Abstract
Background
Transradial approach for neuroangiography is becoming increasingly popular because of the advantages demonstrated by interventional cardiology. Many advantages of radial access could be applied to intraoperative angiography.
Objective
To report our institutional experience with transradial and transulnar intraoperative angiography, and evaluate its safety and feasibility.
Methods
Intraoperative angiography through upper extremity vessels was attempted in 70 consecutive patients between April 2019 and December 2022. Data on patient characteristics and surgical indications, procedural variables, and complications were collected.
Results
Of the 70 patients who underwent intraoperative angiography, 58.6% were female, and the mean age was 52.9 ± 14.0 years. The reason for surgery was aneurysm clipping in 42 (60.0%) cases. In total, 55 patients (78.6%) were positioned supine, 13 (18.6%) prone, and two (2.9%) were positioned three-quarters prone. Access was attempted via the radial artery in 60 (85.7%) patients and the ulnar artery in 10 (14.3%) patients. The procedure was successful in 69 of 70 cases (98.6%), as one required conversion to transfemoral approach due to significant spasm in the proximal right radial artery. The median fluoroscopy time was 8 min. No procedure was aborted, and no patient experienced access-site or angiography-related complications. Intraoperative angiography altered the surgical management in 3 (4.3%) cases. Re-access for follow-up angiography was unsuccessful in three (13.6%) of 22 due to radial artery occlusion.
Conclusions
Our institutional experience supports that transradial and transulnar intraoperative angiography is safe and feasible during neurovascular procedures for various indications and positions.
Introduction
Intraoperative angiography (IOA) is valuable for cerebrovascular neurosurgeons to confirm aneurysm occlusion, parent vessel patency, arteriovenous malformation (AVM) resection, and dural arteriovenous fistula (dAVF) ligation.1,2 This allows surgeons to detect and revise any lesion residual within the operating room.1,3
IOA has been traditionally performed via the femoral artery. Postoperatively, the patient must remain supine with a straightened leg for up to 6 hours, which complicates postoperative recovery. Interventional cardiology has transitioned from femoral to radial artery access, as it reduces morbidity, mortality, and hospital costs and is preferred by patients.4–7 These advantages could be applied to neuroangiography.8,9,10–17,18 Similarly, many of the advantages of radial access could be transferred to IOA.3,19–21 Therefore, we aimed to present our experience with transradial IOA, describe the technique, and evaluate its safety and feasibility in various indications.
Methods
Study design
We conducted a retrospective study at our academic institution after Institutional Review Board approval. Because of the retrospective design of the study, individual consent was waived. Consecutive patients 18 years of age or older who underwent IOA for any neurosurgical indication with attempted vascular access to the upper extremity (transradial, either proximal or distal [at the anatomical snuffbox], and transulnar approach) between April 2019 to December 2022 were included.
Data collected included basic demographics, body mass index, smoking status, surgical indication (lesion location, rupture status), Spetzler-Martin (SM) grade 22 for AVMs, Cognard dAVF classification 23 for dAVFs, access site, patient positioning, contrast dose, fluoroscopy time, radiation exposure, number of vessels selected (excluding access site), conversion to femoral approach, any angiography-related and access site complications, and procedural abortion. We also recorded any previous angiography with access at the same site, and any repeat angiography at the same site at a later date focusing on success and artery patency.
Technique
After general anesthesia induction, access is obtained in the supine position before final positioning for the surgery in a hybrid biplane operating room (Siemens Artis Q or ARTIS icono) for the majority of cases. The wrist is prepped and draped using a sterile technique. A radiolucent carbon fiber radial arm board is typically used (STARBoard, Adept Medical, Auckland, New Zealand). The access site is evaluated using ultrasound, and alternative access is used if the anatomy is unfavorable. The skin overlying the radial or ulnar artery (whichever is larger) is locally anesthetized with 1% plain lidocaine. A small skin incision is occasionally made. Using a standard radial access kit, the micropuncture needle is advanced into the artery under real-time ultrasound guidance. A 5-French sheath is placed. A cocktail of 5 mg of verapamil, 200 mcg of nitroglycerin, and 2000 units of heparin is administered through the sheath. Heparin is withheld in ruptured cases. The patient's head is positioned on a radiolucent Mayfield head frame, and the wrist is positioned and taped against the hip (Figure 1A). In the prone position, the arms are tucked internally rotated by the patient's side with the thumbs pointing upward. After surgery, an intraoperative diagnostic angiogram is performed (Figure 1C-F). A 0.035'' Glidewire and a catheter are used to select vessels for angiography. Typically, a 5 Fr Simmons 2 Glide Catheter (Terumo) was used for carotid artery selection, and an Angle Taper Glide Catheter (Terumo) for vertebral artery selection. After the procedure, the catheter and sheath are removed, and hemostasis is achieved by placement of a TR band compression device (Figure 1B). The patient is then transferred to the neurological intensive care unit (ICU) intubated and sedated.
