Abstract
Background
Endovascular thrombectomy (EVT) is standard of care for acute ischemic stroke. Stent assisted EVT with aspiration (SOLUMBRA) technique has remained a mainstay approach. There is growing evidence that A Direct Aspiration First Pass Technique (ADAPT) is a safe, efficient and effective approach for EVT, offering several advantages. This study describes and reports initial institutional experience in the use of a standardized scientific based aspiration only technique: CANADAPT.
Methods
Single center prospective cohort study was performed on consecutive patients treated for large/medium vessel ischemic stroke with CANADAPT. Intravenous thrombolytics were administered according to routine practice, independent of the decision to proceed with EVT. A sequential stepwise aspiration only technique was then applied, CANADAPT, consisting of three maneuvers, A, B and C. The reperfusion success rate, number of passes, use of rescue technique, complication rate and procedural cost were determined.
Results
Twenty-two patients were included in this case series representing M1 (17, 77%), M1/2 (2, 9%), carotid-T (2, 9%) and basilar (1, 5%) occlusions. First pass recanalization was achieved in 11 (50%) of patients. A further four patients had successful reperfusion with a second pass of CANADAPT (total 68% success rate). Only one patient had successful reperfusion with the aspiration catheter at the clot interface (CANADAPT A). All others required some withdrawal of the aspiration catheter for reperfusion (CANADAPT B and C). Seven patients had SOLUMBRA rescue. Of these, five patients (22% of total patients) had further successful reperfusion. Overall median procedural time was 23 min for first recanalization and 30 min for final recanalization. The cost per procedure was $6630 ± 1069 for CANADAPT, and $13,530 ± 2706 for SOLUMBRA techniques.
Conclusions
CANADAPT represents a standardized scientific-based approach to aspiration only thrombectomy intervention. This initial study demonstrates the safety, efficiency and efficacy of this technique for use in EVT.
Introduction
Endovascular thrombectomy (EVT) is the standard of care for large vessel intracranial occlusions in the setting of acute ischemic stroke, 1 demonstrating a profound treatment advance in those patients facing an otherwise devastating clinical outcome. Furthermore, these interventions are increasingly employed with medium vessel ischemic strokes, an ongoing area of investigation. 2 Despite this significant advance in treatment of ischemic stroke, there remains considerable heterogeneity of interventional approaches and techniques employed for EVT. Within this context, stent assisted EVT or stent with concomitant use of aspiration catheter (SOLUMBRA) techniques have remained mainstay approaches in these patients. There has however been growing evidence that A Direct Aspiration first Pass Technique (ADAPT), without employing a stent, is a safe, efficient and effective approach for thrombectomy, offering several advantages over stent only and SOLUMBRA interventions. Initial ADAPT trials have demonstrated non-inferiority of clinical outcomes compared to traditional stent retriever approaches.3–6 Recent advances in techniques and dedicated aspiration catheter systems for this purpose have further advanced the attractiveness of ADAPT in first pass approaches.
Within this context, increasing inner diameter lumen, manoeuvrability and conformability of aspiration catheters 7 in conjunction with increasingly robust continuous aspiration pumps, affords greater potential for ADAPT in acute ischemic stroke. Aspiration catheters are specifically designed to safely navigate the neurovascular system to deliver the largest possible inner diameter lumen to the interface of a thrombus in large and medium size intracranial arteries for use with aspiration thrombectomy. These advances provide several potential benefits compared to stent assisted interventions, such as reduced procedural time, 8 improved revascularization rates 9 and clinical outcomes 4 as well as reduced intervention costs. 10 However, even among ADAPT interventions, there is not a consensus on an optimal approach and many aspects of the procedure are left to the discretion of individual interventionalists. This includes choices of type and size of aspiration catheters, use of continuous pump aspiration, positioning of the aspiration catheter with respect to the thrombus (close proximity, at the interface, ‘bump’ techniques), time for clot engagement and catheter positioning/methodology when withdrawing the aspiration catheter. Varying these factors likely have a substantial influence on the efficacy and effectiveness of ADAPT, including first pass success, reperfusion/clinical outcome, need for alternate interventions such as SOLUMBRA, procedural time and cost of interventions. Furthermore, there is a general paucity of literature and guidelines with respect to EVT methodology thus, further limiting opportunities to provide optimal standard protocols.
We describe a specific ADAPT technique for use in EVT that represents a paradigm shift at our institution. Our initial institutional experience with an interventional technique employing stepwise standardized physics-based catheter position/technique during aspiration first EVT is detailed, termed CANADAPT. The effectiveness and efficacy of using CANADAPT as a first pass technique in acute ischemic stroke is highlighted in our initial patient population with discussion of the relative theory and physics.
