynamics (OR, 14.67; p = 0.020; 95% CI: 1.52–151.34). It was also associated with postoperative events, including coil extrusion (OR, 89.03, p = 0.007, 95% CI: 4.25–356.7), hemodynamic instability (OR, 27.66, p = 0.007, 95% CI: 2.72–410.74), new or worsening neurological deficit (OR, 382.97; p = 0.001; 95% CI: 1.58–2629.08), and critical laboratory derangement (OR, 130.19, p = 0.019, 95% CI: 2.32–1174.32). On the multivariable model, independent predictors for prolonged LOS were neurological deficit (OR,100.68; 95% CI, 1.32–210.32; p < 0.001), hemodynamic instability (OR,27.84; 95% CI, 6.51–134.88; p < 0.001), critical laboratory derangement (OR,14.51; 95% CI, 1.31–153.96; p = 0.022), intraoperative coil extrusion (OR,17.81; 95% CI, 2.56–120.21; p = 0.002), and intraoperative thrombus formation and/or altered intracranial hemodynamics (OR,11.87; 95%CI, 2.73–50.52, p < 0.001). No association was found with preprocedural variables or procedural techniques (Table 2).
Multivariate logistic regression analysis for predictors of prolonged LOS.

Timeframe of intraoperative and postoperative indications for NCU-level care
Intra- and postoperative indications for NCU-level care were defined as respiratory distress or the need for close neurologic, hemodynamic, or cardiac monitoring per the discretion of clinicians at our institution, bedside interventions requiring an intensive level of care, and/or artificial life support systems only available in an intensive care unit. Of 209 patients included in the analysis, 47/209 patients (22%) had at least one intra- and/or postoperative indication for NCU-level care identified within 24 h of the procedure end time, with 26 of these 47 patients having a prolonged LOS. Of these 47 patients, 24/47 (51%) were identified as having an NCU indication at procedure end time due to isolated intraoperative events, 18/47 (38%) with an isolated postoperative indication, and 5/47 (11%) as having both. Of the 23 patients with postoperative indications for NCU-level care, 20/23 (87%) were identified within a six-hour postoperative window and 3/23 (13%) were identified after six hours. Within the six-hour window, 7/20 (35%) demonstrated hemodynamic instability, 5/20 (25%) new or worsening neurologic deficit, 5/20 (25%) critical laboratory derangements, and 3/20 (15%) respiratory distress.
There were three patients who had postoperative indications identified beyond the six-hour postoperative window. The first patient developed delayed onset left hemi-body numbness, for which an MRI was conducted, revealing a small punctate infarct within the right thalamus. This event did not necessitate significant interventions other than increasing the aspirin dose from 81 to 325 mg and arranging physical therapy/occupational therapy consultation. The second patient experienced new-onset atrial fibrillation with a rapid ventricular rate, which reverted to sinus rhythm following metoprolol and amiodarone bolus administration. This patient remained stable post-episode and was discharged on apixaban 5 mg BID with scheduled follow-ups with cardiologists for further evaluation. The third patient developed refractory hypotension in the setting of adrenal insufficiency. Blood pressure normalized after hydrocortisone injection and normal saline fluid bolus administration without the need for vasopressors. None of these patients required ICU-specific interventions, such as mechanical ventilation, non-invasive positive pressure ventilation for respiratory failure, intracranial pressure (ICP) monitoring, external ventricular drain (EVD) placement, hypertonic fluid administration for cerebral edema, continuous vasoactive medication infusion, or status epilepticus management. 14 Additionally, these delayed postoperative complications did not extend their LOS, and all patients were discharged within 24 h post-UIA treatments.
Potential healthcare cost savings
The median variable cost per patient for the current NCU protocol was $31,505 (IQR, $26,331–$37,053) vs. SDU protocol $29,514 (IQR, $24,746–35,011; p = 0.061) vs. floor protocol $26,768 (IQR, $22,214–$34,107; p < 0.001). Total variable costs for the current NCU protocol were $6,211,497 vs. SDU protocol $5,921,912 (4.89% savings) vs. floor protocol $5,509,052 (12.75% savings) (Figure 1).
