Abstract
Background
Endovascular treatment devices require compatible guide catheters to navigate complex vessels and anatomy. The Fubuki XF Long Sheath guide catheter (Fubuki XF) was developed with a 0.090-inch internal diameter with hydrophilic coating, an atraumatic rounded tip, and enhanced trackability and support with gradual shaft transition zones.
Methods
We retrospectively analyzed a prospectively maintained database of neuroendovascular patients treated using Fubuki XF at our center (July 2022─May 2023). Baseline/procedural characteristics were collected. Outcomes of interest included technical success (procedure completion with Fubuki XF without alternative guide catheter use) and peri-procedural complications.
Results
This study included 60 patients (43.3% [26/60] female; mean age: 69.6 ± 9.7) presenting with stenosis (45.0% [27/60]), unruptured aneurysms (31.7% [19/60]), ruptured aneurysm (1.7% [1/60]), arteriovenous fistula (5.0% [3/60]), arteriovenous malformation (3.3% [2/60]), chronic subdural hematoma (3.3% [2/60]), stroke/emboli (6.7% [4/60]), vasospasm (1.7% [1/60]), or carotid web (5.0% [1/60]). Fubuki XF was used to deliver endovascular treatment devices for stenting (43.3% [26/60]), flow diversion (23.3% [14/60]), embolization (11.7% [7/60]), coiling (10.0% [6/60]), balloon angioplasty (10.0% [6/60]), and mechanical thrombectomy (1.7% [1/60]). The Fubuki XF tip was placed in the internal carotid artery in 38.3% (23/60) of cases. Technical success was achieved in all cases. One V1 non-flow-limiting dissection (not related to Fubuki XF) and one failed closure occurred (1.7% [1/60] each). No iatrogenic strokes or intraprocedural ruptures occurred.
Conclusion
We used Fubuki XF to safely and effectively deliver a variety of compatible neuroendovascular devices. Fubuki XF was stable in all cases and locations, and there were no device-related complications or dissections.
Introduction
Long sheath guide catheters are used in a variety of endovascular procedures to treat neurovascular conditions, such as acute ischemic stroke (AIS),1,2 aneurysms,3,4 and stenosis.5,6 With the multitude of endovascular treatment devices used to treat these conditions, including stents, coils, liquid embolic agents, and flow diverters, guide catheters must be compatible and provide adequate support and flexibility when used in a variety of techniques such as CAPTIVE, 7 ARTS, 8 SAVE, 9 Solumbra, 10 and ADAPT. 10 Guide catheter flexibility and trackability can impact procedural time, patient safety, and clinical outcomes.11,12 The guide catheter tip shape can also affect its ability to navigate tortuous vasculature. 13 However, studies specifically assessing the safety and efficacy of guide catheters on patient outcomes are few and are typically inferred from studies assessing treatment devices. 14
The Fubuki XF Long Sheath guide catheter (Fubuki XF, Asahi Intecc, USA) is a flexible guide catheter with optimum pushability, a hydrophilic coating for smooth navigation, and a large lumen guide with a small puncture site. Here, we describe our experience with Fubuki XF used in a variety of endovascular procedures, including balloon angioplasty, coiling, flow diversion, stenting, and mechanical thrombectomy in patients with neurovascular conditions including aneurysms, AIS, intracranial stenosis, arteriovenous fistula (AVF), arteriovenous malformation (AVM), and carotid web.
Methods
Ethics
The data for this study was retrospectively analyzed from a prospectively maintained database with approval from our local institutional review board. Patients provided written informed consent to be included in the study. Consent for publication of un identifiable information was waived due to the retrospective nature of the study.
Patient selection
All patients who underwent neuroendovascular treatment using Fubuki XF at Valley Baptist Medical Center between July 2022 and May 2023 were selected from a prospectively maintained endovascular database.