Methods
Patient selection
A single center prospective cohort study was performed on consecutive patients treated for large/medium vessel ischemic stroke with CANADAPT from March to October 2023. Potential patients were identified through a comprehensive assessment by stroke neurology and neuroradiology. Briefly, upon activation of the acute stroke protocol, patients were assessed by the stroke neurology team who assigned an NIHSS and clarified baseline status and symptom onset. Concurrent standard imaging protocol consisted of an unenhanced CT head to rule out intracranial hemorrhage and determine ASPECT score, a multiphase CT angiogram to determine occlusion site and quality of collateral arterial supply to the affected territory as well as cerebral perfusion imaging to identify degree of core infarct and salvageable penumbra. Each case was considered by the neurointerventionalist and stroke neurologist on call for thrombectomy candidacy. This generally included clinical assessment of deficits, ASPECTS score, collaterals, mismatch on perfusion or delayed CT angiogram to indicate ischemic penumbra as well as vascular access assessment. Intravenous thrombolytics were administered as deemed appropriate by the stroke neurology service and this was independent of the decision to proceed with endovascular thrombectomy. Patients that underwent thrombectomy were excluded from this case series if they had a chronic occlusion (carotid and/or intracranial) or if they had tandem occlusions.
CANADAPT – endovascular thrombectomy technique
CANADAPT was performed in our study by a team of four neurointerventional staff radiologists along with three neuroradiology fellows. Once the decision to proceed with endovascular thrombectomy was made and after informed consent, the patient was urgently transferred to the neurointerventional suite and positioned supine on the table. The patient was given general anesthesia or conscious sedation at the discretion of the anesthesiology and neuroradiology teams. Using a common femoral artery approach, an 8 Fr short sheath or 70 cm Raabe sheath was inserted. A 8 French 90 cm BMX 96 guide catheter (or NeuronMax, Penumbra, Inc.), 6 French 125 cm Berenstein or Simmons select catheter (Merit Medical, South Jordan, USA), and a 0.035 inch 180 cm Terumo Glidewire Advantage (Terumo Medical Canada, Inc., Vaughan, Canada) were advanced to the aortic arch. The guide catheter was positioned within the ipsilateral distal cervical or petrous internal carotid artery before withdrawing the select catheter and Glidewire. At this point, biplane cerebral angiography through the guide catheter was performed to assess the occlusion and neurovasculature.
A coaxial system of a RED 62, 68, or 72 aspiration catheter (Penumbra, Inc.), Phenom 21 (Medtronic, Minneapolis, MN, or Velocity [Penumbra, Inc.]) microcatheter, and Traxcess 14 EX microwire (MicroVention, Aliso Viejo, USA) were advanced with the aspiration catheter to the clot interface before removal of the microwire and microcatheter. Care was taken to not traverse the thrombus with the microcatheter/microwire during this step. Selection of aspiration catheter was at the discretion of the treating neurointerventionalist and generally was based on clot location and calibre of the intracranial vessels. No formal vessel measurements to guide aspiration catheter selection were performed.
A sequential stepwise aspiration approach was then applied (Table 1, Figure 1), CANADAPT. To begin, any 3-way valve/co-pilot system was removed from the end of the aspiration catheter. Absence of spontaneous blood return confirmed occlusive positioning. Continuous pump aspiration (Penumbra Engine, Penumbra, Inc.) was then applied directly to the catheter for 60 s. If during this time, there was free flow of blood return through the aspiration tubing, the aspiration catheter was left in position, the pump switched off, and an angiogram was performed through the guide catheter. If there was angiographic improvement, this maneuver was labeled ‘CANADAPT 1A’, where 1 indicates that it is the first pass and A denotes the maneuver type (Figure 1, Table 1). If further intervention was required, a second pass was attempted with minimal repositioning required of the aspiration catheter to the thrombus interface.