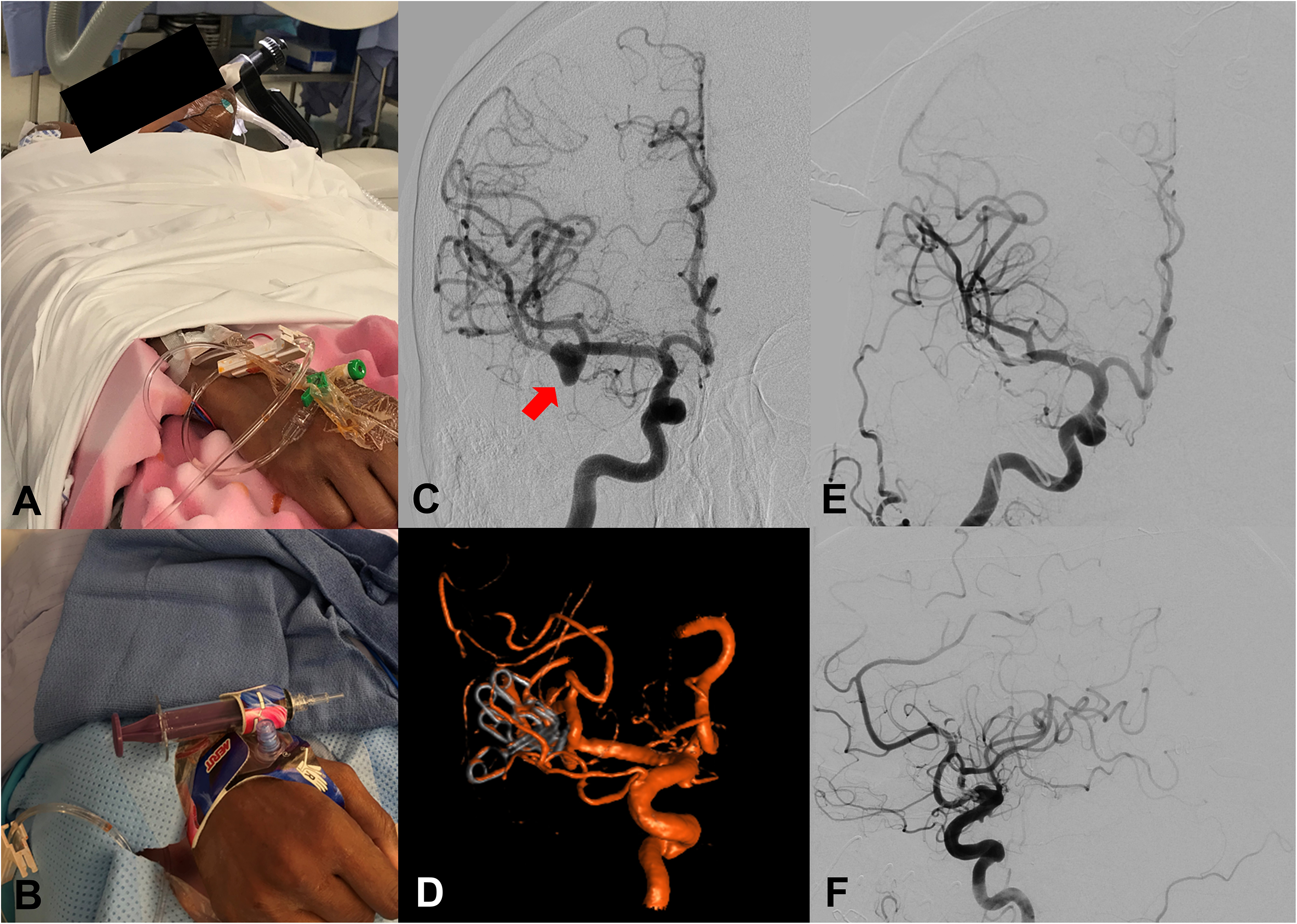
A patient with a 9 mm unruptured right MCA bifurcation aneurysm with a wide neck and associated calcification. The patient underwent right pterional craniotomy for clipping of the aneurysm with IOA. (A) Photograph of patient positioning and right radial sheath. (B) Photograph of the TR band compression device after the procedure and removal of the catheter and sheath. (C) Anteroposterior projection of the unruptured right MCA bifurcation aneurysm (red arrow). (D) 3D rotational angiogram reconstruction, (E) anteroposterior, and (F) lateral projections during the IOA demonstrating complete occlusion of the clipped right MCA bifurcation aneurysm. MCA: middle cerebral artery; IOA: intraoperative angiography; 3D: three-dimensional.
