Abstract
Introduction
Superbore 0.088″ catheters provide a platform for optimizing aspiration efficiency and flow control during stroke mechanical thrombectomy procedures. New superbore catheters have the distal flexibility necessary to navigate complex neurovascular anatomy while providing the proximal support of traditional 8F catheters. The safety and feasibility of Zoom 88™ superbore angled-tip catheters in the middle cerebral artery (MCA) segments smaller than the catheter diameter have not been previously described.
Methods
Twenty consecutive cases of acute MCA mechanical thrombectomy were retrospectively identified from the senior authors’ prospectively maintained Institutional Review Board-approved database, in which the Zoom 88 (Imperative Care, Campbell, CA) catheter was successfully navigated to at least the M1 segment. Patient demographics, procedural details, and periprocedural information were analyzed. Rates and averages (standard errors) are generally reported.
Results
The average National Institutes of Health Stroke Scale at presentation and age were 15 ± 2 and 73 ± 3 years, respectively. The M1 and M2 occlusions were evenly distributed. The average M1 measurements before thrombectomy ranged from 2.36 ± 0.07 mm proximally to 2.00 ± 0.11 mm distally, and after thrombectomy, they ranged from 2.34 ± 0.07 mm proximally to 1.97 ± 0.10 mm distally. First-pass modified thrombolysis in cerebral infarction (mTICI) 2C/3 recanalization was achieved in 40% of cases, and final mTICI 2C/3 recanalization was achieved in 90% of cases. A single case of mild vasospasm was managed with verapamil. No hemorrhagic or periprocedural complications were noted.
Conclusion
Superbore 0.088″ catheters with flexible distal segments can be safely navigated to the MCA to augment mechanical thrombectomy even when the MCA segment is smaller than the catheter.
Introduction
Acute ischemic stroke (AIS) is a significant cause of morbidity and mortality worldwide.1 Medical management of AIS with antithrombotic and thrombolytic medications has strict guidelines and significant side effects.2 The endovascular management of AIS has witnessed substantial advancements over the past decade, leading to mechanical thrombectomy becoming a safer and more effective treatment option.3,4 In 2013, the largest available reperfusion catheter was the 5MAX ACE (Penumbra, Alameda, CA) with a 0.060″ inner diameter (ID). After introduction, this catheter increased in adoption due to its combination of navigability for a large bore size and ability to provide direct aspiration.5 Further advancements introduced even larger catheters in 2015: the ACE64 and ACE68 with IDs of 0.064″ and 0.068″, respectively. Today reperfusion catheters boast even larger IDs between 0.071″ and 0.074″. These catheters provide support for delivering other devices to treat distal vessel occlusions and have led to improved revascularization rates due to the increased aspiration force.6,7
Superbore 0.088″ catheters designed for intracranial use are the most recent advancement in endovascular technology for ischemic stroke. If advanced to the M1 segment of the middle cerebral artery (MCA), superbore catheters can achieve distal flow arrest due to their large outer diameter (OD) relative to the diameter of the M1-MCA.8 This size advantage may minimize the risk of embolic showers from clot fragmentation, potentially improving neurological outcome.9 Applying aspiration when positioned near the face of the clot, and when other smaller reperfusion catheters are used, also has the potential to increase first-pass success rates and improve the final degree of recanalization.10 Furthermore, compared to the proximal position attained with contemporary balloon guide catheters, utilization of 088 superbore catheters in the MCA along with 071 and 055 thrombectomy catheters reduces the distance between the thrombectomy and flow-arrest catheters, consequently lowering the likelihood of clot fragmentation.
Zoom 88 (Imperative Care, Campbell, CA) is a 0.088″ ID, 2.74 mm OD, a catheter designed for acute mechanical thrombectomy. The catheter has an angled distal tip that is thought to enhance suctioning force on thrombus due to higher surface area. The safety and feasibility of using Zoom 88 within MCA segments smaller than the catheter diameter have not been established. We herein report our initial experience navigating Zoom 88 to the MCA segments and using it to support AIS thrombectomy. To the best of our knowledge, this is the first known report of its kind.