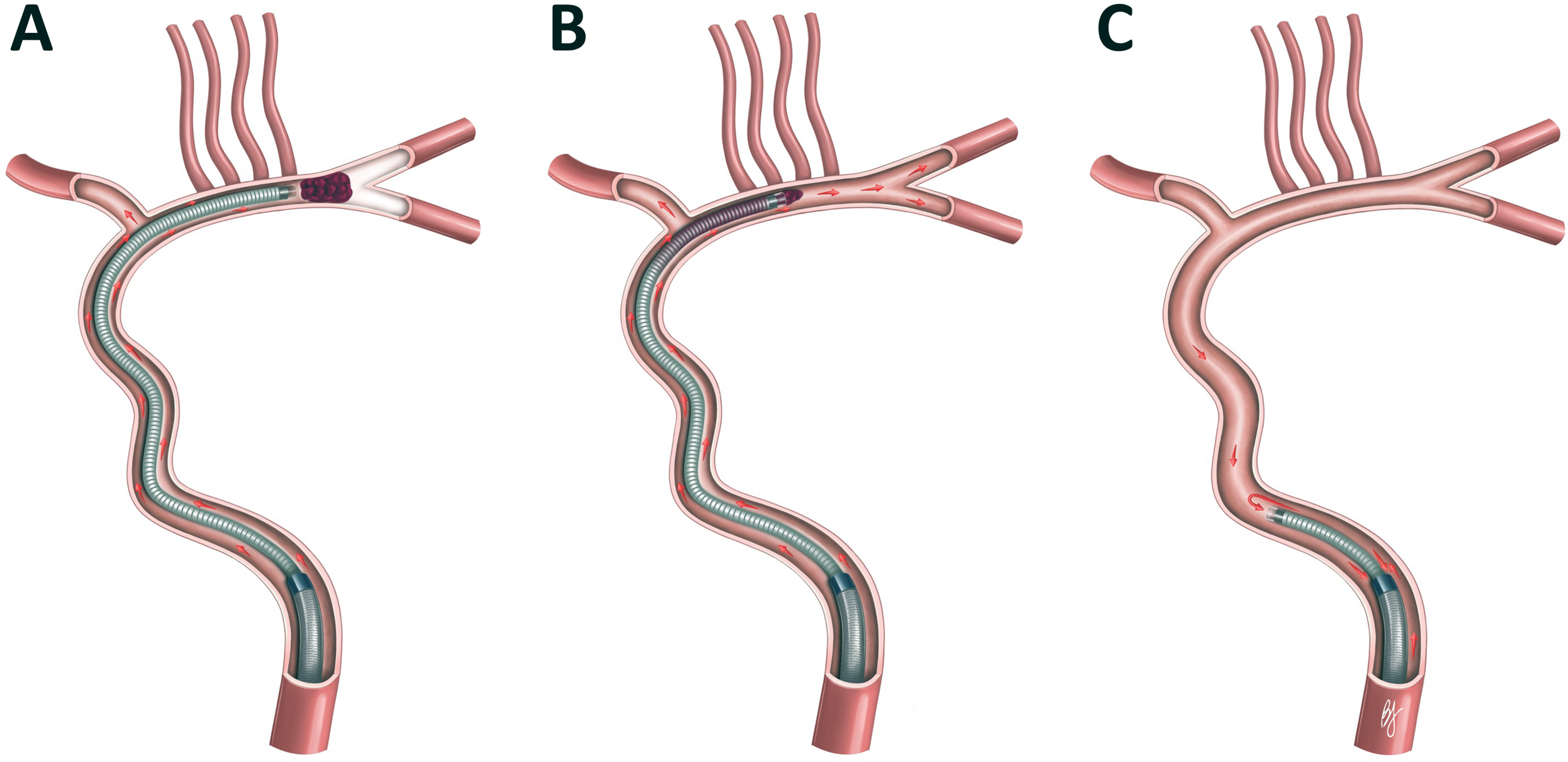
CANADAPT maneuvers.
CANADAPT nomenclature.

If there was no free flow after the first maneuver, the aspiration catheter was slowly withdrawn, by millimetric movements, under continuous pump aspiration until it entered a known patent segment of vessel, ideally in the same territory (e.g., distal M1 to proximal M1). Aspiration continued in this position for an additional 60 s. If there was free blood return, the catheter was left in position, the pump switched off, and an angiogram through the guide catheter was performed. If there was angiographic improvement, this was labeled “CANADAPT 1B” (Table 1). If further intervention was required, a second pass starting at the CANADAPT A maneuver positioning, at the clot interface, was attempted.
If there was no free blood return after the B maneuver, pump aspiration was continued through the aspiration catheter with additional manual aspiration directly applied through the guiding catheter side arm while slowly withdrawing the aspiration catheter. After complete removal of the aspiration catheter, an angiogram was then performed through the guiding catheter. If there was angiographic improvement, this was considered “CANADAPT 1C” (Table 1). Any further intervention consisted of a second pass of CANADAPT starting at maneuver A positioning or a SOLUMBRA rescue technique (Solitaire [Medtronic] and Embotrap [J&J Med Tech] stents were used). Up to 2 CANADAPT passes were performed before switching to SOLUMBRA.
If there was no angiographic improvement despite sequential A, B and C maneuvers, this was determined to be a CANDAPT 1X. An additional CANADAPT pass or switch to SOLUMBRA technique was then considered at the discretion of the treating neuroradiologist. Notably, the CANADAPT maneuvers are a sequential approach as opposed to predetermined independent techniques for EVT.
Outcome measurements
Our primary outcomes were successful recanalization defined as mTICI score (modified thrombolysis in cerebral ischemia) of 2B-3, procedural time defined as femoral puncture to recanalization, and number of passes to achieve reperfusion. Secondary outcomes were intraprocedural complications and cost of the intervention.
Ethics
This initiative to develop a standardized ADAPT technique for our institution to improve speed and efficacy of intervention for acute stroke was deemed quality improvement and an exemption was obtained from our local research ethics board.
Results
A total of 22 patients were included in this case series (Table 2). The average age was 74.2 ± 10.5 years with a slight female predominance (14 patients, 63%). ASPECT scores were generally favourable (8.3 ± 1.6) with moderate NIHS scores (13.8 ± 5.6). The occluded vessel was typically M1 (17, 77%), with two M1/2 (9%), two carotid-T (9%) and one basilar (5%) occlusion.