Data collection
Baseline clinical characteristics were recorded, including demographic information, comorbidities, lesion characteristics, and target vessel location. Procedural characteristics of interest included procedure type, fluoroscopy time, operative room time, access location, and target treatment device. Procedural outcome data included technical success, defined as completion of the procedure with the use of Fubuki XF without using an alternative guide catheter. Safety outcome data included incidence of dissection, iatrogenic stroke, or intraprocedural rupture.
Fubuki XF device
Fubuki XF is a flexible guide catheter with an atraumatic straight or angled tip and flexible distal shaft with gradual transition zones to minimize vascular injuries. A well-balanced proximal shaft with a tungsten and stainless-steel braid to increase rigidity enables enhanced distal trackability and optimum pushability. A hydrophilic coating and resin allow smooth navigation and quick distal placement with a radiopaque marker and visible shaft. Visibility is improved with radiopaque markers on the tip and shaft. Additionally, Fubuki XF is a 6Fr long sheath and has a large 0.090-inch inner diameter allowing additional space for devices, with 80-, 90-, 100-, and 110-cm working lengths. All procedures in this study used the 90-cm length Fubuki XF.
Endovascular procedures
General anesthesia was used for all embolization procedures, while conscious sedation was used for mechanical thrombectomy and carotid stenting. Appropriate systemic anticoagulation with heparin was administered in all embolization cases. Radial cocktail was administered in all radial access cases, including heparin, nitroglycerine, and verapamil. Antiplatelet boluses with Brilinta and/or Aggrastat were given during the procedure when needed.
Mechanical thrombectomy
The Solumbra approach 15 was used to cross the lesion and included the Fubuki XF 0.090, a distal access catheter (DAC)/aspiration catheter, and microcatheter. Fubuki XF was navigated into the vertical petrous or proximal V3 vertebral segments, followed by deployment of a stent retriever for 5–7 min, which was then pulled back under aspiration along with the DAC. Stroke patients did not receive heparin; however, 2000 units/bag were connected to flush lines, and GP2b/3a inhibitors were not used. After 60 min of intervention time, the procedure was terminated.
Carotid stenting
Fluoroscopic guidance was used during cannulation and mapping of the common carotid artery (CCA) with a triaxial catheter system and Fubuki XF. Both the guide wire and guide catheter were advanced into the distal CCA. A distal embolic protection device was inserted into the distal cervical segment of the internal carotid artery (ICA) after which the device was deployed and the delivery system was removed. To pre-dilate the lesion, a small balloon was inserted and inflated, followed by stent deployment. Post-dilation was performed and the distal embolic protection device was retrieved. If the procedure was successful, the catheters and sheaths were taken out, and an arteriotomy closure device was utilized to close the site of the incision.
Liquid embolization
Under fluoroscopic guidance, liquid embolic agents were introduced using a triaxial catheter system with Fubuki XF. Upon contact with blood, the agent precipitated into an embolus in situ and catheters were removed. The duration of the injection was approximately 30 min.
Flow diversion
Flow diverters were inserted into the parent artery through a microcatheter using a triaxial guide catheter system with the Fubuki XF. The entire process was guided by fluoroscopy with additional computed tomography angiography employed at the surgeon's discretion. If successful, the delivery catheter was removed and the procedure was completed.
Endovascular coiling
A triaxial catheter system with Fubuki XF for proximal support was employed to guide the insertion of a detachable platinum coil into the aneurysm under fluoroscopic guidance. Once the coil was positioned inside the aneurysm, it detached from the catheter, effectively blocking blood flow to the aneurysm. If required, multiple coils were utilized.
Balloon angioplasty
Under fluoroscopic guidance, Fubuki XF was used to advance a balloon guide catheter into the target vessel. The balloon was then inflated at the site of the blockage for intervals of 30–60 s at a pressure of 4–6 atm, until recanalization of the vessel was achieved. When flow was restored to the target vessel, the procedure was terminated and the introducer sheath was removed.
