Abstract
Background
The appropriate choice of perioperative sedation during endovascular thrombectomy for ischemic stroke is unknown. Few studies have evaluated the role of nursing-administered conscious sedation supervised by a trained interventionalist.
Objective
To compare the safety and efficacy of endovascular thrombectomy for ischemic stroke performed with nursing-administered conscious sedation supervised by a trained interventionalist with monitored anesthesia care supervised by an anesthesiologist.
Methods
A retrospective review of a prospectively collected stroke registry was performed. The primary outcome was functional independence at 90 days, defined as a modified Rankin score of 0–2. Propensity score matching was performed to control for known confounders including patient comorbidities, access type, and direct-to-suite transfers.
Results
A total of 355 patients underwent endovascular thrombectomy for large vessel occlusion between 2018 and 2022. Thirty five patients were excluded as they arrived at the endovascular suite intubated. Three hundred and twenty patients were included in our study, 155 who underwent endovascular thrombectomy with nursing-administered conscious sedation and 165 who underwent endovascular thrombectomy with monitored anesthesia care. After propensity score matching, there were 111 patients in each group. There was no difference in modified Rankin score 0–2 at 90 days (26.1% vs 35.1%, p = 0.190). Patients undergoing monitored anesthesia care received significantly more vasoactive medications (23.4% vs 49.5%, p < 0.001) and had a lower intraoperative minimum systolic blood pressure (134 vs 123 mmHg, p < 0.046). There was no difference in procedural efficacy, safety, intubation rates, and postoperative complications.
Conclusion
Perioperative sedation with nursing-administered conscious sedation may be safe and effective in patients undergoing endovascular thrombectomy for ischemic stroke.
Introduction
Multiple studies have demonstrated safety and efficacy of endovascular thrombectomy (EVT) in the treatment of anterior circulation large vessel occlusion (LVO) strokes up to 24 h from stroke onset.1,2 The appropriate choice of perioperative sedation and airway management during mechanical thrombectomy, however, is unknown. There is large variability in sedation practice ranging from utilization of local anesthesia only, local anesthesia with nursing-administered conscious sedation (NACS) provided under the supervision of a sedation-trained physician operator, monitored anesthesia care (MAC) administered under the supervision of an anesthesiologist, to the utilization of general endotracheal anesthesia. 3
Decisions on perioperative sedation in patients undergoing emergent mechanical thrombectomy are typically based on institutional resources and availability of providers in an emergent setting. Balancing procedural efficacy and patient safety with efficient resource utilization is especially important as the global burden of acute ischemic stroke and access to EVT increases. 4
While local anesthesia only, MAC, and general endotracheal anesthesia are well studied, few studies evaluate NACS. 5 Our institution transitioned from NACS to MAC in 2020. We performed a single-center propensity score-matched retrospective cohort study of prospectively collected data from our comprehensive stroke center comparing patients from the two-year period prior to and after this institutional protocol change. We hypothesized that MAC would allow for faster reperfusion times and better outcomes compared to NACS.
Methods
Study design and protocol
A retrospective cohort study of a prospectively maintained stroke database of patients who underwent EVT for LVO at a single comprehensive stroke center from 2018 to 2022 was conducted. Approval was obtained from the institutional review board with a wavier for informed consent. The majority of patients who underwent NACS had their procedure between 2018 and 2020 while the majority of patients who underwent MAC had their procedure between 2020 and 2022.
Patients were triaged and identified as candidates for EVT as determined by a non-contrast computerized tomographic (CT) with evidence of a LVO on CT angiogram. Intravenous thrombolytic was administered for eligible patients within the recommended time window as per guidelines. Endovascular thrombectomy was performed by four experienced neuro-endovascular surgeons utilizing stent retrievers and/or aspiration per the physician's discretion. NACS was performed by hospital-trained nursing staff (not a nurse anesthetist) and overseen by the operating surgeon who had institutional privileges for moderate sedation. Nursing staff provided intravenous sedation, typically midazolam and/or fentanyl. The nurse functions under the supervision of a physician with moderate sedation privileges, in this case, the neuro-endovascular physician. MAC was performed by dedicated anesthesia providers supervised by an attending anesthesiologist. Sedative medications utilized by anesthesia providers include intravenous midazolam, fentanyl and in certain cases dexmedetomidine or propofol. Patients from certain “spoke” centers could be transferred directly to the neuro-interventional suite via a direct-to-suite (DTS) protocol, bypassing the emergency department. Patients who were intubated prior to arrival to the neuro-interventional suite were excluded. Blood pressure parameters for both groups were based on intravenous thrombolytic use with a goal of 140–180 mmHg in patients who had received thrombolytics and 140–200 mmHg in those who had not. Medications typically utilized were intravenous labetalol (5–10 mg) and/or hydralazine (5–10 mg) and if needed nicardipine or clevidipine drips (titration to blood pressure parameters). Upon completion of EVT, patients were taken to a neuro-critical care unit under the care of a multi-disciplinary stroke team with a protocolized blood pressure goal based on reperfusion status.