Results of CANADAPT.
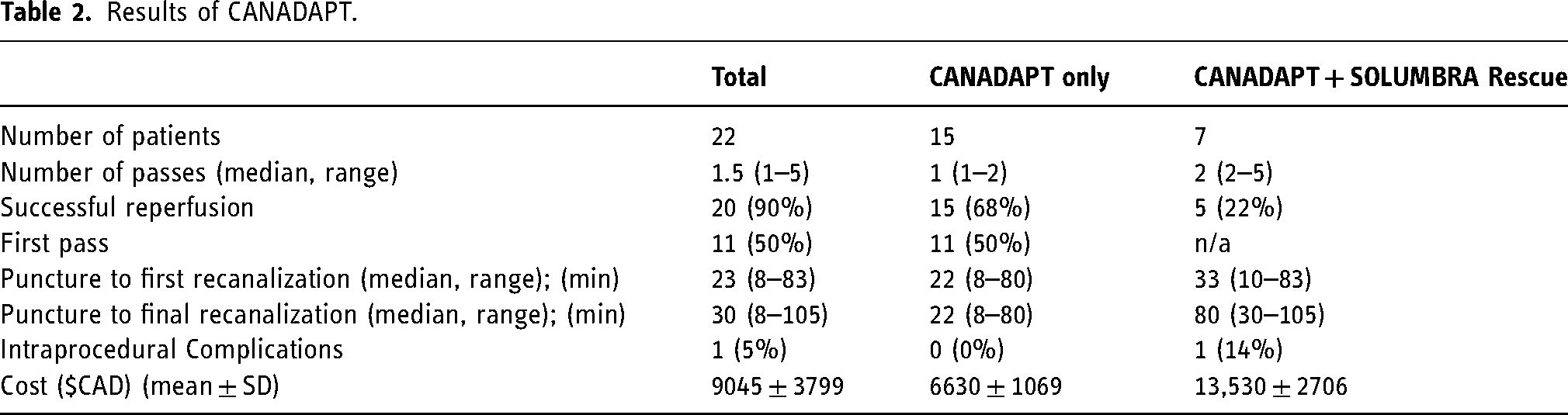
First pass successful recanalization was achieved in 11 (50%) of patients. A further 4 patients had successful reperfusion with a second pass of CANADAPT (total 68% success rate with CANADAPT only; Table 2). There was only one CANADAPT procedure in which maneuver A was successful, where clot was completely aspirated through the tubing and free flow was achieved through the catheter. On the first pass, free flow could be achieved in 35% of cases with maneuver B, by slowly withdrawing the aspiration catheter into a patent vessel (Table 3). However, the remainder of successful CANADAPT passes required complete removal of the aspiration catheter, with the clot lodged at likely the distal aspect of the tip (CANADAPT maneuver C, Table 3). With respect to aspiration catheters, RED 72 was typically used (12 patients, 55%) followed by RED 68 (8 patients, 36%) and RED 62 (2 patients, 9%).
CANADAPT maneuver results.
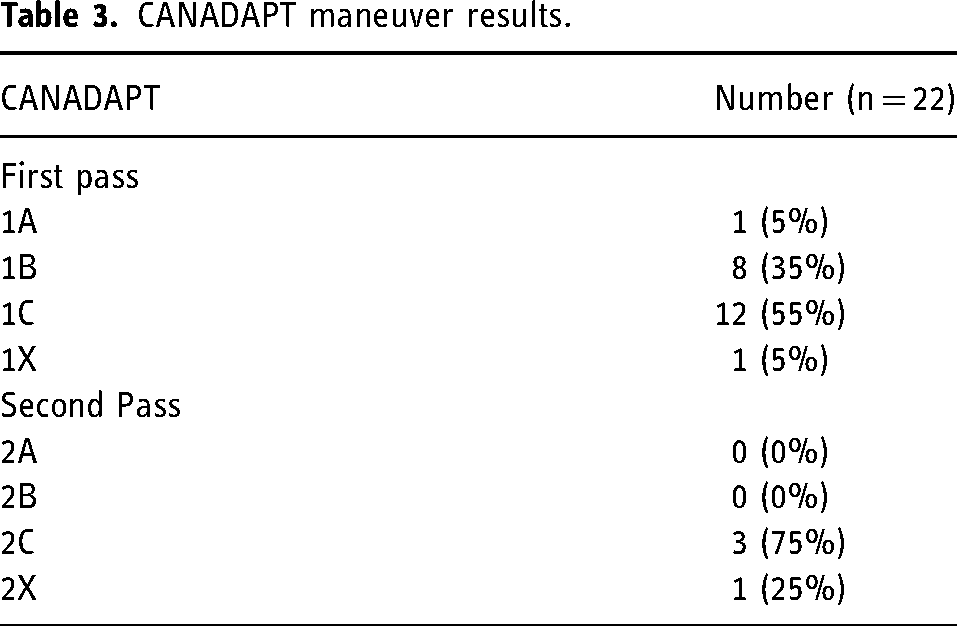
There were 7 patients who had SOLUMBRA rescue: 1 patient had two attempted CANADAPT passes without angiographic improvement (CANADAPT 1X and 2X) and 6 patients had partial recanalization after one CANADAPT pass. Of these, 5 patients (22% of total patients and 71% of rescue patients) ultimately had successful reperfusion with rescue technique (TICI 2B-3).