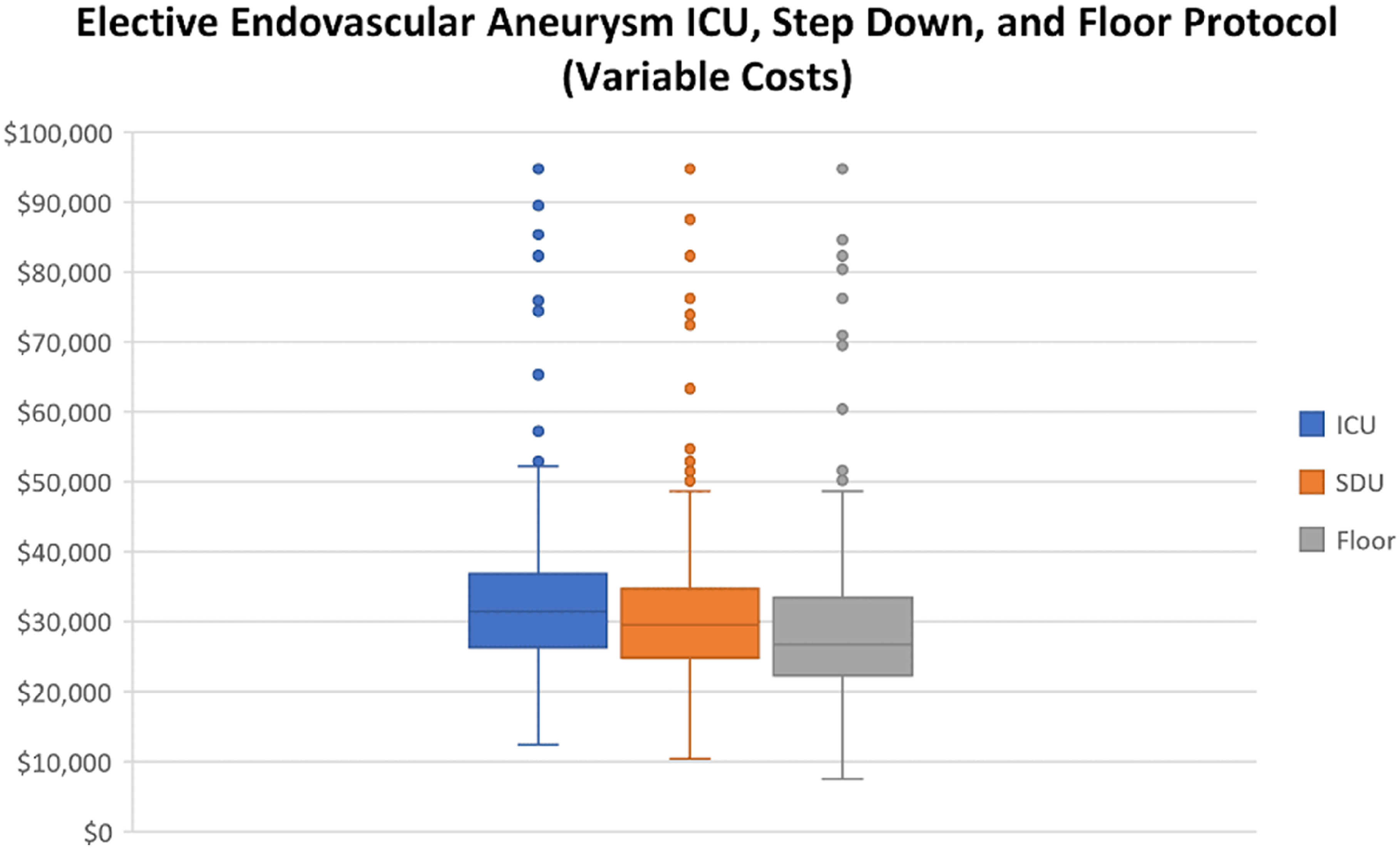
Whisker-box plot displaying cost differences among NCU, SDU, and floor protocols.
Discussion
Our study suggests that the intraoperative and postoperative events, both indications for NCU-level care post-UIA treatment, are the only significant predictors for prolonged LOS, while preprocedural variables or type of treatment had no association with LOS. Further, we demonstrated that a majority of these intra- and post-operative indications for NCU-level care were identified within a six-hour postoperative window. Our cost analysis showed that admitting uncomplicated patients without intra- or postoperative events to SDU or floor rather than ICU after a six-hour PACU observation period would be a logical avenue for cost reduction, resulting in cost savings of 4.89% and 12.75%, respectively.
Our findings that intraoperative and postoperative events are the independent predictors for prolonged LOS following UIA treatment are consistent with previous studies.12,13,15,16 Various studies have shown that postoperative complications following UIA treatment are likely to occur shortly after the procedure.11,17–19 Kameda-Smith et al. 17 found that most complications were detected within six hours after anesthesia reversal. In a retrospective study of 687 elective UIA treatments by Arias EJ et al., 18 74% of complications occurred within four hours of the procedure, 14.8% within 4–12 h, and 11.1% after 12 h. This study also found that patients with complications occurring after 12 h were discharged without deficits. Our findings align with this study, as 87% (20/23) of postoperative events occurred within the six-hour window. The three postoperative events that occurred outside the six-hour PACU observation window were delayed neurological deficit, new-onset atrial fibrillation with RVR, and refractory hypotension. While NCU may be best suited to identify and manage these postoperative events—with its lowest patient-to-nurse ratio of 2:1 (3:1 in the SDU and 4:1 on the floor), Q1 hour neurological checks (Q2 hours in the SDU and Q4 hours on the floor), and capabilities for continuous infusions of IV vasoactive medications via both peripheral and central lines, mechanical ventilation, and EVD placement—it is important to note that all of these patients did not require NCU-specific care 14 and were discharged within 24 h post-treatment. This demonstrates that patients without intra- and postoperative events identified within the six-hour PACU observation period can be safely managed in an SDU or floor setting.