Materials and methods
Patient inclusion
A prospectively maintained Institutional Review Board (IRB) approved institutional database of the senior authors was retrospectively reviewed. Patient charts from April to August 2023 were reviewed. We did not collect individual patient consent as all data was deidentified and stored in a secure IRB-approved database per our local IRB protocols. Consecutive cases with Zoom 88 navigation to the MCA-M1 or beyond in the setting of mechanical thrombectomy for AIS were selected for analysis. Patients were excluded if Zoom 88 was not used in the MCA or the occlusion location was not in the MCA.
Procedural technique
All procedures were performed in an emergency setting under general anesthesia using femoral access. An 8 French short sheath was introduced into the femoral artery using the Seldinger technique and maintained under continuous flush. Under real-time fluoroscopy, a Neuron Select (Penumbra, Alameda, CA) and Zoom 88 were advanced into the aortic arch over a 0.035″ guidewire. The common carotid artery was selected, and the Zoom 88 was advanced over the Neuron Select and guidewire into the internal carotid artery. For aspiration thrombectomy cases, Zoom 88 was navigated triaxially over Zoom 71 and Zoom 35 (ID 0.071″ and 0.035″, respectively) to the face of the clot in the MCA. The Penumbra Aspiration Pump or Zoom Pump and Zoom POD were used to apply a vacuum through all the catheters and aspirate the clot. If a stentriever was used, Zoom 71 or 55 with Zoom 35 and a microwire were advanced through Zoom 88 to the face of the clot; the Zoom 35 was removed and a 0.021″ microcatheter was utilized to deploy a stentriever. Zoom 88 was advanced over the stentriever to the clot, before removing the stentriever under vacuum aspiration through the Zoom 88. Follow-up angiography was performed to confirm revascularization before concluding the case.
Data collection
The patient charts were reviewed for baseline characteristics including age, sex, comorbidities, and presenting National Institutes of Health Stroke Scale (NIHSS). Administration of thrombolytic and thrombus location with laterality were evaluated. The MCA-M1 diameter was measured at proximal, mid, and distal locations using the anterior–posterior angiography view on digital subtraction angiography (DSA) before and after thrombectomy. All measurements were taken using autocalibration to limit magnification errors. Procedural data included system description, total fluoroscopy time, total procedural contrast (mL), and radiation dose (mGy). Proximal internal carotid artery (ICA) tortuosity was characterized by petrous and cavernous grading.11 Outcomes, including modified thrombolysis in cerebral infarction (mTICI) reperfusion grade and the number of passes, were obtained. All cases were reviewed for catheter-related vessel injury.
Statistics
One-sided paired t-tests were used to assess if the proximal, middle, or distal M1 diameters significantly increased between prethrombectomy and postthrombectomy measurements. A p-value of .01, corresponding to a 99% confidence level, was considered statistically significant. All calculations were completed using Minitab 21.4.0 statistical software (Minitab Inc., State College, PA). Discrete datapoints, including patient demographics, baseline characteristics, effectiveness, and safety endpoints were summarized with rates. The mean and standard error were generally reported for continuous data.
Results
Between April 2023 and August 2023, 20 consecutive MCA thrombectomy cases using angled-tip catheters were identified. Baseline characteristics of study patients are presented in Table 1. Shortly, slightly over half (n = 12, 60%) of patients were female and the average presenting NIHSS and patient ages were 15.1 ± 1.97 and 73.2 ± 3.05 years, respectively. Thrombolytics were administered to a minority of patients. The M1 and M2 occlusions were evenly distributed. The most common petrous grade tortuosity was a hockey stick shape (n = 8, 40%), followed by some recurve (n = 6, 30%) and a question mark (n = 6, 30%). The most common cavernous grade was 1a (n = 8, 40%), followed by 1b (n = 5, 25%), 4 (n = 5, 25%), 2 (n = 1, 5%), and 3 (n = 1, 5%). There was a single case of mild vasospasm in the proximal ICA due to tortuous anatomy that was successfully managed with 10 mg of verapamil.
