Abstract
Background
Radial arterial access has gained interest for neurovascular procedures in recent years. Although there are no randomized control trials for neurointervention procedures using radial access, there is growing literature demonstrating its feasibility and favorable outcomes. Equipment technical improvements, like the recently introduced BENCHMARK™ BMX®81 System, have made radial navigation safer, with improved maneuverability and support for a variety of procedures. We present a multicenter case series highlighting our institutional radial access experience comparing the BMX®81 with alternative catheters.
Methods
Multicenter retrospective cohort study of 80 patients who underwent neurovascular procedures through a radial approach. In half of the cases a BENCHMARK™ BMX®81 System was used. The comparison group consisted of the BENCHMARK™071 and 96, Neuron MAX®088 and BALLAST™ systems. Procedures included endovascular thrombectomy, carotid and brachiocephalic artery stenting, middle meningeal artery embolization, flow diverter stenting, vertebral artery sacrifice, aneurysm coiling, and WEB™ device deployment.
Results
In our series, the BMX®81 was successful in the navigation of the anatomy to the target location in 95% of cases. No radial access or BMX®81 related complications were identified. There was no significant difference in fluoroscopy time between the BMX81 and the comparison group. Four patients in the comparison group had catheter-related complications due to vasospasm. Eighty-six percent of BMX®81 cases had satisfactory outcomes and no technical difficulties. The remainder presented technical difficulties, but none of these were considered secondary to the puncture site or support structure.
Conclusions
The BENCHMARK™ BMX®81 System is a recently developed guiding catheter which has design and size features supporting radial access for a variety of neurovascular interventions. Early multicenter experience highlights the ease of use and versatility of this new catheter as an alternative to transfemoral access as well as other catheters used for radial access.
Introduction
Radial arterial access has been used for more than 70 years with the first report in 1948 when Radner et al. described a transradial access (TRA) through arterial surgical exposure. 1 This route has been used extensively in interventional cardiology, becoming the standard access for percutaneous cardiovascular interventions. 2 Due to less tortuous vasculature and a larger patient population, interventional access techniques have often been advanced by the interventional cardiology field before adoption by neurointerventional specialists. 3 Although transfemoral access (TFA) is the continued preferred access choice for most neurointerventionalists, the TRA approach has had growing interest as a first choice for neurovascular procedures in the recent years. 4 In some centers today, more than 90% of diagnostic and interventional procedures are performed using this route. Ruiz-Rodriguez et al. published a meta-analysis in 2016 summarizing outcomes for radial versus femoral access for patients with acute coronary syndrome, concluding a statistically significant lower rate for major access site complications and mortality with radial access. 5 Although there are no randomized controlled trials for neurointervention procedures using radial access, there is growing literature demonstrating the feasibility and favorable outcomes for a variety of procedures.6–8 Furthermore, if TFA is the preferred first choice access, some patients with complex aortic arch anatomy or disease of the abdominal aorta, iliac and femoral arteries demand an alternative route for successful treatment. 9 The marked improvements of specifically designed equipment have made radial navigation safer and allowed larger catheters to be used to provide stability, improved maneuverability, and support for a variety of neurointerventional procedures.
The recently introduced BENCHMARK™ BMX®81 System (Penumbra, Alameda, CA, USA), consists of a laser-cut stainless steel hypotube design with titrated transitions and is engineered to balance support and distal flexibility, positioning this catheter for application in a range of neurointerventional procedures. The stainless steel hypotube allows for a 7Fr outer diameter (OD) and a 0.081-inch inner diameter (ID) without compromising catheter support. In this article, we present a multicentre case series highlighting our early institutional experience with the BENCHMARK™ BMX®81 and we compared them with alternative catheters that were previously used for radial access at our institutions.
Materials and methods
This retrospective cohort study identified 40 patients from two national sites that underwent neurovascular interventions between April 2023 and February 2024 using the BENCHMARK™ BMX®81 System from a radial approach. For comparison, we selected the last consecutive 40 cases in our institutions, in which alternative catheter options were used with radial access, from July 2019 to February 2024. The comparison group consisted of the following catheters: BENCHMARK™071 (Penumbra, Alameda, CA, USA), BENCHMARK™ BMX®96 (Penumbra, Alameda, CA, USA), Neuron MAX®088 (Penumbra, Alameda, CA, USA) and BALLAST™ 088 (Balt USA LLC, Irvine, CA, USA). Clinical records, previous imaging and procedural reports/images were reviewed as well as follow-up imaging, clinical status, and subsequent procedures, when required. The primary outcome was technical success, defined as successfully completing the intended neurointervention procedure and the access catheter successfully reaching the target vessel. Diagnostic angiography cases were not included.
Informed consent was obtained from all patients for their respective neurointerventional procedure as per standard institutional practices. Patients were assessed in advance of their procedure for suitability for radial access using manual palpation and/or ultrasound assessment of the radial artery. Patients with a radial artery size of smaller than 2 mm were excluded. Patients were positioned in supine position in the neuroangiography biplane suite (Allura Clarity Xper FD20/20 X-ray system, Philips Medical Systems, Eindhoven, The Netherlands or ARTIS Icono, Siemens Healthineers, Erlangen, Germany). General anesthesia or conscious sedation was provided based on the usual institutional practices with consideration of patient, technical, and anesthetic factors. In the BMX®81 group, 29 patients (72.5% of total) underwent general anesthesia for their neurointerventional procedure, while 11 patients (27.5%) were treated under conscious sedation. In the comparison group, 26 patients (65%) underwent general anesthesia, while 14 (35%) were treated under conscious sedation.
The right radial artery was selected for access in all patients in both series. This access site was prepped and draped in usual institutional fashion including placement of a premolded cushion underneath the wrist to provide a degree of extension during the procedure. The right radial artery was accessed at the proximal site in all patients with no distal radial access (dRA) (snuffbox approach) or initial ulnar artery access performed in this cohort. Under ultrasound guidance, the radial artery was accessed using single wall puncture technique with a radial access needle and a radial access wire was advanced, after which a 6 or 7 French Glidesheath Slender™ radial sheath (Terumo Medical Corporation, Somerset, NJ, USA) was placed using the Seldinger technique. With the sheath in place, standard angiography was performed to characterize radial artery size as well as course of the arterial system in the upper extremity, including assessment for anatomical variants such as a brachioradial artery or radial artery loops. Through the short sheath, a cocktail consisting of 2.5 mg verapamil, 100–200 mcg nitroglycerin and 2000–3000 units of heparin was hemodiluted to 20 mL and slowly administered.