The lack of an association between prolonged LOS and preprocedural variables such as demographics, comorbidities, and neurovascular comorbidities index (NCI) in our cohort contrasts with existing literature. Koo et al., 16 in a retrospective analysis of the National Inpatient Sample (NIS) database of 46,880 UIA patients who underwent the elective treatment, demonstrated that in addition to in-hospital complications, female gender, black and Hispanic race, and preadmission comorbidities were also predictive of prolonged LOS. Likewise, Patel et al., 20 in another study that also utilized the NIS database, demonstrated that NCI was associated with prolonged LOS, poor functional outcomes, and in-hospital mortality. This discrepancy could be because our study included only the patients who underwent endovascular treatment, while the NIS database included both endovascular treatment and neurosurgical clipping. Moreover, the relatively small sample size of our cohort compared to the NIS database could have also limited the statistical power.
Based on our cost analysis, utilizing an SDU or floor protocol instead of a default NCU protocol following a six-hour PACU observation period could lead to a reduction in healthcare costs, which is in line with existing literature. Stetler et al. 13 found that admitting patients to an SDU or telemetry unit immediately after treatment without an observation period resulted in significant cost savings of 32% and 57%, respectively. Additionally, Burrows et al. 11 demonstrated that patients admitted to an SDU following UIA treatment had comparable outcomes to those admitted to an NCU. Eisen et al. 12 add that most patients undergoing UIA treatment who did not experience intraoperative complications can be safely managed without NCU admission if frequent and reliable neurological assessments are available during a PACU observation period. As such, our study further supports the feasibility and safety of SDU or floor admission for uncomplicated patients without intra- and postoperative complications following UIA treatment. Nevertheless, it is crucial to note that such decisions should be made based on clinical assessments on a case-by-case basis, with safety being the primary consideration.
Our study is not without limitations. First, this is a retrospective cohort study conducted in a single center. As such, the results may not be generalizable to other settings. Adequately powered prospective studies are warranted to identify, with certainty, patients who can be safely managed in SDU or floor settings and those who would benefit from NCU-level care. Second, the relatively small number of patients who had intra- and postoperative indications for NCU-level care post-UIA treatment may have limited the statistical power of our analyses and prevented us from identifying other potential predictors. Third, a prolonged LOS can also be defined as greater than 48 h and employing a 24-h threshold could potentially bias the results. However, a 24-h cut-off is a more stringent criterion, and even with this stricter criterion, our study demonstrates that 179/209 (86%) of patients were discharged from the hospital within 24 h post-treatment. This further strengthens our conclusion that patients who do not exhibit intra- or postoperative NCU indications within the six-hour postoperative window can be safely managed in SDU or floor. Additionally, we were unable to assess the predictors of readmission following treatment due to the high morbidity profiles of our patient cohort, which complicated the isolation of readmissions specifically related to their UIA treatments. However, this is an important area that warrants further investigation and should be considered in future studies. Fourth, the analysis of the impact of NCI on prolonged LOS was performed without dichotomization, which may account for an insignificant association between NCI and prolonged LOS in our cohort. As multiple previous publications have suggested the potential relationship between NCI and post-treatment LOS and outcomes,20–22 a future study is needed to identify the optimal NCI threshold for identifying those who warrant NCU-level care post-UIA treatment. Third, although we conducted a multivariable analysis for prolonged LOS, we were unable to perform a similar analysis for NCU-specific care. Franco et al. 14 define NCU-specific care as any intervention, aside from hourly neurologic assessments, that should be administered in an NCU setting, such as mechanical ventilation, ICP monitoring, EVD placement, hypertonic fluid administration for cerebral edema, continuous infusion of vasoactive medications, and management of status epilepticus. Among the 209 patients included in our analysis, only five required ICU-specific care. Due to the small number of patients requiring NCU-specific care, a multivariable analysis was not feasible. Finally, our study is constrained by challenges associated with obtaining precise financial data and conducting cost analysis accurately. However, we ensured the collection of cost data rather than the charge or reimbursement data to maintain a comprehensive record of incurred expenses across all protocols. It is also very important to note that cost variations between institutions may impact the generalizability of the numerical findings presented in this study. However, instead of quantifying the extent of expenditure reduction associated with either SDU or floor protocols, our aim is to propose that NCU admission may be deemed unnecessary, particularly for patients without intra- or post-operative complications within a six-hour PACU observation period.
Conclusion
Our analysis suggests that intraoperative and postoperative events, both being indications for NCU-level care at our institution, are independent predictors of prolonged LOS following elective endovascular treatment of UIAs rather than pre-operative indicators. Further, most patients identified as having an indication for NCU-level care were identified intraoperatively or within six hours of procedure end time. Thus, selective admission following a close observation period rather than default NCU admission may be a logical avenue for cost reduction.
Footnotes
Acknowledgements
None.
Author contributions
Designed and conceptualized the study (SGR, SA, LV, MTF, MRF, RVC), collected data (SGR, SA, AMH), analyzed the data (SGR, SA, YK), performed cost analysis (CL), wrote the original draft (SA), critically revised the manuscript (SGR, SA, CL, LV, NM, AMH, MTF, MRF, RVC).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and informed consent requirements
The study was approved by Vanderbilt University Institutional Review Board (IRB #230342) and the requirements for informed consent were waived.