Statistical analysis
Categorical variables were presented as percentages. Continuous variables were presented as mean ± standard deviation, or median and interquartile range (IQR), depending on normality. All descriptive statistics were performed using the Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC.
Results
Patient characteristics and surgical indications
We included 70 consecutive patients who underwent IOA with attempted access through the upper extremity at our institution between April 2019 and December 2022. The mean age was 52.9 ± 14.0 years, and 58.6% were female.
The reason for surgery was aneurysm clipping in 42 (60.0%) cases, AVM resection in 14 (20.0%) cases, dAVF resection in 11 (15.7%) cases, decompression for bow hunter syndrome in 2 (2.9%) cases, meningioma resection in 1 (1.4%) case, and bypass surgery for Moyamoya disease in 1 (1.4%) case. Of them, one patient underwent both aneurysm clipping and AVM resection.
Of the 42 patients who underwent aneurysm clipping (15 [35.7%] for ruptured aneurysm), 16 (38.1%) had an aneurysm in the middle cerebral artery. Of the 14 resected AVMs, all were cranial, 11 (78.6%) were ruptured, and most of them were SM grade II (71.4%). Of the 11 dAVFs resected, 5 (45.5%) were ruptured, 1 (9.1%) thoracic spinal dAVF, and most of them were Cognard type IV (40.0%). Further details on patient characteristics and surgical indications are given in Table 1.
Patient characteristics and surgical indications.

a.: artery; ACA: anterior cerebral artery; ACoA: anterior communicating artery; AVM: arteriovenous malformation; dAVF: dural arteriovenous fistula; ICA: internal carotid artery; IQR: interquartile range; MCA: middle cerebral artery; min: minutes; PCA: posterior cerebral artery; PCoA: posterior communicating artery; PICA: posterior inferior cerebellar artery; SD: standard deviation.
*One patient underwent surgery for both aneurysm clipping and AVM resection.
**One of the 11 dAVFs was a thoracic spinal dAVF.
Postoperative vascular imaging was required before discharge in 12 patients (17.1%), mainly for vasospasm.
Procedural characteristics
The procedure was successful in 69 of 70 cases (98.6%), as one case required conversion to femoral access. Fifty-five patients (78.6%) were positioned supine, 13 (18.6%) prone, and two patients (2.9%) were placed in the three-quarters position. Access was attempted via the radial artery in 60 (85.7%) patients and via the ulnar artery in 10 (14.3%) patients. Of the radial cases, 52 (74.3%) were on the right arm, and 29 (41.4%) were at the distal radial site. A median of 1.0 (1.0–2.0) target vessel was selected, and the median fluoroscopy time was 8 (5.4–11.4) minutes. The majority of cases were targeted single-vessel angiographies, whereas some cases required more vessels to be selected depending on the location of pathology. This is applied to lesions with supply from multiple circulations. The contralateral vertebral artery was consistently able to be cannulated when rarely needed. More details on procedural characteristics are presented in Table 2.
Procedural characteristics.

AVM: arteriovenous malformation; dAVF: dural arteriovenous fistula; IQR: interquartile range.
*One conversion to femoral access.
Change in surgical management
IOA altered the surgical management in 3 (4.3%) cases. The first case was an SM grade 2 corpus callosum ruptured AVM resection where the IOA demonstrated residual arteriovenous shunting, and resection was continued. The second case was an unruptured enlarging right distal middle cerebral artery aneurysm clipping in which the IOA demonstrated an aneurysm residual that required clip repositioning. The third case was a ruptured right posterior communicating artery aneurysm clipping in which the IOA demonstrated that the internal carotid artery was kinked with the limited flow and required clip repositioning.