Statistical analysis
The statistics for a specific metric were calculated based on the set of patients for whom the metric was available. Categorical data are presented as n/N (%) for each category. For symmetrical distributions, continuous data are reported as mean ± standard deviation (SD) or median ± interquartile range (IQR). Numerical data are reported as mean ± SD or n (%) as appropriate.
Results
Patient characteristics
A total of 60 patients (26/60 [43.3%] female) were included in this study. Table 1 presents patient baseline and lesion characteristics. Mean patient age was 69.6 ± 9.7 years. The most common comorbidities were hypertension (42/60 [70.0%]), diabetes mellitus (39/60 [65.0%]), and hypercholesterolemia (27/60 [45.0%]). A total of 27 patients (45.0%) presented with stenosis, 19 (31.7%) presented with unruptured aneurysms, 1 (1.7%) presented with a ruptured aneurysm, 3 (5.0%) with AVF, 2 (3.3%) with AVM, 2 (3.3%) with chronic subdural hematoma, 4 (6.7%) with stroke or emboli, 1 (1.7%) with vasospasm, and 1 with carotid web (5.0%). A total of 40.0% (24/60) patients had one vessel treated, while 60.0% (36/60) had more than one vessel treated with a total of 173 lesions among the 60 patients. Most lesions were located in the anterior circulation (99/173 [57.2%]), with the remaining lesions in the posterior circulation (74/173 [42.8%]). Lesion locations included the CCA (34/173 [19.7%]), ICA (55/173 [31.8%]), vertebral artery (VA) (43/173 [24.9%]), external carotid artery (ECA) (10/173 [5.8%]), and the superior cerebellar artery (SCA) (31/173 [17.9%]).
Patient and lesion characteristics.

Data are mean ± SD or n (%). SD: standard deviation; SAH: subarachnoid hemorrhage; ICH: intracerebral hemorrhage; AVF: arteriovenous fistula; AVM: arteriovenous malformation; TICI: thrombolysis in cerebral infarction; CCA: common carotid artery; ICA: internal carotid artery; VA: vertebral artery; ECA: external carotid artery; SCA: superior cerebellar artery.
Procedure characteristics
Procedural characteristics are summarized in Table 2. A total of 43.3% (26/60) patients underwent extracranial or intracranial stenting, while 23.3% (14/60) had flow diversion, 11.7% (7/60) liquid embolization, 10.0% (6/60) coiling, 10.0% (6/60) balloon angioplasty alone, and 1.7% (1/60) mechanical thrombectomy. The Fubuki XF tip reached the target vessel in all cases, which was the CCA in 41.7% (25/60), ICA in 38.3% (23/60), VA in 16.7% (10/60), ECA in 1.7% (1/60), and transverse sinus in 1.7% (1/60) of cases. In the ICA, Fubuki XF reached the distal cervical segment in 43.5% (10/23), petrous segment in 34.8% (8/23), and proximal cervical segment in 21.7% (5/23) of cases. Median fluoroscopy time was 26 ± 20.4 min.
Procedure characteristics.

Data are median ± IQR or n (%). CCA: common carotid artery; ICA: internal carotid artery; VA: vertebral artery; ECA: external carotid artery; IQR: interquartile range.
Technical success was achieved in 100% of patients. One (1/60 [1.7%]) V1 non-flow-limiting dissection occurred in a patient who underwent balloon-assisted coiling for an aneurysm, which was not caused by Fubuki XF. One failed closure occurred (1/60 [1.7%]) in a patient who underwent flow diversion with Pipeline Shield. There were no instances of iatrogenic stroke or intraprocedural rupture.
Illustrative cases
Case 1: angioplasty of distal cervical ICA
A patient in their 60s was referred for symptomatic left supraclinoid recurrent stenosis. During the angioplasty procedure, vessel tortuosity was encountered. Fubuki XF was placed in the distal cervical ICA providing adequate support and enabling a successful angioplasty procedure (Figure 1(a)). There was no evidence of post-procedure dissection or spasm.
