Abstract
Background
Endovascular embolization of the middle meningeal artery (MMA) has emerged as an adjunctive and stand-alone modality for the management of chronic subdural hematomas (cSDH). We report our experience utilizing proximal MMA coil embolization to augment cSDH devascularization in MMA embolization.
Methods
MMA embolization cases with adjunctive proximal MMA coiling were retrospectively identified from a prospectively maintained IRB-approved database of the senior authors.
Results
Of the 137 cases, all patients (n = 89, 100%) were symptomatic and underwent an MMA embolization procedure for cSDH. 50 of the patients underwent bilateral embolizations, with 53% (n = 72) for left-sided and 47% (n = 65) for right-sided cSDH. The anterior MMA branch was embolized in 19 (14%), posterior in 16 (12%), and both in 102 (74.5%) cases. Penetration of the liquid embolic to the contralateral MMA or into the falx was present in 38 (28%) and 31 (23%) cases, respectively, and 46 (34%) cases had ophthalmic or petrous collateral (n = 41, 30%) branches. MMA branches coiled include the primary trunk (25.5%, n = 35), primary and anterior or posterior MMA trunks (20%, n = 28), or primary with the anterior and posterior trunks (54%, n = 74). A mild ipsilateral facial nerve palsy was reported, which remained stable at discharge and follow-up. Absence of anterograde flow in the MMA occurred in 137 (100%) cases, and no cases required periprocedural rescue surgery for cSDH evacuation. The average follow-up length was 170 ± 17.9 days, cSDH was reduced by 4.24 ± 0.5(mm) and the midline shift by 1.46 ± 0.27(mm). Complete resolution was achieved in 63 (46.0%) cases.
Conclusion
Proximal MMA coil embolization is a safe technique for providing additional embolization/occlusion of the MMA in cSDH embolization procedures. Further studies are needed to evaluate the potential added efficacy of this technique.
Introduction
Chronic subdural hematoma (cSDH) is a common neurological condition that can result in significant morbidity and mortality. 1 The traditional treatment options, especially for a large cSDH with significant mass effect, include surgical evacuation with burr hole drainage or craniotomy. 2 However, endovascular management, particularly embolization of the middle meningeal artery (MMA), has emerged as an alternative and potentially adjunctive option. 3 Despite its believed efficacy, there are several challenges in achieving successful and safe embolization, particularly when using liquid embolic agents (nBCA or Onyx). 4
One of the major challenges of liquid embolization of the MMA is the risk of non-target embolization (NTE) complications such as blindness and cranial neuropathies.5,6 As the MMA is a branch of the external carotid artery, it supplies blood to the meninges and scalp, and therefore, has numerous collateral branches to the orbit and cranial nerves around its proximal course. 7 The use of liquid embolic agents in the proximal portion of the MMA may cause NTE of these branches, resulting in severe adverse events ranging from blindness and cranial nerve injuries to even potentially pial artery NTE and stroke.5–8 According to a recent systematic review, the risk of complications with liquid embolization of the MMA ranges from 2.2% to 16.7%, with blindness being the most common devastating complication. 9
In addition to the safety concerns, there is a need to prevent recanalization after embolization with particles, glue, or onyx. 4 The collateralized nature of the MMA tree can cause recanalization and recurrence of cSDH, leading to the need for repeat procedures. 10 According to a recent study, the rate of recanalization after MMA embolization with liquid or particle embolics was as high as 3.5%, highlighting the need for more effective embolization techniques. 9
The use of proximal MMA coil placement in embolization procedures for cSDH has recently been suggested as a potentially safe approach, 11 simultaneously addressing both the challenges of liquid embolization and recanalization. With the known rates of complications associated with liquid embolic agents in the proximal MMA and the need for preventing recanalization, 12 further understanding in this area is necessary to establish the role of proximal MMA coil placement in the management of cSDH. We herein report our initial experience with the safety of adjunctive proximal MMA coil embolization in patients undergoing liquid embolic embolization for cSDH. We believe this to be the first known report of its kind.
