Abstract
Background
Predicting outcome after endovascular treatment for acute ischemic stroke is challenging. We aim to investigate differences between predicted and observed outcomes in patients with acute ischemic stroke treated with endovascular treatment and to evaluate the performance of a validated outcome prediction score.
Patients and methods
MR PREDICTS is an outcome prediction tool based on a logistic regression model designed to predict the treatment benefit of endovascular treatment based on the MR CLEAN and HERMES populations. ESCAPE-NA1 is a randomized trial of nerinetide vs. placebo in patients with acute stroke and large vessel occlusion. We applied MR PREDICTS to patients in the control arm of ESCAPE-NA1. Model performance was assessed by calculating its discriminative ability and calibration.
Results
Overall, 556/1105 patients (50.3%) in the ESCAPE-NA1-trial were randomized to the control arm, 435/556 (78.2%) were treated within 6 h of symptom onset. Good outcome (modified Rankin scale 0–2) at 3 months was achieved in 275/435 patients (63.2%), the predicted probability of good outcome was 52.5%. Baseline characteristics were similar in the study and model derivation cohort except for age (ESCAPE-NA1: mean: 70 y vs. HERMES: 66 y), hypertension (72% vs. 57%), and collaterals (good collaterals, 15% vs. 44%). Compared to HERMES we observed higher rates of successful reperfusion (TICI 2b-3, ESCAPE-NA1: 87% vs. HERMES: 71%) and faster times from symptom onset to reperfusion (median: 201 min vs. 286 min). Model performance was good, indicated by a c-statistic of 0.76 (95%confidence interval: 0.71–0.81).
Conclusion
Outcome-prediction using models created from HERMES data, based on information available in the emergency department underestimated the actual outcome in patients with acute ischemic stroke and large vessel occlusion receiving endovascular treatment despite overall good model performance, which might be explained by differences in quality of and time to reperfusion. These findings underline the importance of timely and successful reperfusion for functional outcomes in acute stroke patients.
Introduction
Endovascular treatment (EVT) is an established treatment option for patients with acute ischemic stroke and large vessel occlusion (LVO) and substantially improves the outcome post stroke. In a meta-analysis of five randomized controlled trials testing EVT vs. standard of care, 46% of patients in the intervention arm achieved good functional outcomes defined as a modified Rankin Scale (mRS) 0–2 at day 90. 1 However, predicting post-stroke outcome for an individual stroke patient is still an ongoing challenge. Not only treatment itself but also additional clinical and procedural variables such as age, stroke severity, premorbid conditions, or time from onset to treatment influence functional outcome in an individual stroke patient.2,3
Taking these variables into account, the MR PREDICTS tool has been developed to predict outcomes in patients with ischemic stroke. 4 The MR PREDICTS model aims to evaluate a patient's potential benefit with EVT based on clinical and radiological characteristics available on initial assessment in the emergency department and to support clinical decision making in the acute stroke setting.4,5 Model derivation was based on the study population of MR CLEAN 6 and was further updated on the HERMES dataset, using pooled data from six randomized controlled trials. 7
Since the implementation of EVT for acute stroke with LVO in routine clinical care based on the results of the aforementioned randomized controlled trials in 2015, there has been increasing experience and technical advances in the field of EVT. 8 We hypothesize, that these improvements might have further increased chances of good functional outcome in patients with acute ischemic stroke. This could lead to an underestimation of good functional outcomes and treatment benefit based on currently available outcome prediction tools. ESCAPE-NA1 is a recent acute ischemic stroke trial comprising 1105 patients receiving EVT and either the intravenous neuroprotectant nerinetide or placebo. 9 We applied the MR PREDICTS model to this recent large dataset of acute stroke patients treated with endovascular thrombectomy and aimed to compare the predicted and observed outcome at 3 months post-stroke and to assess model performance of the MR PREDICTS tool in the ESCAPE-NA1 dataset.
