Abstract
Subdural hematoma (SDH) refers to the collection of blood between the dura matter and the arachnoid membrane. Advancements in imaging technology have enabled the categorization of SDH based on specific imaging characteristics, causative factors, and the onset of symptoms. Given that the prognosis of SDHs varies significantly and is contingent upon the size and chronicity of the hemorrhage, a comprehensive understanding of its subtypes may carry crucial treatment implications. For example, an acute SDH classically results from severe traumatic brain injury and appears as a homogenous, crescent-shaped hyperdense extra-axial collection. If not treated, over the course of 1–3 weeks, this hematoma will evolve into a sub-acute phenotype as a consequence of subdural effusion and demonstrate mixed-density hemorrhage on imaging. Chronic SDH (cSDH) becomes the end result of an untreated SDH, with neo-membranization and neo-angiogenesis from branches of the middle meningeal artery driving a mass-like growth pattern. This review article aims to elucidate the complex anatomical features of the end-stage cSDH, with a particular focus on reconceptualization of this entity based on its mass-like growth patterns, and how this is driving a shift towards endovascular treatment.
Introduction
Subdural hematoma (SDH) refers to the collection of blood between the dura matter and the arachnoid membrane. Advancements in imaging technology have enabled the categorization of SDH based on specific imaging characteristics, causative factors, and the onset of symptoms. Given that the prognosis of SDHs varies significantly contingent upon the size and chronicity of the hemorrhage, a comprehensive understanding of subtypes and potential is crucial for treatment. For example, while acute SDH occurs classically secondary to a severe traumatic brain injury and presents as a homogenous crescent-shaped hyperdense extra-axial collection, if allowed to age untreated over the course of 1–3 weeks, this hematoma will evolve into a sub-acute phenotype as a consequence of subdural effusion and demonstrate mixed-density hemorrhage on imaging. Chronic SDH (cSDH) becomes the end result of an untreated SDH, with neo-membranization and neo-angiogenesis from branches of the middle meningeal artery (MMA) driving a mass-like growth pattern. This review article aims to elucidate the complex anatomical features of the end-stage cSDH, with a particular focus on reconceptualization of this entity based on its mass-like growth patterns, and how this is driving a shift toward endovascular treatment.