In the BMX®81 group, initial access was obtained with a 6 French Glidesheath Slender™ radial sheath in 14 cases, which was then exchanged for a BMX®81 using a 260-cm radial InQwire (Merit Medical, South Jordan, UT, USA). For the remaining 26 cases, a small skin nick was made, and initial access was obtained through a 7 French Glidesheath Slender™ radial sheath. The BMX®81 was then advanced through this Slender sheath over 125 cm 5 French Bernstein or Simmons catheters and 0.035-inch Terumo Glidewire Advantage® wire (Terumo Medical Corporation, Somerset, NJ, USA). In the comparison group, initial access was obtained with a 6 French Glidesheath Slender™ radial sheath in 14 cases and a BENCHMARK™071 access system was advanced through the sheath. For the remaining 26 cases, the 6 French Slender sheath was exchanged for the BENCHMARK™071, BENCHMARK™ BMX®96, Neuron MAX®088 and BALLAST™ 088 using a 260-cm radial InQwire, 180 cm Terumo Glidewire® or Glidewire Advantage® (Terumo Medical Corporation, Somerset, NJ, USA).
Once the BMX®81 catheter was in a satisfactory position, selective angiograms were performed. The catheters and wires required for treatment, were advanced coaxially through the BMX®81 to the target location. Neurointerventional procedures included endovascular thrombectomy, carotid stenting, brachiocephalic artery stenting, middle meningeal artery (MMA) embolization, flow diverter stent deployment, vertebral artery sacrifice, balloon and stent-assisted coiling as well as WEB™ (MicroVention, Aliso Viejo, CA, USA) device deployment. Procedures performed in the comparison group included aneurysm coiling, flow diverter stenting, carotid artery stenting, dural AV fistula embolization and endovascular thrombectomy.
At the conclusion of the procedure, arterial puncture site hemostasis was achieved with the placement of Terumo TR Band® radial compression device (Terumo Medical Corporation, Somerset, NJ, USA). The TR Band® was attached firmly to the wrist at the puncture site and was inflated to 15–18 mL, then 1–5 mL were withdrawn until a small drop of blood was seen at the puncture site. The balloon was then re-inflated by 2 mL to ensure adequate occlusion pressure. The TR band was subsequently deflated with the following schedule: Removal of 2 mL after 30 minutes, a further 2 mL after 45 minutes, 3 mL after 60 minutes, 3 mL after 75 minutes, and full deflation after 90 minutes. If at any time point, blood was noted to accumulate at the puncture site, the immediately withdrawn quantity of air was readministered into the band and the time point reset. When hemostasis was achieved with the TR band fully deflated, the band was then removed and Tegaderm dressing was used to cover the puncture site.
Results
BMX®81 group
Forty patients were included in this cohort with ages ranging from 23 to 92 years old with a mean age of 65.9 years (standard deviation [SD] ± 14.2), of which 25 patients were female (62.5%) and 15 (37.5%) were male (Table 1). Depending on the procedure, the BMX®81 was advanced into the carotid artery in 29 cases (72.5%), into the vertebral artery in 10 cases (25%), and into the brachiocephalic artery in 1 case (2.5%).
Demographics and neurointerventional procedures.
Twenty (50%) of the cases were unruptured aneurysm embolizations, of which nine (22.5% of total) were managed with flow diverter stenting, two (5%) balloon-assisted coiling, three (7.5%) stent-assisted coiling and four (10%) WEB™ device deployments. One WEB™ device deployment for a basilar tip aneurysm was converted to femoral access, due to challenging anatomy to access the dominant right vertebral artery from a radial approach.
Nine patients (22.5% of total) presented with clinically significant carotid stenosis and were referred for endovascular treatment. Of these patients, six cases (15% of total) underwent carotid stenting: three (7.5%) were treated with right ICA-CCA stenting and angioplasty; two (5%) were treated with left ICA-CCA stenting and angioplasty; one (2.5%) was treated with left ICA stenting. Of the three (7.5%) aborted stenting procedures, one case (2.5%) was due to severe stenosis of the brachiocephalic artery, origin of the left CCA and the left subclavian artery, which prevented access. The other two patients (5%) had lower degree of stenosis on angiography compared to the cross-sectional work up and therefore stenting was not warranted. One patient in our series (2.5%) underwent brachiocephalic artery stenting due to severe stenosis, which required combined transradial and TFA.
In relation to acute stroke, three patients (7.5%) were treated for large vessel occlusions, two (5%) involving the right M1 MCA and 1 (2.5%) involving the basilar artery. These patients were successfully treated with aspiration technique. Two patients (5%) were treated for medium vessel occlusions. One of those patients had a right M2 MCA occlusion and was treated with one pass of combined stent retriever and aspiration technique. The other patient had a left P2–P3 PCA occlusion and was successfully treated with one pass of aspiration only.
With respect to dural AV fistulas, three patients (7.5%) had right-sided fistulas that were successfully treated transarterially with liquid EVOH embolization through the right MMA. One patient (2.5%) on anticoagulation was treated for a chronic subdural hematoma with MMA embolization. One patient (2.5%) was treated with right vertebral artery sacrifice due to Bow Hunter's syndrome with posterior circulation strokes.
In the 40 cases included in this cohort, there were no radial puncture or BMX®81 related complications. Particularly, no hemorrhage, ischemia, vascular injury, or catheter entrapment occurred. Of the 36 patients in which the procedure was performed, 31 patients (86.1%) had no technical difficulties and the procedures had satisfactory outcomes. Of the 5 patients with difficulties, one was related to coil tail prolapse into the parent artery, during the treatment of an unruptured aneurysm. The patient was placed on ASA and there were no further complications. In one case, the complication was due to thrombosis of a side branch at the conclusion of aneurysm coiling, which resolved after the administration of a bolus of a glycoprotein IIb/IIIa inhibitor integrilin (eptifbatide). In the remaining three cases, complications were reported because of technical difficulties deploying a flow diverter stent. One of these patients had a left paraophthalmic aneurysm with difficult stent deployment and developed a non-hemorrhagic left anterior choroidal territory infarct. Another patient required balloon angioplasty to achieve good flow diverter wall apposition. For the last patient, an initial flow diverter stent had difficulty correctly deploying and was exchanged for another stent, which was subsequently deployed without issue. None of these technical difficulties were considered secondary to the radial access puncture or support structure.