Methods
Study population—ESCAPE-NA1
ESCAPE-NA1 is a multicenter, randomized, double-blinded, placebo-controlled trial investigating the effect of intravenous nerinetide vs. placebo on functional outcomes in patients with acute ischemic stroke due to LVO. Overall, 1105 patients were enrolled between March 2017 and August 2019. Eligibility was defined by age (>18 years), baseline National Institute of Health Stroke Scale (NIHSS, >5), premorbid functional independence (Barthel-Index >90), and time from stroke onset (enrollment within 12 h of symptom onset). Proximal intracranial vessel occlusion (intracranial ICA or M1) and baseline Alberta Stroke Program Early Computed Tomography Score (ASPECTS, limited to 5–10), as well as, moderate-to-good collateral filling were assessed on non-contrast computed tomography (CT) and multiphase CT angiography (mCTA). All patients received EVT. Functional outcome after stroke was measured on the mRS at 90 days. Study approval was provided by local ethics boards; informed consent was obtained from the patient or their legal representative prior to enrollment in the trial. 9
Access to the ESCAPE-NA1 data supporting these findings is planned for the future but is not currently publicly available.
MR PREDICTS
MR PREDICTS is an outcome prediction tool designed to predict functional outcome on the mRS at 90 days for patients presenting with acute ischemic stroke and LVO based on baseline clinical and radiological characteristics. The outcome prediction is based on a multivariable logistic regression model and was initially developed in the MR CLEAN trial population. 5 The derivation cohort consists of 500 acute stroke patients presenting within 6 h of symptom onset with a proximal intracranial vessel occlusion in the anterior circulation (ICA, M1, M2). 6 MR PREDICTS was further updated on the HERMES population and validated in the MR CLEAN registry cohort. 7 The following variables are included in the updated MR PREDICTS model: EVT, age, baseline NIHSS, premorbid mRS, diabetes mellitus, systolic blood pressure (SBP), blood glucose, intravenous thrombolysis, ASPECTS, vessel occlusion location, collateral score and time from onset to groin puncture. Intercepts for each category on the mRS scale are provided by the model. The regression equation as well as the intercepts for the different mRS-categories can be found in the supplementary material. The prediction model is available as a web application via https://mrpredicts.shinyapps.io/temp_1/; an example is provided in the supplementary material (Supplemental Figure I). 10
Data analysis
The analysis in the ESCAPE-NA1 population was limited to patients in the control arm treated within 6 h of symptom onset to account for differences in patient inclusion criteria between trials and the effect of nerinetide. As collateral scoring in the derivation cohort was based on single-phase CTA, collateral score readings in ESCAPE-NA1 were repeated by two readers (M.M., N.S.) based on the first phase of baseline mCTAs. Collaterals were scored on a 4-point scale, ranging from 0 (= absent collaterals) to 3 (= collaterals filling 100% of the occluded territory). 11
Baseline, workflow, and outcome variables for the study population (ESCAPE-NA1 control arm, limited to patients treated within 6 h) were descriptively compared to the HERMES population, 1 which comprises the population of the most recent model update. Probabilities for each mRS-score were calculated for each patient in the study population based on the MR PREDICTS regression equation. 7 For comparison of predicted and observed outcomes we calculated the mean of each category of the predicted mRS and the averaged distribution of the observed mRS at 90 days. Model performance was assessed by calculating discrimination (quantified by Harrel's concordance statistic (c-statistic) 12 ) and calibration (quantified by the calibration intercept and slope, graphically assessed via calibration plots) 13 ; good model performance is characterized by a c-statistic and slope approaching 1, and an intercept close to 0. Discrimination and calibration were calculated for good functional outcome (mRS 0–2) as well as mRS 0–1 and mRS 0–3. To explore the influence of potential explanatory variables for differences in predicted and observed outcomes currently available literature was evaluated for variables associated with functional outcomes in patients with acute ischemic stroke receiving EVT. A list of predictors of outcome is provided in the supplementary material (supplementary table 1). Additionally, all published data on the HERMES population was searched for data availability on these variables. Variables with available data were descriptively compared in the study and model derivation cohort.
Baseline characteristics for the ESCAPE-NA1-control and HERMES trial populations.