Baseline characteristics of study patients, procedural characteristics, and vessel diameters.
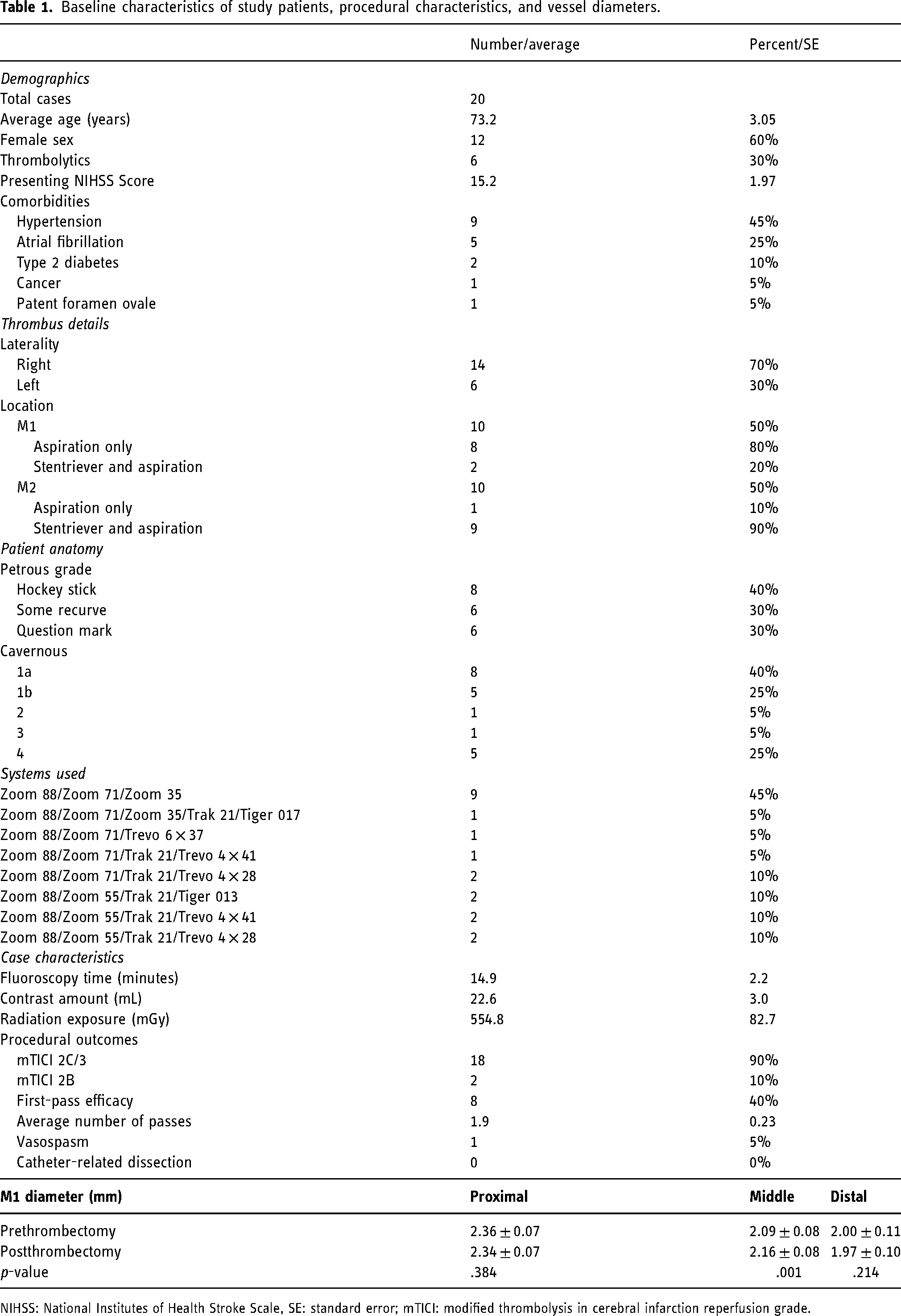
NIHSS: National Institutes of Health Stroke Scale, SE: standard error; mTICI: modified thrombolysis in cerebral infarction reperfusion grade.