The BMX®81 catheters used were all straight tip catheters. Thirty-three (82.5%) were 95 cm in length, six (15%) were 105 cm, and one (2.5%) was 115 cm in length. In 26 cases (65%), the BMX®81 catheter was introduced through a radial 7 French Glidesheath Slender™ radial sheath. In 14 cases (35%), it was used as a primary sheath. All 40 cases were started as a right radial approach, but one case was immediately converted to a right ulnar approach due to anatomical variant and vasospasm.
With respect to access workflow, in the cases in which the guide catheter was positioned in the carotid artery, the time from puncture to the initial radial angiogram was between 3 and 9 minutes, with a mean time of 5.1 minutes (SD ± 1.7). The time from the first radial angiogram to the angiogram with the BMX®81 positioned in the target artery, was between 6 and 35 minutes with a mean time of 14.5 minutes (SD ± 7.0). The radiation dose (dose area product [DAP]) for these cases was between 2222 and 410,056 mGy/cm2 with an average of 84,221.5 mGy/cm2. The fluoroscopy time (FT) was between 3.86 and 149.5 minutes, with an average of 37.4 minutes. In the cases in which the guide catheter was positioned in the right vertebral artery, the time from puncture to radial angiogram was between 3 and 6 minutes with a mean time of 4.3 minutes (SD ± 1.17). The time from radial to target artery was between 4 and 27 minutes, with a mean time of 13.2 minutes (SD ± 6.76). There was no significant difference in time from radial to target artery between the carotid and vertebral arteries (p = .60). The DAP for these cases was between 5993 and 321,424 mGy/cm2 with an average of 98,467.9 mGy/cm2. The FT was between 10.06 and 83.24 minutes, with an average of 36.4 minutes.
Comparison group
40 patients were included in this cohort with ages ranging from 42 to 91 years old with a mean age of 70.1 years (SD ± 13.5), of which 19 patients were female (47.5%) and 21 (52.5%) were male.
Depending on the procedure, the guide catheters were positioned in a carotid artery in 25 cases (62.5%), in a vertebral artery in 14 cases (35%), and in the right subclavian artery at the vertebral artery origin in one case (2.5%).
The catheters used were the BENCHMARK™071 in 14 cases (35%), BENCHMARK™ BMX®96 in 24 cases (60%), Neuron MAX®088 in one case (2.5%) and BALLAST™ 088 in the remaining case (2.5%).
Fourteen patients (35%) were treated for aneurysms. Nine (22.5%) were unruptured, of which seven (17.5% of total) were managed with flow diverter stenting, and two (5%) with balloon-assisted coiling. Five patients (12.5%) with ruptured aneurysms were treated with balloon-assisted coiling. Four patients (10% of total) were treated for clinically significant carotid stenosis, two on the right side and two on the left side. They were treated with ICA-CCA angioplasty and stenting. In relation to acute stroke, sixteen patients (40%) were treated for large vessel occlusions, eleven (27.5%) involving the M1 MCA segment and five (12.5%) involving the basilar artery. Four patients (10%) were treated for medium vessel occlusions. With respect to dural AV fistulas, two patients (5%) had right-sided fistulas that were successfully treated transarterially with liquid EVOH embolization through the right MMA.
In the 40 cases included in this comparison group cohort, there were four (10%) access catheter-related complications. Three of these were related to radial sheath entrapment at the end of the procedure. This happened with the BENCHMARK™071, the BMX®96 and the BALLAST™ 088 (one time with each). These were treated with systemic vasodilators (nitroglycerin and verapamil) and in the case of the BMX®96, with periarterial nitroglycerin injection from the brachial to the radial artery. Two of these cases resulted in vessel injury, which subsequently required surgical management. In one case with the BMX®96, the patient was treated for a left MCA occlusion, with the access catheter positioned in the left ICA, which showed evidence of FMD. After the recanalization of the vessel, the left ICA developed severe, refractory vasospasm, which required ICA-CCA stenting and angioplasty. Two patients had complications that were not related to the access catheter. In both cases, the patients were being treated for medium vessel occlusions, there was perforation of the target artery, which required arterial sacrifice.
With respect to access workflow, in the cases in which the guide catheter was positioned in the carotid artery, the time from puncture to radial angiogram was between 4 and 19 minutes, with a mean time of 8.2 minutes (SD ± 3.9). The time from radial to target artery, was between 4 and 45 minutes with a mean time of 17.3 minutes (SD ± 10.7). The DAP for these cases was between 4408 and 375,392 mGy/cm2 with an average of 86,743.8 mGy/cm2. The FT was between 6.06 and 163.3 minutes, with an average of 37.0 minutes. In the cases in which the guide catheter was positioned in the right vertebral artery, the time from puncture to radial angiogram was between 4 and 16 minutes with a mean time of 7.4 minutes (SD ± 3.7). The time from radial to target artery was between 3 and 36 minutes, with a mean time of 18.4 minutes (SD ± 9.46). The DAP for these cases was between 16,208 and 286,412 mGy/cm2 with an average of 119,441.1 mGy/cm2. The FT was between 3.48 and 60.3 minutes, with an average of 24.0 minutes.