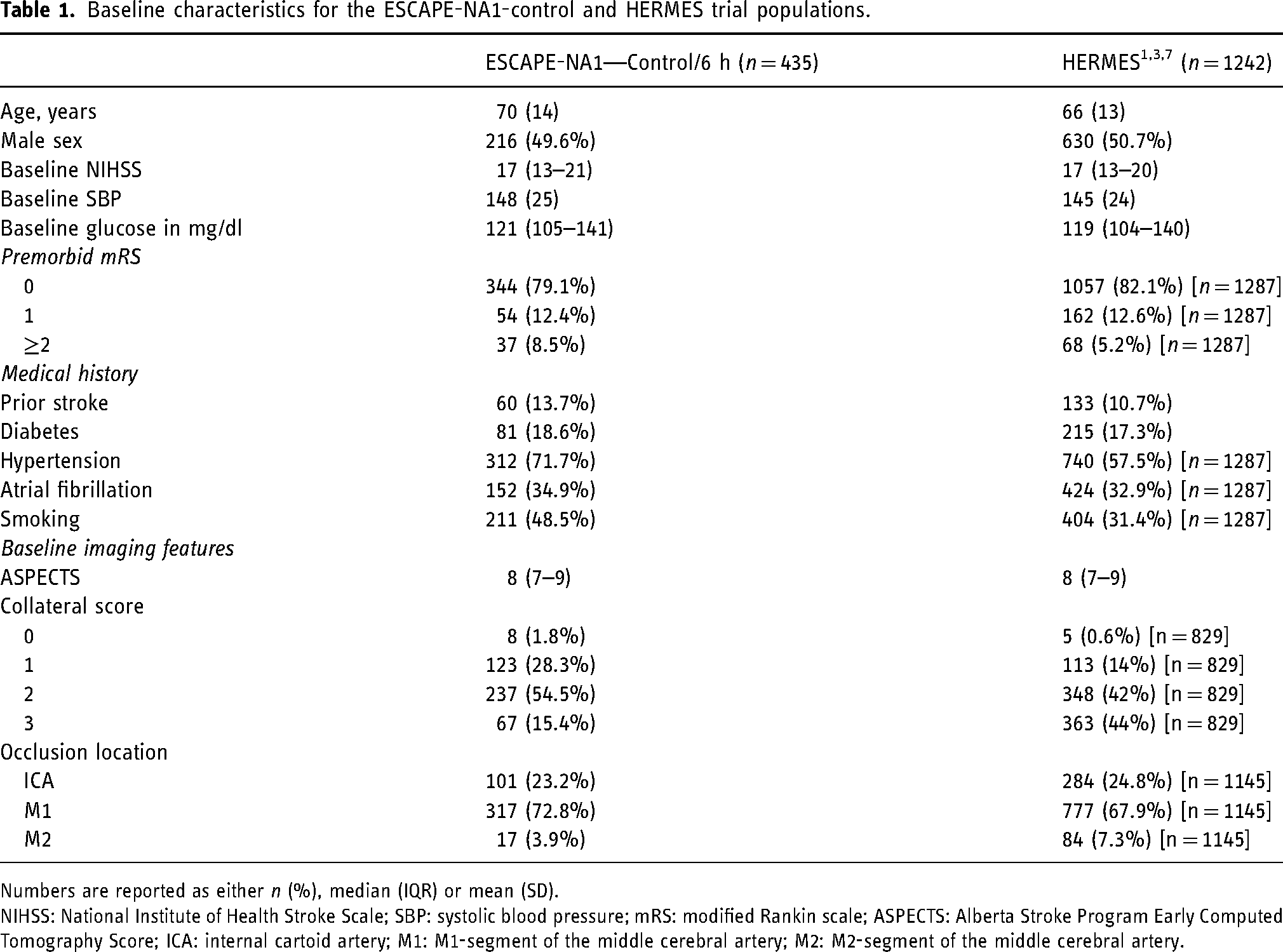
Numbers are reported as either n (%), median (IQR) or mean (SD).
NIHSS: National Institute of Health Stroke Scale; SBP: systolic blood pressure; mRS: modified Rankin scale; ASPECTS: Alberta Stroke Program Early Computed Tomography Score; ICA: internal cartoid artery; M1: M1-segment of the middle cerebral artery; M2: M2-segment of the middle cerebral artery.
Statistical analyses were performed in the statistical environment R (version 3.6.1), missing values for independent variables were imputed using predictive mean matching for numerical variables and polytomous logistic regression for categorical variables embedded in the R package “mice”. 14
Results
Overall, 556 of 1105 (50.3%) patients were randomized to the control arm of the ESCAPE-NA1-trial. After the exclusion of 115 patients with time from onset to groin puncture of more than 6 h and exclusion of 6 patients with missing data on functional outcome at 3 months, 435 patients (78.2%) were included in this analysis. Baseline variables for the analyzed cohort and corresponding numbers for the most recent model update cohort (i.e., HERMES population) are provided in Table 1. In comparison to the HERMES population, patients included in this analysis were older (median age, ESCAPE-NA1: 70 years vs. HERMES: 66 years) and presented more frequently with premorbid disability, history of hypertension and history of smoking. In addition, the number of patients was with good collaterals based on single-phase CTA was lower in the ESCAPE-NA1-population.