Data collection
Basic demographic information including age, sex, weight, medical comorbidities (history of hypertension, diabetes mellitus, smoking history, atrial fibrillation, or hyperlipidemia), admitting National Institute of Health Stroke Scale (NIHSS), premorbid modified Rankin score (mRS), site of LVO, stroke side, utilization of intravenous thrombolytics, and DTS transfer status were prospectively collected in our stroke center database. Discharge mRS and NIHSS as well as 90-day follow up were prospectively collected by stroke neurologists not involved in the procedure.
Procedural characteristics including utilization of the DTS process, access site, door to puncture time, angiography suite to puncture time, puncture to first pass time, puncture to successful reperfusion (thrombolysis in cerebral infarction (TICI) 2b or 3) time, total procedural time, complications including symptomatic groin hematoma (requiring transfusion, vascular surgery consultation or operative intervention), intracranial vessel perforation, emboli of new territory, and post-operative intracerebral hemorrhage (parenchymal hematoma type 1 (PH1) and type 2 (PH2) based on the Heidelberg Bleeding classification 6 ) were also collected. Procedural times were generally recorded by nursing staff intraoperatively or immediately postoperatively based on puncture time and arterial access closure.
Perioperative characteristics including blood pressure parameters (first blood pressure on arrival to the interventional suite, minimum and maximum systolic blood pressure) and anesthesia complications (loss of airway/conversion to general endotracheal anesthesia), intra-operative use of any vasoactive medications (including norepinephrine, labetalol, hydralazine, etc.) and post-operative pneumonia (defined as any pneumonia requiring antibiotic treatment) were recorded.
Study outcomes
The primary outcome was functional independence at 90 days (as defined by a mRS of 0–2). Prespecified secondary outcomes included successful reperfusion (defined as TICI 2b or 3), discharge disposition (home/rehab versus skilled nursing facility/death), discharge NIHSS, discharge mRS, discharge mRS 0–2, and mRS at 90 days. Procedural data were also examined as secondary outcomes including door-to-groin puncture time, angiography suite to puncture time, puncture to first pass, puncture to successful reperfusion (TICI 2b or 3), total procedural time and safety data including anesthesia and neuro-interventional complications.
Statistical analysis
Demographic characteristics in patients who underwent EVT with NACS and MAC are presented as means with standard deviations or medians with interquartile ranges, proportions for categorical data and medians with ranges for ordinal data. Univariate comparisons were performed with unpaired t-tests for interval data, Fischer's exact or χ2 test for categorical data, and Wilcoxon rank-sum test for ordinal data. Variables were found to be statistically significant with a p value less than 0.05. Characteristics found to be statistically significant and other clinically relevant variables were included in propensity score matching with a tolerance set to 0.1. Analyses were performed using SPSS Statistics (version 29.0).
Results
Patient characteristics
A total of 355 patients underwent EVT for LVO between 2018 and 2022. 35 patients were excluded as they arrived to the endovascular suite intubated. We included 320 patients in our study, 155 who underwent EVT with NACS and 165 who underwent EVT with MAC. Baseline demographics and comorbidities are reported in Table 1.
Demographic and comorbidity information of included patients undergoing EVT for large vessel occlusion.