Two thrombectomy techniques were used, aspiration only for nine cases (45%) and stentriever with aspiration in 11 cases (55%). Of the M1 occlusions, aspiration only was used in 80% (n = 8) of cases and aspiration with stentriever in 2 (20%) cases. Ninety percent (n = 9) of M2 occlusions were managed with a concomitant aspiration and stentriever technique, and 1 (10%) case used aspiration only with the placement of Zoom 88 into the M1. The average fluoroscopy time was 14.9 ± 2.2 min, radiation doses were 554.8 ± 82.7 mGy, and contrast volume was 22.6 ± 3.0 mL (Table 1). First-pass mTICI ≥2C recanalization was achieved in 40% (n = 8) of cases and the average number of passes was 1.9 ± 0.23. Final mTICI ≥2B recanalization was achieved in all cases (Table 1). Proximal and distal M1 diameters did not significantly differ between prethrombectomy and postthrombectomy; a small (0.07 mm) but statistically significant increase in the middle M1 diameter was observed postthrombectomy (Table 1).
Discussion
Over the last two decades, there has been a consistent trend toward developing larger bore aspiration catheters that can navigate the tortuous anatomy of neurovasculature. The first stroke aspiration catheters were released in 2007 and had IDs between 0.026″ to 0.041″. Over the following years, aspiration catheters continued to increase in size with devices like the 0.060″ ID 5MAX ACE released in 2013, 0.068″ ID ACE68 in 2015, and now there are multiple large bore aspiration catheters with IDs between 0.070″ and 0.074″ available. The primary incentives for these increases in aspiration catheter ID include enhancing the clot engagement force exerted by the catheter while simultaneously decreasing the force needed to ingest the clot. These features have resulted in faster procedures, fewer passes to achieve successful reperfusion, and improvements in the achieved level of reperfusion.6,12 Despite the evident clinical benefits associated with the use of larger diameter catheters for neurovascular interventions, concerns persist regarding the safety of employing 0.088″ catheters in M1 vessels that are nominally narrower than the catheter itself.
Guide catheters of 0.088″ ID have long been used as neurovascular guides to support the delivery of interventional devices. Early use of 0.088″ catheters in the neurovasculature focused on stiffer catheters (6F Shuttle, Cook Medical, Bloomington, IN) that were utilized to deliver carotid stents and distal cerebral protection devices, with the Shuttle typically positioned near the common carotid artery bifurcation.13 While these stiffer catheters are sometimes used to support mechanical thrombectomy procedures,14 0.088″ catheters with softer distal tips (Neuron Max) were developed which allowed for more consistent navigation to the distal cervical segment of the internal carotid artery and delivery of thrombectomy catheters and other devices directly into the intracranial vasculature.15
In recent years, newer 0.088″ catheters with longer flexible segments have been developed which can be consistently navigated past the cervical ICA and into more distal neurovasculature. The infinity-long sheath (Stryker) was designed with a 9.5 cm distal flexible segment that enables it to track past challenging tortuosity in the cervical ICA and the TracStar Large Distal Platform (Imperative Care) was designed with a 14 cm distal flexible segment that enables it to consistently track intracranially into the cavernous and clinoid segments of the ICA.16–18 The Zoom 88 catheters used in our study have an even longer 18 cm distal flexible segment and have previously been reported reaching locations up to the M1 segment of the MCA.19–21
The ability to track 0.088″ catheters intracranially into the distal ICA and MCA, combined with prior observed improvements in reperfusion success rates using larger ID aspiration catheters, will likely see a natural progression toward performing aspiration thrombectomy with superbore 0.088″ catheters. While no superbore 0.088″ catheters are currently indicated for reperfusion, initial reports have shown promising results associated with using these catheters to support thrombectomy. Nogueira et al. found promising results in a series of early cases using the Zoom 88 and TracStar LDP catheters within the ICA, M1, and Basilar arteries, with an observed first pass mTICI 3 rate of 60%.20 However, the authors noted that navigation of the 0.088″ catheters into the more distal aspects of the M1 segment was avoided due to concerns about the vessel diameter (2.6 mm) being smaller than the OD of the catheter (2.74 mm).