Further comparison of our study population to the model update cohort (HERMES population) showed substantial differences in both workflow and radiological outcome parameters (Table 2). Time from onset to reperfusion was faster in the ESCAPE-NA1 cohort compared to the HERMES population (median time from onset to reperfusion: ESCPAE-NA1: 201 min vs. HERMES: 286 min). Time from onset to door was comparable in both cohorts (ESCAPE-NA1: 92 min vs. HERMES: 99 min), whereas procedural times (e.g., time from groin puncture to reperfusion) were faster in the ESCAPE-NA1 population (Table 2). Rates of successful reperfusion, defined as TICI 2b-3, were higher in the ESCAPE-NA1 population (final TICI 2b-3: ESCAPE-NA1: 87% vs. HERMES: 71%).
Workflow and outcome characteristics within EVT populations in ESCAPE-NA1-control and HERMES.
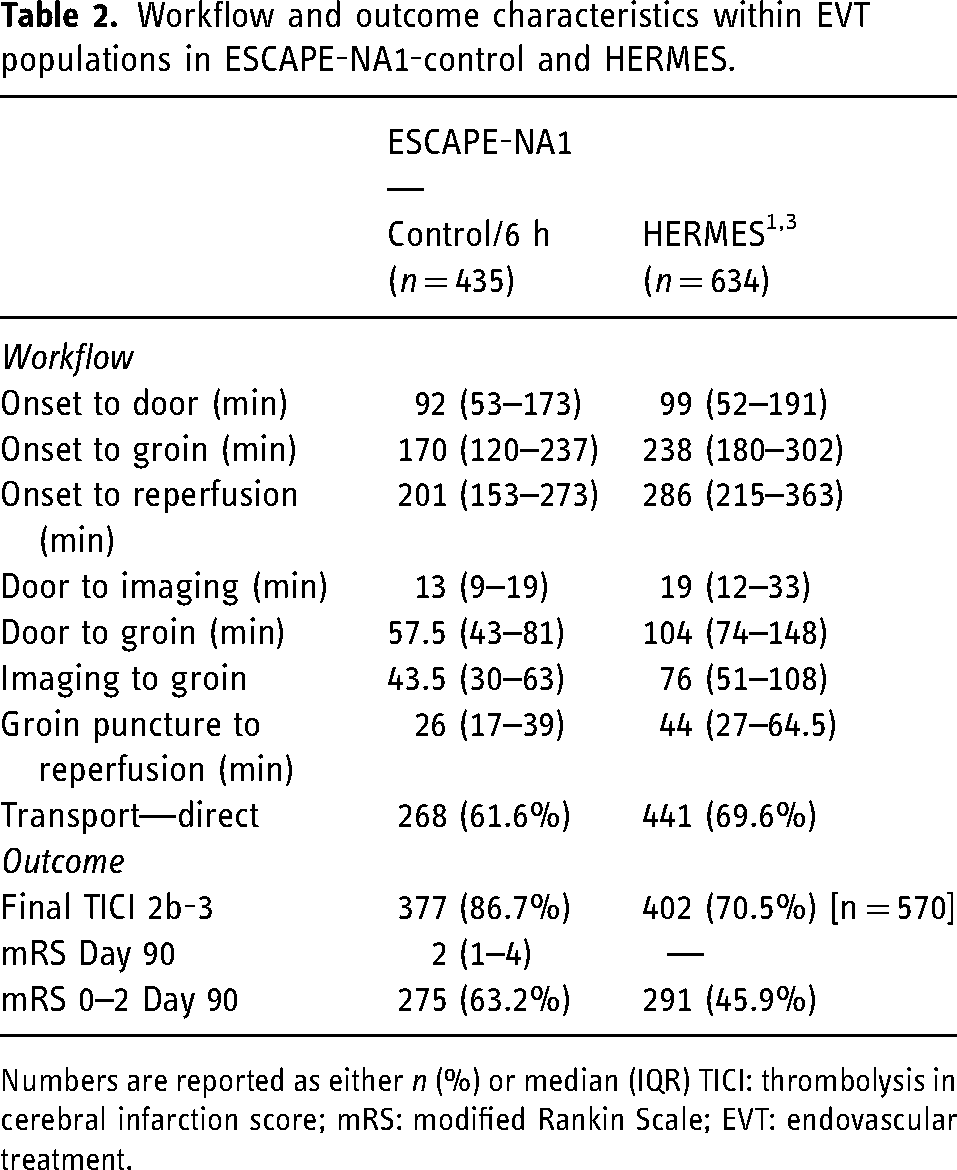
Numbers are reported as either n (%) or median (IQR) TICI: thrombolysis in cerebral infarction score; mRS: modified Rankin Scale; EVT: endovascular treatment.