Discussion
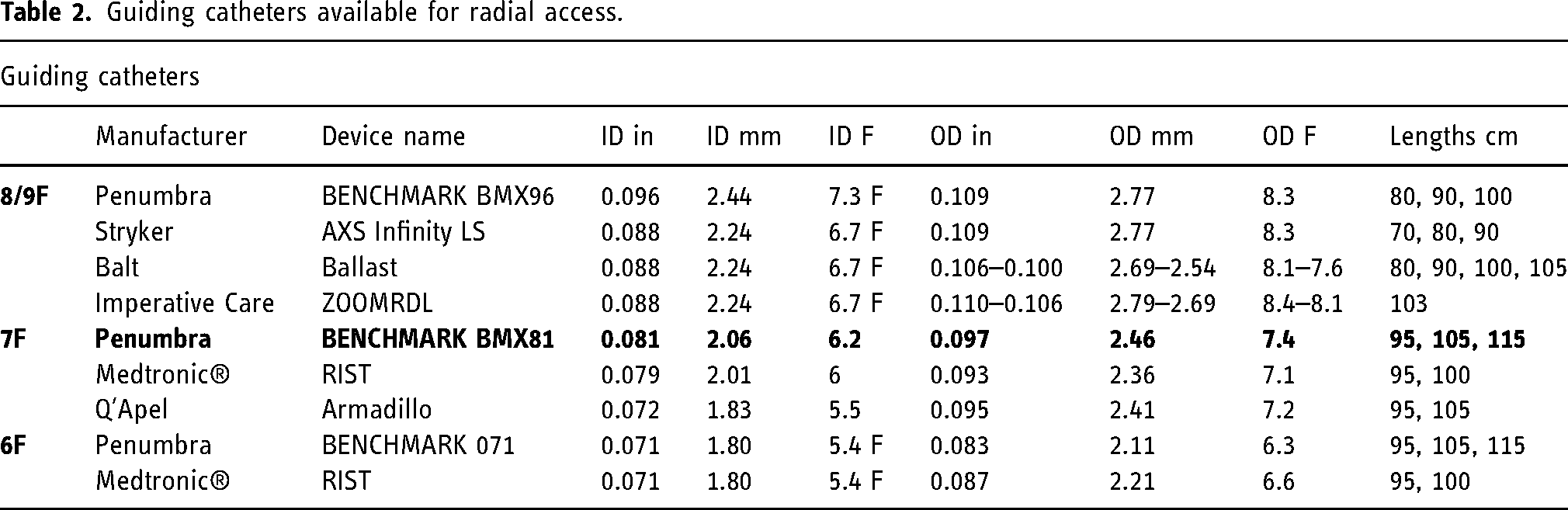
This case series highlights the use of the 7Fr guide BMX®81 catheter for TRA, allowing for safe and simple arterial navigation as well as satisfactory support for neurointervention. There are several alternative guiding catheters available that could be used for radial access (Table 2). The advantage of using an 8 or 9F guide, is that it supports larger bore 0.068–0.072-inch (ID) aspiration catheters. Additionally, these guides can even sometimes be used for treatment of extensive clot burden. 10 Complication rates associated with radial artery access (1–8%) are similar to what is reported for femoral artery access (2–8%) 11 although these can be typically <1% in high volume centers with experienced operators. 12 From literature and site experience, larger guides increase the risk for radial artery spasm (RAS) and subsequent guiding catheter entrapment, which are relatively common complications of radial neurointerventional approaches. RAS has been reported in up to 30% of the cases and can result in procedural delays and radial artery complications. In the scenario of RAS, it is important to follow a stepwise approach treatment algorithm 13 which ranges from minor intervention (such as administering intraarterial vasodilators), to more invasive interventions (such as injecting subcutaneous nitroglycerin along the catheter tract, brachial plexus block and surgical removal of the retained catheter). 14 In our radial access case series, there were two 8Fr guides (BMX®96 and Ballast™) and one 7Fr guide (Benchmark 071) resulting in radial sheath entrapment versus none in the BMX®81 group. Some guiding catheters have a larger OD proximally, to provide support, with tapering more distally (i.e., Ballast proximal OD is 0.106 inch and distally is 0.100 inch). The smaller profile of the BMX®81 can provide a crucial benefit in minimizing complications such as spasm or catheter entrapment. Additionally, hydrophilic coating on the guiding catheter theoretically would allow for easier advancement distally and reduced risk for RAS. In case of the BMX®81, the distal 18 cm consists of hydrophilic coating followed by a 16 cm distal flexible transition zone for atraumatic deliverability in tortuous anatomy (Figure 1). Finally, the one-piece design of the BMX®81 laser-cut stainless steel hypotube offers an advantage over traditional guide catheters by providing superior column strength and support. Distally, the design provides a construct with kink resistance, deliverability, and support. The OD is .097-inch (7.4 French), and internal diameter is 0.081-inch (6.2 French); it therefore is compatible with 7 French Glidesheath Slender™ radial sheaths and may be used with catheters up to 5 French. It is compatible with 0.062-inch (ID) aspiration catheters, although it does not support the larger bore 0.068–0.072-inch (ID) catheters. Our current cohort series demonstrates that a wide range of neurovascular interventions can be performed with the BMX®81, given that it can accommodate two microcatheters or a microcatheter and balloon catheter while also leaving ample area to perform angiograms (Figure 2). This shares one of the main advantages of using a larger 8 or 9F guide system and avoids the main disadvantage of using a smaller 6F guide system which could only accommodate one microcatheter. When a microcatheter and balloon catheter are inside BMX®81, the calculated available area for contrast injection is equivalent to that of a stand-alone catheter with .064-inch ID. The BMX®81 has the largest ID of all 7Fr radial access guiders (i.e., RIST (Medtronic, Minneapolis, MN, USA) has an ID of 0.079).