Within our cohort, we found that the Zoom 88 catheter was able to successfully navigate simple and tortuous anatomy in the petrous and cavernous ICA before being advanced to or beyond the M1 (Table 1). Figures 1 and 2 show the more challenging anatomies the angled-tip catheters navigated in this study, which included multiple >180° bends and loops. Despite the relatively large difference in size between the 2.74 mm OD Zoom 88 catheters and the smaller diameter M1 vessels encountered in this study, all procedures were completed without noted dissections or significant vascular injury (Figure 3). This establishes the technical feasibility and safety of utilizing 0.088″ catheters for thrombectomy within these narrower-diameter MCA vessels. We also found that the average diameter of the M1 vessels did not appear to have a clinically significant increase after completing the thrombectomy procedures, though small numeric increases between 0.1 mm and 0.2 mm were observed in some cases (Figures 1 and 2). These findings further support that there was minimal damage to the vessel wall and that superbore angled-tip catheters can be safely used within smaller-diameter vessels (Supplementary Video 1). We believe that the excellent safety observed in this cohort may be associated with flexibility within the distal end of the catheter.
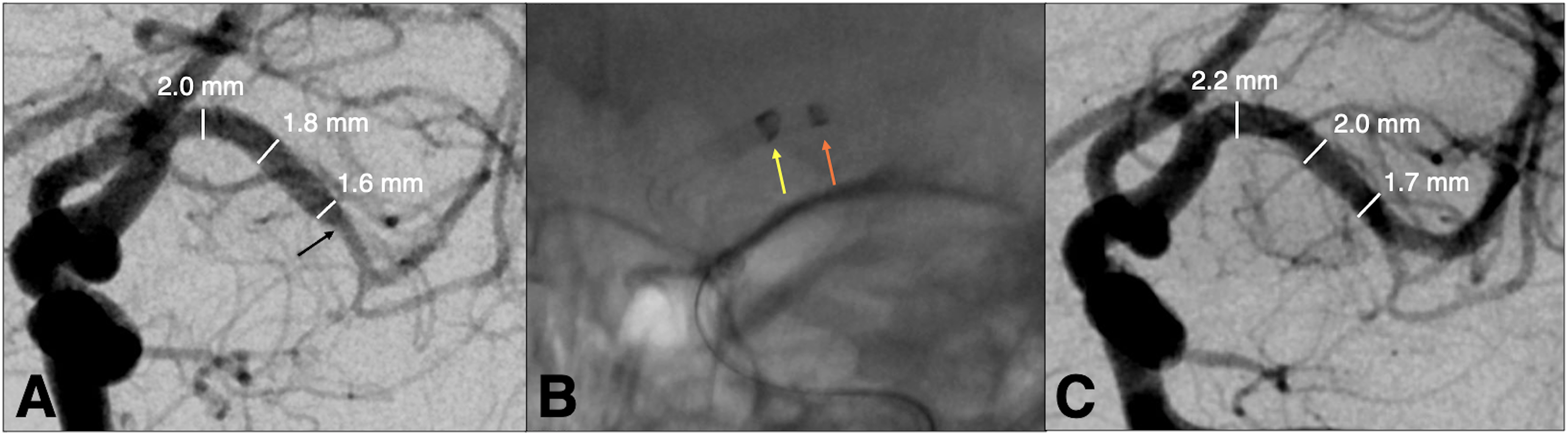
(A) An adult patient with NIHSS of 26 presents with a distal M1 occlusion (black arrow). (B) Navigation of the Zoom 88 (2.74 mm OD) (yellow arrow) with Zoom 71 (orange arrow) to the 2 mm M1 for direct thrombus aspiration. (C) Postangiography demonstrating TICI 3 with M1 enlargement post Zoom 88 thrombectomy.