Good functional outcome was achieved in 275 of 435 (63.2%) patients in the study population. In comparison, the mean predicted probability for good functional outcome as per MR PREDICTS was 52.5% for the overall study cohort, resulting in an absolute difference of 10.7% between predicted and observed outcomes. Poor functional outcome and mortality (mRS 5–6) occurred in 21.1% of the study population compared to a mean predicted probability of 15.1%. The averaged distribution of predicted probabilities and observed outcomes in our study population per mRS-category can be found in Figure 1.
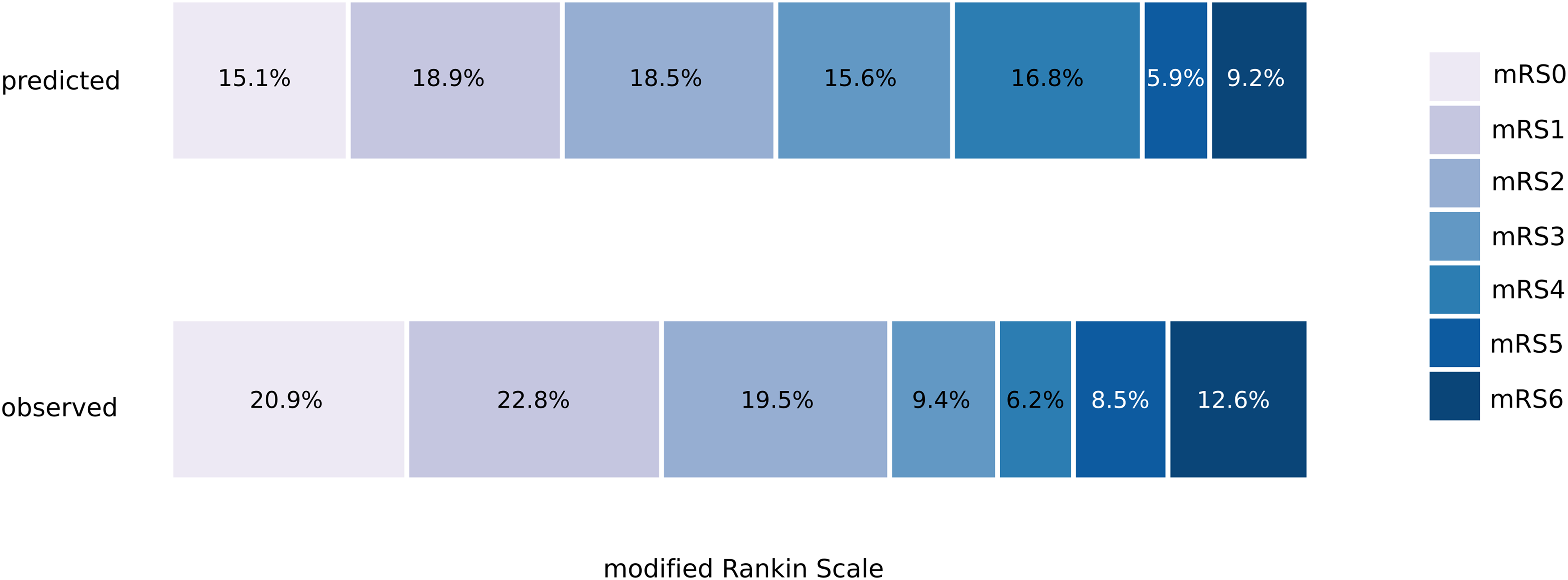
Predicted vs. observed outcome. Averaged predicted probabilities for 90-day modified Rankin scale (mRS) scores and distribution of the observed functional outcome at 90 days for patients enrolled in the control arm of ESCAPE-NA1 treated within 6 h of symptom onset.