Overall median procedural time (groin puncture to recanalization) was 23 min (range 8–80 min) for first recanalization and 30 min (range 8–105 min) for final recanalization. For patients who received CANADAPT only, median final recanalization time was 22 min (8–80 min). The longest of these cases, that took 80 min, was secondary to difficulty navigating the guide catheter around challenging neuroanatomy. There was one procedural complication of a direct cavernous fistula after SOLUMBRA rescue technique in a challenging case with a heavily calcified embolus in the proximal M1.
Based on procedural material cost analysis. The cost per procedure was $6630 ± 1069 for CANADAPT, which essentially doubled when SOLUMBRA rescue techniques had to be subsequently employed ($13,530 ± 2706).
Discussion
ADAPT is a promising technique for endovascular thrombectomy. The described CANADAPT approach employs an initial scientific based stepwise standardization of aspiration catheter position/technique to effectively and efficiently perform aspiration first thrombectomy as an initial pass approach for large and some medium vessel occlusions.
In our initial clinical experience with this approach, our overall success rate was 50% for first pass only thrombectomy with an acceptable angiographic result, similar to other ADAPT studies.5,11 Using CANADAPT in a second pass approach yielded a further 18% success rate (overall success rate of 68%). Thus, the CANADAPT approach can yield good angiographic results in most patients presenting with large/proximal medium vessel occlusions. Furthermore, there was one case of basilar occlusion with a successful aspiration result suggesting promise of this technique for posterior circulation occlusions. However, despite the initial angiographic improvements with CANADAPT, rescue interventions such as SOLUMBRA technique, remain important approaches to achieve desirable angiographic reperfusion results. In our initial study, a combination of CANADAPT and rescue SOLUMBRA approaches yielded 90% success rate in achieving good reperfusion results, similar to other proposed standardized approaches for EVT. 11
With respect to CANADAPT, most of the angiographic success occurred with the B and C maneuvers requiring some withdrawal of the aspiration catheter from the clot interface. This increased success rate over the CANADAPT A position (solely at the clot interface) is theorized to relate to the fact that effective aspiration needs a positive (systolic) pressure to push the clot and a negative (aspiration pump) pressure to pull the clot (Figure 2). Systolic pressure in the CANADAPT A position is minimal because it only acts on the front face of the clot, between the catheter and the vessel wall. This is reflected in the relatively low rate of successful clot aspiration with this maneuver, and furthermore, this is in fact the traditional approach to aspiration only thrombectomy attempts. CANADAPT A can rather be considered as the clot engagement stage where the ingestion of the clot starts as a preparation for the B and C maneuver. By bringing back the aspiration catheter into patent vessel and waiting for 60 s (CANADAPT B), the systolic pressure exposure beyond and around the clot is theorized to increase the amount of push on the clot. This systolic positive pressure push effect, added with the negative pressure pull effect arising from the aspiration pump, is thought to aid in successful encapsulation and aspiration of the clot (Case example in Figure 3). This effect may explain the observation that the use of balloon guide catheters, thus arresting anterograde vessel flow in EVT intervention, has been found associated with lower reperfusion rates, decreased first pass effect rates as well as longer procedure times, when compared with standard mechanical thrombectomy. 12 Thus, we hypothesize that optimizing the systolic push effect during EVT is a valuable component to successful clot aspiration. This theory would likely benefit from in vitro investigation to further support the proposed physics underlying negative and positive pressure interactions for successful aspiration thrombectomy. Our methodology uses 60 s in maneuver ‘A’ and 60 s in ‘B’. Future investigation into technique refinement may benefit from increasing time applied to the ‘B’ step, which may help in further increasing success of the 1B maneuver. Obtaining thrombus clearance in the B maneuver provides significant advantage in that if successful in this step, the aspiration catheter does not need to be fully removed and thus, re-prepared and re-navigated around anatomy for further intervention, if required. Furthermore, using a large diameter guide catheter, such as the BMX 96, allows for immediate angiography without the need to remove the more distal aspiration catheter with this maneuver.