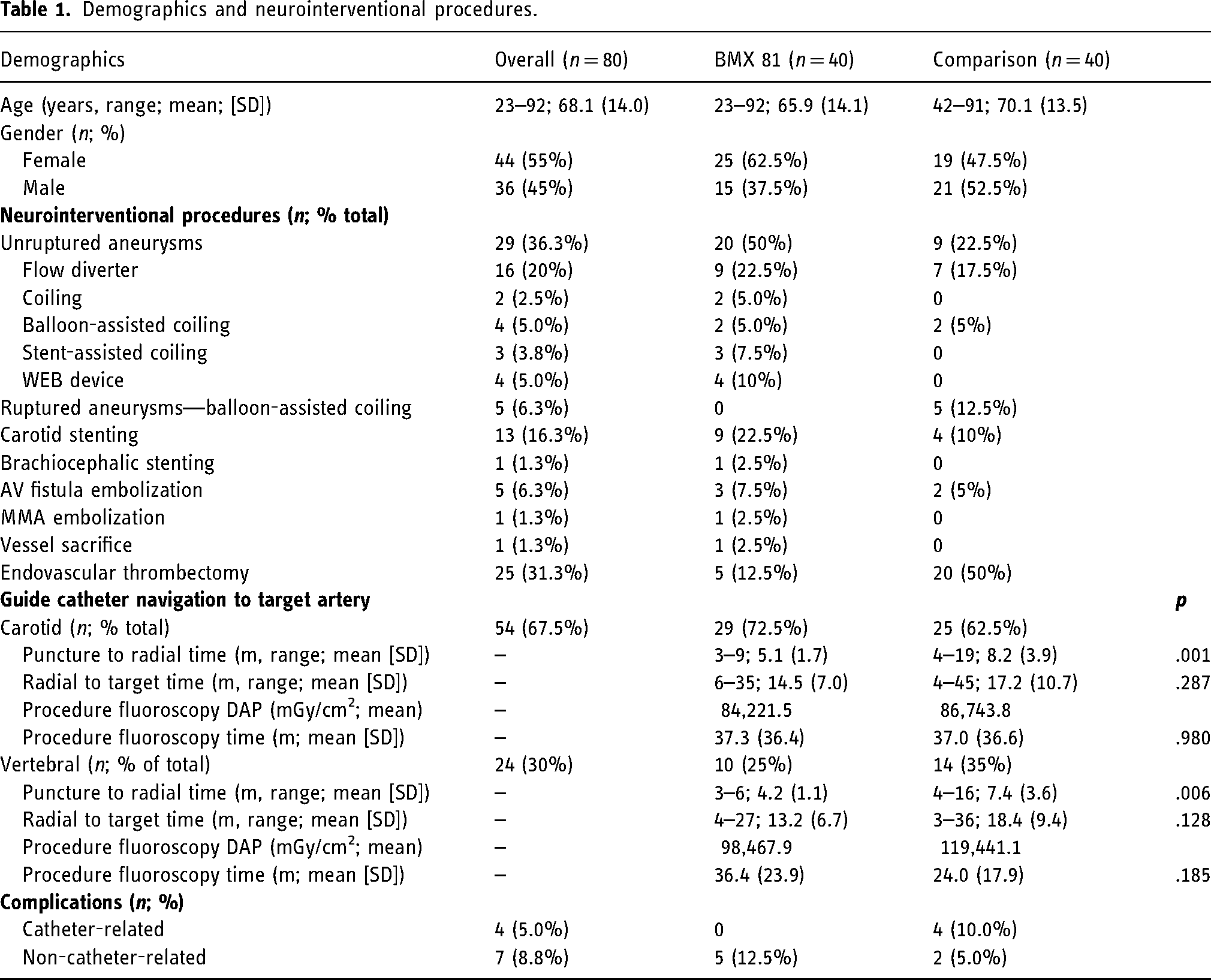
Ap fluoroscopy (A) and corresponding right common carotid artery roadmap (B) demonstrating how the BMX®81 guiding catheter successfully navigated a tortuous origin of the right common carotid artery from the brachiocephalic artery (white arrow) as well as a tortuous course of the proximal cervical right internal carotid artery (black arrow). From this support, the aspiration catheter was able to navigate to the occlusion in the M1 segment of the right MCA (arrowhead).
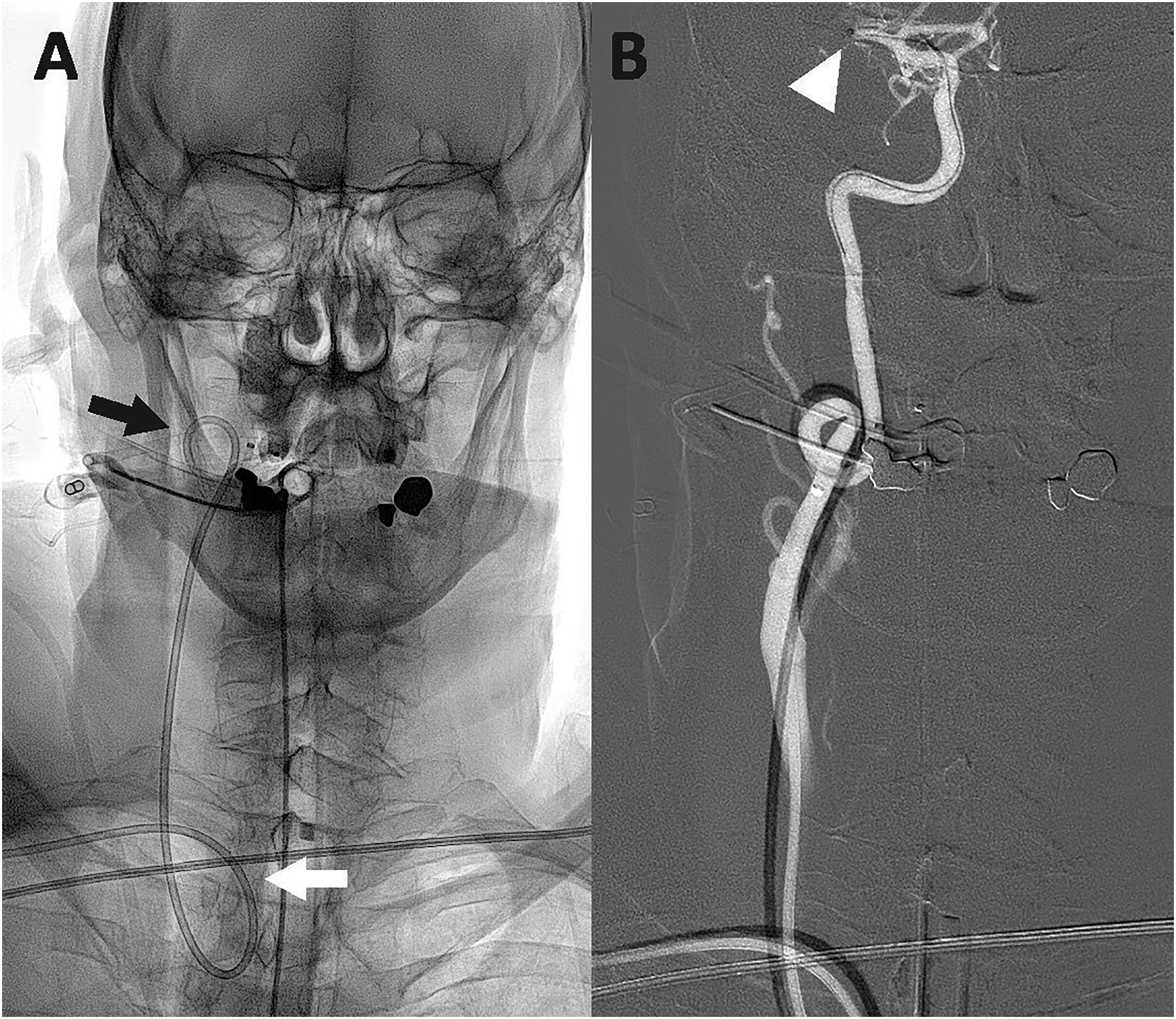
Inner and outer diameter representations (A) of the BMX®81 guiding catheter, a SL-10 coiling microcatheter (Stryker, Portage, MI, USA) and scepter C balloon microcatheter (MicroVention, Aliso Viejo, CA, USA). (B) During neurointerventional procedures, both the coiling microcatheter and balloon microcatheter can be advanced simultaneously through the BMX®81 guide catheter with a residual guiding catheter lumen equivalent to a 0.064-inch inner diameter, suitable for intraprocedural angiography.