Materials and methods
Patient inclusion
We retrospectively reviewed a prospectively maintained Institutional Review Board-approved database of the senior authors. All individual identifying information, including age and sex, have been meticulously anonymized to achieve appropriate anonymity in the manuscript. From June 2020 to April 2023, consecutive patients with cSDH who underwent MMA embolization with adjunctive proximal MMA coiling were identified. Individual patient consent was not required as all data collected was de-identified and stored in a secure database in keeping with IRB protocols. cSDH was defined as subdural collections that were isodense or hypodense on CT imaging, with radiographic appearance of SDH. cSDHs with a dural vascular territory not amenable to embolization were excluded (e.g., tentorial and interhemispheric SDHs).
Procedural MMA embolization details
The decision to proceed with embolization as treatment of the cSDH was based on surgeon preference and institutional practice patterns. Informed consent for surgical and procedural interventions was discussed and obtained from all patients. General consideration for embolization included symptomatic recurrent cSDH with or without prior surgical evacuation, asymptomatic cSDH with significant mass effect or concerning midline shift, or cSDH in patients requiring continued antiplatelet or anticoagulant therapy for other medical comorbidities. All embolizations were performed under general anesthesia, with at least 2000U of systemic heparin administered as a bolus at the start of the case, through 6-, 7-, or 8-French femoral access. All cases utilized a tri-axial catheter platform as previously described. 13 Guide sheaths utilized for access included: Armadillo (Q'Apel Medical, Fremont, California, USA), Infinity (Stryker Neurovascular, Fremont, California, USA), Ballast (Balt USA, Irvine, California, USA), Benchmark (Penumbra, Alameda, California, USA), Wahoo (Q'Apel Medical, Fremont, California, USA), Rist (Medtronic Neurovascular, Irvine, CA), and Tracstar (Imperative Care, Campbell, California, USA). Distal access catheters included: Phenom plus (Medtronic Neurovascular, Irvine, California, USA), Offset (Stryker Neurovascular), AXS Catalyst 5 (Cat5, Stryker Neurovascular), and Vecta46 (Stryker Neurovascular). Microcatheters included: SL-10 (Stryker Neurovascular), Duo 156 (Microvention, Aliso Viejo, California, USA), Prowler (Cerenovus, Irvine, California, USA), Echelon 10 (Medtronic Neurovascular, Irvine, California, USA), XT-27 (Stryker Neurovascular), and Apollo and Marathon (Medtronic Neurovascular, Irvine, California, USA). Microwires included: Synchro2 (Stryker Neurovascular), Transcend (Stryker Neurovascular), X-Pedion (Medtronic Neurovascular, Irvine, California, USA), and Aristotle 14 (Scientia Vascular, West Valley City, Utah, USA).
After obtaining femoral access, a guide catheter was navigated into the proximal external carotid artery. Under roadmap guidance, a distal access catheter was advanced into the distal internal maxillary artery. Next, a microcatheter was introduced and navigated over a microwire into the proximal MMA. The anterior division was selected, and digital subtraction angiography was performed to identify critical ophthalmic collaterals. If a liquid embolic (Onyx, Medtronic Neurovascular; or TRUFILL® N-butyl cyanoacrylate, Cerenovus) was used, it was injected at this time, followed by removal of the microcatheter. A second microcatheter was introduced over the microwire into the posterior division, and catheter angiography was performed to identify dangerous petrous collaterals. If a liquid embolic was used, it was injected at this time and the microcatheter was removed. A third microcatheter was advanced over the microwire into the proximal MMA and coil embolization occurred in the primary trunk with or without the anterior and/or posterior divisions. If coil-only embolization was used, only a single microcatheter was used to navigate into the proximal MMA. If coil embolization was used in conjunction with a liquid embolysate, a total of three microcatheters were used as described above. Final angiography was performed to evaluate MMA embolization. Technical success was defined as the absence of anterograde flow in the intracranial MMA trunk.