cSDH—a late-stage phenotype
cSDH development occurs from aging of an acute subdural hemorrhage (Figure 1) caused by damage via two major pathways: traumatic and atraumatic events.
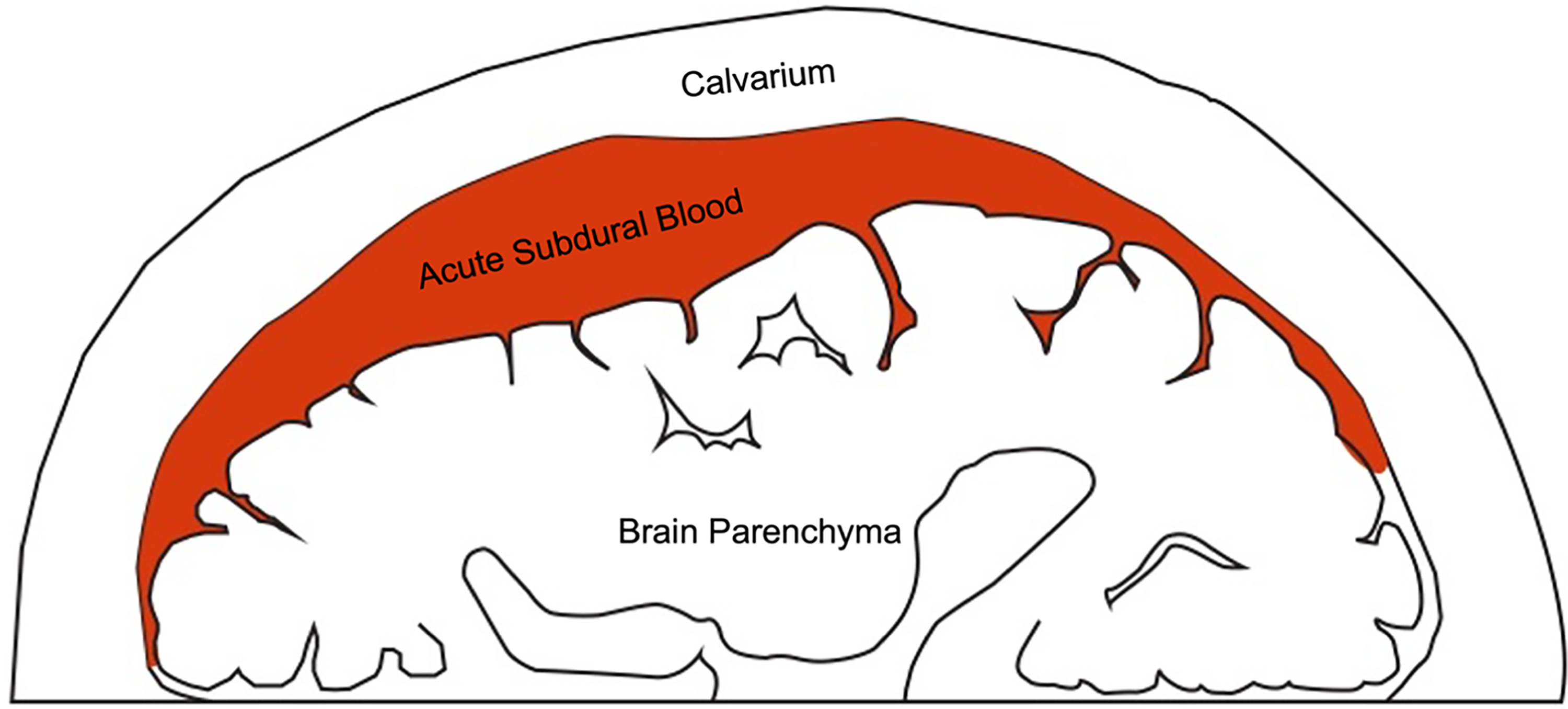
Acute subdural hematoma most often occurs via head trauma. Elderly individuals are at an increased risk due to a larger subdural potential space and higher prevalence of coagulopathies.
Traumatic cSDH has a predilection for elderly individuals who are at higher anatomical risk for subdural hemorrhage due to a larger subdural potential space from a combination of age-related cerebral atrophy, focal encephalomalacia from prior insult, and/or pre-existing chronic subdural hygromas.1–5 These anatomical risk factors are exacerbated by a higher percentage of coagulopathies from anticoagulant use for non-related co-morbidities.1,6,7 In these circumstances, light physical trauma can cause an outsized disruption of the dura and leakage of blood products into the subdural space which can remain occult until transformation to cSDH is complete. Traumatic cSDH formation in pediatric and young adult populations can occur as well due to the progression of acute SDH most often from non-accidental trauma or sports-related injuries,8–13 and although rare, can also occur secondary to rupture of congenital arachnoid cysts for predisposed populations.14–20
Atraumatic cSDH has been documented as a late-stage complication of intracranial hypotension, which causes tearing of subdural bridging veins resulting in occult SDH development. This phenomenon has most often been attributed to cerebral spinal fluid (CSF) leak following spinal anesthesia, CSF diversion therapies such as ventriculoperitoneal shunt implantation for the treatment of normal pressure hydrocephalus, or more rarely from spinal meningeal cysts and fistulas.1,21–29 However, select evidence has also demonstrated atraumatic cSDH formation in patients aged ≥69-year-old secondary to spontaneous intracranial hypotension. 30
The duality of cSDH expansion demonstrates mass-like features
cSDH anatomy and growth involves not only hemodynamic elements, but also a complex interplay with biochemical factors. In contrast to acute and sub-acute SDH, a cSDH will not eventually stop expanding when the opposing pressures of the intracranial vault exceed the hemodynamic intra-vascular pressures of a bleeding vessel characteristic of an active hemorrhage. This apparent hemodynamic contradiction in the pattern of expansion is driven by a separate biochemical pathway that causes cSDH to continue growth in a mass-like manner.