Evaluation of model performance in predicting good functional outcome (mRS 0–2) resulted in a c-statistic of 0.76 (95%CI 0.71–0.81) with a calibration slope of 1.00 (95%CI 0.76–1.23) and an intercept of 0.56 (95%CI 0.34–0.77, Figure 2). Performance for mRS 0–1 resulted in similar parameters, whereas performance parameters improved for mRS 0–3 (mRS 0–1: c-statistic 0.75 (95%CI 0.70–0.79), slope 0.97 (95%CI 0.73–1.20), intercept 0.51 (95%CI 0.30–0.72) and mRS 0–3: c-statistic 0.77 (95%CI 0.71–0.82), slope 0.99 (95% CI 0.75–1.23), intercept 0.27 (95%CI 0.04–0.51); Supplemental Figures II and III).
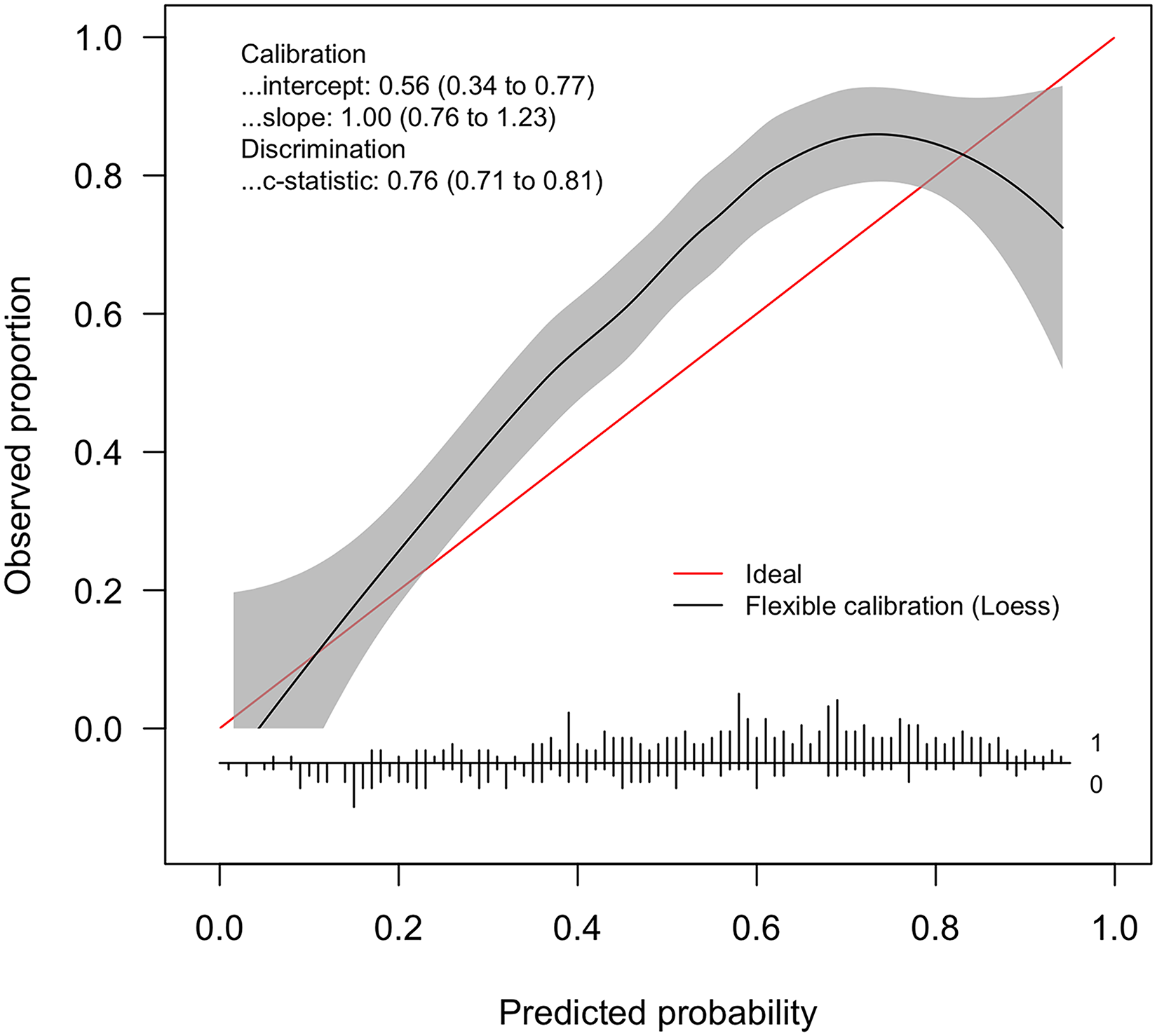
Calibration plot for good functional outcome (mRS 0–2). Distribution of predicted probabilities vs. observed proportions of mRS 0–2; the bar chart indicates patients with (1) and without (0) the outcome of mRS 0–2.