Data collection
Patient charts were reviewed for demographic and clinical data, including age, sex, comorbidities, use of antiplatelet, anticoagulant, or aspirin medications, and presenting symptoms. The presence of recurrent cSDH, prior embolization surgeries, including burr hole or craniotomy with accompanying laterality, were also obtained. Radiographic data obtained includes cSDH laterality, widest diameter of SDH (mm) at presentation, and midline shift (mm). Procedural data were also recorded, including embolization laterality, proximal coil use, type of embolysate (10:1 TRUFILL® glue hyper-dilution, Onyx, particles, or a combination thereof), MMA branches embolized (anterior vs posterior vs both), total fluoroscopy time, total procedural contrast (cc/mL), and radiation dose (mGy). Additional procedural characteristics include contralateral MMA or falx liquid embolic penetration, target coil occlusion (defined as extending from the planum sphenoidale to the foramen spinosum on lateral x-ray projection), presence of ophthalmic or petrous collaterals, and whether the absence of anterograde flow in MMA occurred. In most patients, the lack of anterograde flow was determined at the final control angiography at the completion of the initial procedure. Although scheduled follow-up angiography was not conducted at the senior authors’ institutions, follow-up magnetic resonance angiography (MRA) revealed the MMA patency status in several cases. Procedural outcomes, including complications, and the incidence of either planned pre-surgery embolization or periprocedural rescue surgery for cSDH evacuation, were assessed at 30-days follow-up.
Except for age, cSDH thickness, and midline shift (MLS), counts and percentages of each category were obtained for all previously mentioned data. The average patient age, standard error of the mean, and range were all calculated.
Results
A total of 89 patients undergoing 137 consecutive MMA embolization procedures (mean age 74.5 ± 1.0 years, 34% females) over a 34-month study period (June 2020 to April 2023) were included. Patients presented after trauma or fall (47%, n = 64), altered mental status (14%, n = 19), headache (12%, n = 17), incidental (7%, n = 9), weakness (6%, n = 8), or other symptoms (15%, n = 20) (Table 1). Patients were taking dual antiplatelet therapy, 14 (32%, n = 44), aspirin (27%, n = 37), oral anticoagulant, (11%, n = 15), or no anticoagulant or antiplatelet medications (30%, n = 41). In total, 99 cases (72%) were managed without surgery, while those with recurrent SDH (near 50%, n = 68) had previous treatment of burr holes (19%, n = 26), craniotomy (8%, n = 11), or burr hole with craniotomy (1%, n = 1). Of the surgeries, 60.5% (n = 23) were on the left side, 32% (n = 12) on the right, and 8% (n = 3) were bilateral (Table 1). One case (1%) had a planned MMA embolization prior to a burr hole evacuation of the cSDH.
The characteristics of 137 cases (89 patients) of proximal coil embolization for cSDH.

cSDH = chronic subdural hematoma; DAPT = dual antiplatelet therapy; ASA = acetylsalicylic acid; OAC = oral anticoagulant. Values and percentages are based on the number of cases unless otherwise indicated.
*Percentages based on the number of surgeries performed.
The average cSDH thickness is 10.0 ± 0.5 mm wide with a midline shift of 1.80 ± 0.3 mm (Table 2). The average radiation is 1160 ± 50 mGy, fluoroscopy time is 42.3 ± 3.2 min, and contrast is 32.6 ± 1.4 cc/mL. 50 patients (56%) underwent bilateral embolizations for 72 left-sided cSDH (53%) and 65 right-sided cSDH (47%). 50% (n = 68) and 50% (n = 69) of cases were right and left embolizations, respectively. Embolization using liquid agents was performed in the proximal anterior MMA branch in 19 (14%), posterior in 16 (12%), and both in 102 (74.5%) cases. Utilization of only proximal coil embolization without liquid embolysate was performed in 17 cases (12%). For the remainder of embolizations with coils and adjunctive liquid embolysate (n = 120 cases, 88%): onyx in 33 (27.5%) cases, glue in 87 (72.5%) cases, 48 (55%) of which were glue 10:1 hyperdilution. Evidence of liquid embolic penetration into the contralateral MMA was present in 38 (28%) cases, liquid embolic penetration into the falx in 31 (23%) cases, and evidence of critical ophthalmic (n = 46, 34%) and petrous (n = 41, 30%) intracranial anastomoses (Table 2).
Radiographic, procedural, and safety characteristics of the MMA embolizations.