Similar in structure to a benign vascular neoplasm, cSDH presents as an intact mass complete with encapsulating neomembranes, organized microenvironmental architecture, chronic inflammation driven by cytokine secretion, and angiogenesis in a process mirroring tumor tropism.1,8,31–35 Analysis of surgically resected cSDH demonstrates a gelatinous mass of liquid or semi-solid consistency organized around a mixture of collagen fibrils supplied by neovascular networks composed of capillaries, sinusoids, and larger caliber vessels from branches of the MMA, and encapsulated by a discrete outer membrane capable of undergoing calcific degeneration1,31–41 (Figure 2). Although leakage of extravascular blood products into a nidus from fragile neovascular basement membranes continues to contribute a small component to cSDH expansion, the major driver of growth occurs biochemically from the delivery of nutrients in a tropic mass-like manner.1,8,33,35,42 The minimal contribution of late-stage hemorrhage to cSDH expansion is further supported by the sometimes absent signal detection on magnetic resonance imaging T2*, gradient-echo, and susceptibility-weighted sequences which are highly sensitive for iron deposition from extravascular blood products. 8
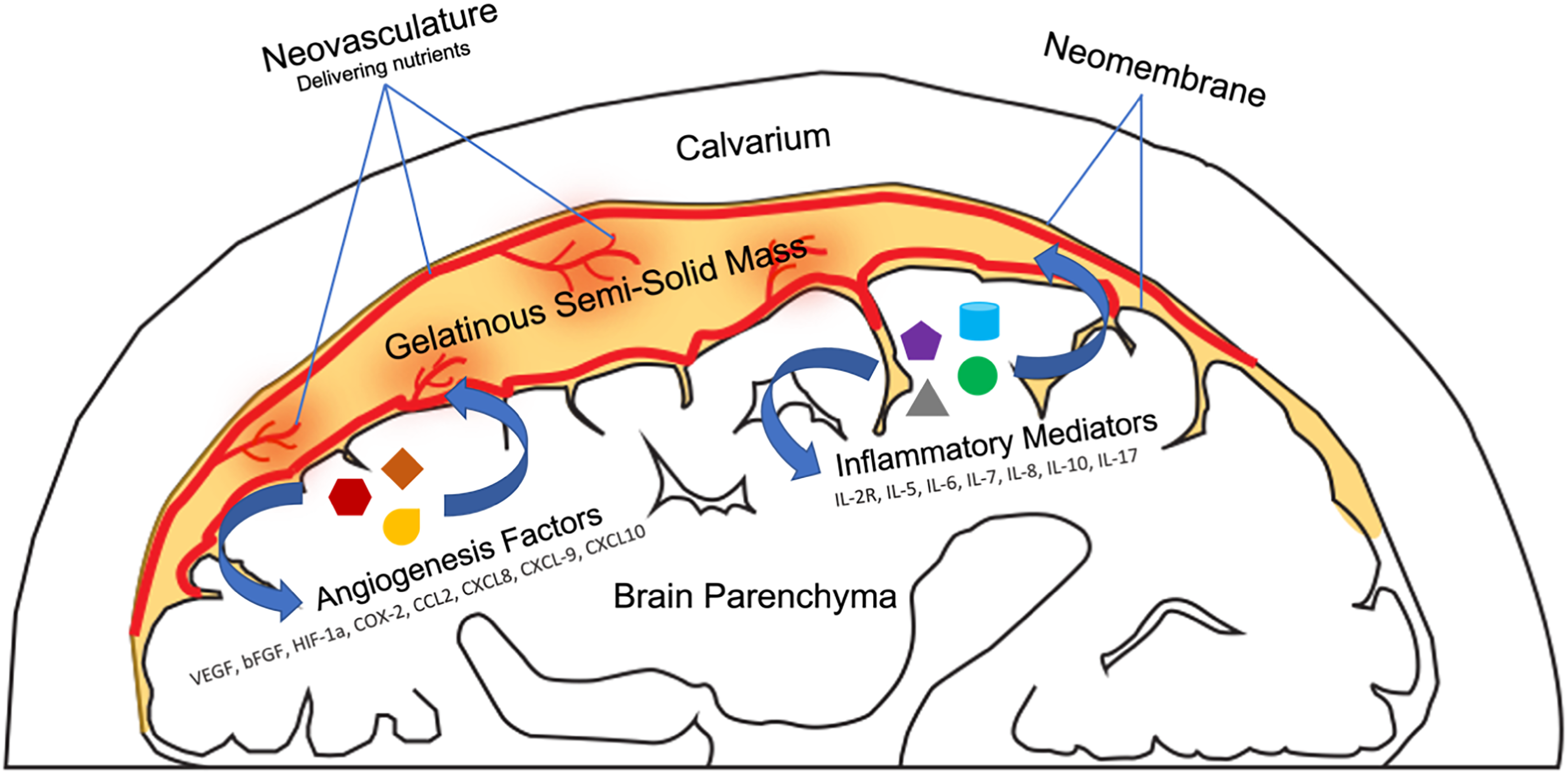