Discussion
Prediction of functional outcome in our study population of patients with acute ischemic stroke and LVO based on a validated prediction model showed good model performance, but a substantial difference between predicted and observed outcome. Probabilities of good functional outcomes as well as the risk of mortality and vegetative state were underestimated by the model.
Overall, 63.2% of patients enrolled in the control arm of ESCAPE-NA1 and treated within 6 h of symptom onset achieved good functional outcomes (mRS 0–2) at 90 days. In comparison, pooled data from five randomized trials showed good functional outcomes in 46% of patients receiving EVT. 1 More recent randomized controlled trials enrolling specific subgroups of patients with acute ischemic stroke and LVO (e.g., patients with late presentation) report similar results on functional outcome with EVT.15,16 The applied prediction tool underestimated both probabilities of good and poor functional outcomes in our study cohort. This finding underlines the complexity and limitations of prediction tools. At the time of applying the prediction model—i.e., in the setting of an acute presentation of a patient with ischemic stroke—the available information is restricted to the patient's baseline characteristics. However, there are various additional factors apart from baseline characteristics that potentially influence functional outcome in a patient with acute ischemic stroke but are not known at the time of presentation and initial decision making (e.g., quality of reperfusion, time to reperfusion). Consequently, the influence of these variables on outcome is not covered by prediction tools that are based on information available at the time of acute presentation. Furthermore, factors such as differences in the system of care or patient selection are known to affect model calibration and performance. 17 As an example, differences in patient selection based on radiological characteristics such as collateral status in the trials compared in this analysis might have affected model performance. To account for these differences in patient populations, model updating is an important next step to obtain reliable estimations in a particular setting.
In our study cohort successful reperfusion, defined as TICI 2b-3, was achieved in 87% of included patients. The number of successful reperfusions in our cohort is comparable to those reported in recent trials testing different technical approaches for thrombectomy—the ASTER and COMPASS trial both reported TICI 2b-3 in more than 80% of all patients.18,19 Additionally, registry-based data show similar rates of successful reperfusion in patients with LVO and EVT with stent retrievers (87.9% TICI 2b-3 with 56.6% good functional outcome in the STRATIS registry). 20 However, in comparison to the prediction model derivation cohort, we observed higher rates of successful reperfusion in our study population (ESCAPE-NA1: 87% vs. HERMES: 71% TICI 2b-3). Information on the quality of reperfusion is not available at the baseline assessment of an acute stroke patient but is known to be associated with good functional outcome post-EVT in acute stroke patients.21,22 The difference in frequencies of successful reperfusion between the model derivation cohort and our study population might have therefore contributed to the differences between predicted and observed outcomes.
In addition to the quality of reperfusion, minimizing time from symptom onset to reperfusion is critical in achieving good outcome after ischemic stroke. 3 Secondary analyses from randomized controlled trials showed the importance of time to treatment and its influence on functional outcome, with every hour of delay substantially reducing the probability of functional independence. 23 Especially, intra-hospital delays in treatment have been found to be an important predictor of poor outcomes in patients receiving EVT. 24 Different strategies to improve workflow, such as early notification or direct transfer of suspected stroke patients to imaging or the angio-suite have been shown to reduce times to treatment and improve outcome 25 and there is data showing improvement in treatment times over the last years. 8 In our study cohort, time from symptom onset to reperfusion was overall 85 min shorter compared to the HERMES population. Time intervals from symptom onset to door (i.e., presentation to the hospital) are comparable between these two cohorts whereas in-hospital and procedural times were shorter, suggesting improvement mainly after a patient's presentation to the hospital. This finding could be partially explained by lower numbers of transferred patients in the ESCAPE-NA1 population but also indicates improvement of in-hospital workflow and procedure duration. Comparable to quality of reperfusion, information on time to reperfusion is not available at the initial presentation of an acute stroke patient, but is known to substantially influence functional outcome. 3 Improvements in treatment times in our study population compared to the model update cohort might also contribute to better outcomes than expected based on the prediction model.