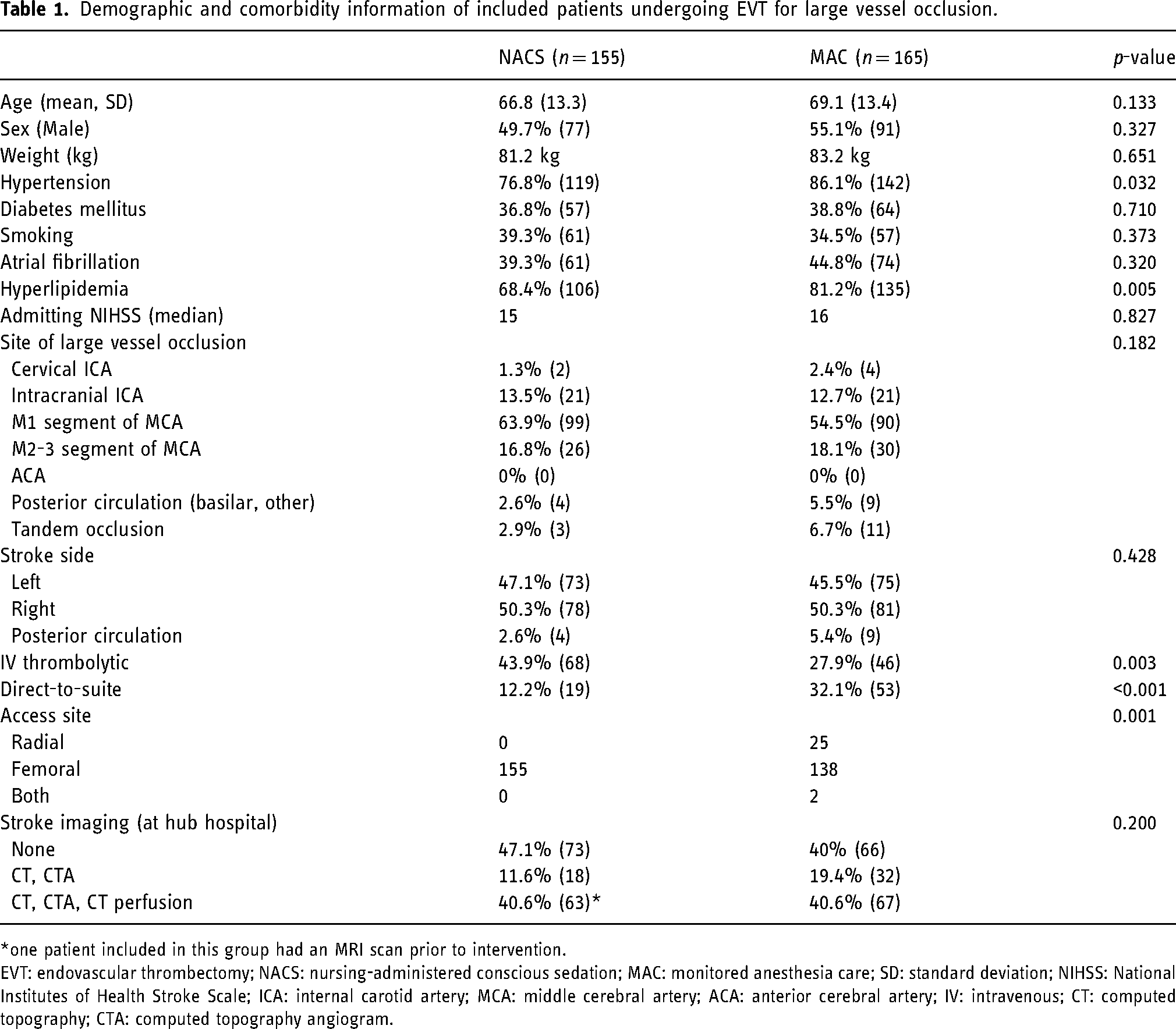
one patient included in this group had an MRI scan prior to intervention.
EVT: endovascular thrombectomy; NACS: nursing-administered conscious sedation; MAC: monitored anesthesia care; SD: standard deviation; NIHSS: National Institutes of Health Stroke Scale; ICA: internal carotid artery; MCA: middle cerebral artery; ACA: anterior cerebral artery; IV: intravenous; CT: computed topography; CTA: computed topography angiogram.
The average age (66.8 in NACS group versus 69.1 in MAC group, p = 0.133) and gender (49.7% in NACS group versus 55.1% male in MAC group, p = 0.327) were similar between the groups. Baseline demographics were similar except for hypertension (76.8% in the NACS group versus 86.1% in MAC group, p = 0.032) and hyperlipidemia (68.4% in NACS group versus 81.2% in MAC group, p = 0.005), which were more common in the MAC group. The median admitting NIHSS was similar in both groups (15 in NACS group versus 16 in MAC group, p = 0.827). There was no difference in the location of LVO, with the majority of strokes occurring in the middle cerebral artery (p = 0.182). There was no difference in the laterality of stroke (p = 0.428). Intravenous thrombolytic administration was higher in the NAC compared to the MAC group (43.9% versus 27.9%, p = 0.003). There were significantly more DTS transfers within the MAC group (32.1% versus 12.2%, p < 0.001). More patients had EVT via a radial artery approach in the MAC group, p = 0.001. There was no difference in the percentage of patients receiving imaging prior to intervention (p = 0.200).