Duality of chronic subdural hematoma (cSDH) expansion occurs via both hemodynamic elements of hemorrhage, and biochemical factors involving cytokine signaling cascades and angiogenesis that promote growth in a mass-like manner.
Given the duality of cSDH expansion involving both hemodynamic and biochemical factors, it is unsurprising that endovascular embolization of the MMA (Figure 3), which provides neovascular supply to the expanding mass, results in an improved remission profile compared with either traditional burr hole evacuation or conservative management. Burr hole evacuation of cSDH decreases the extrinsic intracranial tamponade pressure and promotes further extravascular hemorrhage into the subdural space from leaky neovascular basement membranes resulting in re-accumulation, while leaving cSDH to tamponade on its own also leads to continued expansion as the growth phenotype shifts in a primarily biochemically driven mass-like manner. MMA embolization solves this duality by arresting blood supply to the cSDH, which allows for progressive involution over time by preventing the delivery of essential nutrients and cytokinetic factors to the established mass, while also allowing for a continued extrinsic tamponade.43–45 The result of MMA embolization closely resembles the pathophysiological contraction of a benign vascular neoplasm following embolization, as opposed to the arrest of contrast extravasation from active bleeding (Figure 4).
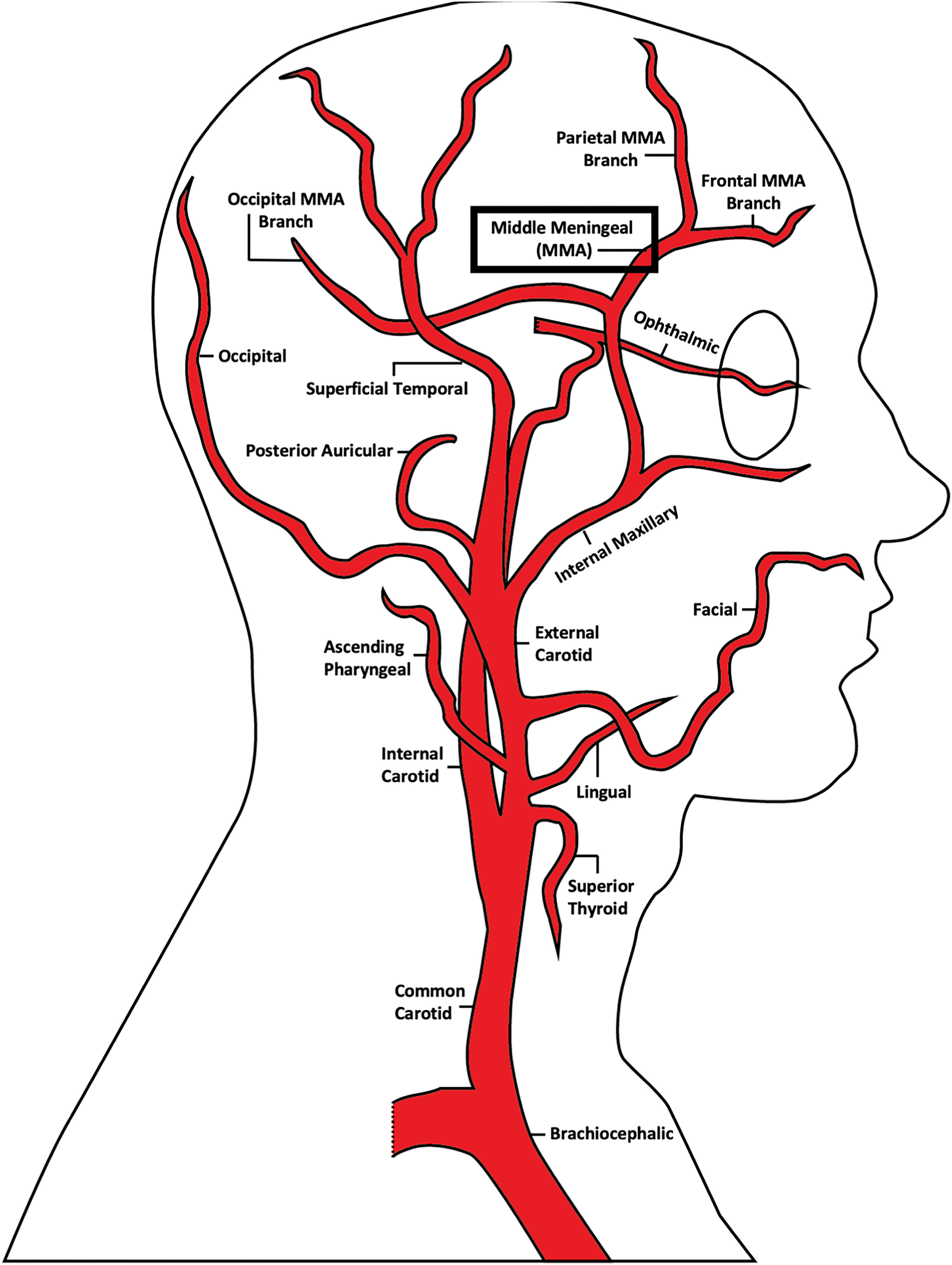
Vascular anatomy of the head and neck with a focus on branches of the external carotid artery, including the middle meningeal artery, whose branches contribute the major neovascular supply to chronic subdural hemorrhages.
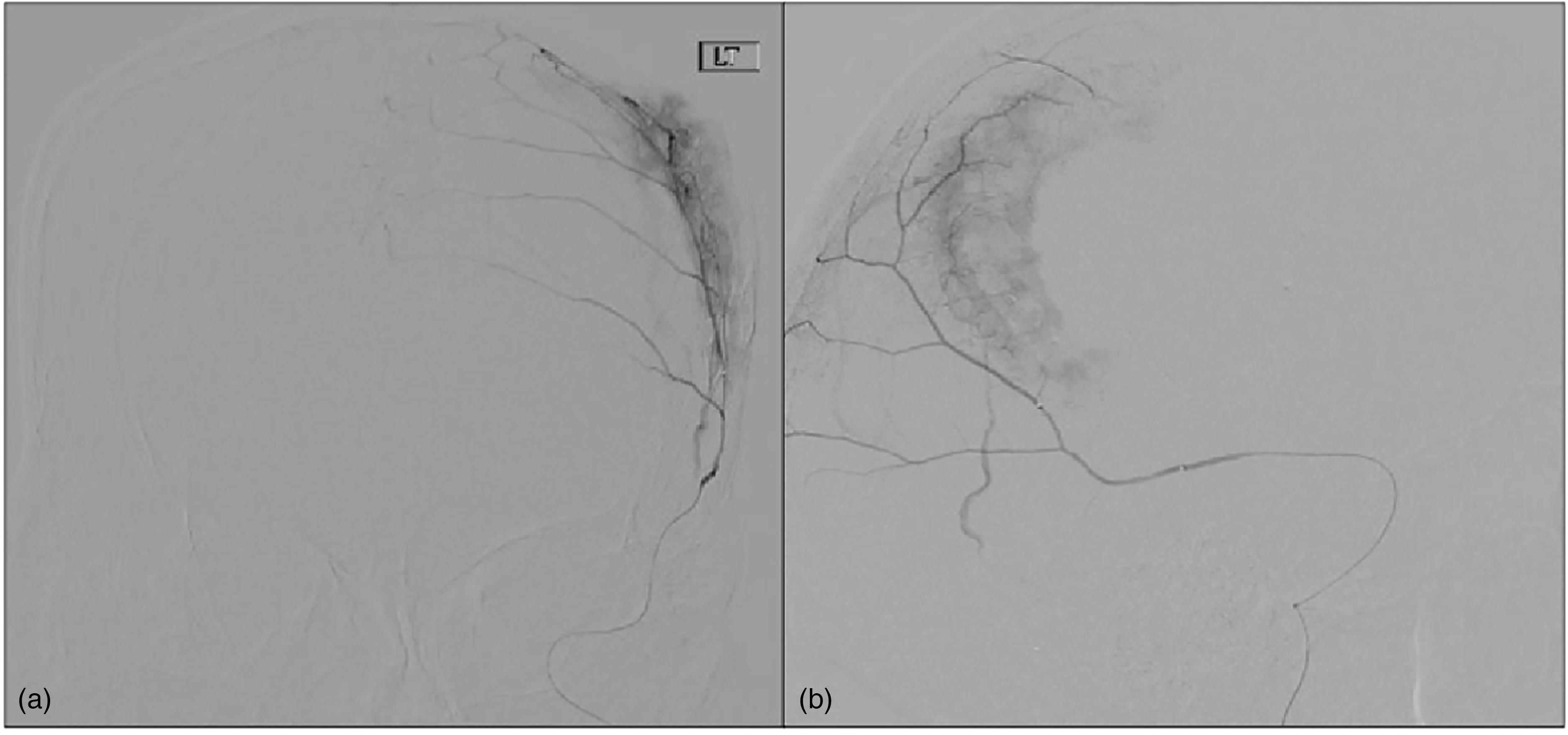
(a) Anterior–posterior and (b) lateral views of super-selective middle meningeal artery angiography demonstrating a contained and well-circumscribed blush in the region of a known chronic subdural hematoma (cSDH) similar to a tumor blush, and less similar to contrast extravasation typically seen with active bleeding.
Future directions for cSDH intervention
MMA embolization may benefit from exploration of embolics that target specific cytokinetic factors potentiating the biochemically driven portion of cSDH growth. Given that analyses of CSF have demonstrated an increase in the amount of vascular endothelial growth factor (VEGF) in trabecular-subtype cSDH, next-generation MMA embolics for this subgroup may involve the delivery of microparticles linked to anti-VEGF drugs such as aflibercept, bevacizumab, ranibizumab, and/or pegaptanib.1,46–48 Higher levels of pro- and anti-inflammatory mediators interleukin (IL)-2R, IL-5, IL-6, IL-7, IL-8, IL-10, and IL-17; and angiogenesis factors basic fibroblast growth factor, HIF-1a, COX-2, CCL2, CXCL8, CXCL9, and CXCL10 in cSDH CSF warrant their exploration and development as targeted embolics as well.1,33,35 Expansion of high throughput processing of cSDH to include lysates of neomembranes can broaden the discovery of more prospective biochemical drug targets, further increasing the opportunity for the development of moiety-linked embolic material.
Conclusion
There is growing evidence of the duality of cSDH expansion involving not only hemodynamic factors of hemorrhage, but also an additional even greater biochemical contribution that promotes growth with mass-like features similar to a benign vascular neoplasm. Conceptualization of cSDH treatment should therefore evolve, with MMA embolization reconceptualized as a tumor de-vascularization intervention in this paradigm. Further elucidation of mimics and cSDH-adjacent phenomena such as subdural hygromas may provide a greater understanding of the complexity of non-acute subdural proteinaceous collections, and lead to the development of better future treatments.49,50
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