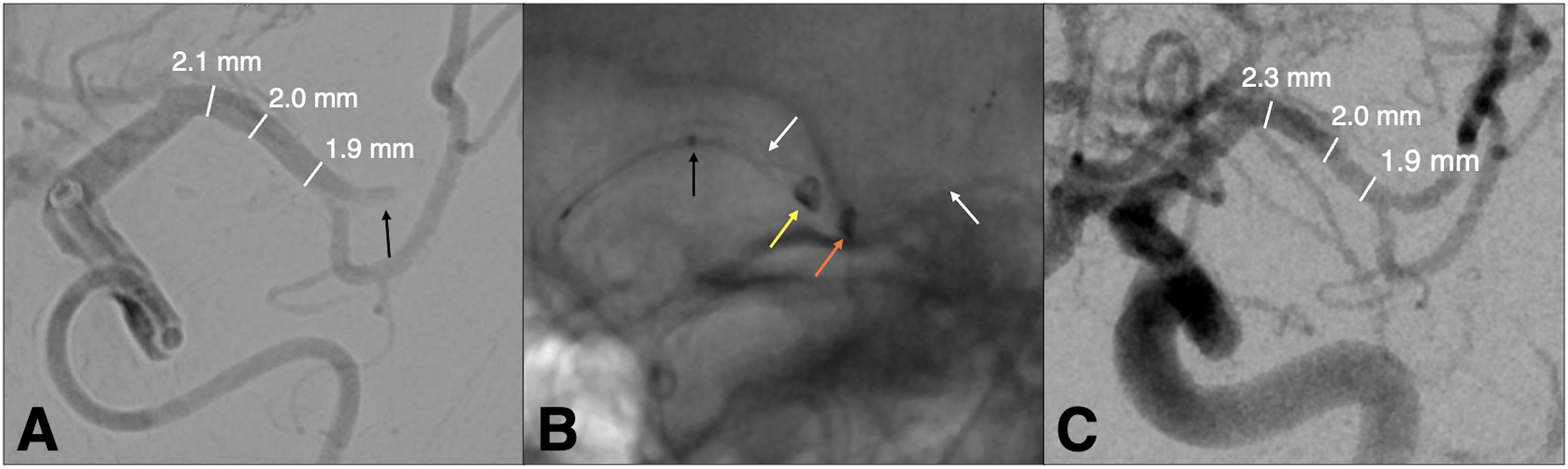
(A) A sexagenarian patient with NIHSS of 20 presents with a thrombus in the superior division of the proximal left M2 (black arrow). (B) Navigation of Zoom 88 (yellow arrow) with Zoom 71 (orange arrow) to the M1 with deployment of Trevo 4 × 28 (white arrows) stentriever in the superior M2 via the Trak 21 microcatheter (black arrow). (C) Postangiography demonstrating TICI 3 with enlargement of proximal M1 post Zoom 88 stentriever-assisted thrombectomy.