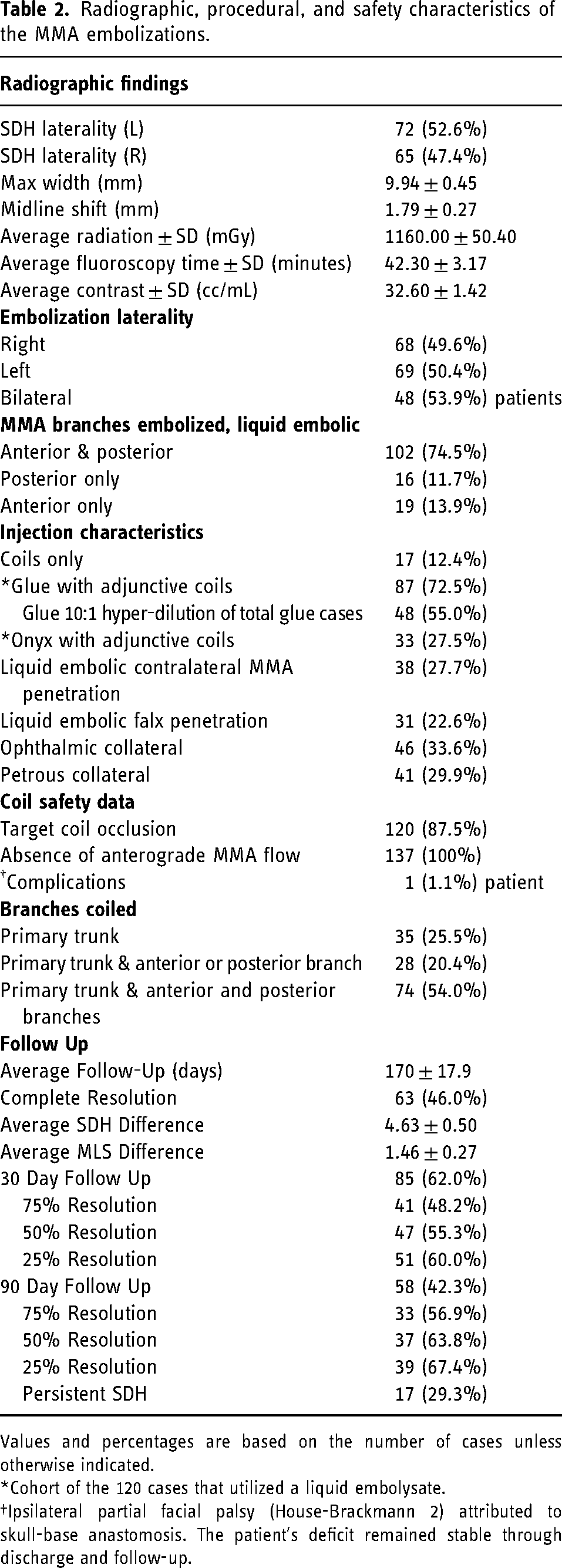
Values and percentages are based on the number of cases unless otherwise indicated.
*Cohort of the 120 cases that utilized a liquid embolysate.
Ipsilateral partial facial palsy (House-Brackmann 2) attributed to skull-base anastomosis. The patient's deficit remained stable through discharge and follow-up.
MMA branches coiled include the primary trunk (25.5%, n = 35), primary and the anterior or posterior branches (20%, n = 28), or primary with the anterior and posterior branches (54%, n = 74) (Table 2). One postprocedural complication was noted, an ipsilateral partial facial palsy (House-Brackmann 2) attributed to non-target embolization of skull-base collaterals. The House-Brackmann scale remained at 2 through discharge and their most recent follow-up 68 days postoperatively. Target coil occlusion was achieved in 88% (n = 120) of cases. In the remaining 12% (n = 17) of cases, coils were placed in the proximal MMA branch without spanning the planum sphenoidale. The absence of anterograde flow in the MMA occurred in 100% (n = 137) of cases. Of the 24 (27%) patients who had follow-up MRA available, 100% showed a lack of recanalization of the MMA. No cases required periprocedural rescue surgery for evacuation of cSDH.