Procedural and outcome data
There was no difference in functional independence at 90 days as defined by mRS of 0–2 (29.7% in NACS group versus 35.2% in MAC group, p = 0.340). A summary of procedural, safety, and outcome measures is reported in Table 2.
Procedural information and outcome data of included patients undergoing EVT for large vessel occlusion.
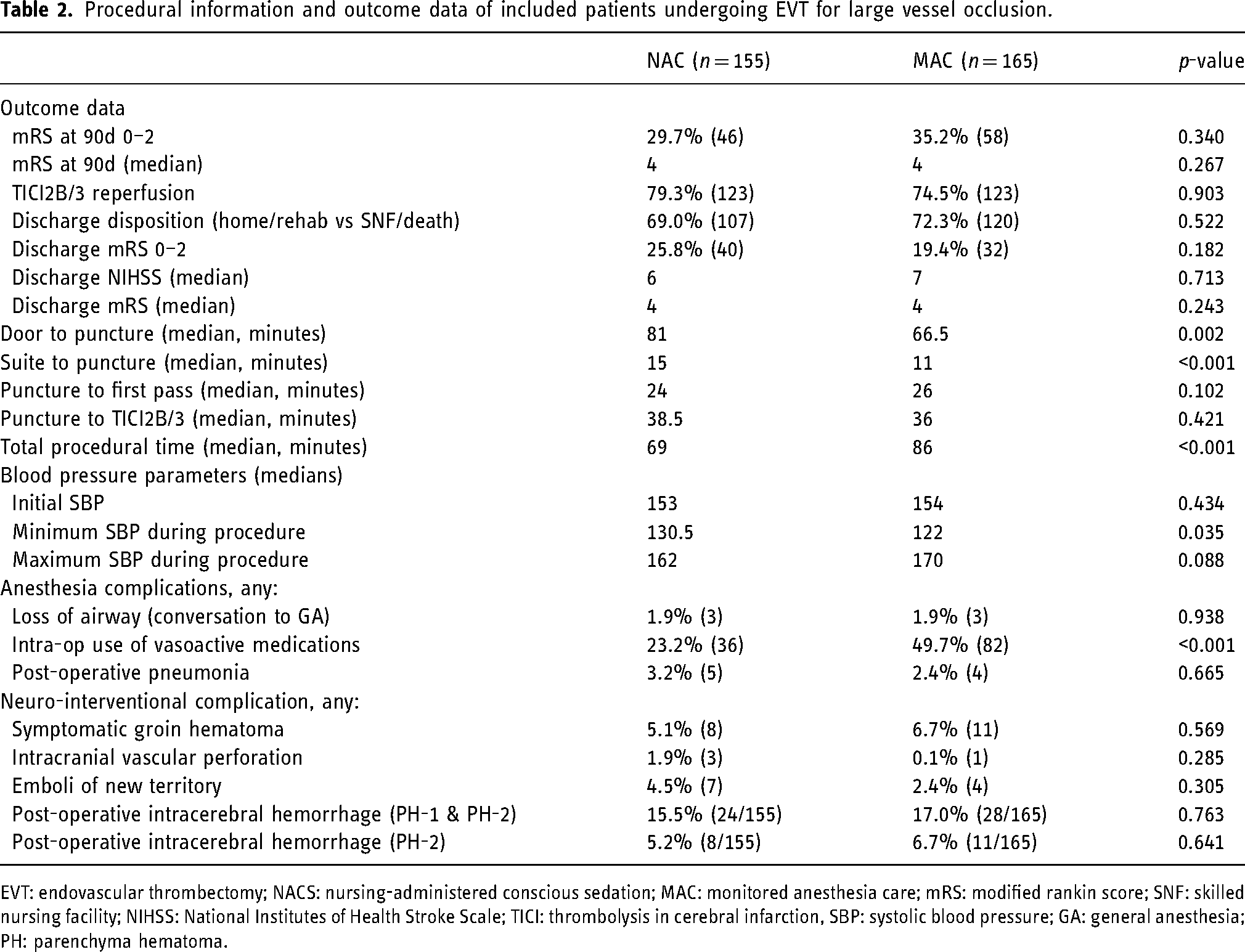
EVT: endovascular thrombectomy; NACS: nursing-administered conscious sedation; MAC: monitored anesthesia care; mRS: modified rankin score; SNF: skilled nursing facility; NIHSS: National Institutes of Health Stroke Scale; TICI: thrombolysis in cerebral infarction, SBP: systolic blood pressure; GA: general anesthesia; PH: parenchyma hematoma.