Limitations
This study has several limitations. First, we restricted our patient sample to patients enrolled in the control arm of the ESCAPE-NA1 trial and patients treated within 6 h which resulted in a limited amount of available patient data. However, this step was taken to avoid a potential influence of treatment with nerinetide and to comply with inclusion criteria for the initial model derivation cohort. Second, apart from quality of reperfusion and speed of reperfusion there are additional variables that potentially influence functional outcome but were not available for our analysis. Some of these additional variables are known but not measured (e.g., accessibility and quality of rehabilitation, management of complications, secondary stroke prevention), others might be completely unknown, and we were therefore not able to analyze their potential influence on functional outcome.
Conclusion
Outcome prediction based on a prediction model using baseline clinical and radiological variables in a recent cohort of patients with large vessel acute ischemic stroke receiving EVT underestimated probabilities of good functional outcome even though model performance measures indicate overall good discrimination and calibration. Our findings underline the complexity of prediction tools applied in the acute stroke setting based on limited availability and information of patient characteristics at the time of application. Substantial differences both in quality of reperfusion as well as time to reperfusion between the model derivation cohort and our study population could explain the discrepancy between predicted and observed outcomes and support the importance of timely and successful reperfusions. The knowledge of these additional patient characteristics throughout the course of stroke treatment and their implementation in updated prediction models is needed to further improve outcome prediction of patients with acute ischemic stroke and LVO.
Supplemental Material
sj-docx-1-ine-10.1177_15910199231221491 - Supplemental material for Predicting outcome in acute stroke with large vessel occlusion—application and validation of MR PREDICTS in the ESCAPE-NA1 population
Supplemental material, sj-docx-1-ine-10.1177_15910199231221491 for Predicting outcome in acute stroke with large vessel occlusion—application and validation of MR PREDICTS in the ESCAPE-NA1 population by Martha Marko, Mayank Goyal, Johanna M Ospel, Nishita Singh, Esmee Venema, Raul G Nogueira, Andrew M Demchuk, Ryan A McTaggart, Alexandre Y Poppe, Bijoy K Menon, Charlotte Zerna, Maxim Mulder, Diederik WJ Dippel, Hester F Lingsma, Bob Roozenbeek, Michael Tymianski and Michael D Hill in Interventional Neuroradiology
Footnotes
Author contribution
MM contributed to the conceptualization and statistical analysis, and drafted the manuscript. MG, JMO, NS, EV, and MDH contributed to the conceptualization, analysis, and critical revision of the manuscript. The remaining authors contributed to the critical revision of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG: personal fees from Mentice, Medtronic, Microvention, Stryker; patent to systems of acute stroke diagnosis. JMO: funds from the University of Basel Research Foundation, Julia Bangerter Rhyner Foundation, Freiwillige Akademische Gesellschaft Basel. RGN: fees/compensation from Stryker, Medtronic, Cerenovus/Neuravi, Phenox, Anaconda, Genentech, Biogen, Prolong Pharmaceuticals, Brainomix, Viz.ai, Corindus Vascular Robotics, Vesalio, and Ceretrieve; member of the Physician Advisory Board for Cerenovus/Neuravi. ADM: grants from NoNO; honoraria from Medtronic; patent Circle NVI. BKM: shares in Circle NVI; patent for systems of triage in acute stroke. DWJD: grants from Dutch Heart Foundation, Dutch Brain Foundation, The Netherlands Organisation for Health Research and Development, Health Holland Top Sector Life Sciences & Health, AngioCare BV, Medtronic/Covidien/EV3®, MEDAC Gmbh/LAMEPRO, Penumbra, Stryker, Top Medical/Concentric and Thrombolytic Science. MT: CEO of NoNO; patents owned by NoNO. MDH: grants from Canadian Institutes for Health Research, Alberta Innovates, NoNO, Heart & Stroke Foundation of Canada, National Institutes of Neurological Disorders and Stroke, Covidien, Boehringer-Ingleheim, Stryker, and Medtronic; fees from Merck; patent for systems of acute stroke diagnosis; stock in Calgary Scientific. All other authors report no disclosures.
Ethics approval
Study approval for the ESCAPE-NA-1 trial was provided by local ethics boards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from the patient or their legal representative prior to enrollment in the trial.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