The average length of follow-up was 170 ± 17.9 days, cSDH was 4.55 ± 0.48, midline shift was 0.75 ± 0.15. Sixty-three cases (46%) had complete resolution, and 54 (39%) had >50% cSDH reduction. Of the patients with 90 days or greater follow-up (n = 58), 29% (n = 17) had persistent cSDH at 90 days. None of these patients required repeat procedures as the cSDH was small and they were all asymptomatic. 2
Discussion
In this study, we successfully performed proximal coiling of the MMA to achieve absence of anterograde flow of the proximal trunk. Technical success occurred in 100% of patients. Well over half of cases had dangerous ophthalmic or petrous collaterals present on angiography (Figure 1). Nearly 90% of cases had coils that extended from the planum sphenoidale (as planned) to the foramen spinosum on lateral x-ray projection, and in half of the cases the primary MMA trunk plus the anterior and posterior branches were coiled. The absence of MMA anterograde flow was achieved in all patients. This series represents the largest number of consecutive MMA embolization cases augmented with proximal coils occlusion in the literature.
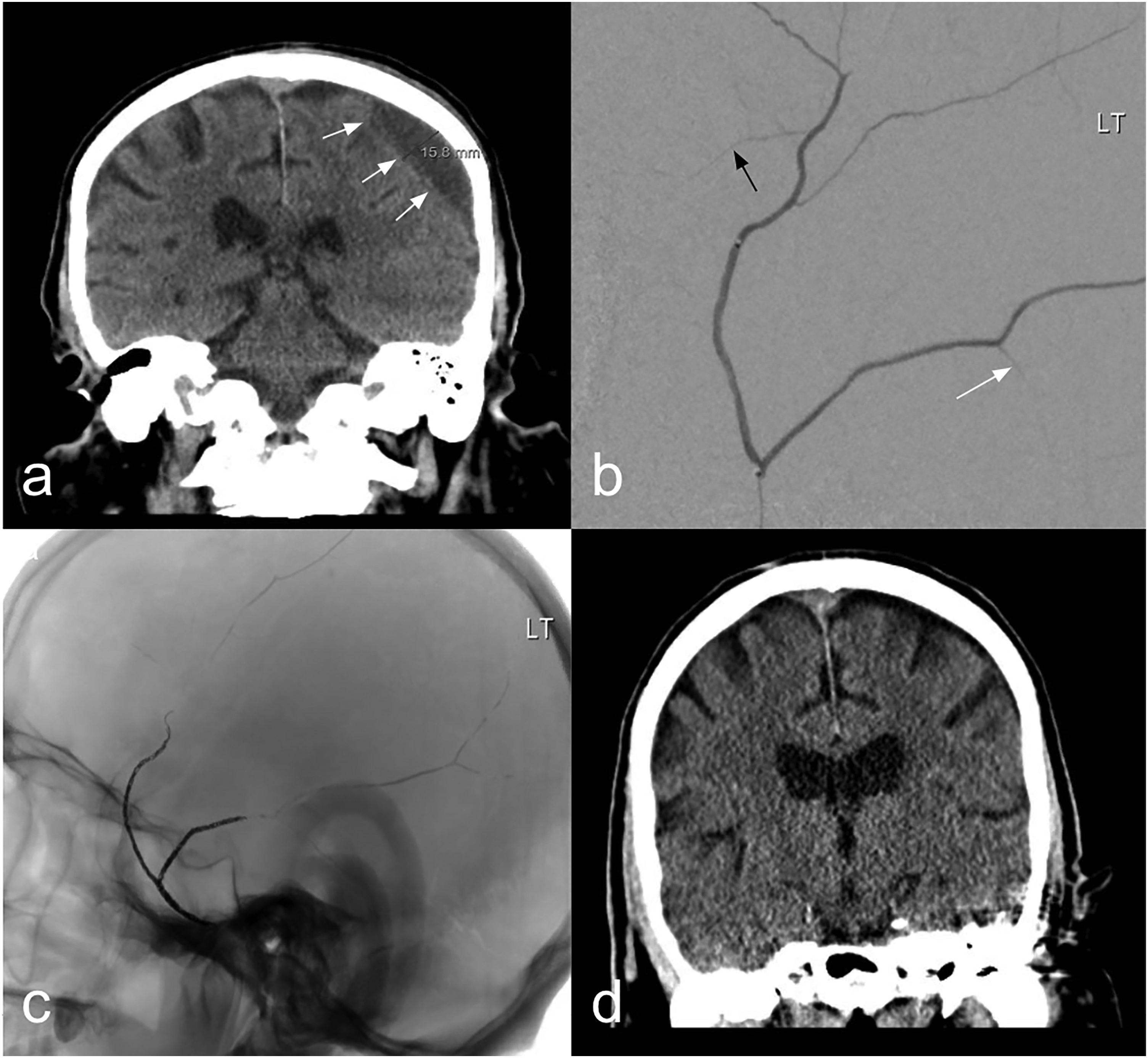
a) Coronal CT demonstrating a preoperative 15.8 mm chronic subdural hematoma (cSDH). (b) Ophthalmic (black) and skull-base temporal bone (white) collateral vessels seen as branches off the anterior and posterior MMA branches, respectively. (c) Native view lateral angiography demonstrating the proximal MMA coiling with distal glue embolization of the anterior and posterior middle meningeal artery branches. (d) Seven-month post-op coronal CT illustrating complete resolution of the cSDH.