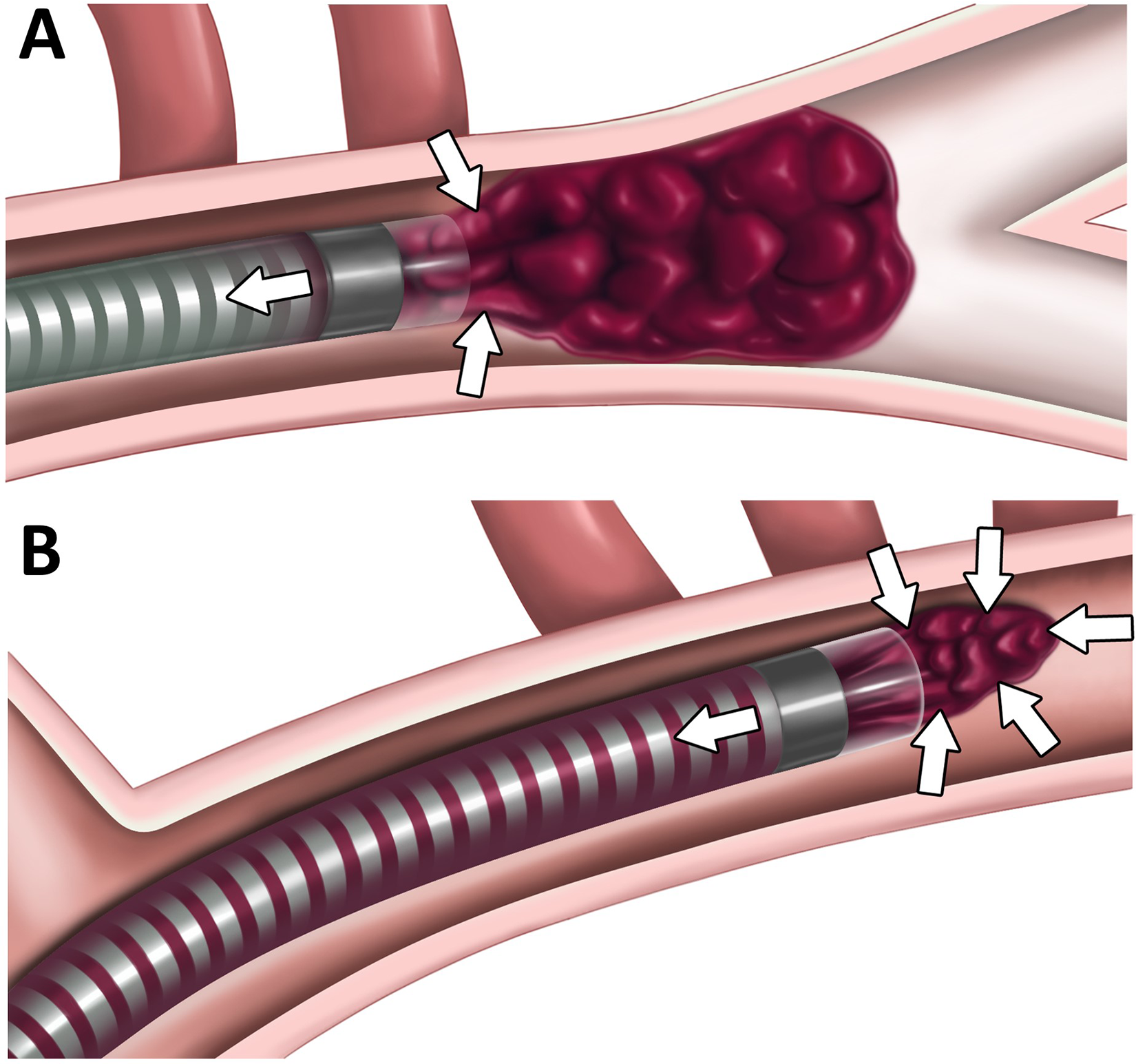
Physics behind effective aspiration.