There was no difference in successful reperfusion (TICI2B or 3), discharge disposition, discharge NIHSS, discharge mRS, functional independence (mRS of 0–2) at discharge or 90-day mRS. Door to groin puncture times were longer in the NACS group (81 versus 66.5 min, p < 0.001). Patients undergoing MAC had significantly faster median angiography suite to puncture times (11 versus 15 min, p < 0.001) but longer overall total procedural times (86 versus 69 min, p < 0.001). There was no difference in puncture to first pass or puncture to successful reperfusion (TICI2B or 3). There was no difference in initial systolic blood pressure (SBP). Patients undergoing MAC had a significantly lower minimum SBP (122 compared to 130.5, p = 0.035) compared to those undergoing NACS. There was no difference in maximum SBP. There was no difference in airway complications including loss of airway (conversion to general endotracheal anesthesia) (p = 0.938) or post-operative pneumonia requiring antibiotics (p = 0.665). Patients who underwent MAC were administered significantly more intra-operative vasoactive medications (49.7% versus 23.2%, p < 0.001). There was no difference in neuro-interventional complications including symptomatic groin hematoma, intracranial vessel perforation, emboli to new territory, or post-operative intracerebral hemorrhage (PH-1 or PH-2).
Propensity score matching
Given significant differences between the cohorts in potential confounding variables, propensity score matching was utilized to balance the cohorts. Matching was performed with a tolerance of 0.1. Variables matched include age, sex, hypertension, hyperlipidemia, admitting NIHSS, site of LVO, stroke side, administration of IV thrombolytics, DTS status, and access site. Matching appropriately balanced variables with a 30.6% reduction in the overall cohort (222 from 320 patients). Demographic and comorbidity data of the propensity score matched cohort are reported in Table 3.
Demographic and comorbidity information of included patients undergoing EVT for LVO after propensity score matching.
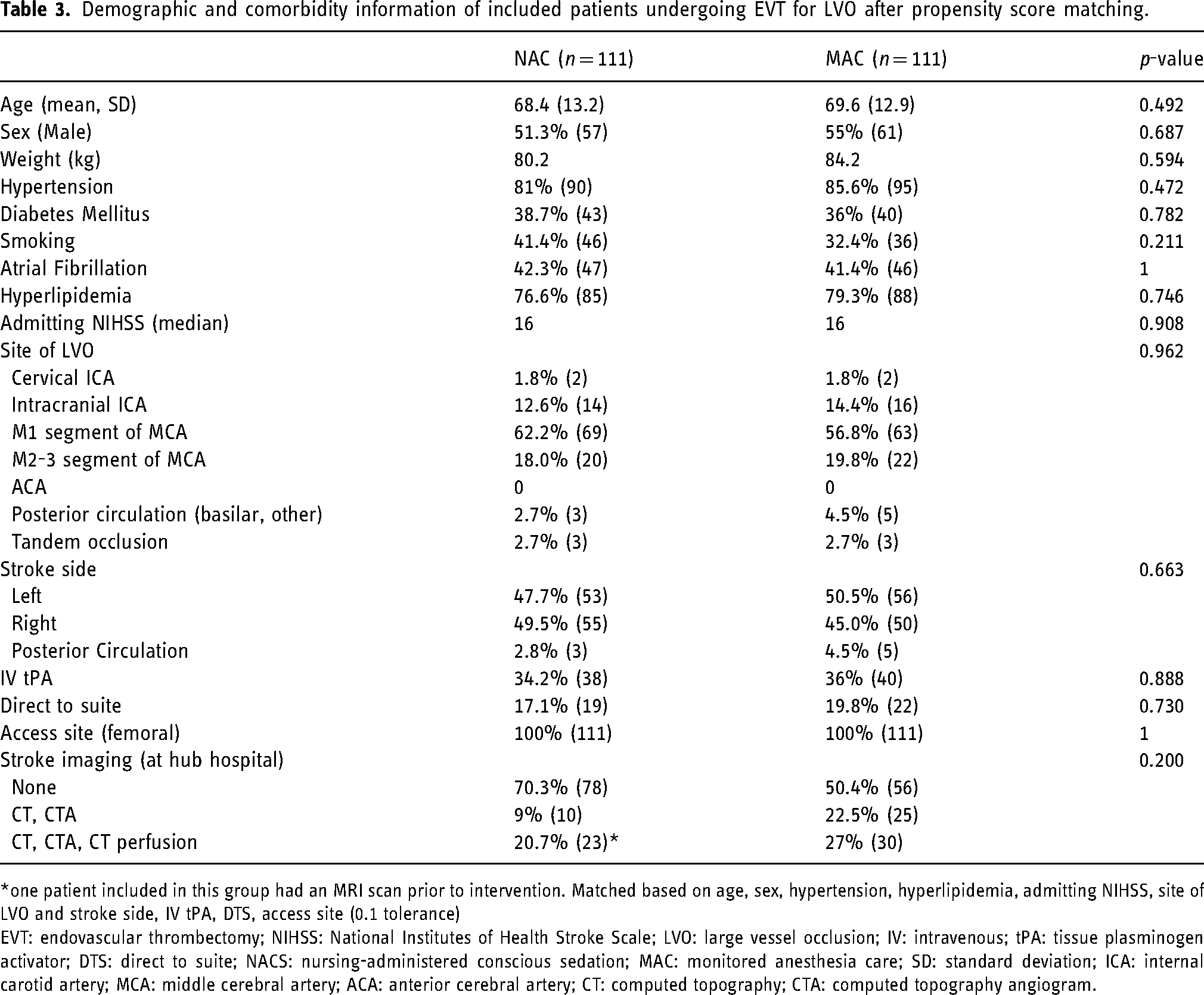
one patient included in this group had an MRI scan prior to intervention. Matched based on age, sex, hypertension, hyperlipidemia, admitting NIHSS, site of LVO and stroke side, IV tPA, DTS, access site (0.1 tolerance)