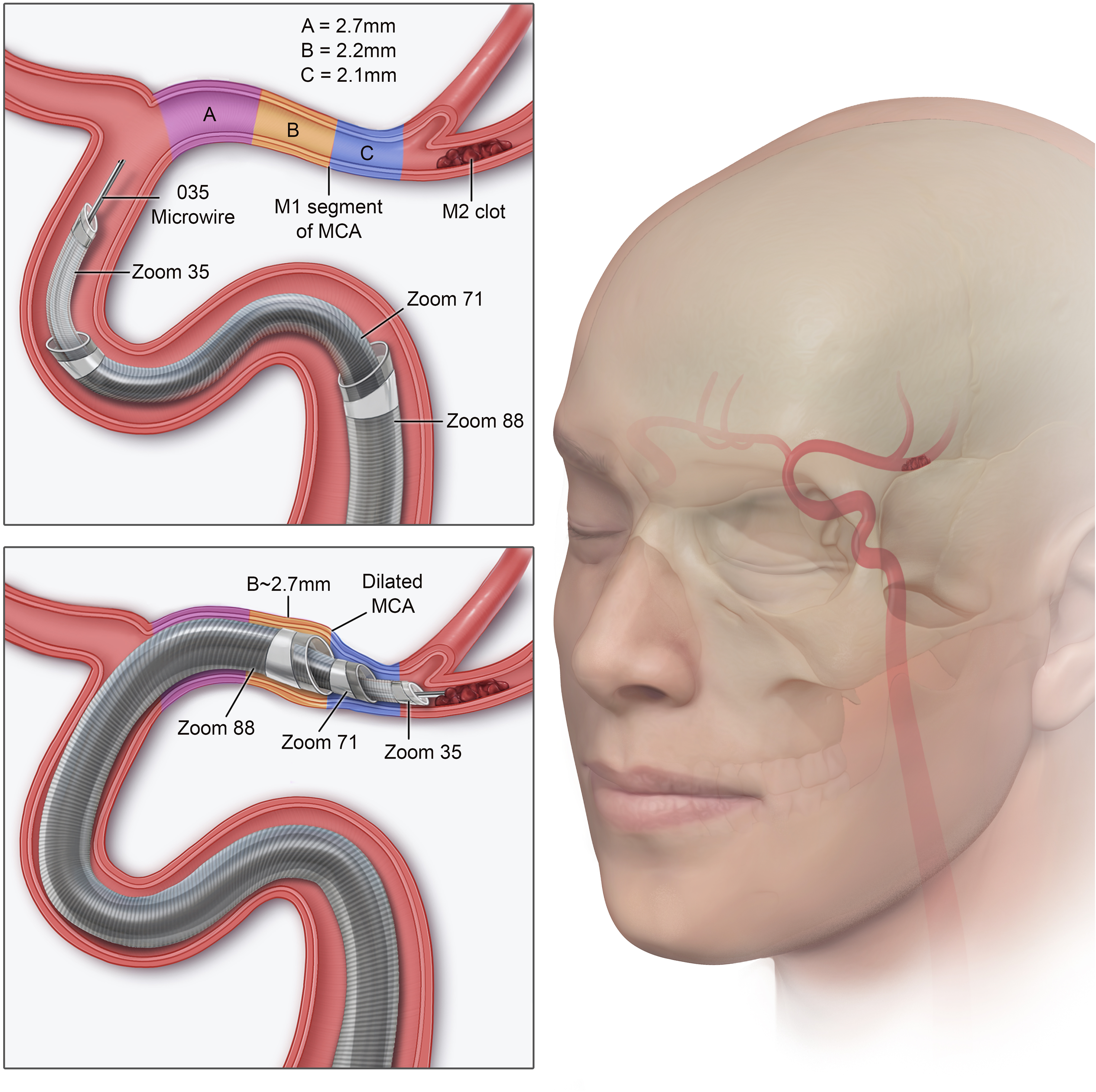
Illustration depicting Zoom system placement of superbore Zoom 88, Zoom 71, and Zoom 35 catheters for an M2 segment occlusion. Using the system designed for coaxial tracking with minimal endoluminal step-off, the Zoom 88 superbore catheter is placed safely within an M1 segment that measures less than 2.7 mm (size of Zoom 88). Copyright Tess Marhofer. Published with permission.