Technical failures and complications
One (1.4%) patient in the supine position required conversion to femoral access due to significant sheath spasm in the right radial artery (wrist), even though the cocktail mentioned above was administered. This patient had no other attempted access at the same site before IOA. No procedure was aborted, and no patient developed access-site or angiography-related complications such hematoma, arterial dissection, pseudoaneurysm, or stroke.
Access at the same site
Eighteen patients underwent access at the same site (all transradial, 10 distal radial access) for DSA prior to undergoing IOA. Access for IOA was successful at the same site in all cases. In 9 additional patients, the previous access was proximal, and the access for IOA was successful at the distal radial site.
Twenty-two patients underwent follow-up digital subtraction angiography (DSA) with attempted re-access at the same site (all transradial, 11 distal radial access). Re-access for follow-up DSA was unsuccessful in three (13.6%) patients (1 distal radial access) due to radial artery occlusion on ultrasound. Four additional patients with distal and 2 with proximal transradial approach for IOA underwent successful follow-up angiography via the proximal and distal radial arteries, respectively.
Discussion
IOA is a useful adjunct in neurovascular surgery, allowing determination of technical success, lesion obliteration, and parent vessel compromise before closing.1,2,24 Further, if done with high quality, it can eliminate the need for postoperative DSA. Our results demonstrate that transradial and transulnar approach for IOA is feasible and safe in various cerebrovascular pathologies, not only aneurysms, AVMs, and dAVFs as previously described,3,19,20,24 but also bow hunter syndrome, meningioma, and Moyamoya disease. No access site complications occurred, and transradial IOA was feasible for the supine, prone, and three-quarter prone positions.
Evidence from randomized trials has led the interventional cardiology community to transition to radial artery access.4–6 A growing body of evidence supports radial access for diagnostic angiography and neurointervention.8–17 Large series have demonstrated that IOA via the femoral artery is safe and useful, with complications ranging from 0.4% to 2.6% and changes in surgical management ranging from 8.2% to 12.4%.1,25,26 However, the literature on transradial IOA is limited. Case series have discussed transradial IOA, but included fewer patients, 3 and were limited to AVMs, 20 aneurysms, 19 or AVFs, 27 or included only non-supine patients24,28 Success rates ranged from 88.8% to 100%3,20,27,28; conversion to another access site ranged from 0% to 10%.3,19,20,24,27,28 In our study, which represents the largest series to date, IOA through the radial or ulnar arteries had a success rate of 100% without access site or angiography-related complications.
Complications related to angiography
Access site complications are lower with transradial versus transfemoral approach for neuroendovascular procedures (1.62% vs. 3.31%, P < 0.01). 11 Complications include spasm, occlusion, or perforation of the radial artery leading to forearm hematoma and compartment syndrome. Less common complications include pseudoaneurysm, arteriovenous fistula, granuloma, and sterile abscess formation. 11 In transradial IOA, the only angiography-related complication reported in the literature is a brachial artery dissection requiring an axillary to interosseous artery bypass. 24 In our series, no patients experienced angiography-related complications.
Conversion from radial to femoral access was found to be 4.77% in a systematic review by Joshi et al. 16 for neuroendovascular procedures, all involving severe radial artery spasm. Regarding transradial IOA, Osbun et al. 3 reported 2 (7.4%) cases of conversion to femoral access due to arterial vasospasm and inadvertent venous catheterization, while Chalouhi et al. reported one (10%) conversion to the contralateral radial artery due to chronic occlusion after a previous extensive ICU stay. 19 In our series, one (1.7%) case required conversion to femoral access due to a significant spasm in the radial artery.