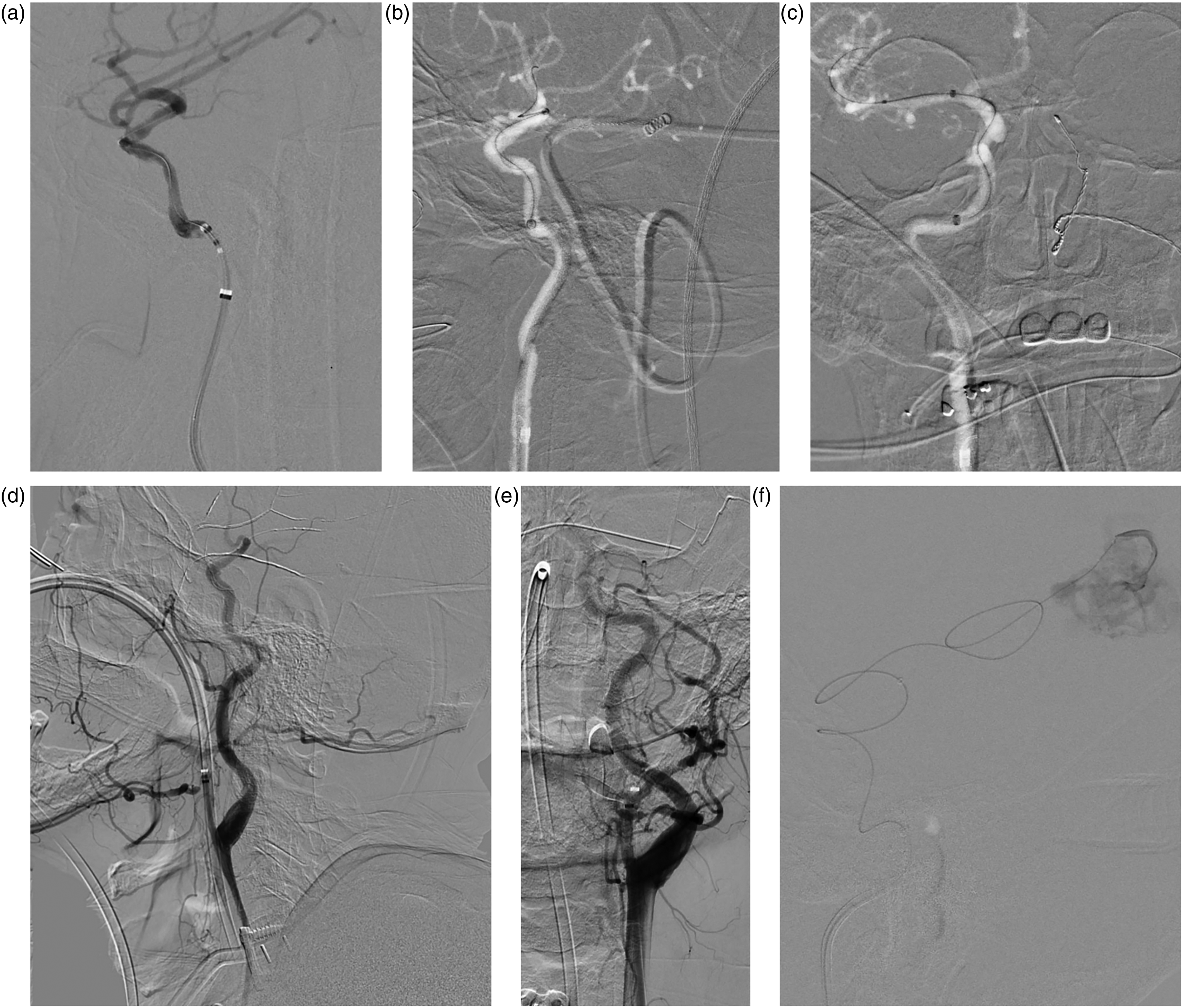
Illustrative cases using Fubuki XF. (a) Fubuki XF parked in distal cervical ICA during ICAD angioplasty with no evidence of post-procedural spasm. (b, c) Fubuki XF placed in horizontal petrous of the ICA during Pipeline deployment without evidence of post-procedural spasm. (d, e) Fubuki XF parked in proximal occipital artery branch of external carotid artery during AVM embolization. (f) Fubuki XF placed in proximal petrous of the ICA during AVM embolization. AVM: arteriovenous malformation; ICA: internal carotid artery; ICAD: intracranial atherosclerotic disease.