EVT: endovascular thrombectomy; NIHSS: National Institutes of Health Stroke Scale; LVO: large vessel occlusion; IV: intravenous; tPA: tissue plasminogen activator; DTS: direct to suite; NACS: nursing-administered conscious sedation; MAC: monitored anesthesia care; SD: standard deviation; ICA: internal carotid artery; MCA: middle cerebral artery; ACA: anterior cerebral artery; CT: computed topography; CTA: computed topography angiogram.
After propensity score matching, there was no difference in functional independence at 90 days (35.1% in MAC group versus 26.1% in NACS group, p = 0.190). There was no difference door to puncture time (76 versus 69 min, p = 0.271). There was a significant difference in time from angiography suite to puncture time (15 versus 12 min, p < 0.001). There was no difference in median puncture to first pass or puncture to successful reperfusion (TICI2B or 3) times. Total median procedural time was longer in the MAC group (88 versus 72 min, p = 0.002).
There was no difference in successful reperfusion (TICI2B or 3), discharge disposition, discharge NIHSS, discharge mRS, or functional independence at discharge (mRS 0–2). A summary of the procedural, outcome and safety data of the propensity score matched cohort is presented in Table 4.
Procedural information and outcome data of included patients undergoing EVT for large vessel occlusion after propensity score matching.
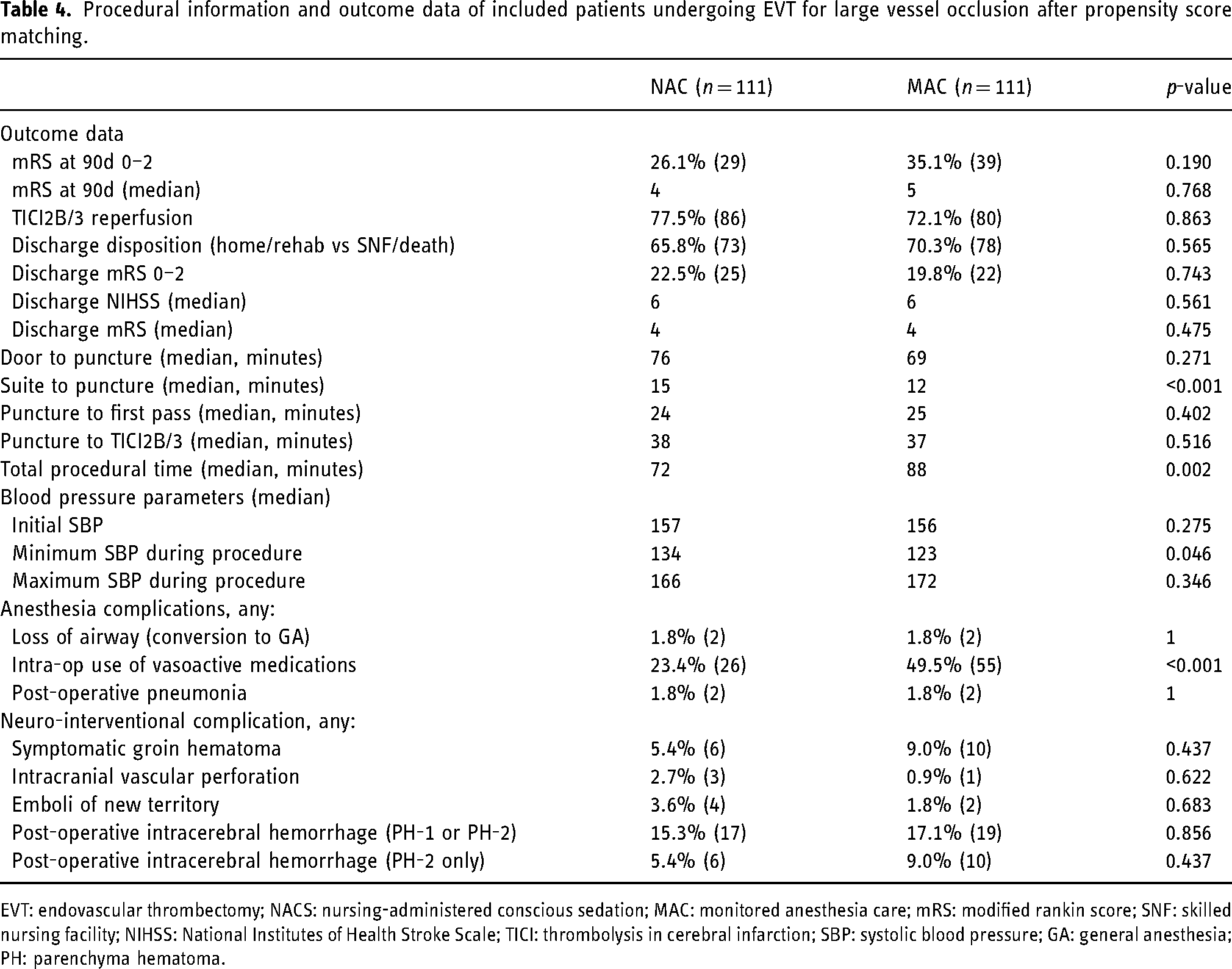
EVT: endovascular thrombectomy; NACS: nursing-administered conscious sedation; MAC: monitored anesthesia care; mRS: modified rankin score; SNF: skilled nursing facility; NIHSS: National Institutes of Health Stroke Scale; TICI: thrombolysis in cerebral infarction; SBP: systolic blood pressure; GA: general anesthesia; PH: parenchyma hematoma.
Within the matched cohort, patients undergoing MAC had a significantly lower intra-operative minimum SBP (123 versus 134 mmHg, p = 0.046). There was no difference in initial or maximum SBP. Patients undergoing MAC had significantly higher intra-operative vasoactive medication administration (49.5% versus 23.4%, p < 0.001). There was no difference in loss of airway or post-operative pneumonia. There was no difference in neuro-interventional complications including post-operative intracerebral hemorrhage (PH-1 or PH-2).
Discussion