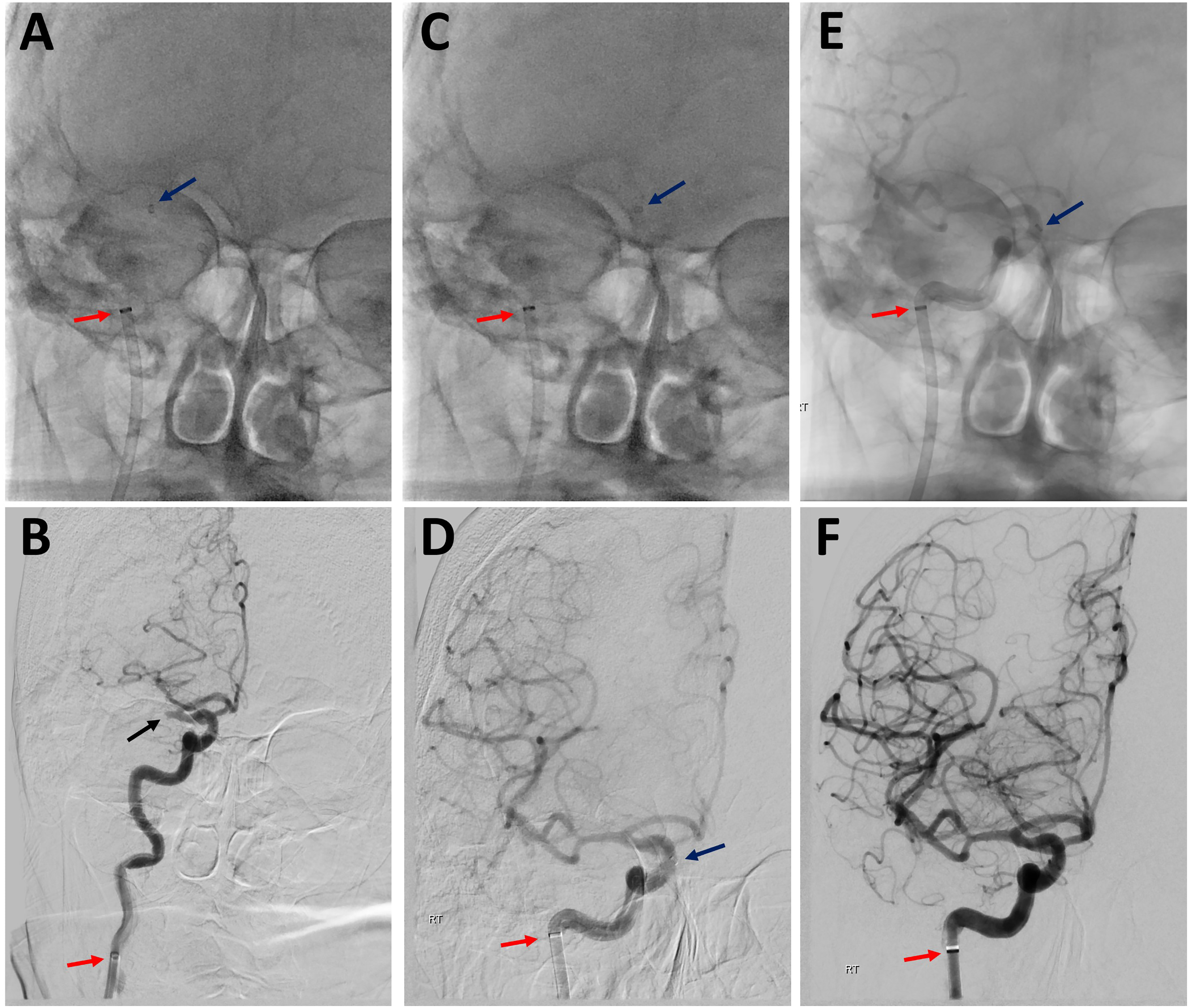
Case example of CANADAPT 1B.
CANADAPT 1C maneuver employs additional manual aspiration on the guiding catheter during aspiration catheter withdrawal under continuous pump aspiration. Both the negative pressure pull effect from the aspiration catheter and the positive pressure push effect generated by the guiding catheter aspiration can aid in successful thrombus aspiration. This is reflected in the high number of successful CANADAPT C maneuvers. The relative forces achieved by these catheters can be represented mathematically by Poiseuille's Law (Figure 4). Thereby even small increases in inner to outer diameter ratios can significantly increase the pull force while maximizing push forces generated by both the aspiration catheter and guiding catheters. With respect to the guiding catheter, use of the largest inner diameter to outer diameter ratio, such as employing the BMX 96 catheter, will optimize this push effect, as long as the catheter is not occlusive within the parent vessel.
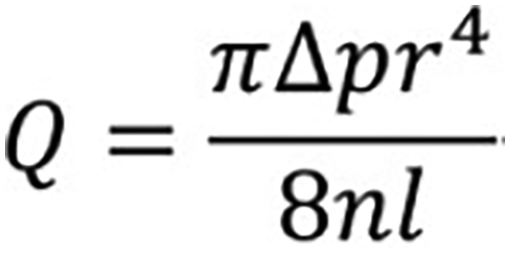
Poiseuille's law rearrangement. This equation governs the relationship between volumetric flow rate (Q) and catheter inner radius (r) to the 4th power. Therefore, small increases in aspiration/guiding catheter inner diameter can translate into significant increases in force for endovascular thrombectomy. n = fluid viscosity, l = length of tubing, Δp = pressure difference.
Direct comparison of CANADAPT to other aspiration and stent assisted thrombectomy approaches is significantly limited by the general paucity of specific methodological data/descriptions involving these techniques. The lack of detailed reports includes crucial EVT steps such as microcatheter/wire and aspiration catheter positioning with respect to the clot interface, aspiration material (such as a specific pump or manual aspiration), aspiration dwell time and aspiration withdrawal methodology. This heterogeneity of reported methodology has been chronicled in the literature, including for the assessment of syringe vs pump aspiration for EVT. 13 Although many of these techniques, including the slow withdrawal described in CANADAPT C, are likely routinely employed in practice, without discrete methodologic data it remains difficult to conclude the most effective practices for EVT. Therefore, the detailed standardized CANADAPT technique is well positioned to provide a systematic approach for EVT that can be continually assessed for further efficiency/efficacy refinements.