In addition to excellent periprocedural safety, the results from our study also showed a high rate of first-pass success and self-adjudicated final mTICI ≥ 2C reperfusion. The final mTICI ≥ 2C rate of 90% observed in our cohort is notably higher than in prior studies such as COMPASS which reported a 56% rate of mTICI ≥ 2C when using the current generation large bore aspiration catheters and stent retrievers.22 While these results should be interpreted with caution given the small sample size in our study, they appear to be in line with the other research demonstrating improved reperfusion success when using intracranially positioned flexible super-bore 0.088″ catheters to support stroke thrombectomy.20 Although first-pass efficacy (FPE) in our study aligns with results from larger trials in literature, it remains uncertain whether the presence of Zoom 88 in MCA confers a notable advantage in terms of FPE.20
Our study has several limitations. As this was a retrospective study without core lab adjudication, case selection, and endpoint assessments may be biased. However, we included consecutive patients treated by a single operator to minimize the potential bias. The relatively small sample size also limits the generalizability of our findings to a broader patient population. Factors such as variations in the aortic arch and tortuosity of proximal vessels might impact the number of cases wherein a 0.088″ catheter can effectively navigate to the MCA, although in our study patients with high-grade tortuous petrous and cavernous internal carotid arteries were incorporated. M1 measurements were taken using an anterior–posterior view on DSA, which subjects the vessel measurements to a magnification error. To limit this, all measurements were taken using the autocalibration technique. Despite these limitations, the analyses presented in our report provide a crucial preliminary insight into the safety and feasibility of positioning and using the Zoom 88 catheters within the M1/M2 arteries as part of mechanical thrombectomy procedures for AIS which has not previously been reported. The Zoom 88 catheters are also being studied as part of the ongoing prospective multicenter clinical investigation of the Zoom Reperfusion System (NCT04129125) which includes independent adjudication of efficacy and safety endpoints. This will further address the limitations noted in our study. Another limitation of our study is the varied techniques used, such as aspiration alone or in combination with a stent retriever. Given our small sample size, the superiority of one technique over the other cannot be determined. Our primary aim is to highlight the feasibility of placing the superbore 0.088 catheters in MCA vessels that are nominally smaller.
Conclusion
Within our study, we demonstrated that it is safe and feasible to use the Zoom 88 catheter to support M1 and M2 stroke thrombectomies even when the diameter of the M1 was smaller than the OD of the Zoom 88. Based on our results, the M1 vessel size does not appear to be a significant limitation to the navigation of superbore angled-tip catheters to the MCA for mechanical thrombectomy procedures.
Supplemental Material
Footnotes
Authors’ Notes
Jessica K Campos and Benjamen M Meyer contributed equally to this work. This material was accepted as an abstract and electronic poster to the 2023 Society of Vascular and Interventional Neurology meeting in Miami, Florida, USA.
Data sharing statement
There is no additional unpublished data from this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alexander L. Coon is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Stryker Neurovascular, Cerenovus, Rapid Medical, Avail MedSystems, Imperative Care, Deinde, InNeuroCo, Q'apel and a proctor for MicroVention-Termo, Stryker Neurovascular, and Medtronic Neurovascular. Li-Mei Lin is a proctor for Medtronic Neurovascular, Stryker Neurovascular, MicroVention-Terumo and a consultant for Medtronic Neurovascular, Stryker Neurovascular, MicroVention-Terumo, Rapid Medical, and Balt. Geoffrey P. Colby is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Rapid Medical, Cerenovus, and Stryker Neurovascular. Matthew Bender is a proctor Stryker Neurovascular. All other authors have no conflict of interest. No author received financial support in conjunction with the generation of this submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