We found no significant difference in functional independence (mRS of 0–2) at 90 days between patients undergoing EVT for acute ischemic stroke with NACS supervised by a neuro-interventionalist versus MAC administered by an anesthesiologist. Similar results were found after propensity score matching of known confounding variables including age, sex, hypertension, hyperlipidemia, admitting NIHSS, site of LVO, stroke side, utilization of IV thrombolytics, DTS transfers, and access site. Patients who underwent EVT with MAC had significantly faster angiography suite to puncture times though there was no difference in door to puncture or groin to reperfusion times. Safety data including reintubation, post-intervention pneumonia, and reperfusion hemorrhage were similar between the two groups.
Several randomized clinical trials and meta-analyses have compared general endotracheal anesthesia with monitored conscious sedation and show mixed results.7–13 Other studies have compared local anesthesia at the puncture site only with monitored conscious sedation based on institutional/provider preference and have shown that local anesthesia may be preferable.14–17 Few studies have evaluated NACS specifically. Slawski et al. retrospectively compared NACS and MAC in a small cohort of 104 patients who underwent EVT between July 2015 and December 2017 and found similar results to our study with no difference in functional outcomes (mRS of 0–2) at 90 days. 5
Interestingly, our results show that patients had faster angiography suite to puncture times with the introduction of MAC. This suggests that interventionists may have had more time to focus on obtaining appropriate endovascular access while an anesthesiologist was able to appropriately manage the patient's comfort and vital signs. Additionally, nursing and technologist staff likely are better able to focus on transferring and preparing the patient and equipment when anesthesia providers are managing the airway and blood pressure. Importantly, there were no differences in the door to puncture, puncture to first pass, or puncture to successful reperfusion (TICI2B or 3) times within the matched cohort. Total procedure time, defined as the time of arterial puncture to placement of closure device, was significantly longer in the MAC group. This may be due to shifting team dynamics with the introduction of anesthesiology or variability in how procedural time is recorded by nursing staff.