Case 2: flow diversion in horizontal petrous segment of the ICA
A patient in their 60s was referred for a subarachnoid hemorrhage with a small right paraclinoid aneurysm measuring 5.8 × 4.7 × 5.1 mm. We performed embolization with flow diversion using Pipeline Shield (Figure 1(b) and (c)). Fubuki XF was placed in the horizontal petrous segment of the ICA providing excellent support with an intermediate catheter and enabling successful deployment of the flow diverter. There was no evidence of dissection or spasm post-procedure.
Case 3: embolization of AVM in ECA
A patient in their 50s presented with a left temporal occipital AVM with a Spetzler-Martin grade IV and feeder arteries in the occipital branch of the ECA. Fubuki XF easily navigated to the ECA and was placed into the proximal portion of the occipital artery (Figure 1(d) and (e)). Embolization was successful without evidence of vasospasm or dissection.
Case 4: embolization of ICA
A patient in their 60s presented with uncontrolled seizures and a left parietal AVM (37 × 29 mm) with multiple feeders from left ACA, MCA, and PCA with deep and superficial drainage with a Spetzler-Martin grade IV. Fubuki XF easily navigated to the horizontal petrous portion of the ICA with an intermediate catheter (Figure 1(f)), and embolization was performed successfully using Scepter XC and Onyx 18. There was no evidence of dissection or spasm post-procedure.
Discussion
In this report, we describe our experience with the Fubuki XF Long Sheath guide catheter used in a variety of endovascular treatment procedures, including balloon angioplasty, coiling, flow diversion, stenting, and mechanical thrombectomy in patients with neurovascular conditions, such as aneurysms, intracranial stenosis, AVF, AVM, and carotid web. The Fubuki XF was successfully navigated to the lesions in a variety of locations in the anterior and posterior circulation, with low complication rates.
In a 2020 case series from the Mayo Clinic, Rinaldo et al. 16 assessed the safety and efficacy of the original Fubuki guide catheter used in 154 mechanical thrombectomy patients, with access site complications including one pseudoaneurysm, persistent bleeding at femoral access site, and limb ischemia. Fubuki reached the target vessel in 98.1% (151/154) cases and crossed the lesion in 97.4% (150/154) cases, with predictors of guide catheter failure significantly associated with a type III aortic arch. 16 In a 2019 retrospective analysis, Sakamoto et al. 17 assessed the safety and efficacy of a triaxial catheter system that used Fubuki and Axcelguide in 27 CAS patients and reported no peri-procedural complications. Technical success, defined as successful navigation of a 6-French guiding sheath into the CCA of the target side using a triaxial catheter system and successful stent deployment, was achieved in all patients (100% [27/27]). 