Guiding catheters available for radial access.
The analysis of timing metrics showed that the time from puncture to successful radial access was lower in the BMX®81 group with respect to the comparison group (p = .001 for carotid and p = .006 for vertebral interventions). The time from radial access to accessing the target vessel was not statistically significant. Aside from the BMX®81 design, an additional explanation for the faster procedure times could be the increased site experience using the radial access. The overall procedure fluoroscopy times as well as the radiation doses were not significantly different between the groups, albeit there is significant heterogeneity of overall procedural durations given the wide scope of neurointerventional procedures included in this case series.
When the initial upper extremity angiogram is obtained, it is important to recognize anatomical variations like radial loops, which may be associated with a radial recurrent branch (Figure 3). Identifying this pattern is important because inadvertent catheterization of the recurrent branch instead of the main radial artery, can lead to severe vasospasm and consequent additional challenges, or procedural failure from a radial approach. 15 For access in our series, we placed a 7 French Glidesheath Slender™ radial sheath prior to advancing the BMX®81 sheath or used the BMX®81 as primary sheath. In all cases the initial assessment of the radial artery showed an adequate caliber for the catheters selected. When the radial artery is not large enough to support the selected catheters for treatment, the ulnar artery might be considered. This approach has been proven to be a feasible alternative when TRA and dRA are not possible. 16 In one case of our series, we identified a right radial artery loop. While navigating the catheter, severe vasospasm prevented the catheter from advancing. The radial access approach was then aborted, and we continued the procedure through a right ulnar approach, with no challenges or complications (Figure 3). The BMX®81 catheter can also be used as a primary sheath, therefore reducing the size of the access system, which is relevant in smaller radial arteries compared to the larger common femoral artery.
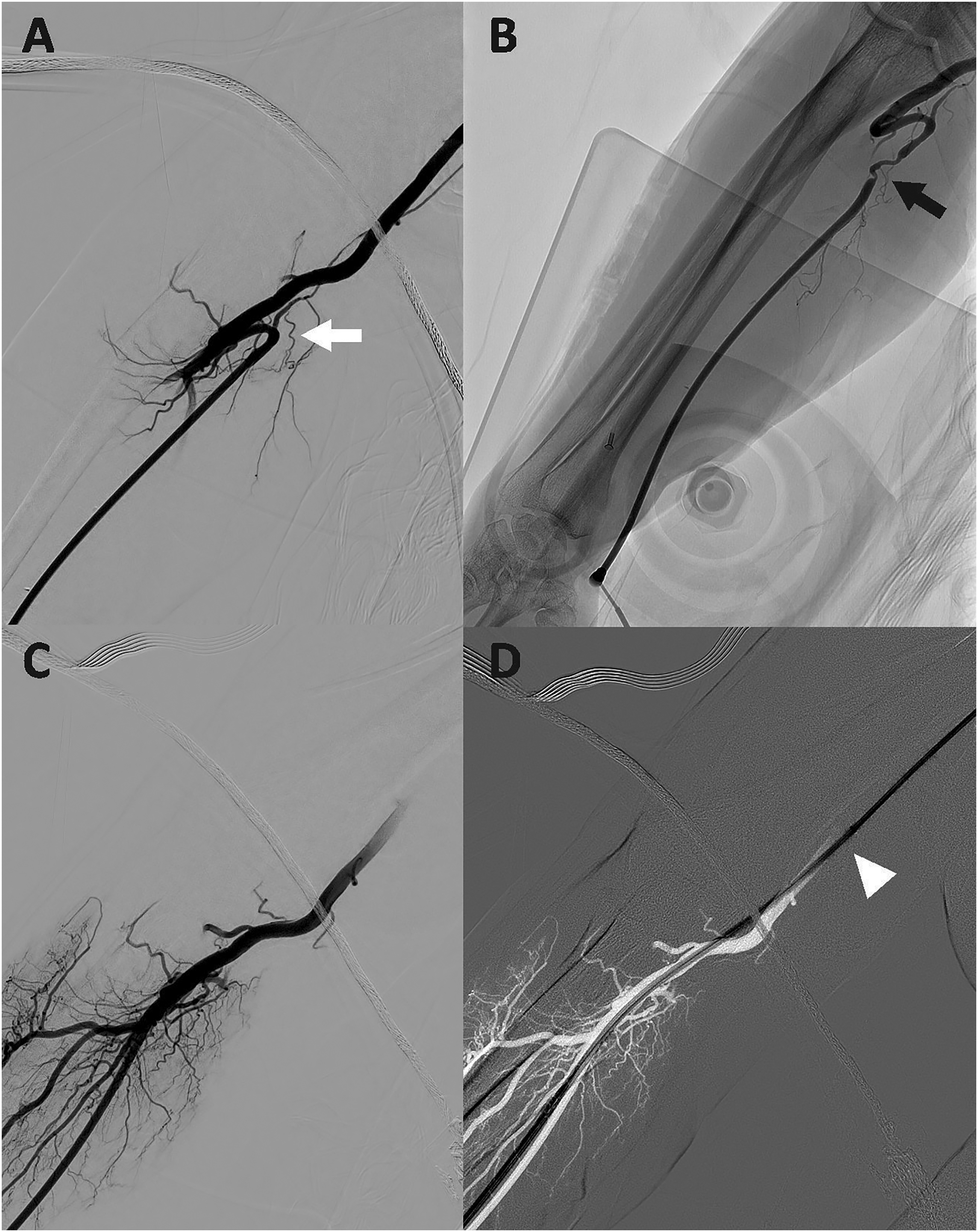
Right radial artery angiogram demonstrating a radial artery loop (A, white arrow) and subsequently vasospasm (B, black arrow) after attempted passing. Right ulnar artery angiogram (C) was performed, and the BMX®81 guiding catheter was successfully advanced to the brachial artery (D, arrowhead).