cSDH likely forms from injury to dural border cells. 10 Damage to these cells leads to the formation of new capillaries continuous with the dura, which are highly permeable and prone to injury due to thin walls and gap junctions.4,10 A state of persistent capillary injury (with significant dural collateral blood flow) can lead to continued expansion of the cSDH. 4 Embolization of the MMA tree has been demonstrated to be effective for initial and recurrent cSDH management.15–17 In a similar pathophysiology to embolization of arteriovenous malformations, coiling may serve as an additional technical augmentation to avoid liquid embolic reflux into dangerous collaterals by decreasing anterograde flow through the MMA tree during embolic embolization and may reduce recanalization by enhancing thrombosis of the MMA trunk. Current literature for the treatment of arteriovenous malformations (AVMs) has shown embolization of AVMs with balloon-guided glue injection assists by reducing inflow into the AVM, while suppression of flow enhances penetration of a liquid embolic injection into upstream vessel helping to reduce AVM recurrence. 18 Proximal flow control has also been seen to aid in reduction of undesirable reflux of the liquid embolic agent. 18 In a similar approach, reduction of flow to the MMA tree during MMA embolization may also enhance embolization. Proximal coiling may improve distal liquid embolic penetration, reduces undesirable reflux into dangerous collaterals, and reduces recanalization by promoting blood stasis to enhance thrombosis of the MMA trunk.
A primary advantage of adjunctive proximal coiling with liquid agents over liquid embolics alone is the ability to avoid embolizing dangerous ophthalmic and skull-base collaterals.5,8 NTE may lead to lasting cranial nerve and/or visual deficits. In our study, ophthalmic or petrous collaterals were present in 34% and 30% of cases, respectively (Figure 1). Presence of these collaterals requires embolization of the MMA trunk with proximal coiling to protect these branches. One complication, an ipsilateral partial facial palsy (House-Brackmann 2), occurred and was attributed to an aggressive glue injection in the posterior division with temporal penetration early in our experience. The patient's neurological status (House-Brackmann 2) remained stable through discharge and follow-up. No complications were reported in this series in coil-only MMA embolizations.
Previous reports of MMA embolization with proximal coils are sparse. Joyce et al. report their multi-center experience of 6 (4%) successful cases of coil-only MMA embolization in the elderly without lasting complications. 19 Madison et al. report a successful pediatric case of coil-only MMA embolization due to robust ophthalmic collaterals. 20 One case series performed coil-only MMA embolization in 12 cases and demonstrated coils to be as safe as particle embolization. 12 There have also been uses of coil-only embolization for traumatic arteriovenous fistulas involving the MMA.21,22 Other studies suggest additional uses for proximal coiling including blocking distal flow of the embolysate, 23 preventing liquid agent reflux through collateral vessels, 24 and enhancing the quantity of embolysate administered to the distal vasculature. 25 Overall, this literature supports our belief that adjunctive proximal coiling avoids embolizing dangerous MMA collaterals with a liquid embosylate to avoid cranial nerve or visual deficits. This series with its 137 embolizations represents a significant contribution to the literature on the safety of proximal MMA coil embolization.