17 In a 2021 retrospective review of prospectively collected data from the Neurological Intervention and Imaging Service of Western Australia, Phillips et al. 18 assessed the use of Fubuki and other long sheath guide catheters for radial access in 130 mechanical thrombectomy procedures with a 90-day mRS ≤ 2 rate of 67.2% and final eTICI 2b/3 rate of 94.6%. Radial artery occlusion occurred in 2.3% (3/130) of patients with no reports of access site complications. 18
Complications related to other commercially available guide catheters are most frequently reported in studies primarily assessing the safety and efficacy of other treatment devices. Bageac et al. 19 assessed safety and efficacy of several 0.088-inch ID guide catheters (Neuron MAX, Ballast 088 Long Sheath, AXS Infinity LS PLUS, Walrus Balloon Guide Catheter, FlowGate2 Balloon Guide Catheter, 8Fr Flexor Shuttle Sheath) in AIS patients who underwent thrombectomy. Complications related to guide catheters included groin hematomas (2.2% [1/45]), vessel dissection (4.4% [2/45]), and flow-limiting vasospasm (2.2% [1/45]). 19 Guide catheter failure, defined as failure to maintain stable positioning within the cervical ICA or use of an alternative guide catheter at any point after initial access was attempted, occurred in 13.3% (6/45) of patients. 19 Dmytriw et al. 20 retrospectively assessed the safety and efficacy of various guide catheters (Flexor Shuttle Sheath, Penumbra Benchmark intracranial access catheter, Penumbra Neuron Max sheath, Flexor Shuttle sheath, and Microvention Chaperon guiding catheter) used in a variety of procedures in pediatric patients (embolization [13/27], intra-arterial chemotherapy [10/27], mechanical thrombectomy [3/27], and diagnostic angiography [1/27]). The technical success rate was 93% (25/27), with the two unsuccessful procedures in patients who underwent intra-arterial chemotherapy where a vascular route could not be accessed, 20 and one sheath-induced vasospasm (3.7%) occurred. 20
A summary of the features, success rates, limitations, and complications of long sheath guide catheters are presented in Table 3. Difficulties experienced when using guide catheters have included catheter prolapse and difficulty navigating past kinks and through tortuous anatomy, 12 although these were not experienced in our series.
Competing long sheath guide catheters.
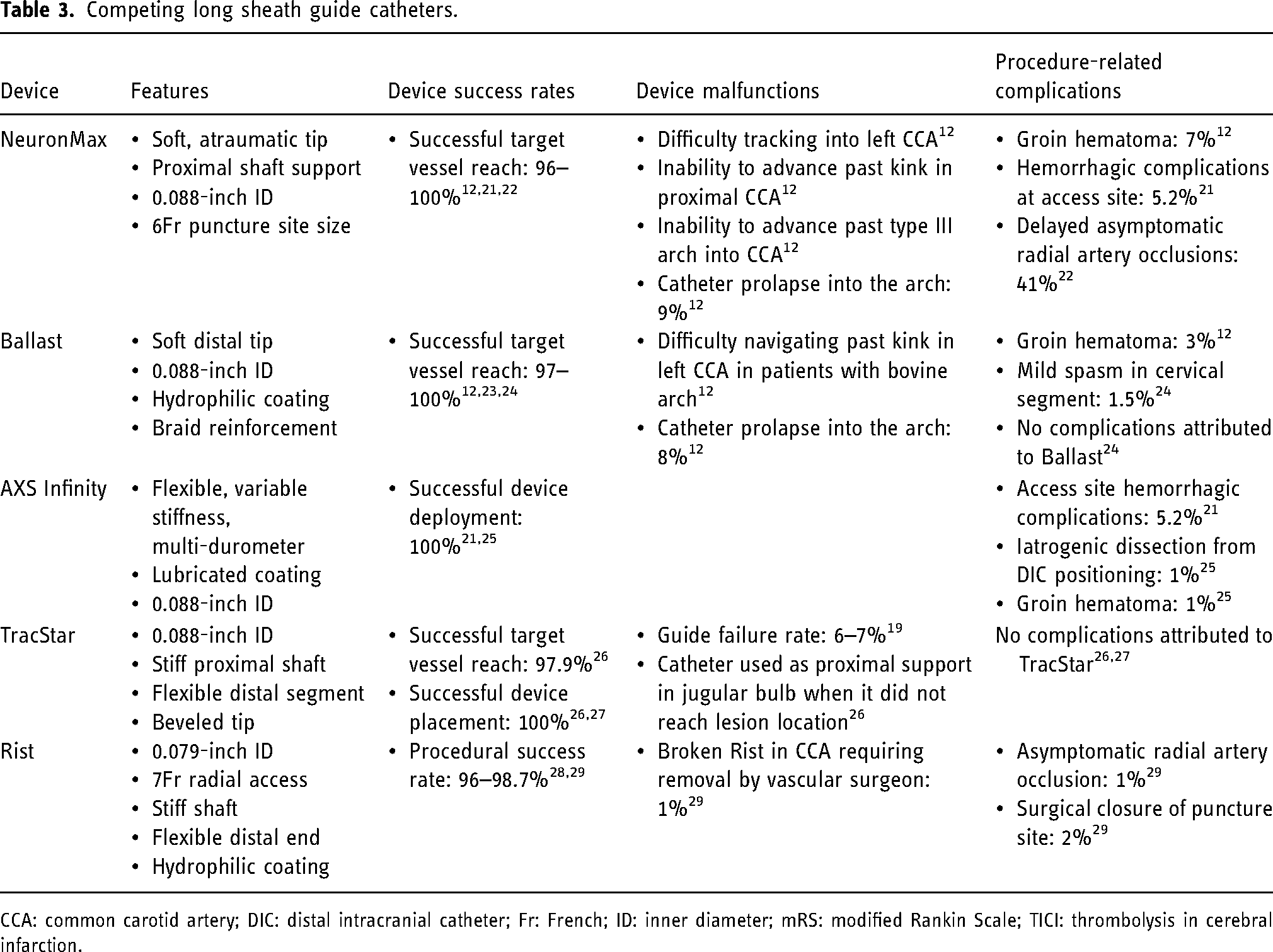
CCA: common carotid artery; DIC: distal intracranial catheter; Fr: French; ID: inner diameter; mRS: modified Rankin Scale; TICI: thrombolysis in cerebral infarction.