TRA has gained traction in the past decade for neurovascular interventions and in some centers, it has become the preferred route over TFA. Even for complex intracranial treatments, radial access has shown similar efficacy and safety profiles as femoral access, with no significant increase in procedural times. 17 Patients have a preference for radial access due to decreased pain, shorter recovery time, faster mobilization and quicker discharge. 12 TRA requires a technical skillset and repetition to recognize the potential anatomical challenges, and the ability to recognize and deal with potential complications. 11 Some authors propose a “radial first” approach for neurointervention, as it is for interventional cardiology. 12 dRA is also becoming more popular among neurointerventionists. 18 Since TRA is becoming the reality for many centers around the world, it is essential to train and become proficient in radial access to minimize technical limitations. Even if a center is not “radial first”, many times the patient's anatomy or vascular disease below the aortic arch, demands the neurointerventional team to find an alternative route, which is commonly the radial access. In order to improve access success and navigability, it is imperative that neurointerventionalists are properly trained and that industry innovation allows for better tools to complete the task. 3
When attempting radial access, it is important to perform an ultrasound examination of the wrist to evaluate the arterial anatomy, particularly, the size and characteristics of the radial artery. An artery with a diameter of less than 2 mm, is generally not acceptable when considering neurointerventional procedures, given the size of the sheaths and catheters required. 19 Assessing the ratio of the radial artery ID to the sheath OD can be beneficial, as a ratio less than 1.0 results in an increased incidence of severe flow reduction and occlusion. 20 dRA approach is a variant technique that is gaining popularity as the caliber of the distal radial artery in the anatomic snuffbox is only slightly smaller than in the distal forearm, and it typically does not require downsizing of sheath size. 21 Furthermore, dRA can reduce the rates of radial artery occlusion and access site hematoma compared with proximal radial access. It is also associated with short recovery times 22 and can provide a more ergonomic set up when left radial access is required thereby reducing exposure to radiation by the operator. The concerns with the dRA approach are related to lack of familiarity with the technique and uncertainty of complications associated with larger access catheters into the radial artery. Initial experience with dRA access for diagnostic angiograms and carotid stenting is promising but this approach would need to be confirmed in larger studies.3,23 Development of new devices designed specifically for wrist access may improve results with dRA. 19 Although no dRA was performed in this series, BMX®81 may be a suitable catheter for this approach.
In our series, the BMX®81 was successful in the navigation of the anatomy to the target location in 95% of cases from a radial approach. In one case we were unable to reach the target vessel and had to convert to a femoral approach. In another case we had to access the ulnar artery due to a radial artery loop with associated vasospasm. The radial approach provided suitable stability for various microcatheters, allowing for a wide range of procedures, including intracranial aneurysm treatment (Figure 4), stroke, dural AV fistula and MMA embolization. The BMX®81 can also safely be used in smaller vessels such as the vertebral artery as demonstrated by our case series. With an OD of 2.46 mm, the BMX®81 is with a 7Fr size situated between the larger 8Fr catheters with an OD of 2.77 mm and the smaller 6Fr catheters with an OD of 2.11 mm (Table 2). The improved navigability of this guide catheter allows for atraumatic access through markedly tortuous anatomy, for supporting intracranial interventions. The BMX®81 lumen allows for the delivery of the largest carotid stent sizes, while providing adequate support for their deployment (Figure 5). After gaining access to the radial artery (mean access time of 4.9 minutes), we had a mean time of 13.2 minutes from the first radial artery run to positioning the BMX®81 at its target vessel. In our series, we experienced no access or catheter-related complications, particularly no hemorrhage, significant vasospasm, or catheter entrapment. Although the number of cases in this series is small, the BMX®81 catheter performance shows that it is safe to use for radial access and can be used for a variety of neurovascular interventions.
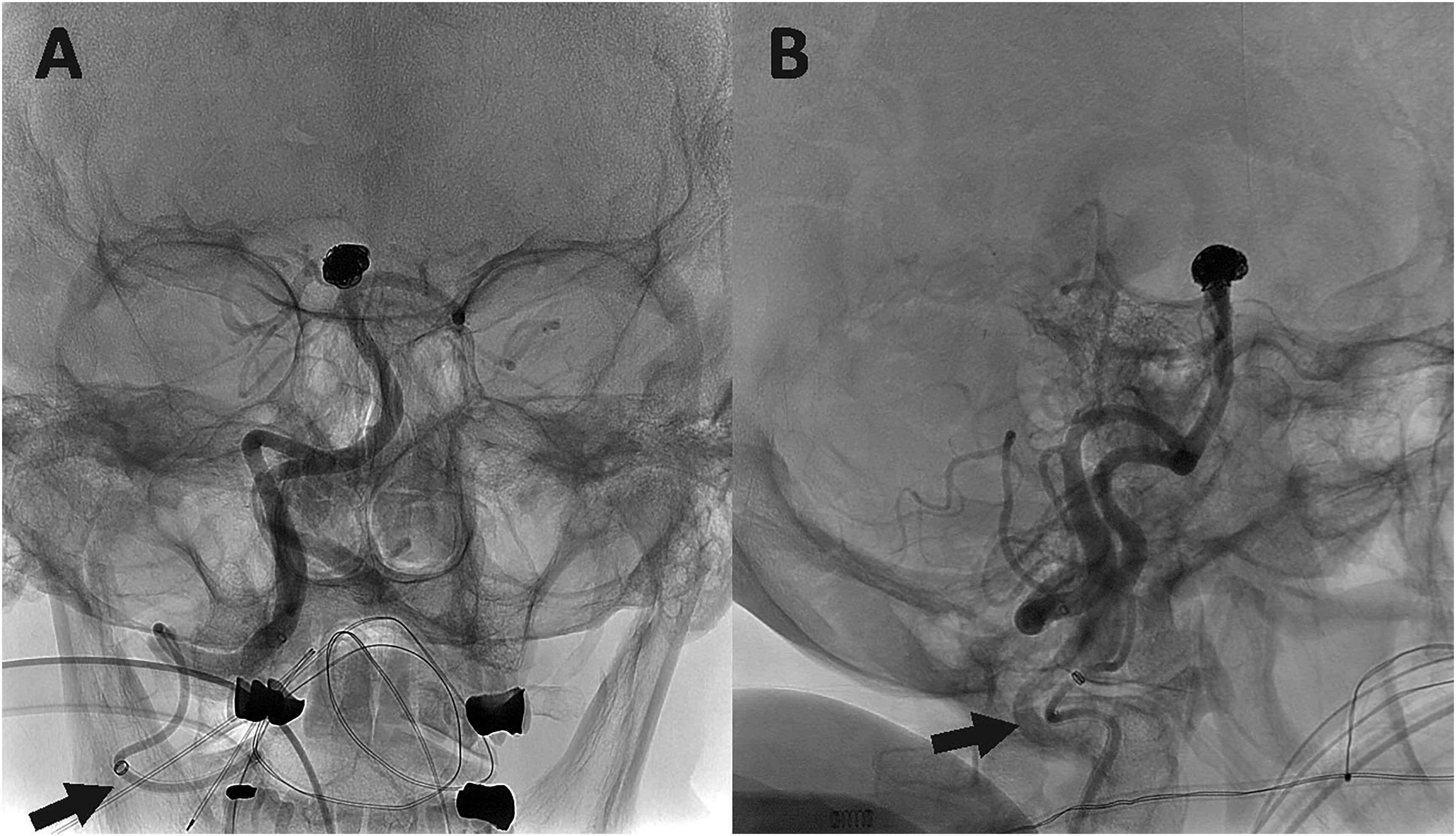
Right vertebral artery AP (A) and lateral (B) angiograms demonstrating the BMX®81 guide catheter navigated into the V3 segment of the right vertebral artery (black arrows), providing support for basilar tip aneurysm coiling.