The collateral nature of the two MMA branches requires the embolization of the primary trunk to prevent hypertrophy and recanalization of the anterior and posterior MMA branch vessels. To prevent this collateral neovascularization, 120 out of 137 cases had coils extending from the planum sphenoidale to the foramen spinosum (Figure 1). Coiling to the planum sphenoidale prevents liquid embolic penetration of dangerous collaterals that exist in this region including ophthalmic, petrous, and pial arteries. 26 Target coiling was performed to the foramen spinosum proximally because this is the opening to the intracranial origin of the MMA. 26 Furthermore, nearly 75% of cases involved coiling the primary trunk with at least the anterior or posterior branch. Proximal coiling disrupts the two primary MMA trunks that supply various collaterals, making future collateral recruitment less likely.12,27 These interventions allowed for the absence of MMA anterograde flow to be achieved in 100% of cases.
Further post-embolization treatment may be required due to the recanalization of MMA vessels. 9 This may be due to the collateralized nature of the MMA tree or due to insufficient embolization. Liquid embolic agents have a particularly high rate of recanalization at 3.5%. 9 A recent study has attempted to decrease the recanalization rate with liquid agents by first dilating the MMA vasculature with coils and then injecting a liquid agent. 25 However, this technique still risks NTE of dangerous MMA collaterals. To decrease recanalization rates in the current study, we placed adjunctive coils from the planum sphenoidale to the foramen spinosum in 120 cases and coiled the primary MMA trunk with the anterior and posterior branches in 74 cases. This allowed us to achieve the absence of anterograde flow MMA in 137 cases with the intent to prevent long-term recanalization. Additive proximal coiling incorporates the penetration of liquid embolics with the added safety of preventing NTE. We hope in future analyses to determine the realized efficacy benefits of this presumed more robust MMA occlusion.
Limitations
The present analysis was a nonrandomized, retrospective study of a prospectively maintained database that only allows for correlations. Although we demonstrated immediate procedural safety of adjunctive proximal coil occlusion of the MMA, the long-term efficacy and safety of this remain unknown. Furthermore, chronic recanalization of the MMA is a poorly understood process. Although the absence of anterograde MMA flow achieved in this study is hypothesized to lend to greater long-term MMA occlusion rates, only longer-term follow-up will be able to demonstrate its efficacy for certain.
Conclusion
Additive proximal coil MMA embolization with liquid embolics is a safe treatment option for cSDH. Further prospective studies are warranted to determine the optimal management for MMA embolization.
Footnotes
Contribution statement
JKC and BMM contributed equally to this work. JKC, BMM, DAZ, and MWK assisted with the conception and design of the manuscript. JKC, BMM, and DAZ drafted and revised the manuscript for important intellectual content. JKC, BMM, JCCdB, and GA assisted with the data acquisition and analysis. MA, KG, NBB, MTB, GPC, LML, and ALC reviewed the important intellectual content presented in the manuscript. ALC and MWK performed treatment procedures and critically revised the important intellectual content. All authors read and approved the final manuscript.
Data sharing statement
There is no additional unpublished data from this study.
Declaration of conflicting interests
Alexander L. Coon, M.D. is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Stryker Neurovascular, Cerenovus, Rapid Medical, Avail MedSystems, Imperative Care, Deinde, InNeuroCo, Q'apel; a proctor for MicroVention-Termo, Stryker Neurovascular, and Medtronic Neurovascular. Li-Mei Lin, M.D. is a proctor for Medtronic Neurovascular, Stryker Neurovascular, MicroVention-Terumo; and a consultant for Medtronic Neurovascular, Stryker Neurovascular, MicroVention-Terumo, Rapid Medical, and Balt. Geoffrey P. Colby, M.D., PhD is a consultant for Medtronic Neurovascular, MicroVention-Terumo, Rapid Medical, Cerenovus, and Stryker Neurovascular. Matthew Bender, M.D. is a proctor Stryker Neurovascular. Narlin B. Beaty, M.D. is a proctor for Medtronic Neurovascular, Stryker Neurovascular, and CMO of NeuroMedica. All other authors have no conflict of interest. No author received financial support in conjunction with the generation of this submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