We experienced no instances of iatrogenic stroke or intraprocedural rupture in patients, one failed closure, and one non-flow-limiting dissection not caused by Fubuki XF. Reports of complications related to other guide catheters included groin hematomas (1–7%),12,19,25 delayed asymptomatic radial artery occlusion (41%), 22 vessel dissection (1–4.4%),19,25 hemorrhagic access site complications (5.2%), 21 mild spasm in cervical segment (1.5%), 24 surgical closure of puncture site (2%), 29 sheath-induced vasospasm (3.7%), 20 and flow-limiting vasospasm (2.2%) 19 (Table 3). In our experience, Fubuki XF has good balance between tractability and support, which played a role in the good outcomes achieved in this series.
Limitations
Limitations of this study include the lack of a comparator group, the retrospective nature of the study, and small sample size; however, our experience with this new guide catheter demonstrates early clinical efficacy. Additional analyses are needed to compare the outcomes of Fubuki XF to the other guide catheters.
Conclusion
This study demonstrates the compatibility, safety, and efficacy of the Fubuki XF Long Sheath guide catheter with a variety of endovascular devices used in stenting, flow diversion, liquid embolization, coiling, balloon angioplasty, and mechanical thrombectomy in patients with various neurovascular indications in both the anterior and posterior circulations. Fubuki XF was successfully navigated to the target vessel in all patients with no complications or dissections related to the device.
Footnotes
Acknowledgements
The authors acknowledge Superior Medical Experts for drafting assistance.
Author contributions
HS and AEH contributed to the design of the work and the acquisition, analysis, and interpretation of data, made critical revisions to the manuscript, and agree to be accountable for all aspects of the work. SM contributed to the design of the work and the acquisition, analysis, and interpretation of data. HP contributed to data analysis, manuscript preparation, and critical revisions. All authors gave final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AEH serves as a consultant and speaker for GE Healthcare, Medtronic, Stryker, Microvention, Penumbra, Balt USA, and Genentech. HP is employed by Superior Medical Experts. HS and SM have no discloses to declare.
Ethics approval
This study was reviewed and approved by the MetroWest institutional review board (IRB, number 2017–149). The patients provided their written informed consent to participate in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this study and manuscript development was provided by Asahi Intecc USA, Inc.