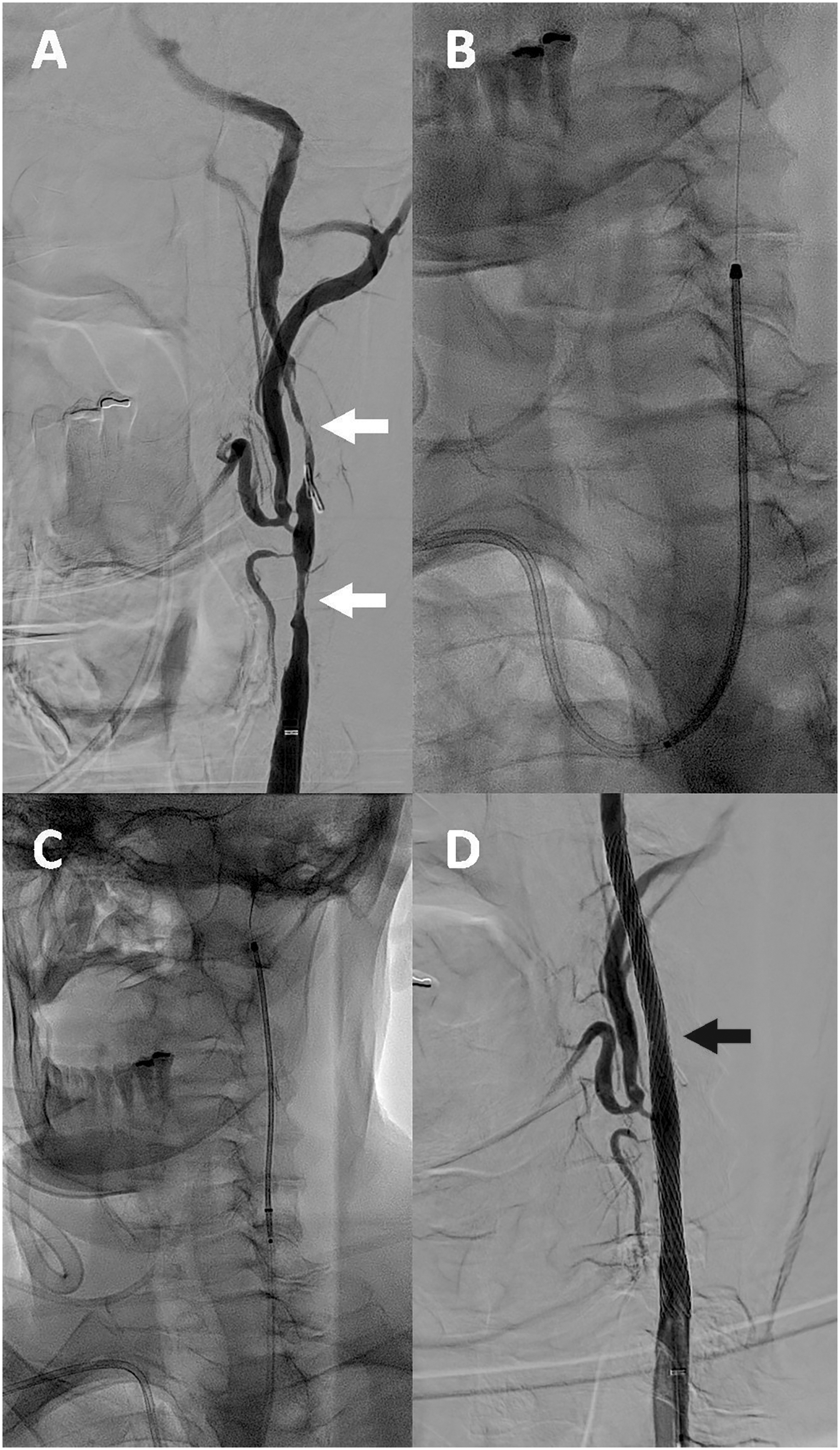
Left common/internal carotid artery AP angiogram in an 80-year-old patient 3 months after endarterectomy demonstrates long-segment left carotid stenosis and irregularity (A, white arrows). A 9 × 50 mm carotid Wallstent (Boston Scientific, Marlborough, MA, USA) was advanced through the BMX 81 guide catheter (B, C) and deployed in the target position with improved flow (D, black arrow).
Conclusion
The BENCHMARK™ BMX®81 System is a recently developed guiding catheter which has design and size features supporting radial access for a variety of neurovascular interventions. Early multicenter experience highlights the ease of use and versatility of this new catheter for first approach radial access in neurointervention as an alternative to other catheters used for radial access.
Supplemental Material
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DV is a consultant for Penumbra.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