Despite similar protocolized blood pressure goals in both groups, patients who were sedated under the management of an anesthesiologist were more likely to be administered vasoactive medications and have a statistically significant difference in minimum SBP compared to their counterparts receiving NACS. Perioperative blood pressure control is an important modifiable factor to ensure adequate cerebral perfusion during mechanical thrombectomy. Patients with poor collateral flow require a greater hypertensive response to maintain perfusion. 18 Furthermore, following recanalization, there is a significant risk of hyper-perfusion due to a dramatic increase in cerebral blood flow with associated brain injury and hemorrhage. 19 While this finding did not lead to worse outcomes at 90 days in our study, previous studies have suggested that aggressive blood pressure reduction was associated with unfavorable outcome, especially in patients undergoing general anesthesia. 20 Notably, there was no difference in post-thrombectomy hemorrhagic transformation (as defined as PH-1 or PH-2) in our study. Our results underscore the difficulty of blood pressure control in these patients. Both groups had minimum SBPs which were below the target goal.
There was no difference in complications or other safety parameters between the NACS and MAC group. Procedural complications including groin complications, emboli to a new territory, and vessel perforation were similar between the groups. Anesthesia-related complications were also similar in both groups including conversion to general anesthesia and post-operative pneumonia requiring antibiotics. Interestingly, our rate of emergent intubation was extremely low in both groups (1.8%), especially compared to other studies which suggest a rate closer to 10%.21,22 This is likely secondary to differences in stroke protocol including the fact that a large proportion of patients undergoing MT within our institution are transferred from spoke centers and patients who require intubation are often done so prior to transfer or on arrival to the emergency department. These intubated patients were excluded from this cohort. The majority of patients within this cohort underwent thrombectomy for anterior circulation LVO. Few patients (16.8% in NACS group and 18.1% in the MAC group) underwent thrombectomy for distal M2-3 occlusions. While numbers were small, there was no difference in outcomes with either MAC or NACS, suggesting that either could be used efficaciously.
While the results of our study suggest equivalent outcomes between MAC and NACS, the authors preferred MAC sedation as the primary modality of care. Our institution has transitioned completely to MAC for mechanical thrombectomy. NACS may be an option in resource-poor environments where anesthesia providers may not be available at all times. In our experience, NACS resulted in an increased cognitive burden on our interventionalists. While the majority of patients did not require much sedation, very agitated patients made performing a successful thrombectomy very difficult and were a significant distraction to the proceduralist. In agitated patients, it was quite difficult to appropriately balance sedation and blood pressure control as interventionalists were not typically able to start sedative drips.
Limitations
While the majority of the data collected for the study was done in a prospectively managed registry, the study does suffer from biases typically seen in single-center retrospective cohort studies. There are several potential known confounding variables and differences between the NACS and MAC group, which we attempted to correct for via propensity score matching. This may impact the generalizability of the data including for patients who underwent intervention via a radial arterial access. Posterior circulation strokes were also poorly represented in our cohort. Specific anesthetic dosing and level of sedation were not recorded and may have allowed for more granular analysis of NACS versus MAC sedation. Unknown confounding variables remain which could significantly alter the results of our study. Finally, this study does not completely account for variation in thrombectomy services provided with the rapid advancement of technology over the 4-year period studied.
Conclusion
The distinction between conscious sedation provided by a sedation-trained provider and an anesthesiologist is an important one as many institutions do not have sufficient resources to adequately staff every stroke intervention with an anesthesiologist at. Our results suggest that proceduralists trained in sedation can safely and effectively supervise nursing-provided sedation to patients undergoing mechanical thrombectomy for ischemic stroke. However, in resource rich environments, MAC may be preferred given faster door to puncture times and lower cognitive burden on interventionalists.
The authors have no relevant acknowledgements, competing interests, or funding associated with this manuscript.
The Human Research Protections Office at Penn State University waived the need for ethics approval and the need to obtain connect for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this study (Study ID 00018998).
Footnotes
Author contributions
VP contributed to conceptualization, methodology, software, formal analysis, data curation, and writing (original draft). CB contributed to methodology, formal analysis, data curation, and writing (original draft). CV, AR, and EC contributed to methodology and data curation. RR, EWC, DAW, and SDS contributed to methodology, data curation, investigation, and writing (review & editing). KMC provided conceptualization, methodology, formal analysis and writing (review and editing).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