Re-access and radial artery spasm
Based on the cardiology literature, heparin may decrease the rate of radial artery occlusion, making future re-access at the same site possible. 29 Although the administration of heparin can be controversial, we administer 2000 units of heparin through the sheath, along with 5 mg of verapamil and 200 mcg of nitroglycerin, but heparin is withheld in ruptured cases. One (1.4%) patient required conversion to femoral access due to significant sheath spasm after proximal radial access. This patient had no other attempted access at the same site prior to IOA. In the rare case of radial artery occlusion after distal radial access, the proximal radial artery can be accessed in the future. Furthermore, because the artery is punctured distal to the palmar collaterals, the distal radial access protects against hand ischemia. 30 While radial artery occlusion is more common than femoral artery occlusion, 11 it is usually asymptomatic. In our series, attempted re-access at the same site was unsuccessful in three (13.6%) of the 22 patients because the artery appeared to be occluded; in all of these three IOAs, heparin was administered through the sheath. Of them, one IOA case was via the distal radial site; in the follow-up DSA, the distal radial artery measured 1.6 mm and the proximal 2.0 mm, both partially occluded, so the ulnar artery was used, which measured 2.6 mm. As an alternative, although the ulnar artery is located deeper than the radial artery, has a less readily palpable pulse, a more difficult hemostatic control, and is immediately adjacent to a large nerve, the transulnar approach is non-inferior to the transradial approach in terms of safety and efficacy, 7 and might even result in fewer local bleeding complications. 31
Potential advantages of transradial IOA
There are several potential advantages of the transradial approach for IOA over the femoral artery.3,19,20,24 Although cerebral angiography through the radial artery has a learning curve, 10 radial access can actually be technically easier than femoral access in select patients, especially in obese patients or tall patients when the base of the operating table blocks the fluoroscopy unit.17,19 The radiolucent arm board under the slightly abducted patient arm allows visualization of catheters and guidewires during live fluoroscopy from the access point to the subclavian artery. The base of the operating table, cardiac monitoring wires, and headholder attachment do not restrict this relatively short and straight path.3,20
The duration of IOA is usually less than 10 min, allowing for prompt surgical adjustments.3,19 Similar to a femoral sheath, the radial sheath can also serve as an arterial line for anesthesiologists during the surgical phase, eliminating the requirement for a separate arterial line. While the diagnostic catheter is in the sheath during IOA, noninvasive blood pressure monitoring may be sufficient. 3 In line with anecdotal evidence from our institution, Hendrix et al. 21 found that dose-area product and fluoroscopy time were comparable between transradial and transfemoral IOA.
Additionally, applying the radial artery band at the end of the procedure to achieve hemostasis is less painful, invasive, time-consuming, and expensive than manual groin compression or a femoral artery closure device.3,7,15,17,19 The transradial approach also allows the bed head to be elevated after extubation to prevent cerebrospinal fluid leak and improve pulmonary function, and mobilize the patient faster.3,7,15,19 Invasive blood pressure monitoring can continue via radial access for up to 24 h postoperatively without requiring a separate arterial line. Radial access allows unrestricted arm movement without complications, whereas coughing or involuntary leg movement can lead to bleeding from a recently closed femoral access site. 3
For posterior vascular lesions requiring non-supine positioning, patients must either be turned back supine, extend the femoral sheath in the prone position and keep it patent with anticoagulation to prevent thrombosis, or use an access site in the upper extremity20,24 or the popliteal fossa. 32 The transradial approach can be used in the prone and three-quarter prone positions, in which transfemoral approach requires long or reinforced sheaths, and sometimes the knee has to be flexed. In the prone position, the wrist rests in a natural position at the patient's side, the arm is relatively free, sterility is improved, access is easy throughout the operation, and the kinking of the femoral sheath is eliminated. Additionally, femoral artery injury, thrombosis, or skin injury due to long operative time and pressure from the patient's weight are avoided.20,24 However, reaching below the end of the thoracic spine with a standard catheter is difficult, interventionalists are less familiar with the transradial approach, 24 and imaging the aortic arch for vessel selection can be challenging when the image is inverted. 3
Limitations
This is a retrospective case series, reflecting the experience of a single cerebrovascular center and may not be generalizable. There is no comparison between transradial and transfemoral IOA; thus, we cannot conclude that transradial IOA is safer. The level of granular data of transfemoral IOA at our center was limited, with many cases having been done during a prior era and a different medical record. Comparisons are made here to previously published IOA studies. A prospective and larger study comparing transradial with transfemoral IOA is needed.
Conclusions
We have described our experience with transradial and transulnar IOA for neurovascular indications. Our results reaffirm that IOA via the radial and ulnar arteries is feasible and safe during surgical treatment of various neurological lesions, with several potential benefits over the transfemoral approach.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