A type of pressure loss is termed ‘Minor Losses’ and occurs at any bend/joint/valve associated with the aspiration system. Therefore, to minimize these losses, pump aspiration should be applied directly to the hub of the aspiration catheter when possible (with removal of any 3-way valve/Co-pilot system). This typically entails modifying the catheter set up once in position at the clot interface to apply pump aspiration, by removing unnecessary connections/connectors. This is crucial to maximize the negative pressure applied via continuous pump aspiration.
It has recently been proposed that exponential infarct growth may be associated with sustained intraprocedural blood pressure drops during EVT. 14 We propose systolic pressure as in fact aiding in the ingestion of clot into the aspiration catheter, therefore future studies examining the effects of increasing blood pressure during EVT on reperfusion success as well as infarct size should be considered.
In 35% of our cases, a SOLUMBRA rescue intervention was performed to improve the angiographic result. This employs the use of aspiration catheters with the concomitant deployment of a stent retriever across the thrombus. This has been shown to be a powerful technique for clot removal however has several disadvantages when compared to aspiration only technique. This includes having to cross through the thrombus in order to deploy the stent thus increasing the possibility of distal emboli as well as navigating a microcatheter/microwire to an angiographically occult vessel, increasing the possibility of traversing pathology, such as an unknown aneurysm, or increasing the risk of parent vessel perforation. Therefore, CANADAPT is well positioned as a relative safe technique for first pass intervention, as demonstrated in our study. In addition, SOLUMBRA technique generally increases procedural time due to navigating to a more distal vessel, deploying the stent and allowing for incorporation before finally withdrawing it. Multiple SOLUMBRA passes also can increase procedural time given the need to reprepare the aspiration catheter, microcatheter/microwire and stent systems. With respect to multiple SOLUMBRA passes, it has been demonstrated that 3 or more passes with a stent retriever are an independent predictor of parenchymal hematoma. 15 Thus, employing even multiple passes of an aspiration only technique may prove valuable in reducing intraprocedural risk. Finally, SOLUMBRA technique substantially increases the material cost of intervention, in our case approximately double the cost, compared to aspiration only technique. This is similar to other cost analysis studies comparing ADAPT to SOLUMBRA techniques.10,16 Based on our preliminary study approximately 2/3 of CANADAPT procedures are angiographically successful without the need for a stent and therefore would significantly reduce the costs associated with an EVT program. Rescue techniques such as SOLUMBRA however remain crucial techniques for endovascular thrombectomy, and as our study shows, is required to achieve acceptable reperfusion results in some patients. However, given the relative disadvantages, we consider CANADAPT as an attractive first pass approach in almost all large/proximal medium vessel EVT.
Limitations of our study include the small sample size and limited clinical follow-up to date of this cohort. Thus, statistical analyses assessing the angiographic success of the different CANADAPT maneuvers as well as patient outcomes were not feasible. Furthermore, the majority of cases were M1 occlusions, an occlusion site with some of the best evidence of effectiveness of ADAPT techniques. It remains unclear if this efficacy may translate to other segment occlusions. Patients with tandem occlusions and those presenting with acute on chronic occlusions were not considered for this study therefore the effectiveness of the CANADAPT approach in these patients remains unknown. Similarly, there was only one patient with a posterior circulation occlusion in our study (basilar occlusion). Despite the good CANADAPT reperfusion outcome in this patient, further study for the use of this technique in the posterior circulation is warranted. Future studies to clarify the long-term patient outcomes of CANADAPT and inclusion of a greater range/territories of occlusions will help further elucidate the role of standardized aspiration only technique for EVT. Choice of catheter with the largest inner diameter to maximize push/pull pressures, while avoiding parent and collateral vessel occlusion, 17 and thus limiting systolic push, is hypothesized to be crucial to optimize the aspiration only approach. Further study may benefit from obtaining accurate vessel measurements to further investigate the optimal catheter choice for a specific patient's intervention.
Implementation of a standardized aspiration approach for EVT is a promising avenue for improving outcomes. We show that CANADAPT is a safe, effective and efficient initial pass approach for EVT.
Conclusions
CANADAPT represents a standardized approach to Aspiration only initial pass endovascular interventions for large/proximal medium vessel occlusions. This initial study demonstrates the safety, efficiency and efficacy of this approach for use in EVT.
Footnotes
Acknowledgements:
The authors would like to thank the interventional neuroradiologists, Dr J. Heidenreich, Dr M. Schmidt, Dr R. Vandorpe and Dr D. Duggan for their aid and expertise in applying the CANADAPT technique as part of this manuscript. We also thank Katrina Boudreau for her help in collecting the procedural cost data.
Declaration of conflicting interests
B.S. is an employee of Penumbra.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
